Abstract
A myopathy affecting the pectoralis major muscle of the commercial broiler has emerged creating remarkable economic losses as well as a potential welfare problem of the birds. We here describe the macroscopic and histologic lesions of this myopathy within 10 pectoralis major muscles of 5- to 6-week-old broilers in Finland. Following macroscopic evaluation and palpation of the muscles, a tissue sample of each was fixed in formalin, processed for histology, and histologically evaluated. The muscles that were macroscopically hard, outbulging, pale, and often accompanied with white striping histologically exhibited moderate to severe polyphasic myodegeneration with regeneration as well as a variable amount of interstitial connective tissue accumulation or fibrosis. All affected cases also exhibited perivenular lymphocyte accumulation. The etiology of this myodegenerative lesion remains yet open. Polyphasic myodegeneration is associated with several previously known etiologies, but palpatory hardness focusing on the pectoralis major, together with perivenular lymphocytes, has not been described in relation to them. The results of this study provide the pathological basis for further studies concerning the etiology of the currently described myopathy.
Prevalence of a novel type of quality defect in the breast meat of broiler chicken has dramatically increased in Finland within the past 3 years, and to the authors’ knowledge, a similar trend also can be seen in several other countries. Macroscopic changes are restricted to the pectoralis major muscle, which shows pale expansive areas of substantial hardness accompanied with white striation; no other skeletal muscles seem to be affected. No particular antemortem signs have been related to this condition. Many of the affected breast fillets are, however, rejected from human consumption, which brings about significant economic losses within the industry and increases its interest to resolve the etiology of this condition and, subsequently, find means to prevent it.
Previously described myopathies that may affect the pectoralis major of the broiler include inherited muscular dystrophy, thermal stress, trauma, and exertional, nutritional, and toxic myopathies.4,9 Hard consistency of the muscle is typically not, however, a major finding within these etiologies. At present, the etiology and the factors causing the rapid increase of this unidentified pectoral muscle disorder in broilers remain obscure. Another recently described problem of unknown etiology—namely, white striping of the pectoralis as well as thigh muscles of the broiler—has also been histologically shown to represent myofiber degeneration. 7 To provide a basis for etiologic studies as well as to find the possible similarities with the white striping myopathy, our aim was to describe the macroscopic and histologic lesions of the novel myopathy, vernacularly referred to as “wooden breast.”
Cases and Methods
Ten affected pectoralis major muscles of commercial broiler chickens (case Nos. 1–10) were obtained from 2 slaughterhouses in Finland and evaluated for macroscopic as well as microscopic abnormalities. Two macroscopically normal pectoralis major muscles served as controls (case Nos. 11 and 12). The examined birds originated from 2 farms both providing 5 affected and a control case for the study. All cases were 5- to 6-week-old Ross 508 broilers of commercial flocks with typical in-farm mortality rates and no perceived clinical symptoms. The birds had not received any vaccinations.
At the slaughterhouse, the detection of wooden breast was conducted on cut-off, cooled (5°C) pectoralis muscles, and therefore, the corresponding carcass and/or the internal organs were not available for evaluation. The muscles were palpated; the shape, consistency, and color as well as presence and type of exudate and/or hemorrhages were evaluated macroscopically. Longitudinal and transverse sections for histology were taken from the macroscopically most severely affected area of each muscle, routinely immersed in formalin, processed in paraffin, cut at a 4-μm thickness, and stained with hematoxylin and eosin (HE). In addition, both controls and 2 affected cases (case Nos. 5 and 6) were stained with Masson trichrome stain. Immunohistochemical staining with the streptavidin-biotin method for anti-CD3 antibody (polyclonal rabbit anti–human A0452; DAKO, Glostrup, Denmark) and anti-CD79a antibody (Monoclonal mouse anti–human HM57 M7051; DAKO) were performed on 2 affected cases (case Nos. 1 and 6).
Results
In macroscopic comparison to the controls (Fig. 1), the consistencies of all affected pectoralis major muscles were remarkably hardened diffusely or on focally extensive areas. The hardened areas were notably pale and slightly outbulging (Fig. 2). The surface was often covered with a thin layer of clear or slightly turbid, moderately viscous material (6/10 cases), as well as scattered petechiae (5/10) or small hemorrhages (1/10). Two cases out of 10 also exhibited poorly demarcated spongy areas with distinctively separated muscle bundles, particularly within the deep layers of the muscle. Parallel to the myofibers, numerous white stripes 0.5 to 3 mm in width, extending to the cut surface, were detected (7/10). According to the experienced meat inspection staff, quality defects similar to wooden breast were not observed in other skeletal muscles of the carcasses within the slaughter batch.
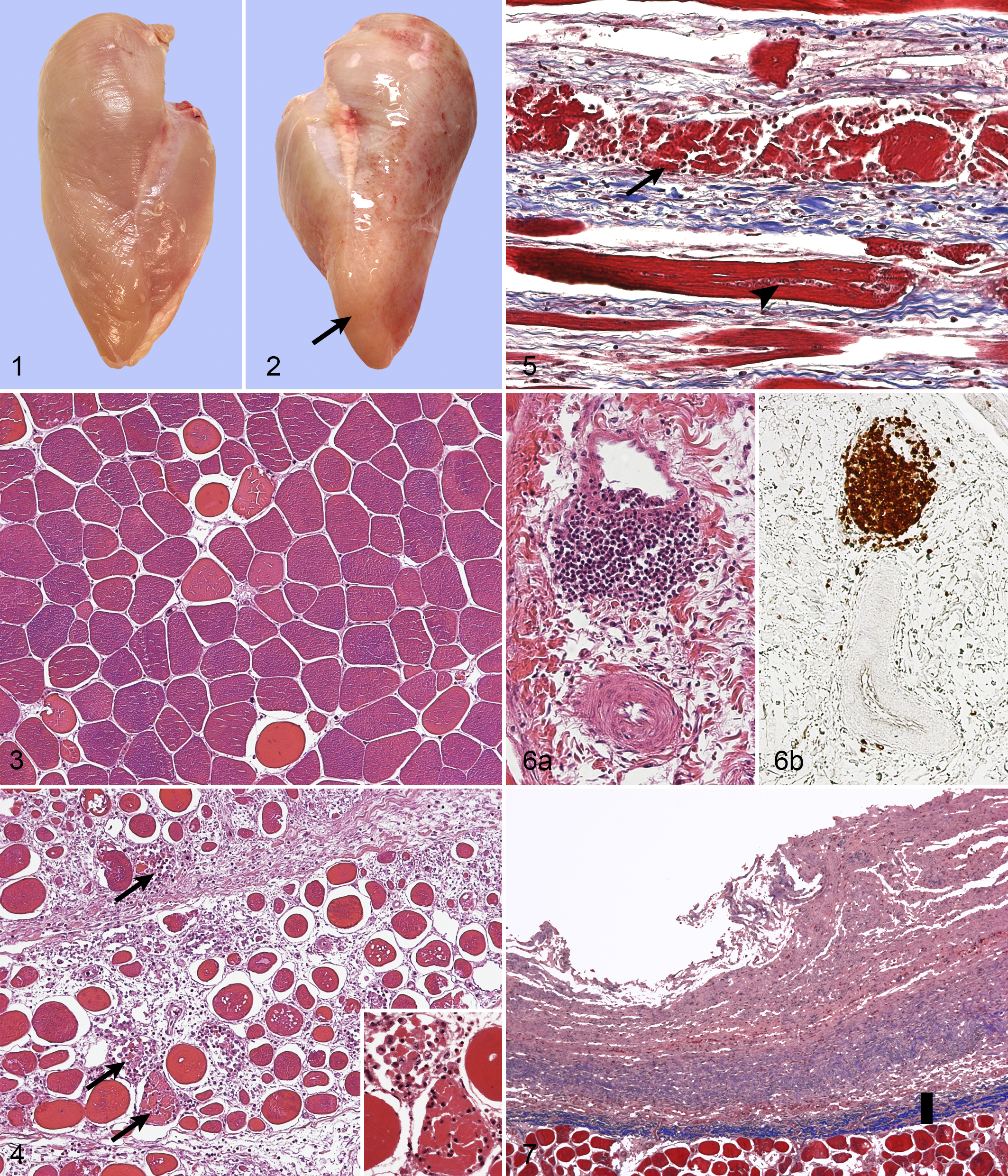
The histologic cross section revealed myofibers of variable diameter and devoid of their characteristic polygonality (Figs. 3, 4). Occasional splitted fibers were seen. Multifocal degeneration and necrosis were characterized by fragmented hypereosinophilic amorphous fibers, loss of striation, and infiltration of inflammatory cells, mainly macrophages and heterophils, within and around the degenerative fiber (Figs. 4, 5). Approximately 12 to 20 degenerative fibers were seen per 10 high-power fields (hpf). The degenerative lesions were often accompanied by thin fibers, slightly basophilic in HE, exhibiting faint cross-striation along with several (up to 15) central oval nuclei in a row, representing regeneration (Fig. 5). The affected areas exhibited diffuse thickening of the interstitium with variable amounts of loose connective tissue, granulation tissue, or collagen-rich connective tissue (fibrosis) separating the muscle fibers (Fig. 5). A moderate multifocal inflammatory cell infiltration, composed mainly of heterophils, macrophages, and occasional lymphocytes, was intermixed with the interstitium together with numerous spindle-shaped 10 × 20–μm cells with abundant lightly eosinophilic cytoplasm and oval nucleus, the latter being interpreted as fibroblasts. The myodegeneration with regeneration, as well as accumulation of interstitial connective tissue with inflammatory cells, was seen in all 10 cases of wooden breast; in the majority of the cases, changes were moderate or severe. Occasional degenerative fibers were seen also in the 2 control cases (0–2 in 10 hpf).
Perivascular inflammatory cell infiltration, usually around the veins only, was seen in all 10 cases of wooden breast (Fig. 6a). The infiltration was irregular, composed of lymphocytes, sometimes having disrupted the vascular wall. The perivascular cells exhibited strong, mainly cytoplasmic, positivity for immunohistochemical staining with anti-CD3 antibody (Fig. 6b). Only occasional cells were positive for anti-CD79a antibody, usually in the interstitium.
The epimysium covering the pectoralis major was, in several wooden breast cases, slightly or moderately thickened by loose connective tissue with abundant amorphous extracellular material, variable amounts of collagen fibers (Fig. 7), mixed inflammatory cell infiltrate, and small vascular profiles with plump endothelium (neovascularization). Moderate to severe edema and accumulation of loose material, most likely loose connective tissue, with moderate numbers of inflammatory cells were also exhibited by the perimysium surrounding the muscle bundles, especially within the areas of macroscopic sponginess.
Perivascular lymphocyte infiltrations or thickened epi-, peri-, or endomysium were not seen in the control cases. Evidence of infectious agents, such as bacteria or parasites, was not detected in either the affected cases or controls.
Discussion
This report describes a myodegenerative disorder of unknown etiology currently emerging in broilers. All 10 macroscopically pale and hardened pectoralis major muscles of broilers evaluated in this study exhibited similar histologic changes: moderate to severe multifocal regenerative myodegeneration and necrosis with a variable amount of interstitial connective tissue accumulation or fibrosis. Fibrosis could explain the characteristic hardness of the affected tissue unless some palpably hard cases were found not to express significant accumulation of collagen.
Previously known etiologies for such morphologic changes include nutritional causes, such as deficiency of selenium and/or vitamin E, excess of their antagonists, and ionophore toxicity; exertional myopathy; hypoxia; and genetic vulnerabilities.4,9 However, strikingly hard consistency, focused to the pectoralis major only and accompanied by fibrosis and perivenular lymphocyte infiltration, has not been described within these etiologies.
Heavy weight and/or rapid growth rate of broilers have been associated as causal factors with 2 recently described myopathies: myodegeneration of the anterior latissimus dorsi (ALD) muscle and macroscopic white striping of pectoralis and thigh muscles.5,10 The main histologic changes in these are very similar to our findings of myodegeneration and necrosis with fibrosis and regeneration within the wooden breast myopathy.7,10 Overlapping of histologic changes with the white striping myopathy is not surprising, since white striping is clearly a feature also in the wooden breast myopathy. Macroscopic similarities between our findings and the ALD myopathy are striking as well: paleness, increased hardness and thickness of the muscle, and exudate on the muscle surface. 10 The ALD myopathy affected most severely the heaviest male birds; occurrence of severe white striping was particularly high in the heavy and rapidly growing birds.5,10 In Finland, the weight of the broilers as well as breast fillets has increased in the past 3 years, together with the occurrence of the hard myopathy of the pectoralis major (Finnish poultry industry, personal communication, 2013). Therefore, it is possible that the fast growth rate, together with the increased breast meat yield, indeed plays a significant role also in the development of the wooden breast myopathy.
Of the other possible etiologic factors, nutritional deficiencies are well-recognized causes of myodegeneration. 9 Selenium and vitamin E deficiencies impair the antioxidant capacity against oxidative stress, especially in the pectoralis major. 1 The genetic selection toward larger breast muscles has, together with the increased total weight and the fast growth rate, probably further increased the susceptibility of the modern broiler to oxidative stress. The effect of increasing nutritional vitamin E supplementation has been shown to have little if any effect on reducing the myofiber damage or white striping in the pectoralis major of broilers.3,6 The role of selenium in the specific myodegeneration of the pectoralis major remains to be studied.
Perivenular infiltration with T lymphocytes was seen in all wooden breast cases of our study, yet it is not a consistent finding in previously described degenerative myopathies. It may, however, represent a component of the inflammatory process associated with the myodegeneration. Of infectious causes, viral infections are especially known to provoke cell-mediated immunity. In broilers, a 793/B variant of the infectious bronchitis virus (IBV) has been associated with pectoral myopathy and chronic phase CD4+ T-cell response, probably secondary to vascular changes due to immune-complex deposition. 2 However, half of the birds studied here (5 + 1) came from a flock recently tested serologically negative for IBV. The 2 control cases within the study were macroscopically normal, yet few occasional degenerative myofibers were seen histologically. That is considered a common finding in broiler breed chicken, particularly within their breast muscles. 8
To our knowledge, this is the first extensive description of a regenerative myodegeneration with fibrosis and substantial hardness occurring in the pectoralis major muscle of broiler chicken. At present, the etiology of this condition is unknown to the scientific community, but recent studies on other emerging myodegenerative problems that affect the muscles of the modern broiler point toward the rapid growth rate as well as the increased final weight of broilers.
Footnotes
Acknowledgements
We gratefully acknowledge HK Ruokatalo Oy and Atria Suomi Oy for providing financial support and research material for this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Sihvo and Immonen were partially funded by HK Ruokatalo Oy and Atria Suomi Oy.
