Abstract
Cardiovascular disease is increasingly recognized as an important cause of morbidity and mortality in captive chimpanzees (Pan troglodytes). This report records 2 cases of sudden cardiac death in closely related subadult captive chimpanzees with marked replacement fibrosis and adipocyte infiltration of the myocardium, which resemble specific atypical forms of the familial human disease arrhythmogenic right ventricular cardiomyopathy. Changes were consistent with left-dominant and biventricular subtypes, which are both phenotypic variants found within human families with familial arrhythmogenic right ventricular cardiomyopathy. Previously reported fibrosing cardiomyopathies in chimpanzees were characterized by nonspecific interstitial fibrosis, in contrast to the replacement fibrofatty infiltration with predilection for the outer myocardium seen in these 2 cases. To the authors’ knowledge, this case report is the first to describe cardiomyopathy resembling arrhythmogenic right ventricular cardiomyopathy in nonhuman primates and the first to describe left-dominant arrhythmogenic cardiomyopathy-type lesions in an animal.
Cardiovascular disease is increasingly recognized as an important cause of morbidity and mortality in captive chimpanzees (Pan troglodytes).7,8,16,17,19,20,26,31,37 In the past 2 decades, cardiovascular disease has overtaken infectious disease as the primary cause of death of captive chimpanzees. 52 Sudden cardiac death (SCD) secondary to fibrosing cardiomyopathy occurs in a significant proportion of these cases. 19,20,52 Fibrosing cardiomyopathies have also been reported in captive western lowland gorillas (Gorilla gorilla gorilla) and orangutans (Pongo pygmaeus). 26,36 Although it is now recognized that cardiomyopathies are a very important disease group of captive great apes, the etiology of these conditions remains a mystery. Proposed initiating factors include genetic susceptibility, viral myocarditis (including but not limited to the picornaviruses, coxsackie A and B viruses, encephalomyocarditis, and influenza viruses), vitamin E/selenium deficiency, hypertension, arteriosclerosis, obesity, stress, and other infectious or toxic agents. 36
Previous reports of myocardial fibrosis in captive chimpanzees describe nonspecific diffuse interstitial fibrosis, 19,20,37,52 and this lesion has been referred to as the “leading cause of death in this species.” 31 The terms myocardial fibrosis, 16,26 idiopathic cardiomyopathy, 37 and interstitial myocardial fibrosis 19,52 have all been used to describe fibrotic lesions in chimpanzee hearts, although they all appear to describe equivalent changes characterized by nonspecific, diffuse interstitial fibrosis of the myocardium. No reports describe examination of chimpanzee hearts according to protocols used to examine human hearts following suspected SCD. Studies of wild chimpanzee populations are very limited, although there is a recent review of full postmortem findings of 11 wild chimpanzees from Gombe, Tanzania. 50 Cardiac disease was not considered a significant pathological finding in any chimpanzee in this study, and cardiac findings were limited to 2 chimpanzees with mild to moderate myocardial fibrosis that had a predilection for periarteriolar myocardium. 50
This report records 2 cases of sudden cardiac death in closely related subadult captive chimpanzees that resemble specific atypical forms of the familial human disease—arrhythmogenic right ventricular cardiomyopathy (ARVC). ARVC is a well-described cardiomyopathy of humans, previously referred to as arrhythmogenic right ventricular dysplasia. 45 Macroscopic findings include dilatation of the right ventricle, replacement of normal myocardium with fibrofatty tissue, and thinning of the right ventricular wall. Microscopic findings are characterized by the replacement of the outer and middle layers of right ventricular myocardium by fibrocytes and adipose tissue. 15 ARVC has an estimated prevalence of 1 in 5000 in humans and is implicated in up to 20% of sudden deaths of young people, and the incidence of familial presentation ranges between 15% and 50%. 10,15 ARVC has also been recognized in dogs, cats, and Polled Hereford cattle. 3,14,46 Although a genetic predisposition is recognized, the pathogenesis of ARVC is not fully understood. This report describes the clinical and pathological features associated with 2 sudden deaths within the captive chimpanzee population at ZSL Whipsnade Zoo in the United Kingdom that closely resemble left-dominant and biventricular variants of ARVC in humans.
Case Histories and Gross Findings
The affected chimpanzee group is within a zoological collection and originally comprised a wild-caught adult male (male A) and two unrelated captive-born adult females (female B and female C) (Fig. 1).
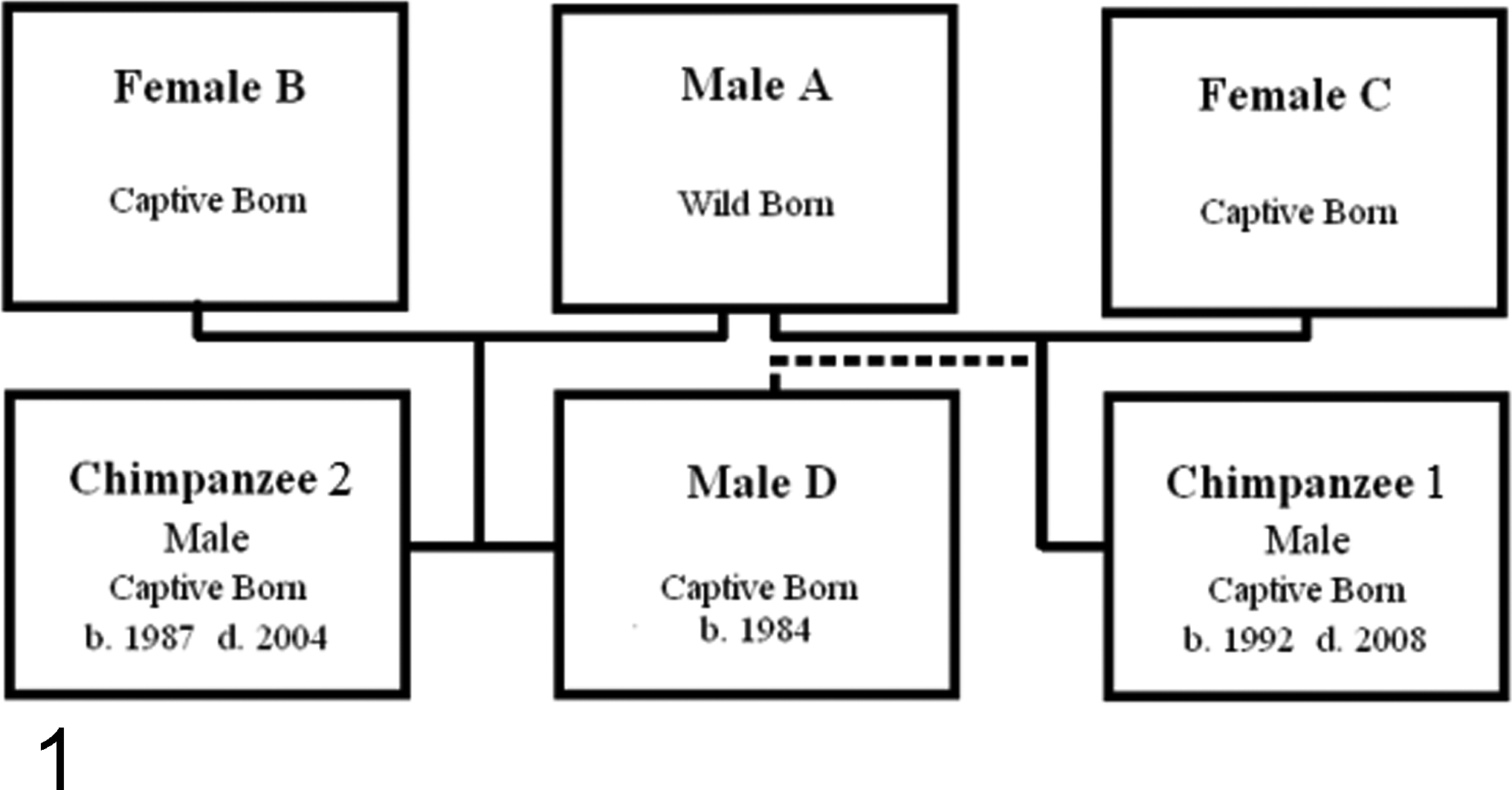
Family tree of affected chimpanzee group. Dotted line indicates possible alternate paternal lineage for chimpanzee No. 1.
Chimpanzee No. 2 was born within the collection in 1987 to male A and female B. Chimpanzee No. 1 was born in 1992 to female C and was probably sired by male A, making him chimpanzee No. 2’s half-brother. It is also possible that he was sired by his half-brother, male D, who was replacing male A as the dominant breeding male around that time. The population was housed in a mixed indoor-outdoor enclosure.
Chimpanzee No. 1
Chimpanzee No. 1 died in June 2008 at 16 years of age. He was observed to collapse in his enclosure with no apparent prior clinical signs or group violence. While under general anesthesia (GA) to investigate the collapse, he succumbed to cardiac arrest, which was nonresponsive to attempts at cardiopulmonary resuscitation, including the administration of intravenous and intracardiac adrenaline (adrenaline [epinephrine], 1 mg; Hamelin Pharmaceuticals, Gloucester, UK), and intravenous doxopram (Dopram-V injection, 100 mg; Fort Dodge, Iowa, USA).
He exhibited no clinical signs of cardiac disease during his life. He was assessed under GA 4 times between 2006 and May 2008, during which no cardiac abnormalities were detected. Throughout life, he did not have any diagnostic cardiac procedures (echocardiography, electrocardiogram, thoracic radiography). Routine blood testing for cardiac markers was initiated in 2007, and in November 2007, he was found to have raised concentrations of both pro–brain natriuretic peptide (BNP) (259 pg/ml; normal in humans <30 pg/ml) and troponin I (0.48 ng/ml; significant in chimpanzees >0.2 ng/ml). At the time of testing, normal values for these markers in chimpanzees were not known—although it has subsequently been demonstrated that his troponin I level was elevated. 9
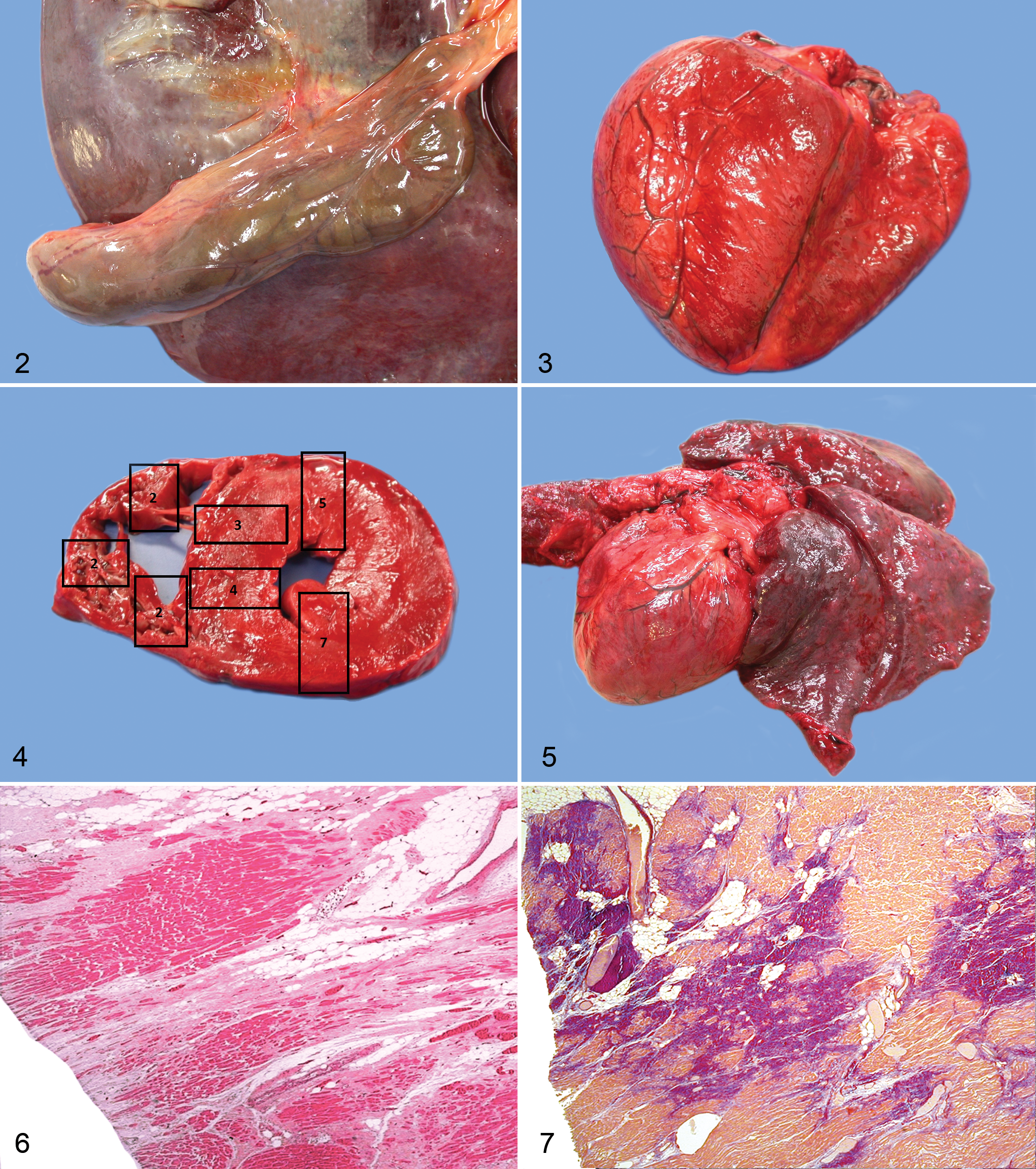
On gross postmortem examination, he was found to be in normal body condition and hydration (60 kg). He had patchy hyperpigmentation diffusely and extensive patchy hair loss over his back, back of neck, thorax, and dorsolateral lumbar region. He had marked straw-colored abdominal and pericardial effusions. Both ventricles of the heart appeared thickened; the left ventricular free wall measured 19 mm and the right 3 mm (normal dimensions for chimpanzee hearts are not established). The left ventricle had a reduced lumen, and fat was visible in the epicardial surface of the right ventricle, particularly the anterior and lateral wall. There was no fat in the left ventricle. A heart weight was not obtained. His major arteries and lungs were grossly normal. There was some patchy hyperemia of the primary and secondary bronchi. His liver was markedly enlarged and bronze colored, and the gallbladder was moderately edematous (Fig. 2). The spleen was congested and slightly enlarged. The kidneys and meninges were both mildly congested. Bacteriological swabs of heart blood produced no growth on routine and chocolate blood agar under aerobic, anaerobic, and carbon dioxide conditions.
Chimpanzee No. 2
In March 2004, chimpanzee No. 2 was found dead at 17 years old with no previous signs of ill health or external injuries. Previous physical examinations in 1993, 1995, and 1999 detected no clinical signs of cardiac disease. His most recent clinical examination under GA was in 1999, during which he was assessed as clinically normal based on physical examination and routine hematology and biochemistry parameters. Throughout life, he did not have any specific cardiac investigations (echocardiography, electrocardiogram, thoracic radiography), and cardiac blood parameters (troponin I, pro-BNP) were never measured.
At gross postmortem examination, he was found to be in normal body condition (64 kg). The heart was globose with a squared apex (Fig. 3). The left ventricular free wall and septum measured 21 mm, and the right ventricular free wall measured 3 mm (Fig. 4). The wall of the left ventricle was assessed as moderately thickened and the right ventricle as mildly dilated. A heart weight was not obtained. There was no evidence of fat in the right or left ventricle. The lungs were markedly congested (Fig. 5). There were multiple tough adhesions between the colon and the abdominal wall. There was multifocal reddening of the base of the ventral neck and inguinal areas, consistent with subcutaneous contusion. The gingiva were cyanotic. Urinalysis revealed moderate proteinuria and was negative for glucose and ketones; the specific gravity was 1.030 and pH was 7.0.
Histological Examination
Routine histological examination revealed significant evidence of cardiac abnormalities and acute cardiac failure in both chimpanzees. Alongside histological examination of routine tissues, an extended examination was performed on the hearts of both chimpanzees. Fifteen heart sections were cut according to a routine protocol for the investigation of cardiomyopathy in humans (Fig. 4). 44 The following cardiac regions were reviewed by one of the authors (M.N.S.): right ventricular outflow tract (2 sections); the lateral right atrium, tricuspid valve leaflet, and right ventricle; the right ventricle (3 sections: anterior, lateral, and posterior); the interventricular septum (2 sections: anterior and posterior); anterior left ventricle; the lateral left atrium, mitral valve leaflet, and left ventricle; the posterior left ventricle and aorta; the sinoatrial node (2 sections); and the atrioventricular node (2 sections).
Chimpanzee No. 1
There was marked replacement fibrosis with fat in the outer half of the right ventricle (RV) throughout all areas sampled (Fig. 6). Sixty percent of normal RV myocardium was replaced by fibrofatty tissue. There was also marked transmural interstitial and transmural replacement fibrosis in the left ventricle (LV) with little fat (Figs. 7, 8) and interstitial diffuse fibrosis in the interventricular septum (IVS) (Fig. 9). Forty percent of normal LV myocardium was replaced by fibrofatty tissue. There was marked myocyte hypertrophy with degenerate myocytes in the LV and IVS (Fig. 8). There was no myocardial disarray in any region. There were rare inflammatory cells, limited to a few foamy histiocytes and lymphocytes surrounding myoctyes in the IVS (posterior > anterior) and occasional eosinophils in the posterior IVS. There were no vascular changes in any region. Staining with elastic van Gieson confirmed the presence of replacement and interstitial fibrosis (Fig. 7).
Within the lungs, there were densely packed erythrocytes in many alveoli (hemorrhage) and occasional intra-alveolar hemosiderophages throughout the lung, although not necessarily associated with hemorrhage. There was minor alveolar hyperinflation multifocally, which alternated with minor foci of atelectasis. There was a minor quantity of intralumenal mucus and occasional erythrocytes within bronchi and bronchioles. There was mild generalized hepatic congestion, and within sinusoids, there were frequent circulating globular eosinophilic bodies (fibrin globules). Kupffer cells, focal clusters of macrophages, and mid-zonal and periacinar hepatocytes contained mild to moderate amounts of granular gold pigment (hemosiderin). The submucosal lymphatic vessels of the gallbladder were dilated. The meningeal blood vessels were sometimes congested. In the spleen, there was mild generalized congestion of the red pulp, with scattered hemosiderophages, and in the kidneys, there was moderate generalized congestion. There was multifocal moderate congestion and possible hemorrhage of the pancreatic interstitium.
Chimpanzee No. 2
Thirty percent of normal RV myocardium was replacement by fibrofatty tissue (Fig. 10). There was mild focal interstitial and replacement fibrosis with fat in the epicardium of the right ventricular outflow tract of the RV. There was marked replacement fibrosis admixed with fat in the outer one-third of the anterior, lateral, and posterior LV (Fig. 11). Affected areas showed replacement of 50% of normal myocardium with fibrofatty tissue. A section of IVS showed widespread transmural replacement and interstitial fibrosis. There was marked myocyte hypertrophy and also degenerate smaller myocytes in the areas of fibrosis and fat in the outer one-third of the LV and IVS. No myocyte disarray or inflammation was noted in any section. Epicardial vessels were normal. Staining with elastic van Gieson confirmed the presence of replacement and interstitial fibrosis (Fig. 10, inset and Fig. 11).
Within the lung, there was generalized moderate to marked congestion, extensive multifocal flooding of alveoli with amorphous eosinophilic fluid (edema), and scattered foci of atelectasis. There was engorgement of tracheal proprial vasculature. Hepatocytes, especially periportally, often contained intracytoplasmic brown pigment granules (iron or lipofuscin), and a few focal sinusoids were locally dilated. There was moderate generalized congestion within the kidney, particularly of the medullary vasculature. There was moderate congestion of a subcutaneous (presumed cervical) lymph node, with mild hemorrhage into the cortex and adjacent adipose tissue.
Age-Matched Controls
For purposes of comparison, 3 additional hearts have been examined. One heart is from a 19-year-old female human (Homo sapiens) who was posthumously diagnosed with ARVC with left-dominant changes. Two hearts from unrelated captive-born 16-year-old male chimpanzees from an Australian zoological collection were also assessed. Routine sections of RV, LV, and IVS were examined, including hematoxylin and eosin and Masson’s trichrome–stained sections.
Human A
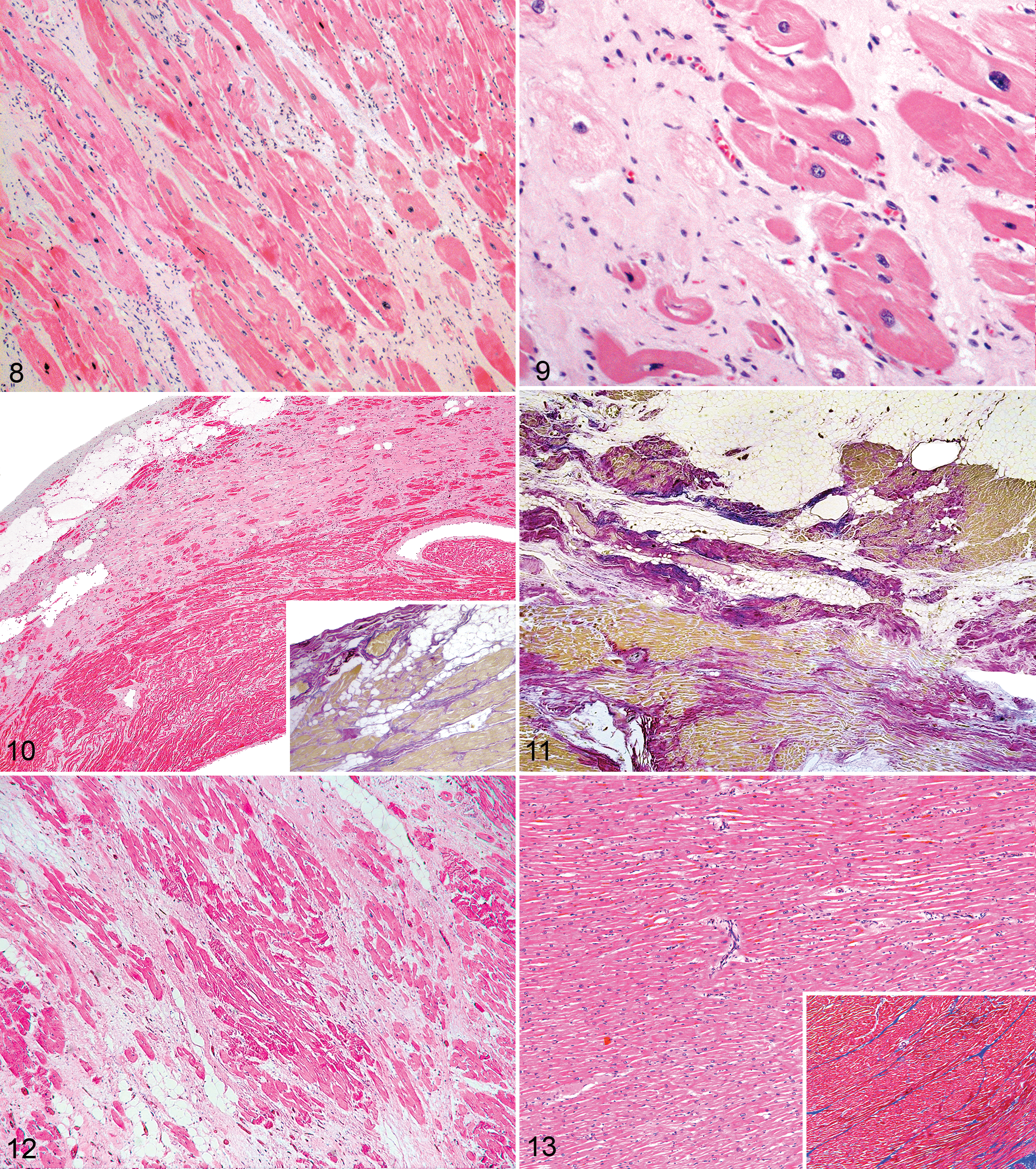
Human A was a 19-year-old Australian female who collapsed suddenly at home following vague symptoms of aching; attempts to resuscitate her were unsuccessful. She had no previous clinical history of heart disease. Full postmortem examination submitted to the NSW State Coroner stated that death was due to acute cardiac arrhythmia secondary to ARVC, and the report indicated significant involvement of the left ventricle, considered consistent with a left-dominant pattern by the reporting forensic pathologist. Histological examination showed extensive mature fibrofatty replacement of the posterior LV wall (Fig. 12). Staining with Masson’s trichrome confirmed the presence of marked replacement and interstitial fibrosis around atrophied myocytes. There were less pronounced fibrofatty foci elsewhere focally in the LV myocardium. The RV myocardium showed moderate to marked fatty replacement, although only very focal associated fibrosis. There was moderate interstitial and replacement fibrosis of the IVS myocardium, where myocyte hypertrophy and degenerate atrophied myocytes were found within areas of marked fibrosis. There were no active inflammatory changes, with only focal mild lymphocyte infiltrates in the LV myocardium. There were no other significant findings on postmortem examination.
Chimpanzee No. 3
Chimpanzee No. 3 was a 16-year-old male in good body condition (79.8 kg) who died during a fight with a cage-mate. He was observed to fall backwards into a moat and sank to the bottom apparently unconscious. He was recovered to the veterinary hospital, and an attempt was made to resuscitate him for 40 minutes using oxygen therapy and cardiopulmonary resuscitation. He was not successfully revived. He had no known prior clinical problems and no history of clinical heart disease. Gross examination and histopathological examination confirmed cause of death was by drowning, characterized by flooding of alveoli with lightly proteinaceous fluid in a patchy distribution, and small aggregates of brown, granular material and plant matter were within alveoli and bronchi. There were no other significant findings. Examination of the heart showed no gross evidence of vascular plaques, valvular disease, or myocardial disease. Histological examination of the LV and RV showed no pathological features. Examination of RV and LV heart sections stained with Masson’s trichrome showed normal distribution of collagen.
Chimpanzee No. 4
Chimpanzee No. 4 was a 16-year-old male in poor body condition (48.0 kg) who was euthanized due to clinical chronic wasting and identification of multifocal purulent lymphadenitis associated with acid-fast bacteria. Annual health checks under GA between 2000 and 2009 revealed no evidence of cardiac disease. Gross and histopathological examination revealed systemic pyogranulomatous mycobacteriosis. The agent of disease was confirmed as Mycobacterium tuberculosis complex using culture and molecular diagnostics. The heart was grossly and histopathologically normal (Fig. 13). Examination of RV, LV, and IVS heart sections stained with Masson’s trichrome showed normal collagen distribution between myocyte bundles and surrounding vessels (Fig. 13, inset).
Immunohistochemistry
Formalin-fixed, paraffin-embedded sections of ventricular myocardium from chimpanzee Nos. 1 and 2 were submitted for immunohistochemical staining using antiplakoglobin and antidesmoplakin antibodies. Examination of these sections showed that chimpanzee No. 1 had apparent loss of plakophilin and reduced desmoplakin, and chimpanzee No. 2 appeared normal for both proteins.
Discussion
Histological examination demonstrated histological cardiac changes that closely resemble ARVC of the biventricular (chimpanzee No. 1) and left-dominant (chimpanzee No. 2) subtypes described in humans. 11,18,42
The features that support a diagnosis of ARVC and distinguish these 2 cases from previous descriptions of great ape fibrosing cardiomyopathies include fibrofatty replacement and interstitial fibrosis of the outer one-third of the myocardium in the right (chimpanzee No. 1) or left (chimpanzee No. 2) ventricles, predominance of replacement over interstitial fibrosis, presence of adipocyte infiltration, the absence of significant inflammation, the lack of evidence of chronic congestive heart failure, the absence of myocyte disarray, and the absence of vascular pathology.
Previously reported cardiomyopathies in chimpanzees include fibrosing cardiomyopathies (interstitial myocardial fibrosis or nonspecific fibrosis), 16,19,20,26,37,52 dilated cardiomyopathy, 47 hypertrophic cardiomyopathy, 16 and nonspecific, viral, and protozoal myocarditis. 29,34,37 None of these are histologically consistent with the lesions observed in chimpanzee Nos. 1 and 2. In contrast, the similarities between the histopathology of chimpanzee Nos. 1 and 2 and human ARVC descriptions are striking. Previous studies in chimpanzees report interstitial fibrosis, as opposed to the predominantly replacement fibrosis in chimpanzee Nos. 1 and 2. 19,20,52 In addition, in previous studies of chimpanzees, fibrosis was distributed haphazardly throughout the cardiac muscle with no described predilection for specific ventricles or regions of the ventricular wall, and there was no reported accompanying adipose infiltration as seen in chimpanzee Nos. 1 and 2. 19,52
There was myocyte hypertrophy of the LV and IVS in these 2 chimpanzees. Interpreting the significance of this finding is complicated by the report that all 10 wild subadult and adult chimpanzees in the field study from Tanzania had marked variation in cardiac myofiber size and megalokaryosis and dyskaryosis. 50 Despite the myocyte hypertrophy, 8 of these chimpanzees had no additional signs of myocardial disease, and only 2 were assessed as having subclinical myocardial fibrosis. 50 Other reports of myocardial fibrosis in apes also commonly refer to a degree of hypertrophy, and it is reported to be a regular finding in “normal” great ape hearts. 19 A study of echocardiographic values of normal gorilla hearts (n = 99) noted that there was progressive LV hypertrophy in all male gorillas older than 11 years. 27 Normal echocardiographic values or postmortem cardiac measurements for chimpanzees are not established. There was no myofiber disarray in the chimpanzees, which is a prerequisite for diagnosis of hypertrophic cardiomyopathy in humans. 49
Terminal Arrhythmia in Chimpanzee Nos. 1 and 2
Although there is no antemortem clinical demonstration of arrhythmia in these 2 chimps, histological findings are supportive of fatal arrhythmia. In chimpanzee No. 2, there was considerable pulmonary edema and mild congestion without evidence of congestive disease (such as hemosiderophages), which implies per-acute cardiac failure. In chimpanzee No. 1, there were mild numbers of intra-alveolar hemosiderophages, implying mild (nonfatal) left-sided failure but no evidence of significant chronic failure. There was no histological evidence for clinically significant right-sided congestive heart failure in either chimpanzee.
An additional factor may have contributed to proarrhythmia in chimpanzee No. 1. From January 2008 until his death, he and others in the group were medicated with sulpiride (Supiride, 300 mg BID; CP Pharmaceuticals, Wrexham, UK) and haloperidol (Haldol Oral Solution, 1 mg SID; Janssen-Cilag, High Wycombe, UK) to treat persistent group aggression. Sulpiride and haloperidol have the potential to prolong the electrocardiographic Q-T interval in humans and are associated with a predisposition to fatal arrhythmias. 1,21,54
Pathogenesis of Arrhythmogenic Right Ventricular Cardiomyopathy
ARVC is characterized by replacement fibrosis with fat of the myocardium. There have been multiple theories regarding its pathogenesis. It seems likely that the disease is multifactorial or has multiple modes of pathogenesis. Although it is primarily thought to be a genetic disease, additional pathogenic mechanisms have been suggested either as causes of ARVC or as environmental factors that facilitate gene expression. These include but are not limited to myocarditis, mechanical stress (intense exercise), and apoptosis. 5,6,23,38,41,48,53
Continuing genetic studies support the theory that desmosomal dysfunction is the ultimate pathway of disease expression in ARVC. 38 Cardiac desmosomes form intercellular junctions between neighboring cardiomyocytes and are essential for normal mechanical and electrical function of myocardial tissue. 35 Various models and theories have attempted to explain how desmosomal dysfunction ultimately occurs.
The primary model is known as the degeneration-inflammation model. 38 Normal desmosomes anchor intermediate filaments to cytoplasmic membranes of adjacent cells, giving tissue its strength. Mutations in desmosomal genes affect the tissues’ ability to withstand mechanical stress, and normal cardiac function may instead cause detachment of cardiomyocytes and necrosis. 38 This may be accompanied by an inflammatory response—after which necrosis and inflammation are replaced by fibrocytes and/or adipocytes. 38,40,41 It is theorized that the predilection for right ventricular changes may be due to the thin wall and high distensibility of the RV. However, to fulfill its normal function, the left ventricle must conversely be capable of withstanding high systolic pressure. Therefore, weakness in desmosomal structures is thought to be capable of causing pathology in both ventricles due to the requirements of their respective physiological functions. 38
Interpretation of familial susceptibility is complicated by the considerable genetic and allelic diversity across individuals with ARVC. 41 Within families with identical mutations, there are marked variations in clinical features and outcomes and age of onset of disease. 38 This is well demonstrated by subtype discordance, where 67% of families include multiple individuals who manifest the different disease patterns (classic, biventricular, or left-dominant subtypes). 43
In humans and Boxer dogs, autosomal dominant or recessive inheritance of ARVC with variable penetrance has been demonstrated. 2,16 Disease-causing mutations are found in genes coding for desmosomal components. Causal mutations have been identified in desmoplakin (DSP), plakoglobin (JUP), plakophilin 2 (PKP2), desmoglein 2 (DSG2), and desmocollin 2 (DSC2). 30,40,41 In addition, extra-desmosomal genes have also been associated with ARVC, including transforming growth factor β3 (TGFβ3), transmembrane protein 43 (TMEM43), and cardiac ryanodine receptor (RyR2). 24,40,41 Only 30% to 40% of people with ARVC have mutations in one of the known disease-causing genes. 41 Phenotypic expression of ARVC is thought to have a complex relationship with causal genotype. It is hypothesized that causal mutations may only manifest as disease when accompanied by other genetic variants or environmental influences. 41,43
The challenges of genetic diagnosis in humans are amplified when applied to chimpanzees. Currently, a fundamental barrier is that the chimpanzee genome is not complete 51 and is missing regions within both desmosomal and sarcomeric genes. Even with full knowledge of the chimpanzee genome, it is an expectation that genetic diagnosis could face the same challenges of highly variable mutational status and phenotypic expression as described in humans. Some preliminary genetic studies have been undertaken on chimpanzee Nos. 1 and 2, although work is currently incomplete. Exploration of potential pathogenic mutations will be an important future step to understanding ARVC-like disease in chimpanzees.
Myocarditis in ARVC
The presence of patchy inflammation is an expected finding in ARVC in humans—the reported prevalence varies from 5.5% to 79%. 5,38,48 A study of 12 Boxer dogs with ARVC demonstrated concurrent myocarditis in 5 dogs (41.7%). 32 Generally, in humans and dogs with ARVC, it is expected that scattered interstitial lymphocytes are seen surrounding necrotic or degenerative myocytes. 3,5,32 This pattern was observed in chimpanzee No. 1. No inflammation was present in chimpanzee No. 2’s myocardium.
The possible role of viral myocarditis in the development of ARVC in humans and nonhuman animals is hypothesized. In previous studies, the prevalence of a viral genome in myocardial biopsy specimens from patients with ARVC ranged from 0% to 75%. 38,39 If or when viral myocarditis occurs in ARVC, detection is problematic as myocardial damage reportedly manifests well after the original insult, when the amount of detectable virus is greatly reduced or absent. 12 Familial predisposition to ARVC could be influenced by anti-infective and autoimmune responses to viral myocarditis, manifesting in clinical ARVC. 12 An alternate theory is that myocarditis associated with ARVC is noninfectious in origin.
Fatal myocarditis is occasionally reported in chimpanzees; viral agents have been identified twice due to encephalomyocarditis 34 and coxsackie B viruses, 29 respectively. Myocarditis in chimpanzees is characterized by intense lymphocytic infiltration of the myocardium and myocardial necrosis. 17,29,34 This is significantly different from the myocardial inflammation of chimpanzee No. 2, whereas chimpanzee No. 1 had no myocardial inflammation.
Left-Dominant Arrhythmogenic Cardiomyopathy
ARVC was originally defined as a condition that primarily affects the right ventricle, although increasingly, cases are being reported demonstrating involvement of the left ventricle. 11,18,22,42 Left-dominant arrhythmogenic cardiomyopathy (LDAC) is now a recognized variant of ARVC in humans, characterized by fibroadipose replacement of the LV with relative sparing of the RV. 42 The regions of myocardium usually affected are the outer one-third of the LV, as seen in chimpanzee No. 2, and the right side of the IVS. The biventricular variant is another recognized subtype defined by bilaterally marked involvement of ventricles, as seen in chimpanzee No. 1’s heart. 33,43 The left-dominant and biventricular variants of this condition are recognized as phenotypes of the same genetic mutation causing ARVC, and variable phenotypes are observed within human families with ARVC. 42 LDAC is primarily observed in human SCD victims but may also be diagnosed in vivo. 42 There has been some suggestion that biventricular ARVC may represent an end stage or a highly progressed manifestation of disease. 11,13,33 ARVC lesions are thought to arise in the subepicardial myocardium and gradually extend toward the endocardium, thus creating the potential for transmural lesions. 5,48 Previous reports of ARVC with left ventricular involvement in animals had concurrent marked right ventricular disease as per descriptions of biventricular ARVC in humans. These include 1 case in the cat and 4 in the dog. 4,28
Diagnostic Immunoanalysis of ARVC in Chimpanzees
Immunohistochemical analysis using anti-plakoglobin markers is currently the most reliable antemortem diagnostic tool for ARVC in humans, although it remains problematic. 2,25 A marked reduction in immunoreactive signal levels for plakoglobin in endomyocardial biopsy specimens is reportedly diagnostic for ARVC in humans. 2 However, there has been some debate regarding the reliability of the test, with the largest study showing a sensitivity of 85% and a specificity of 57%. 25 In addition, interpreting the results of anti-plakoglobin and anti-desmoglein immunoanalysis of chimpanzees has further challenges. These antibodies are not validated for use in this species, and we cannot assume good cross-reactivity or that equivalent optimal methods, such as antibody dilution values, will apply to chimpanzees. Further work, particularly validation of the antibodies, is required to extend the useful application of immunoanalysis to great apes.
Conclusion
To our knowledge, this case report is the first to describe a cardiomyopathy resembling ARVC in nonhuman primates and the first description of left-dominant distribution in animals.
The nomenclature of ARVC is beginning to shift toward the umbrella term arrthymogenic cardiomyopathy 38 to reflect the broader spectrum of disease that is now recognized in humans. These findings in chimpanzees add weight to recent findings that a similar shift may also be appropriate in veterinary medicine. 4,28
Furthering our understanding of these cardiomyopathies will require thorough cardiopathological examination of necropsied chimpanzees and other great apes—as per protocols for suspected human cardiac fatalities. Careful descriptions of the morphological subtypes (replacement vs interstitial) of myocardial fibrosis are also necessary. A parallel multicenter retrospective study implementing such a protocol would also go a long way to progress the assessment of great ape cardiomyopathy.
Identifying potential genetic and environmental risk factors for cardiovascular disease in great apes is an important goal. In humans, diagnosis of ARVC immediately leads to assessment of first-degree family members by a cardiologist, and similar protocols may be appropriate for great apes. New groups such as the Great Ape Heart Project (http://greatapeheartproject.org) and The Primate Heart Project (http://www.primateheartproject.co.uk), allow clinicians, pathologists, and others to share experience, expertise, and data. Such collaboration is critical because of the relative infrequency of deaths or diagnoses in individual captive groups. Increasing our knowledge of this disease is likely to improve our ability to preserve this precious species and will contribute to the collective understanding of a disease that affects both humans and animals.
Footnotes
Acknowledgements
We thank Belinda Clark, Elaine Chew, Karen Barnes, Huy Tran, and Catherine Wong for their technical expertise with gross and histological specimens and Associate Professor Mark Krockenberger for his assistance in facilitating processing of chimpanzee slides. Our grateful thanks are also extended to Dr Cheryl Sangster, DVM (Taronga Zoo, Conservation Society Australia), for her kind assistance with acquiring chimpanzee age-matched control cases and Dr Matthew Orde, MD (Department of Forensic Medicine, NSW Forensic & Analytical Science Service), for his kind assistance in acquiring human age-matched control cases.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
