Abstract
Canine degenerative myelopathy (DM) is a progressive neurodegenerative disease that is frequently found in Pembroke Welsh Corgi (PWC) dogs. Canine DM is potentially a spontaneous animal model for human amyotrophic lateral sclerosis (ALS) because of similar lesions and the involvement of superoxide dismutase 1 (SOD1) mutation. However, the ventral horn lesion in DM has not been characterized in detail. Glutamate excitotoxicity due to deficiency of the glutamine-glutamate cycle has been implicated in neuron death in ALS. Thus, we examined 5 PWC dogs with an SOD1 mutation that were affected by DM, 5 non-DM PWC dogs, and 5 Beagle dogs without neurologic signs to assess the neuronal changes and the expression levels of 2 glial excitatory amino acid transporters (glutamate transporter 1 [GLT-1] and glutamate/aspartate transporter [GLAST]). The number of neurons in the spinal ventral horns of the DM dogs was significantly decreased, whereas no change was found in the cell size. Chromatolysis, lipofuscin-laden neurons, and marked synapse loss were also observed. GLT-1 expression was strikingly decreased in DM dogs, whereas GLAST expression showed no significant change. The results indicate that excitotoxicity related to the reduced expression of GLT-1, but not GLAST, may be involved in neuron loss in DM, as in human ALS, whereas intraneuronal events may differ between the 2 diseases.
Keywords
Canine degenerative myelopathy (DM) is a progressive neurodegenerative disease that causes advancing ataxia and muscle atrophy. 1 Affected dogs initially exhibit hindlimb weakness and paralysis, followed by forelimb ataxia and culminating in paralysis, dysuria, and dyschezia. 7,19 Recently, a superoxide dismutase 1 (SOD1) mutation including c.118G>A was shown to be associated with canine DM in several breeds, including the Pembroke Welsh Corgi (PWC), Boxer, and German Shepherd, 2 followed by a report of c.52A>T mutation in a Bernese Mountain Dog. 40 PWC dogs have been reported to be a favorite breed for studying DM in recent years. 7,19,23 In fact, all cases of canine DM diagnosed at the University of Tokyo between 2009 and 2011 occurred in this breed and were associated with an SOD1 mutation. Histiocytic sarcoma 14 and inflammatory myopathy 38 also commonly occur in PWC in Japan and produce similar clinical signs, which often complicates diagnosis until histopathology is performed.
More than 100 SOD1 gene mutations are involved in human familial amyotrophic lateral sclerosis (ALS), and the pathologic lesions of ALS are similar to those of canine DM, making canine DM a potentially useful animal model of ALS. We previously reported that the overproduction of nitric oxide through inducible nitric oxide synthase (iNOS) and/or the abnormal accumulation of denatured proteins might be involved in the pathogenesis of DM in PWC dogs, 23 as in human ALS. Since lesions of the spinal gray matter are not necessarily required for the diagnosis of canine DM, they have not been examined in sufficient detail, even though loss of the ventral motor neurons is the major event of ALS pathology.
Glutamate is the predominant excitatory amino acid in the central nervous system (CNS). Excitatory amino acid transporters (EAATs) on the cell membrane, which reuptake extracellular glutamate into the cell, modulate homeostasis of synaptic transmission. Of these transporters, astrocytic glutamate transporter 1 (GLT-1; EAAT2) and glutamate/aspartate transporter (GLAST; EAAT1) are responsible for most of the glutamate transport. Glutamine synthetase (GS), which catalyzes the production of glutamine from glutamate and ammonia, is found in astrocytes and oligodendrocytes 8 and may be important for maintenance of the glutamate concentration.
Excessive glutamate exposure is toxic to neurons through massive Ca2+ entry into the cell. It is known that motor neurons are vulnerable to excitotoxicity, 5,39 and it is thought that excitotoxicity and oxidative stress play a major role in motor neuron loss in patients with ALS.
It is also reported that the downregulation of GLT-1 is involved in the disease progression in patients with ALS and in some types of SOD1-transgenic ALS model mice. 4,12,24,28,29 To our knowledge, no studies have examined the involvement of excitotoxicity in canine DM.
The purpose of the present study is to elucidate the severity and morphologic features of neuronal loss and the involvement of GLT-1, GLAST, and GS in canine DM.
Materials and Methods
Animals and Tissue Samples
Spinal cords of 5 PWC dogs pathologically diagnosed with DM (age, 9–14 years; mean, 12.3 years), as well as those of 5 PWC dogs without significant lesion (age, 6–11 years; mean, 8.9 years) and 6 Beagle dogs as control dogs without neurologic signs (age, 1–14 years; mean age unknown), were used. Information on the dogs is summarized in Table 1. Dog Nos. 12 and 13 were already described as DM cases in our previous report. 6 Representative specimens from the cervical (C5–C7), thoracic (T5), and lumbar (L3–L5) cords were used. Only T13 to L3 were available in dog No. 15, T5 to T12 in dog No. 11, and C1 to T11 in dog No. 5. Examined regions are indicated in Table 1.
Dogs Examined in the Present Study.
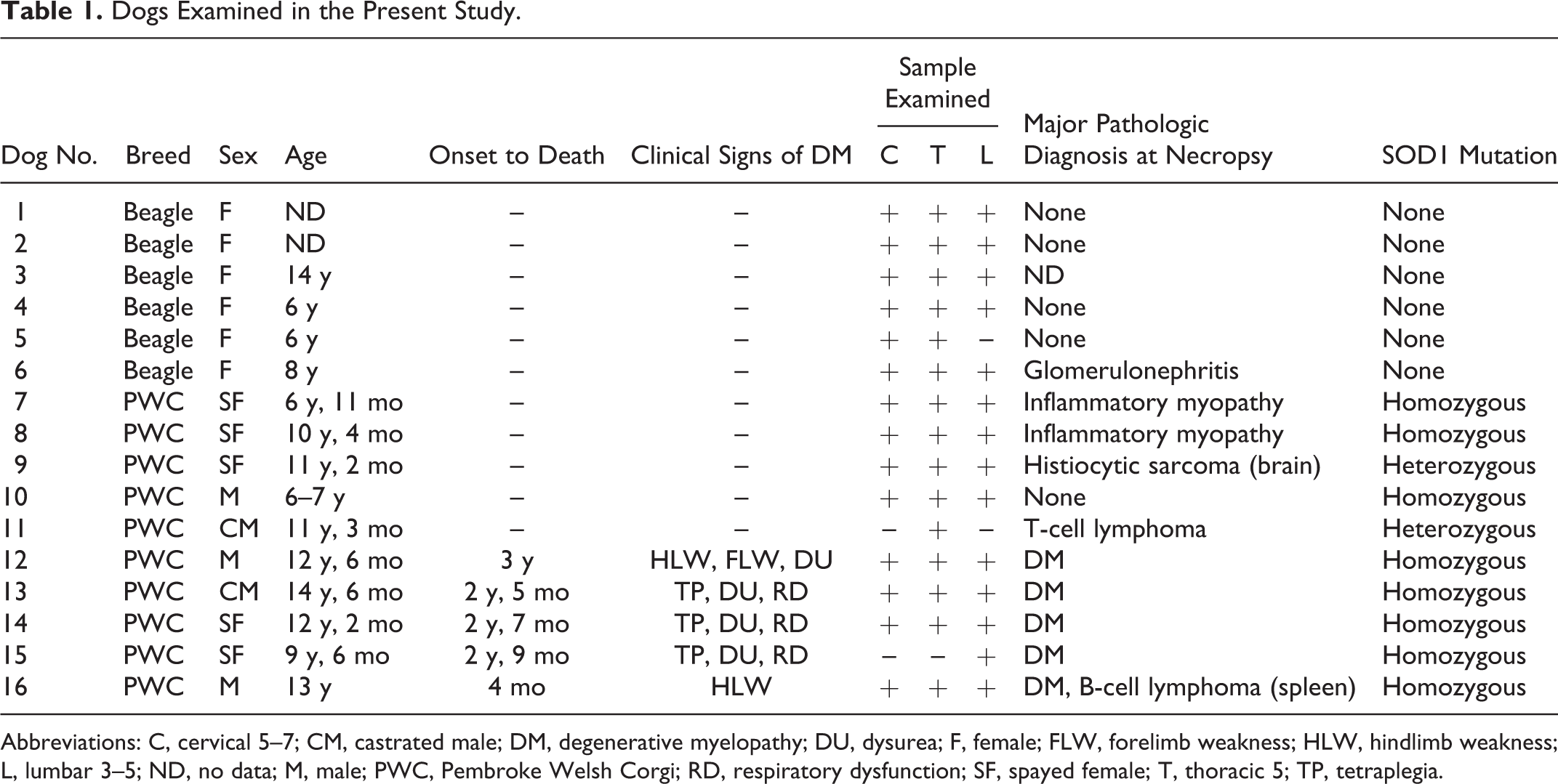
Abbreviations: C, cervical 5–7; CM, castrated male; DM, degenerative myelopathy; DU, dysurea; F, female; FLW, forelimb weakness; HLW, hindlimb weakness; L, lumbar 3–5; ND, no data; M, male; PWC, Pembroke Welsh Corgi; RD, respiratory dysfunction; SF, spayed female; T, thoracic 5; TP, tetraplegia.
Genotyping for SOD1 c.118G>A Mutation
Genomic DNA was extracted from paraffin-embedded tissue of each dog, using a commercial kit (DNA Isolator PS Kit; Wako Pure Chemical Industries, Osaka, Japan) according to the manufacturer’s instructions. The genotypes were determined using a real-time polymerase chain reaction (PCR) method reported previously. 6
Histopathology, Immunohistochemistry, and Immunofluorescence
Tissue samples from spinal cords were fixed in 10% formalin, processed routinely, and embedded in paraffin wax. Then, 4-μm-thick paraffin sections were subjected to hematoxylin and eosin (HE), Luxol fast blue (LFB)–HE, and Kluver-Barrera stains and used for immunohistochemical analysis.
Immunohistochemical analysis was performed using the EnVision polymer method (Dako, Glostrup, Denmark). Table 2 lists the primary antibodies used. Deparaffinized sections were first autoclaved at 120°C for 10 minutes in 10 mM citrate buffer (pH 6.0 or 9.0) for antigen retrieval. Then, the tissue sections were treated with 3% hydrogen peroxide (H2O2)–methanol at room temperature for 5 minutes and incubated in 8% skimmed milk–Tris-buffered saline with 0.2% Tween 20 (TBST) at 37°C for 1 hour to avoid nonspecific reactions. The sections were then incubated at 4°C overnight with one of the primary antibodies (Table 2). After being washed 3 times in TBST, the sections were incubated with Envision horseradish peroxidase (HRP)–labeled polymer anti–mouse or anti–rabbit immunoglobulin G (IgG) (DAKO, Glostrup, Denmark) at 37°C for 50 minutes. Then, the sections were washed with TBS and the reaction visualized with 0.05% 3,3′-diaminobenzidine and 0.03% H2O2 in TBS. Counterstaining was performed with Mayer’s hematoxylin.
Primary Antibodies Used.
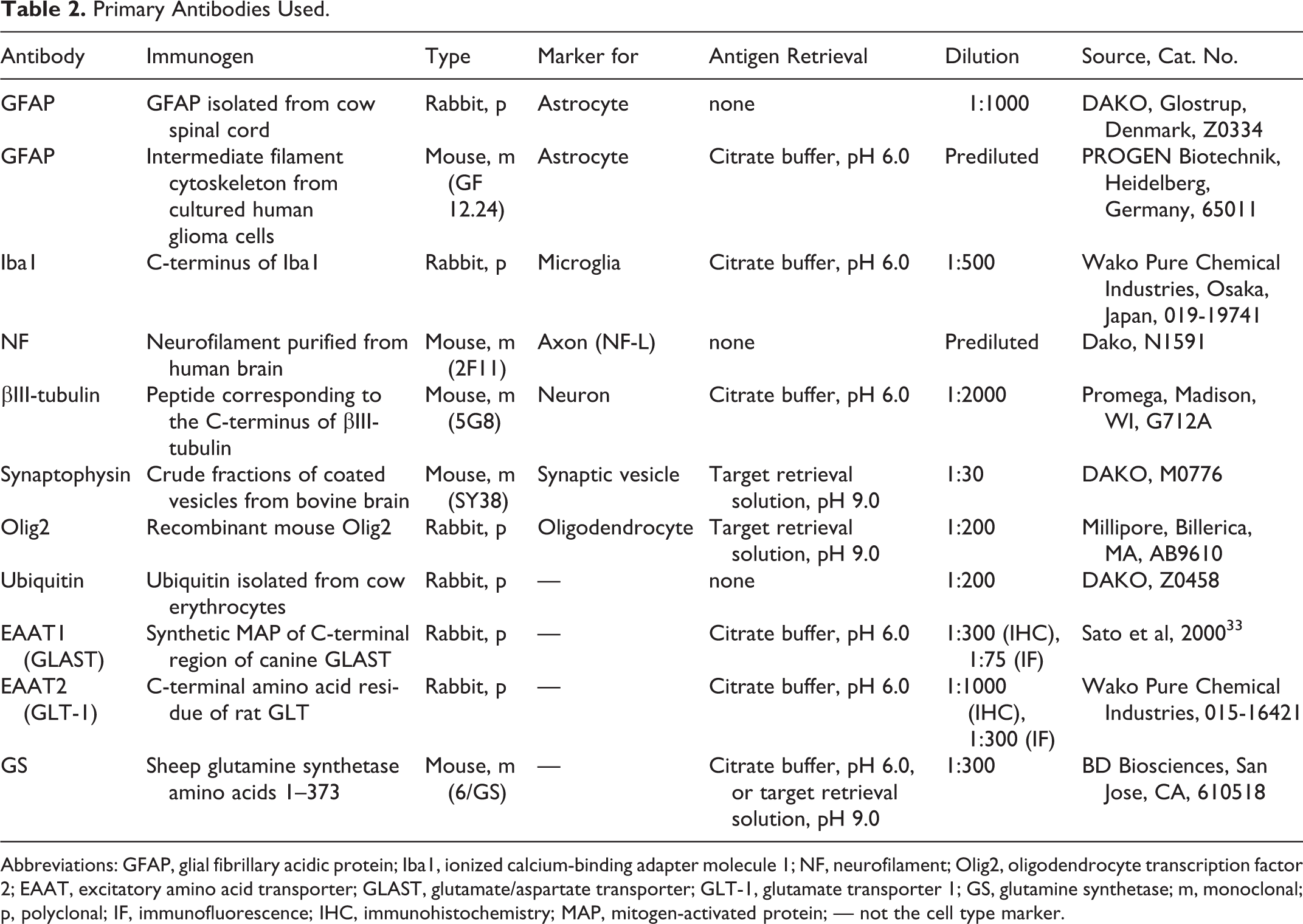
Abbreviations: GFAP, glial fibrillary acidic protein; Iba1, ionized calcium-binding adapter molecule 1; NF, neurofilament; Olig2, oligodendrocyte transcription factor 2; EAAT, excitatory amino acid transporter; GLAST, glutamate/aspartate transporter; GLT-1, glutamate transporter 1; GS, glutamine synthetase; m, monoclonal; p, polyclonal; IF, immunofluorescence; IHC, immunohistochemistry; MAP, mitogen-activated protein; — not the cell type marker.
Ventral motor neurons were defined as βIII-tubulin–positive structures with morphologic features of a neuron, including a minor axis of more than 15 μm in the ventral horn, not including intermediolateral nuclei. Counts of neurons were performed using representative sections.
For double immunofluorescence staining, sections were incubated with a primary antibody and then with another primary antibody (Table 2). The sections were incubated with a cocktail of fluorescein isothiocyanate (FITC)–conjugated anti–mouse IgG (1:100; Invitrogen Life Technologies, Paisley, UK) and Alexa 594–conjugated anti–rabbit IgG (1:100; Vector Laboratories, Burlingame, CA) at room temperature for 1 hour. After being mounted with mounting medium (Vector Laboratories), the specimens were observed and photographed using a DMI3000B microscope (Leica Microsystems, Wetzlar, Germany).
Data Analysis
Macroscopic images of the transverse section of spinal cords were captured using the Nikon COOLSCAN IV ED (Nikon Imaging, Tokyo, Japan). Microscopic images were captured using a Nikon DXM1200F digital camera and ACT-1 software (Nikon Instruments, Tokyo, Japan). Image analysis was conducted using the National Institutes of Health (NIH) Image J software (NIH, Bethesda, MD). Briefly, the color channel was split to RGB 8-bit images, and then the positive area was measured as a limited area with a threshold set 0 to 200. Immunopositive areas for synaptophysin or GLT-1 were measured on a macroscopic image and expressed as the ratio of positive area/total gray matter area. For GLAST, the ratio of positive areas was measured in 6 randomly selected squares (3 in each side) from the ventral horn, dorsal horn, and lateral funiculus on a microscopic image. For GS, the number of positive cells in the random 6 squares from the ventral horn was counted. Then, the average of the 6 areas was calculated.
Data are expressed as the mean ± SEM. For statistical analyses of the data, 1-way analysis of variance (ANOVA) followed by Scheffé’s multiple comparison procedure was used. Differences between groups were considered statistically significant when P < .05.
Results
Among the dogs assessed in the present study, all DM-affected dogs and 3 of 5 non-DM PWC dogs had a c.118G>A homozygous mutation in the SOD1 gene. The other PWC dogs (dog Nos. 9 and 11) had a heterozygous mutation. Beagle dogs did not have the mutation (Table 1).
Histopathologic features of dog Nos. 12 and 13 were described in detail in our previous report. 23 Axonal degeneration and/or loss, as well as demyelination of the white matter, were commonly observed in the spinal cord of dog Nos. 14 to 16 (Figs. 9 and 10), as well as dog Nos. 12 and 13. The lesions were most severe in the thoracic spinal cord of dog Nos. 12, 13, 14, and 16, especially in the dorsolateral funiculi. The severity of these lesions differed among the dogs; the severest were in dog Nos. 14 and 15, whereas it was mild in dog No. 16. The cervical cord of dog No. 16 was almost intact. Proliferation of glial fibrillary acidic protein (GFAP)–positive astrocytes was observed to be correlated with the severity of the axonal loss and demyelination (Fig. 11). Proliferation of calcium-binding adapter molecule 1 (Iba1)–positive microglia throughout the white and gray matter was observed in DM dogs, especially in dog Nos. 12 and 15 (Fig. 12). Myelin loss and axonal degeneration were significant in the spinal ventral roots of dog Nos. 14 and 15, whereas these changes were relatively mild in the other DM dogs.These lesions were not observed in other two groups of dogs (Figs. 1-8).
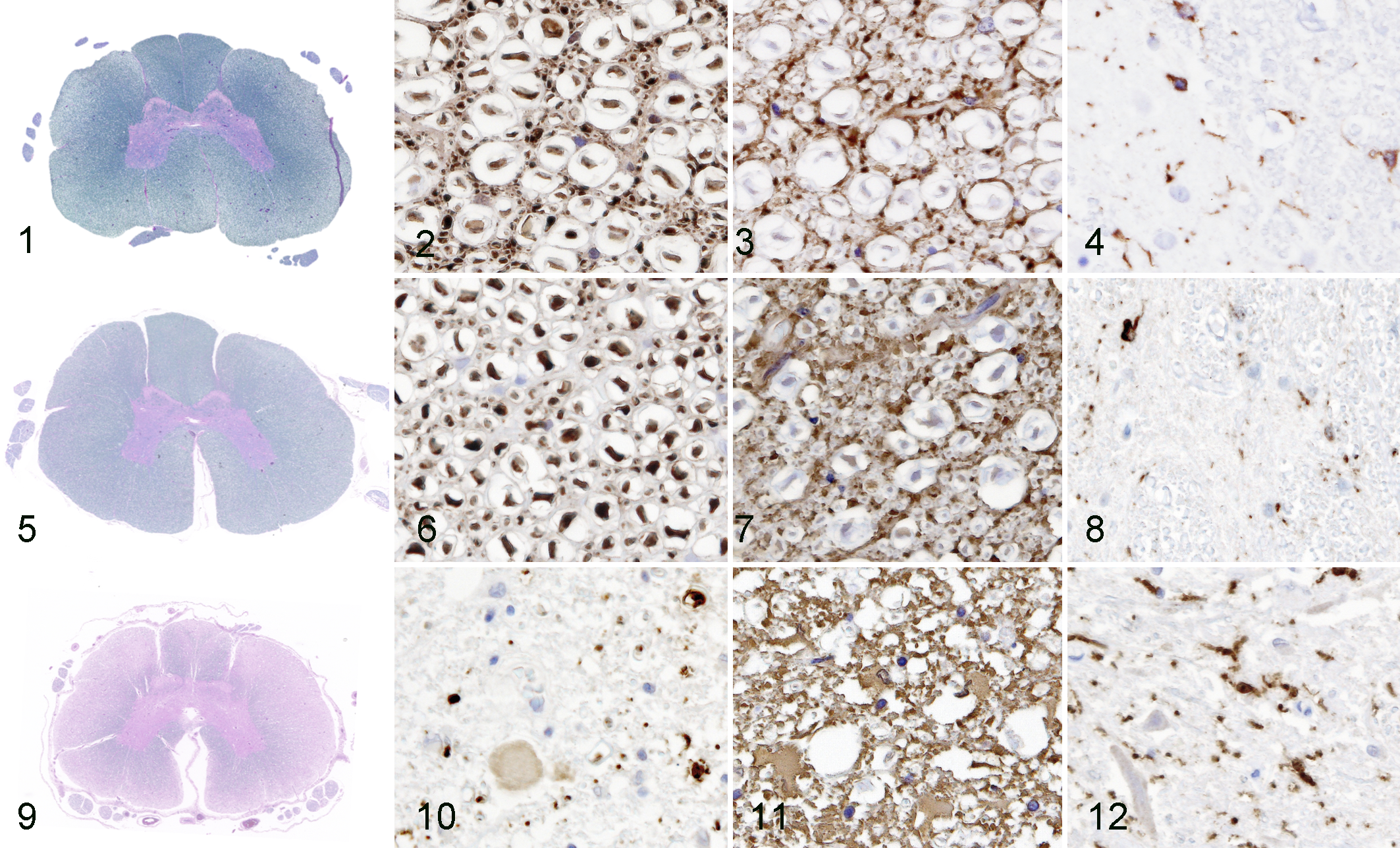
A large amount of lipofuscin was observed frequently in the cytoplasm of neurons in all DM dogs and, to some degree, in aged non-DM PWC dog Nos. 8, 9, and 10 and Beagle dog No. 3. Chromatolysis of large motor neurons was observed in the ventral horn of the cervical, thoracic, and lumbar cords of DM-affected dogs, especially in dogs Nos. 12 and 14. No cytoplasmic inclusion bodies or apoptotic bodies were detected on HE sections in any dog in the present study. Ubiquitin-positive structures were not observed in the surviving neurons, but ubiquitin positivity was identified in degenerated neurons and/or in the neuropil.
There were fewer βIII-tubulin–positive neurons in the ventral horn in DM-affected dogs than in dogs in the other 2 groups (Figs. 13–16, Table 3). The mean numbers of such neurons in the cervical, thoracic, and lumbar cords from DM, non-DM PWC, and Beagle dogs are shown in Table 3. The decrease was statistically significant in thoracic and lumbar cords compared with both non-DM PWC and Beagle dogs. Both large neurons and small neurons survived in the DM sections (Fig. 16). No significant difference in the average size of neuronal soma between DM, non-DM PWC, and Beagle dogs was detected (Fig. 16, Table 3).
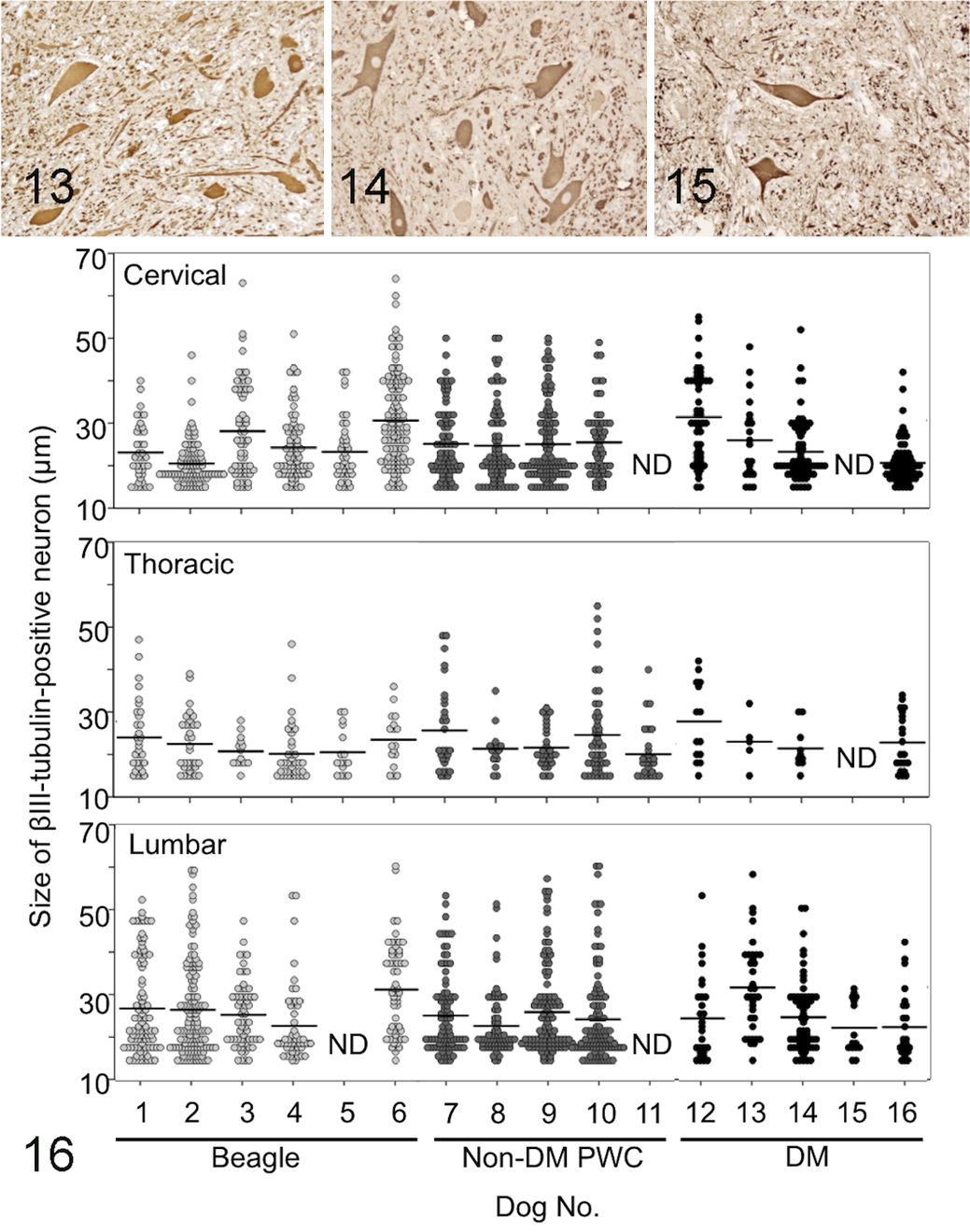
Mean Size and Number ± SEM of βIII-tubulin–Positive Neurons of Beagle, Non–Degenerative Myelopathy (DM) Pembroke Welsh Corgi (PWC), and DM Dogs.
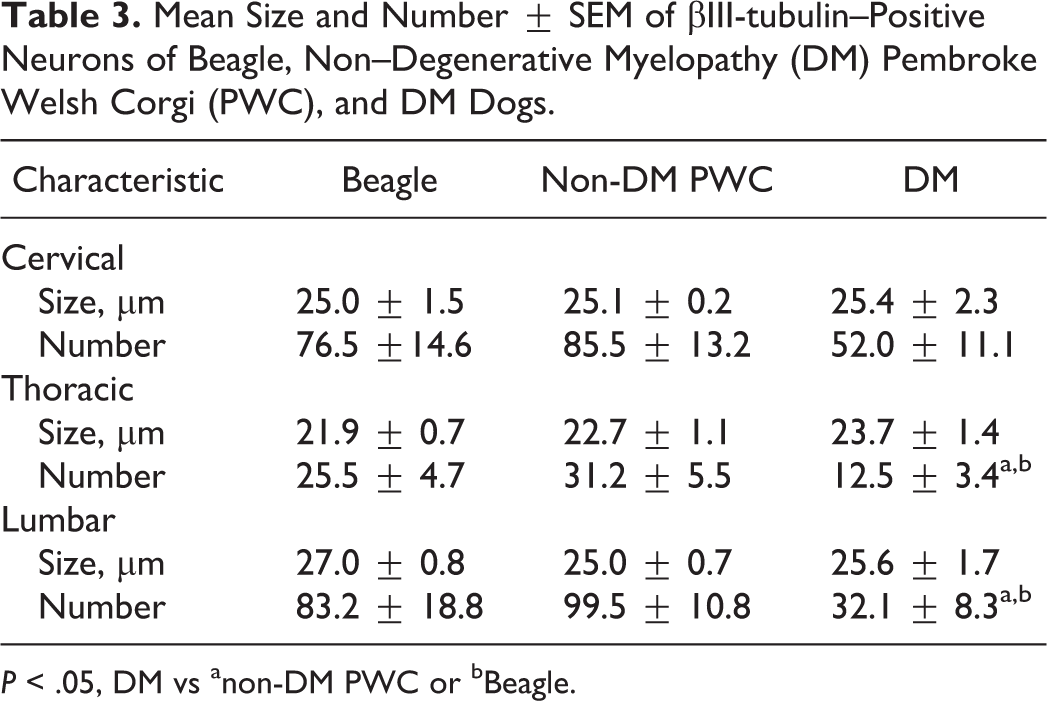
P < .05, DM vs anon-DM PWC or bBeagle.
The ratios of synaptophysin-positive area in the gray matter of DM-affected dogs were significantly decreased to approximately half of those in non-DM PWC dogs or Beagle dogs in the cervical, thoracic, and lumbar cords (Figs. 17–20). The reduction was most prominent in the ventral horn of all regions. Synaptophysin immunoreactivity could not be detected in dog Nos. 1, 2, 7, and 12, presumably because the sample tissues had not been freshly fixed.
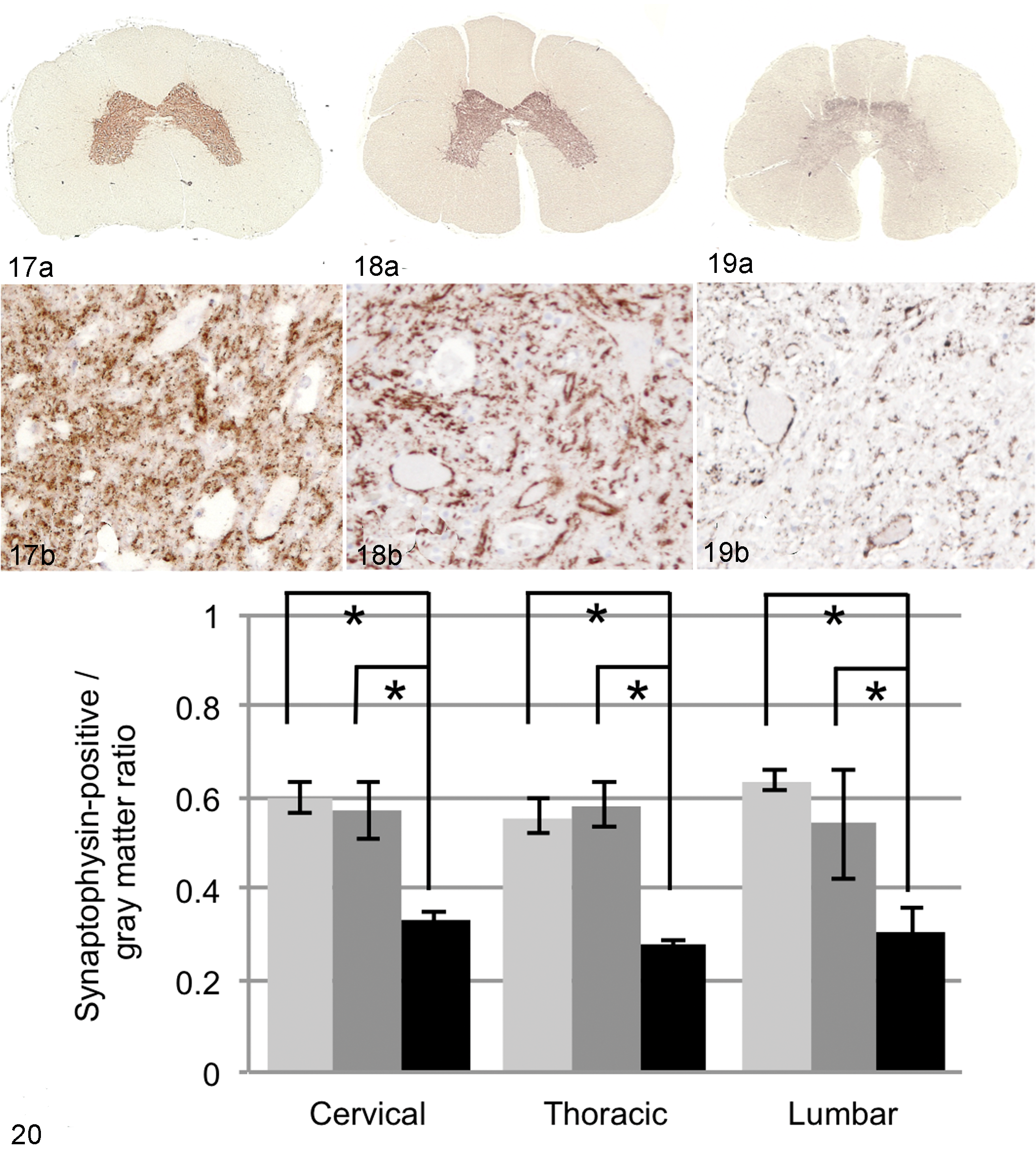
Spinal cord, T5; Beagle dog No. 2. (a) Normal synaptophysin immunoreactivity. (b) Higher magnification of (a), ventral horn. 3,3′-Diaminobenzidine (DAB) and hematoxylin.
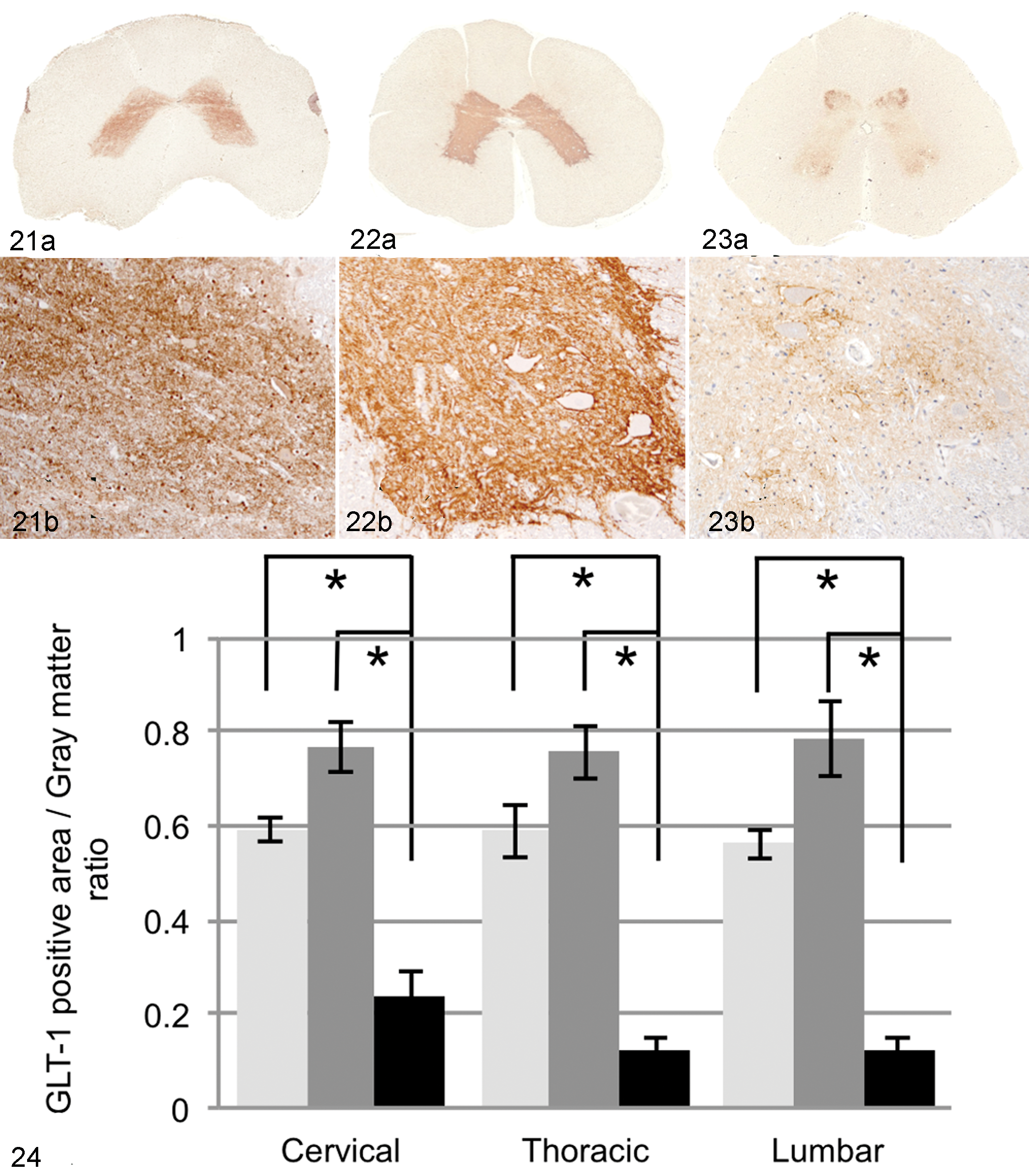
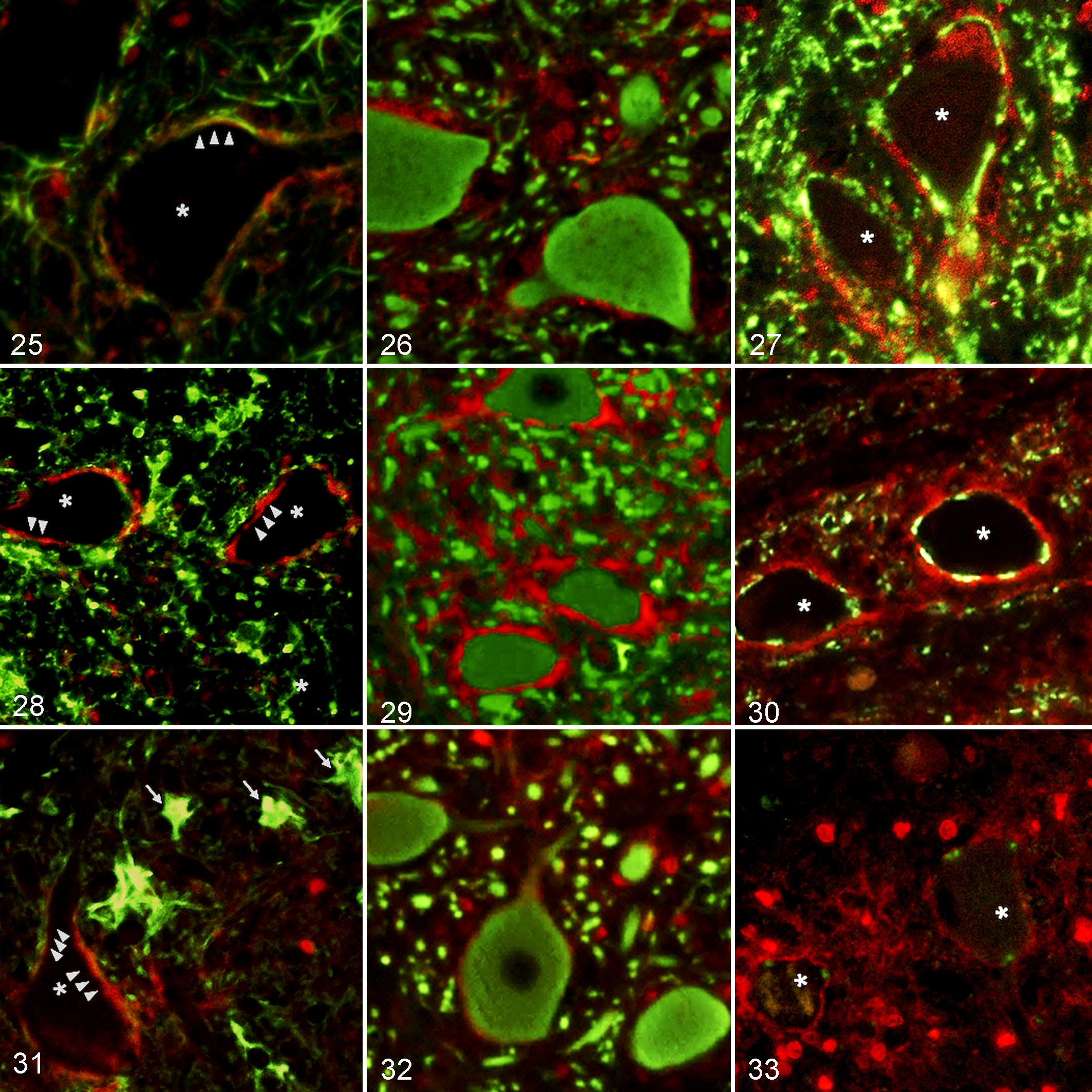
GLT-1 immunoreactivity was distributed throughout the gray matter of the spinal cord of non-DM PWC and Beagle dogs, with the highest expression in the substantia gelatinosa of the dorsal horn and around large ventral motor neurons (Figs. 21–24). Compared with both non-DM PWC and Beagle dogs, the GLT-1 expression in DM dogs was significantly decreased (Fig. 24) in all regions examined, especially in the thoracic and lumbar cords. Results of double immunofluorescence staining indicated that the GLT-1–positive area was mostly localized between the neuron and GFAP-positive process (Figs. 25, 28, 31), whereas no GLT-1–positive staining was observed on the surface of GFAP-positive cells with a large cytoplasm in the gray matter of the DM dogs (Fig. 25). The GLT-1–positive staining was localized especially around βIII-tubulin–positive large neurons (Figs. 26, 29, 32) and synaptophysin-positive synaptic vesicles (Figs. 27, 30, 33), whereas the expression levels were decreased in DM dogs (Figs. 32, 33).
A nonsignificant change of the GLAST expression level in the ventral and dorsal horn was exhibited in DM dogs compared with that in non-DM PWC or Beagle dogs (Figs. 34–37). By double staining, GLAST expression was shown to be co-localized with GFAP in the gray and white matter of DM and the 2 other groups of dogs (data not shown). Astrocytes included in the gliosis lesions in the degenerated white matter or reactive astrocytes in the gray matter showed no significant GLAST immunoreactivity.
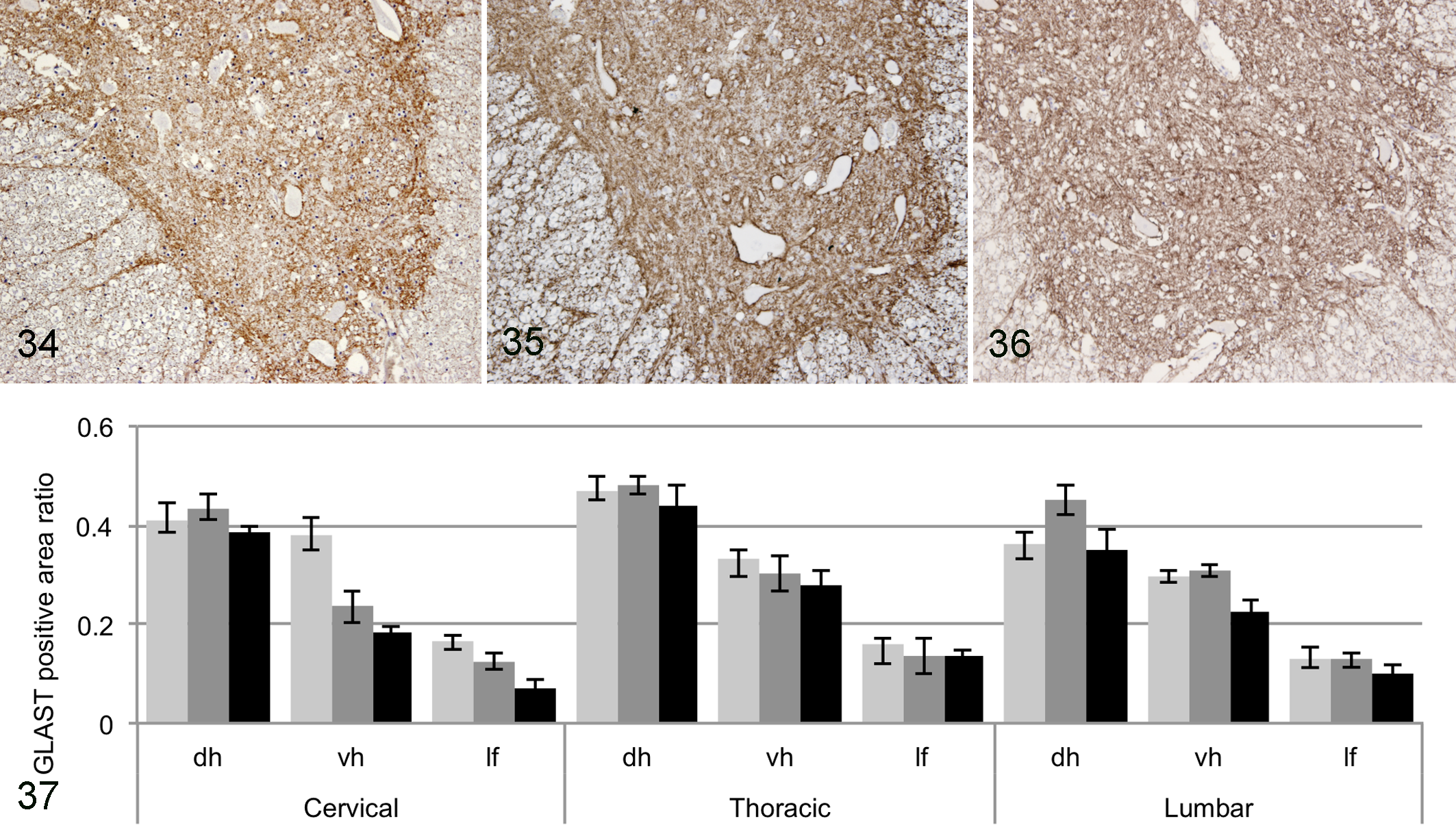
Spinal cord, ventral horn, T5; Beagle dog No. 4. Normal glutamate/aspartate transporter (GLAST) immunoreactivity. 3,3′-Diaminobenzidine (DAB) and hematoxylin.
GS-positive cells were observed throughout the gray matter, and a few were found in the white matter, mostly in the boundary area with the gray matter (Figs. 38–41). By double immunofluorescence, GS immunoreactivity was detected in mostly GFAP-negative and olig2-positive cells with scant cytoplasm (oligodendrocytes) and partly GFAP-positive astrocytes (data not shown). Compared with non-DM PWC or beagle dogs, the average number of GS-positive cells was slightly decreased in DM dogs, but this difference was not statistically significant (Fig. 41).
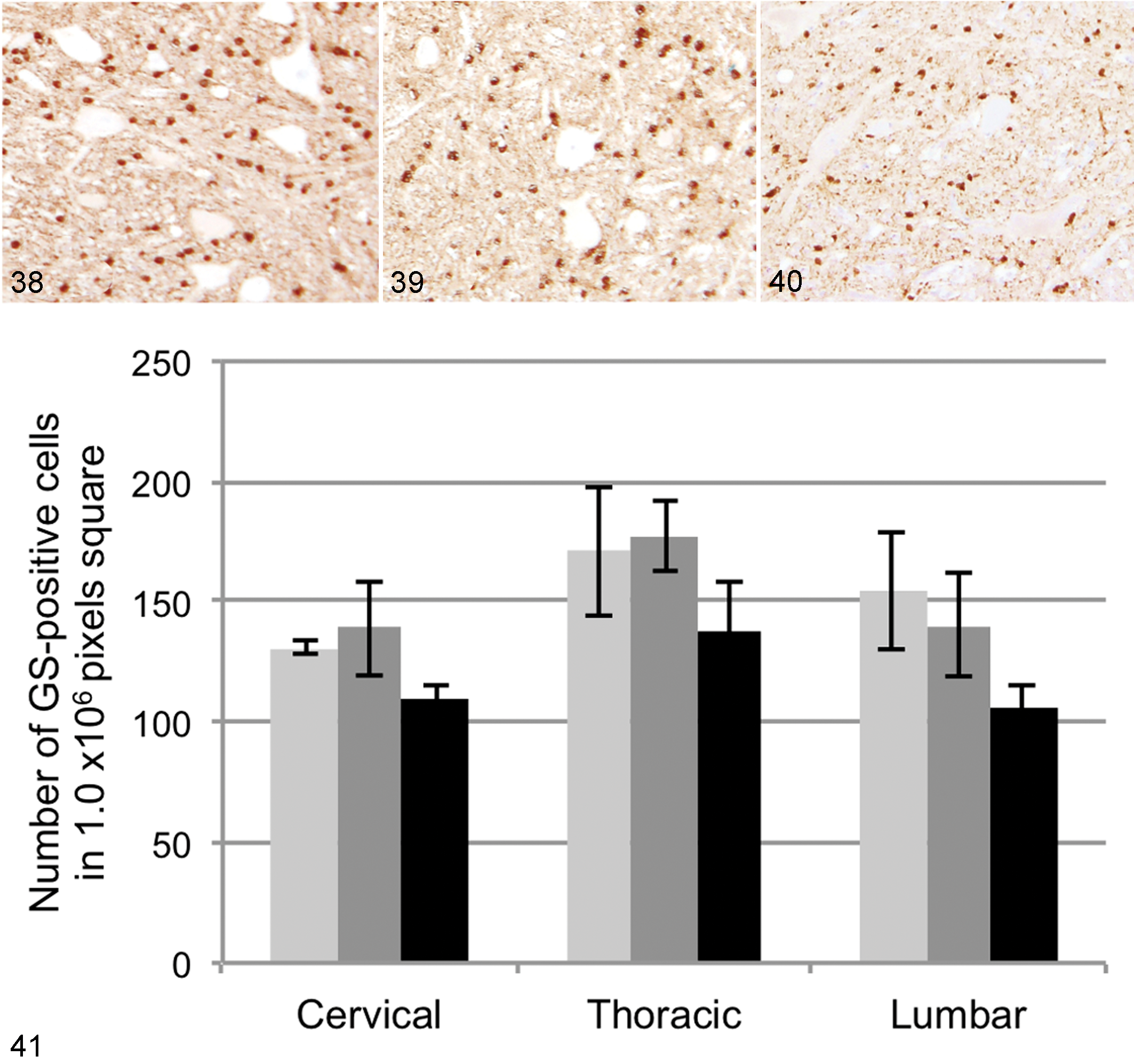
Spinal cord, ventral horn, T5; Beagle dog No. 4. Normal glutamine synthetase (GS) immunoreactivity. 3,3′-Diaminobenzidine (DAB) and hematoxylin.
Discussion
Considering the fact that a high percentage of PWC dogs examined in the present study had an SOD1 mutation, an inherited predisposition to DM might be frequently present in the breed in Japan, irrespective of whether loss or gain of function of the molecule is directly involved in the etiology. Histopathologic lesions observed in the spinal cords of dog Nos. 12 to 16 affected by DM are consistent with those outlined in previous reports describing canine DM, 7,19,21 and the distribution of the spinal lesions was almost identical to that of previous reports on DM in PWC. 7,19 The severity of the pathologic lesions was closely associated with the severity of the clinical signs (Table 1). Whereas no sufficient examination was done on the tissues other than CNS in this study, the ventral root lesions are consistent with the recent reports on peripheral lesions of DM. 34
The pathologic changes in the ventral neurons in DM dogs included chromatolysis, particularly in dog Nos. 12 and 14, and deposition of lipofuscin, which was observed in all DM dogs. Loss of the ventral neurons was more severe in dog Nos. 13 and 15. Chromatolytic or lipofuscin-bearing neurons have been generally observed in motor neurons of patients with ALS. 18,30 Lipofuscin deposition was also observed in aged non-DM PWC dogs, presumably because of normal aging. Because of the difference of ages of the Beagle, non-DM PWC, and DM dogs in this study, the results do not necessarily indicate that the lipofuscin deposition is a consequence of the DM itself.
The loss of synaptophysin immunoreactivity in the present cases of DM indicates that a synaptic loss occurs in DM during the process of neuronal degeneration, which is similar to what occurs in ALS. 15,16,31,32,44 Similar to our previous report 23 and unlike in ALS, inclusion bodies mimicking Bunina bodies, Lewy body–like inclusions, and/or aggregated ubiquitin-positive skein-like inclusions/structures were absent in the surviving neurons of DM-affected dogs. 11 Formation of such aggregations or inclusion bodies indicates the dysfunction of 2 major intracellular degradative pathways (ubiquitin-proteasome system and autophagy system); abnormalities of these systems are thought to be involved in neurodegeneration and ALS. 17,41 Considering the absence of these features, the mechanisms of neuronal death in canine DM are not likely to be the same as in ALS.
The result of size measurement indicates ventral neurons larger than 30 or even 50 μm were still surviving in DM cases. In addition, there was no difference in the average size of neurons between DM dogs and non-DM PWC or Beagle dogs. These facts indicate nonselective (not only large neuron) degeneration of the ventral horn. These findings also suggest that the ventral neuronal loss in the DM-affected spinal cord may not occur through apoptosis, since apoptotic cells usually undergo shrinkage. Moreover, no apoptotic body was observed in the DM dogs in the present study. Although the role of apoptosis in neuronal death in ALS is still controversial, 20,36,42,43 the results of this study suggest that apoptosis is unlikely to be the mechanism of neuronal death in canine DM.
The distribution pattern of GLT-1 in the spinal cords of the present non-DM PWC or Beagle dogs is consistent with that previously outlined in rat, mouse, and human spinal cords. 10,22,26 GLT-1 downregulation is reported in patients with ALS and ALS model mice after disease onset. 4,12,29 The decreased GLT-1 expression level in DM dogs suggests that the deficiency of glial reuptake of excitatory amino acids may play a similar role in DM pathogenesis as it does in patients with ALS and ALS mouse models. 27 Absence of a significant difference between non-DM PWC dogs with the SOD1 mutation and Beagle dogs without the mutation also indicates that the decrease of GLT-1 expression is a secondary event to the disease onset and may be involved in disease progression rather than disease onset, as both a consequence and an exacerbating factor of the disease. In addition, the fact that there was no significant change of the GLAST expression level in any region of the DM dogs in the present study may also support the hypothesis that GLT-1 is responsible for excitotoxicity of the spinal motor neurons in canine DM. There are reports 13,25 indicating that astrocytic nitric oxide production through iNOS is associated with glutamate uptake activity of GLT-1 or glutamate-induced excitotoxicity. Considering our previous finding that reactive astrocytes express iNOS in the gray matter of DM dogs, 23 it is likely that an interaction between NO production and glutamate uptake occurs in DM.
GS localization in astrocytes and oligodendrocytes in canine spinal cords examined in this study coincides with previous reports on cats and rats. 8,37 Although the number of GS-positive cells was slightly decreased in DM dogs compared with those in the 2 other groups, no statistically significant difference among the 3 groups was detected. Alterations in GS levels have been documented in other neurodegenerative disorders, 35 but reports of GS expression in patients with ALS are very limited. 3 In a type of SOD1 transgenic mice, GS expression and/or activity appears to be unchanged in the spinal cord. 9 The role of GS expression in both ALS and canine DM remains undefined, and further studies using a larger number of samples are needed to clarify this issue.
In conclusion, excitotoxicity via the deficiency of the glutamine/glutamate cycle due to reduced expression of astrocytic GLT-1, but not GLAST, may contribute to the striking neuron loss in the ventral horn of DM dogs during disease progression, as in ALS. On the other hand, intraneuronal events such as degradative dysfunction during disease progression seem to differ between DM and ALS.
Footnotes
Acknowledgements
We are grateful to the owners of the examined dogs who participated in this research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
