Abstract
A 1.4-year-old virgin female brown-hooded fancy rat presented for abdominal distention, jaundice, and dyspnea. At physical examination, a firm mass was palpable in the caudoventral abdomen as well as multiple small nodular masses associated with the abdominal viscera. At necropsy, in addition to a large mass replacing the left ovary and myriad nodules studding the peritoneal surface, there was 31 ml of abdominal effusion. By cytology, the abdominal fluid contained numerous pleomorphic vacuolated tumor cells surrounding globular pale eosinophilic to amphophilic acellular material that was strongly periodic acid–Schiff positive. Histologically, the tumor was biphasic with abundant acellular hyaline matrix that was also periodic acid–Schiff positive.
Keywords
Background
Primary ovarian tumors are uncommon in rats and most often are gonadal (sex-cord) stromal tumors, predominantly of the granulosa cell type.1,2,4 In rats, disseminate abdominal tumors are most commonly mesotheliomas, although metastatic ovarian adenocarcinomas and biliary carcinomas can occur. 5 A syndrome of mesothelioma arising from the tunica vaginalis of the testis has been observed in Fischer 344 strain rats.5,8 Mesenteric liposarcoma is uncommon. 5 Nonneoplastic differentials for serosal nodules in a rat include abdominal fat necrosis, chronic peritonitis from intraperitoneal injections, polyarteritis nodosa, and infection with Cysticercus fasciolaris (metacestode of Taenia taeniaeformis).5,8
History and Gross Findings
A 1.4-year-old virgin female brown-hooded fancy rat presented for abdominal distention, jaundice, and dyspnea. This rat had no significant past medical history and was used as an educational animal at a local zoo. During physical examination, the rat was quiet, alert, and responsive. The skin and mucous membranes were diffusely yellow. The urine visible in the cage bedding was also bright yellow. At rest, the rat was noted to have an increased respiratory effort, which progressed to dyspnea when the rat was examined. By abdominal palpation, a firm, round, mass, estimated to be 2.5 × 2.5 × 2.5 cm, was located in the caudoventral abdomen. A diffusely nodular texture to the abdominal viscera and generalized distension of the abdomen were also noted. Other than the rat’s poor body condition score, estimated to be 1.5 on a scale of 1 to 5, there were no other significant findings. The likelihood of a neoplastic process combined with poor body condition in this geriatric rat prompted humane euthanasia.
At necropsy, the rat was in poor body condition with a distended abdomen. Mucus membranes and unpigmented skin were intensely yellow (jaundice). Approximately 31 ml of reddish-yellow–tinged, odorless, cloudy fluid was present in the abdominal cavity. The greater and lesser omentum were infiltrated and expanded by myriad 1- to 5-mm, discrete yellow firm masses, creating an irregular cobblestoned appearance. Similar small masses covered the surface of the liver and spleen. There was a large 4 × 3 × 2.5–cm firm, solid, ovoid, smooth, well-demarcated brown mass with smaller nodular yellow masses replacing the left ovary (Fig. 1). There were numerous small, 1- to 3-mm, distinct, smooth yellow masses associated with the right ovary.
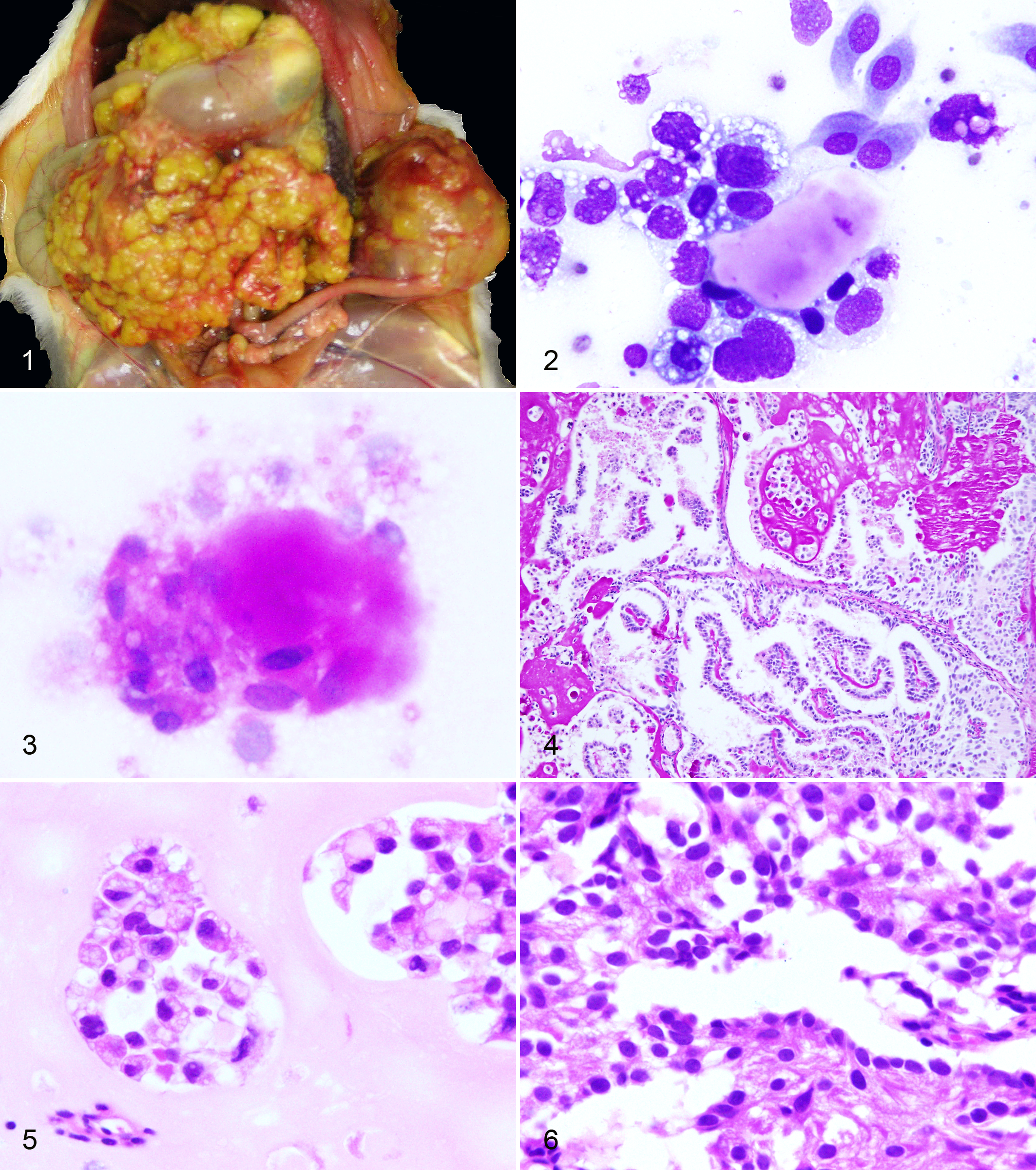
Differential Diagnoses
Grossly, the differentials for this process include mesothelioma, yolk sac carcinoma, ovarian adenocarcinoma, malignant granulosa cell tumor, and biliary carcinoma (cholangiocarcinoma).
Microscopic Findings
Cytology of Wright-Giemsa–stained abdominal fluid was highly cellular with a large population of pleomorphic cells with scant to moderate amounts of coarsely vacuolated cytoplasm, large slightly eccentric and irregular nuclei, and coarsely stippled chromatin (Fig. 2). These cells were found individually and in small clusters, and they frequently surrounded large pale eosinophilic to amphophilic and amorphous acellular globules. Periodic acid–Schiff (PAS) staining was strongly positive in both the globules and the cytoplasm of the pleomorphic cells (Fig. 3). There were also large numbers of plump squamoid and spindloid mesothelial cells in small and large clusters (reactive change).
Histologically the tumor was large, invasive, and biphasic (Figs. 4–6). The more abundant population consisted of irregular nests and cords of polyhedral cells with a moderate to abundant brightly eosinophilic cytoplasm containing clear vacuoles and large central irregular round to oval nuclei with coarsely stippled to vesicular chromatin (resembling parietal yolk sac endoderm). These cells were surrounded by large amounts of pale eosinophilic acellular amorphous to faintly fibrillar hyaline matrix material (resembling Reichert’s membrane; Figs. 4, 5). PAS staining was intensely positive in both the cytoplasm and the matrix (Fig. 4). The second population of cells formed papillary or solid structures, frequently surrounded by the first population. The cells were stellate with scant pale eosinophilic cytoplasm, large round central nuclei, and finely stippled chromatin (resembling visceral yolk sac endoderm; Fig. 6). Occasionally, there were small cysts lined by visceral cells and surrounded by parietal cells and hyaline matrix. Rare cysts were lined by cells with large apical mucin globules resembling goblet cells. In addition to both ovaries, tumor implants were present on the surface of the liver, kidneys, adrenals, stomach, and small and large intestines.
Diagnosis
Based on the characteristic histology and PAS staining, this was diagnosed as a primary ovarian yolk sac carcinoma, a malignant germ cell tumor. The cytologic characteristics of the neoplastic effusion are consistent with this diagnosis.
Discussion
Yolk sac carcinomas were first described as gonadal germ cell tumors in humans and were named for their resemblance to the human embryonic yolk sac. 3 A typical diagnostic feature is the presence of glomeruloid papillary visceral endodermal structures (Schiller-Duval bodies) resembling the endodermal sinuses of rats, for which they were also called endodermal sinus tumors.
Spontaneous yolk sac carcinoma is extremely rare in the rat and can be of uterine or ovarian (germ cell) origin. Yolk sac carcinoma of the testis has been reported in the rat as well. 6 Uterine yolk sac tumors, derived from products of gestation, can be experimentally induced in rats by 1 of 4 methods involving transplantation of extraembryonic tissues or fetectomy.3,12 Whereas germ cell tumors in the ovary are syngeneic with the dam (and always female), products of gestation may differ in 50% of alleles (and can be male). By immunohistochemistry, visceral cells are positive for a-fetoprotein (AFP), although AFP reaction has also been reported in parietal cells.3,9,12 Tumor cells are also positive for keratin, while PAS-positive material is positive for laminin.6,9 In humans, the most specific markers for yolk sac tumors are AFP and glypican 3. 7 Transcoelomic metastases with surface implantation and minimal invasion are typical, and ascitic fluid commonly contains parietal cells with hyaline material as well as visceral cells. 12
Differentials for the acellular globules surrounded by cells observed in the abdominal fluid cytology include collagen balls and psammoma bodies. 11 Collagen balls (serosal balls) are associated with benign mesothelial cells and composed of type I and III collagen as documented by immunostaining.10,13 Collagen balls are Masson’s trichrome positive and weakly PAS positive.10,11 Immunocytochemistry, if available, would be diagnostic. Collagen balls are seen at a low rate (4.5%–18%) in peritoneal washings, typically only occurring as a single small ball per slide, and are reported to be rare in spontaneous effusions.10,13 Psammoma bodies are mineralized concretions most commonly associated with the cytology of ovarian epithelial tumors. 11 Globules were not mineralized in this case.
Although rare, yolk sac carcinoma should be included in the differential diagnosis for ascites and carcinomatosis in rats. This case is unique in that diagnostic cytopathology findings have not been previously illustrated for this tumor.
Footnotes
Acknowledgements
Thanks to Weifang Lin and Ellen Mullady for histologic specimen preparation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
