Abstract
Canine degenerative myelopathy (DM) is a progressive neurodegenerative disease frequently found in Pembroke Welsh Corgi (PWC) dogs, and it has clinical and pathologic similarities to human amyotrophic lateral sclerosis. Autophagy is a major intracellular protein degradation system. Abnormalities of autophagy—resulting in cell death through mechanisms called type II programmed cell death—have recently been reported to occur in various neurodegenerative diseases, including amyotrophic lateral sclerosis. Thus, the distribution and expression levels of proteins involved in autophagy were examined in the spinal cords of 8 PWC dogs suffering from DM with superoxide dismutase mutation, 5 non-DM PWC dogs, and 6 Beagle dogs without neurologic signs. There was no significant difference in the ratio of neurons with microtubule-associated protein light chain 3 (LC3)–positive somata relative to those that were LC3 negative among the 3 groups, whereas the number of LC3-positive neurites was significantly increased in DM dogs. Punctate LC3 immunoreactivity did not colocalize with a lysosome marker, LAMP2 (lysosome-associated membrane protein 2). NBR1 (neighbor of BRCA gene 1) was localized mostly in reactive astrocytes, whereas there were p62 (p62/A170/SQSTM1)–positive foci in the neuropil of the spinal cord of DM dogs. Western blotting revealed in DM dogs the decreased expression of Beclin1 and Atg16 L, which are molecules involved in formation of the isolation membrane. These findings suggest that altered autophagosome degradation may result in LC3 and p62 accumulation in the DM spinal cord, whereas the early stage of membrane formation is likely to be downregulated.
Keywords
Canine degenerative myelopathy (DM) is a progressive neurodegenerative disease that causes progressive ataxia and muscle atrophy and that results in fatal paralysis, dysuria, and dyschezia. 1,6,11 Based on the similarity of clinical findings and possible involvement of superoxide dismutase (SOD1) mutation, 2,19,23 canine DM may be a spontaneous animal model of human amyotrophic lateral sclerosis (ALS), though only few ALS cases have SOD1 mutations.
In Japan, most recent cases of DM with SOD1 mutation c.118G>A have appeared in Pembroke Welsh Corgi (PWC) dogs. 3,17,18 Most PWC dogs do not develop DM even though they have a homozygous or heterozygous SOD1 mutation, thereby indicating that the mutation is only one of the risk factors. 3
We previously reported the involvement of oxidative stress, abnormal deposition of ubiquitinated protein, 17 marked neuronal loss, and involvement of excitotoxicity 18 in the spinal cord of PWC dogs with canine DM. These results clarified the similarities and differences of pathologic features between human ALS and canine DM in PWC dogs.
Macroautophagy, hereafter autophagy, is 1 of the 2 major protein degradation systems in mammalian cells, the other being the ubiquitin-proteasome system. Autophagy is subclassified into 2 types: inducible autophagy and basal autophagy. The former is upregulated in response to several conditions, such as nutrient starvation, oxidative stress, hypoxia, and excitotoxicity. 9 The latter is important for the constitutive turnover of cytosolic components via the degradation of denatured proteins, and its dysfunction is thought to result in neurodegeneration. 22
Protein degradation by autophagy begins with the formation of the Beclin1-Vps34 PI 3-kinase complex and then involves the Atg12-Atg5-Atg16 L complex in the pathway for the formation of a double membrane (the so-called isolation membrane). After the conversion of microtubule-associated protein light chain 3 (LC3) from LC3-I to LC3-II, the isolation membrane wraps around target substances, such as denatured proteins, organelles, and lipofuscin, using adaptor proteins such as p62/A170/SQSTM1 (p62) and neighbor of BRCA gene 1 (NBR1). The isolation membrane finally forms autophagosomes. Then, substances in the autolysosomes—autophagosomes fused with lysosomes—are degraded by lysosomal enzymes. LC3 is a marker for autophagosomes and autolysosomes. 12 Dysregulation of the autophagy system leads cells to programmed cell death other than apoptosis, so-called type II programmed cell death. 10
Recently, basal autophagy deficiency has been shown to be involved in various neurodegenerative diseases, such as Alzheimer disease, Huntington disease, Parkinson disease, and ALS. 4,5,14,15 In sporadic ALS cases, there are LC3-positive autophagosomes in motor neurons and p62-positive skein-like inclusions and/or round bodies in the ventral horn. 21 These features indicate that these inclusion bodies and aggregates observed in ALS cases are target substances of the autophagic process; moreover, deficiency of the process may cause the formation of these inclusions. Yet, in SOD1G93A transgenic mice (an ALS model), massive accumulation of LC3 13 and p62 7 in the ventral neurons was observed. The acceleration of the disease by the upregulated formation of autophagosomes 3 was also reported. These reports indicate that loss of the formation-degradation balance of autophagosomes would be an aggravating factor of ALS. 26 Such deficiency of basal autophagy may result in type II programmed cell death, while the role of apoptosis as a mechanism of neuronal death in ALS 24,25 or DM 17 is still controversial. We previously reported that there were ubiquitin-positive foci in the spinal neuropil in canine DM. 17 Based on the existence of the denatured proteins, the deficiency of basal autophagy as a protein degradation system is suspected.
In the present study, the distribution and expression levels of several autophagy-related molecules are examined in the spinal cords of PWC dogs with or without DM, as well as in beagle dogs, and are compared with those reported in ALS cases and ALS model mice.
Materials and Methods
Animals and Tissue Samples
Spinal cords were used of 8 PWC dogs histopathologically diagnosed with DM (9 to 16 years old), 5 PWC dogs without significant lesions (non-DM, 6 to 11 years old), and 6 Beagle dogs as a control without any neuronal signs (1 to 14 years old). Information on the dogs is summarized in Supplemental Table 1. Representative specimens from the cervical (C5 to C7), thoracic (T5), and lumbar (L3 to L5) cords were used. Examined regions are indicated in Supplemental Table 1.
SOD1 Direct Gene Sequence Analysis
Genomic DNA was extracted from paraffin-embedded tissue from each dog, using a commercial kit (DNA Isolator PS Kit; Wako Pure Chemical Industries, Osaka, Japan) according to the manufacturer’s instructions. The genotypes were determined using direct sequence analysis. Briefly, polymerase chain reaction was carried out targeting a sequence around c.118G>A in exon 2 of the canine SOD1 gene with forward (5′-TTT TGT GCT TTT CTT TGA GTG AAG-3′) and reverse (5′-GAT CAT TTC CCT AAG GCT GAC-3′) primers designed on the basis of GenBank data (NW_003726114 and AF346417). The 270–base pair polymerase chain reaction fragment was purified by agarose gel electrophoresis and sequenced in the forward direction using the BigDye Terminator 3.1 mix (Applied Biosystems, Foster City, CA) and an ABI Prism 3130 Genetic Analyzer (Applied Biosystems).
Histopathology, Immunohistochemistry, and Immunofluorescence
Parts of tissue samples from the spinal cords were fixed in 10% buffered formalin, processed routinely, and embedded in paraffin wax. Four-micrometer-thick paraffin sections were subjected to hematoxylin and eosin, Luxol fast blue–hematoxylin and eosin, and Klüver-Barrera stains and to immunohistochemical analysis.
The immunohistochemical analysis was performed using the Envision polymer method. Supplemental Table 2 lists the primary antibodies used. Deparaffinized sections were first autoclaved at 120°C for 10 minutes in 10mM citrate buffer (pH 6.0) for antigen retrieval. Then, the tissue sections were treated with 3% hydrogen peroxide–methanol at room temperature for 5 minutes and incubated in 8% skimmed milk/Tris-buffered saline with 0.2% Tween 20 (TBST) at 37°C for 1 hour to avoid nonspecific reactions. The sections were then incubated at 4°C overnight with 1 of the primary antibodies (Supplemental Table 2). After being washed 3 times in TBST, the sections were incubated with Envision horseradish peroxidase–labeled polymer anti-mouse or anti-rabbit IgG (Dako) at 37°C for 40 minutes. Then, the sections were washed with TBS, and the reaction was visualized with 0.05% 3,3′-diaminobenzidine and 0.03% hydrogen peroxide in TBS. Counterstaining was performed with Mayer’s hematoxylin.
Ventral motor neurons were defined as a βIII-tubulin-positive structure with morphologic features of a neuronal cell, including a minor axis of more than 15 μm in the ventral horn. 18 Counts of immunopositive neurons or neurites in the ventral horn, not including intermediolateral nuclei, were performed using representative sections of each dog and shown as the number per section.
For double immunofluorescence staining, sections were incubated with a primary antibody and then with another primary antibody (Supplemental table 2). The sections were incubated with a cocktail of FITC-conjugated anti-mouse IgG (1:100, Vector Laboratories, Burlingame, CA) or anti-goat IgG (1:100, Invitrogen Life Technologies, Paisley, UK) and Alexa 594-conjugated anti-rabbit IgG (1:100, Vector Laboratories) at room temperature for 1 hour. After being mounted with mounting medium (Vector Laboratories), the specimens were observed and photographed using a DMI3000B microscope (Leica Microsystems, Wetzlar, Germany).
Microscopic images were captured using the Nikon DM-U3 digital camera and NIS-Elements software (Nikon Instruments, Tokyo, Japan). The number of immunopositive neurons/neurites for active-caspase 3 or LC3 in the ventral horn was counted with an optical microscope.
Western Blotting
The other parts of spinal cord samples of several dogs (Supplemental Table 1) were frozen and used for Western blotting. After being separated into the gray and white matter, the samples were homogenized in a 10mM Tris-HCl (pH 7.8) buffer containing 150mM NaCl, 1mM EDTA, 2mM Na3VO4, 10mM NaF, 1% NP-40, and 5% protease inhibitor cocktail (Roche Applied Science, Penzberg, Germany) using the Bio Masher II (Nippi, Tokyo, Japan) and centrifuged at 12 000 × g and 4°C for 20 minutes. Supernatants were collected as the tissue protein samples. Protein concentrations were determined by the Lowry method using the DC Protein assay kit (Bio-Rad, Hercules, CA) according to the manufacturer’s protocol, with bovine serum albumin as a standard. An equal volume of Laemmli sample buffer (Bio-Rad) was added to the samples, followed by mixing and boiling at 100°C for 5 minutes. The samples were separated by SDS-polyacrylamide gel electrophoresis, and the protein bands were blotted to polyvinylidene fluoride membrane (GE Healthcare, Buckinghamshire, UK). After blocking with 5% skimmed milk dissolved in TBST, the membrane was reacted with antibody against Atg16 L, Beclin1, p62, LC3, or β-actin, followed by a reaction with anti-rabbit or anti-mouse IgG antibody conjugated with peroxidase. Proteins reactive with those antibodies were detected with the aid of ECL detection reagents (ECL Prime, GE Healthcare) and analyzed with a Lumino image analyzer (Chemi-Doc, Bio-Rad). Positive bands were quantified as expression levels relative to the β-actin band using Image Lab software (Bio-Rad), except LC3, which was represented as the ratio of the expression level of LC3-II (16 kDa) to that of LC3-I (18 kDa). Because the samples were not obtained at once, the assays on each dog were performed with the sample of dog No. 1, and the results were shown as the ratio versus dog No. 1.
Data Analysis
Data are expressed as the mean ± SEM or scatter plots. For statistical analyses of the data, 1-way analysis of variance followed by the Tukey-Kramer multiple comparison procedure was used. Differences between groups were considered statistically significant when P < .05. For correlation coefficient (r) between groups, the Pearson product-moment correlation coefficient procedure was used.
Results
Punctate immunoreactivity for LC3 was found in the neuronal somata, neurites, and neuropil of the gray matter (Figs. 1a, 2a, 3a). Lipofuscin granules in the neuronal somata were partially positive for LC3. LC3-positive cells were also observed in the white matter in all dogs (Figs. 1b, 3b, 3b). The result of double immunofluorescence revealed that LC3 colocalized with βIII-tubulin, including neuronal somata and neurites, but not with lysosome-associated membrane protein 2 (LAMP2; Fig. 3c, 3d). The number of neurons with LC3 immunoreactivity in the somata (LC3-positive neurons) in the ventral horns was not significantly different among the 3 groups in the cervical and thoracic cord, whereas there were significantly fewer in DM dogs than in Beagles or non-DM dogs in the lumbar cord (Fig. 4). There were no differences in the ratio of these LC3-positive neurons to βIII-tubulin-positive neurons among the 3 groups in cervical, thoracic, and lumbar cords (Fig. 5). However, the number of neuronal neurites with LC3 immunoreactivity (LC3-positive neurites) was significantly larger in the spinal gray matter of DM dogs compared with that in Beagles or non-DM PWC dogs (Fig. 6).
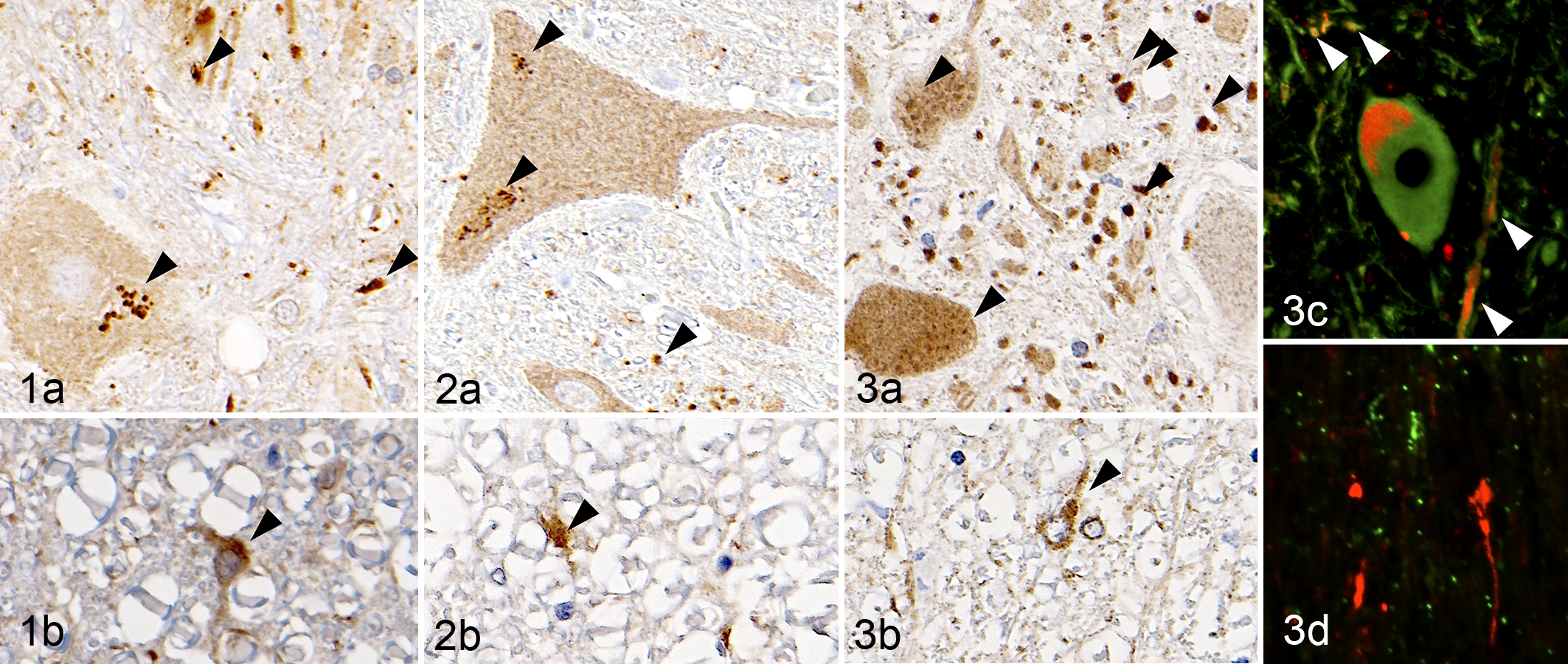
Control, spinal cord, T5: (a) ventral horn and (b) ventral funiculi. Beagle dog No. 2. Punctate microtubule-associated protein light chain 3 (LC3) immunoreactivity (arrowhead) was observed in the neuronal somata and neurites. Immunohistochemistry (IHC) for LC3.
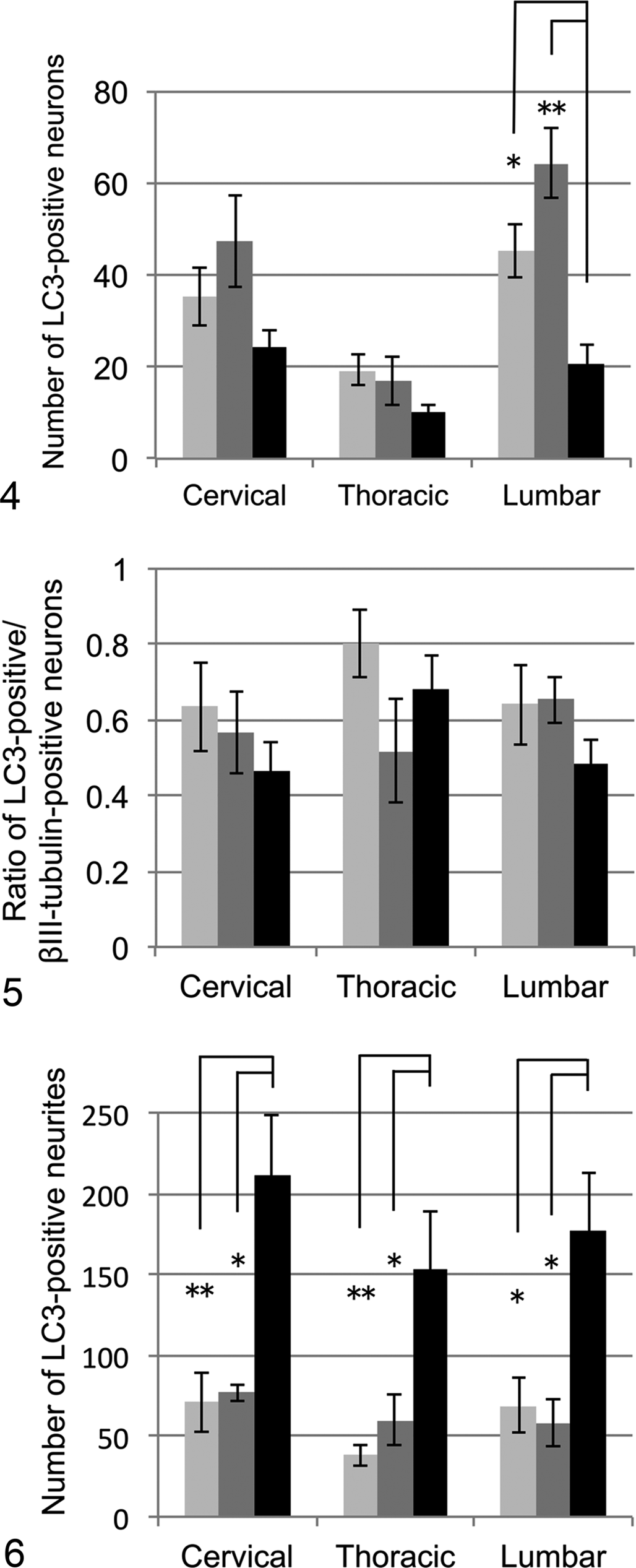
The number of microtubule-associated protein light chain 3 (LC3)–positive neuronal somata in the ventral horn. Dogs: Beagle (light gray bars), Pembroke Welsh corgi (PWC) dogs without degenerative myelopathy (non-DM; gray bars) and with DM (black bars). No significant differences were observed among the 3 groups in the cervical and thoracic cord, whereas the level was significantly low in the lumbar cord of DM dogs. *
P < .05. **
P < .01.
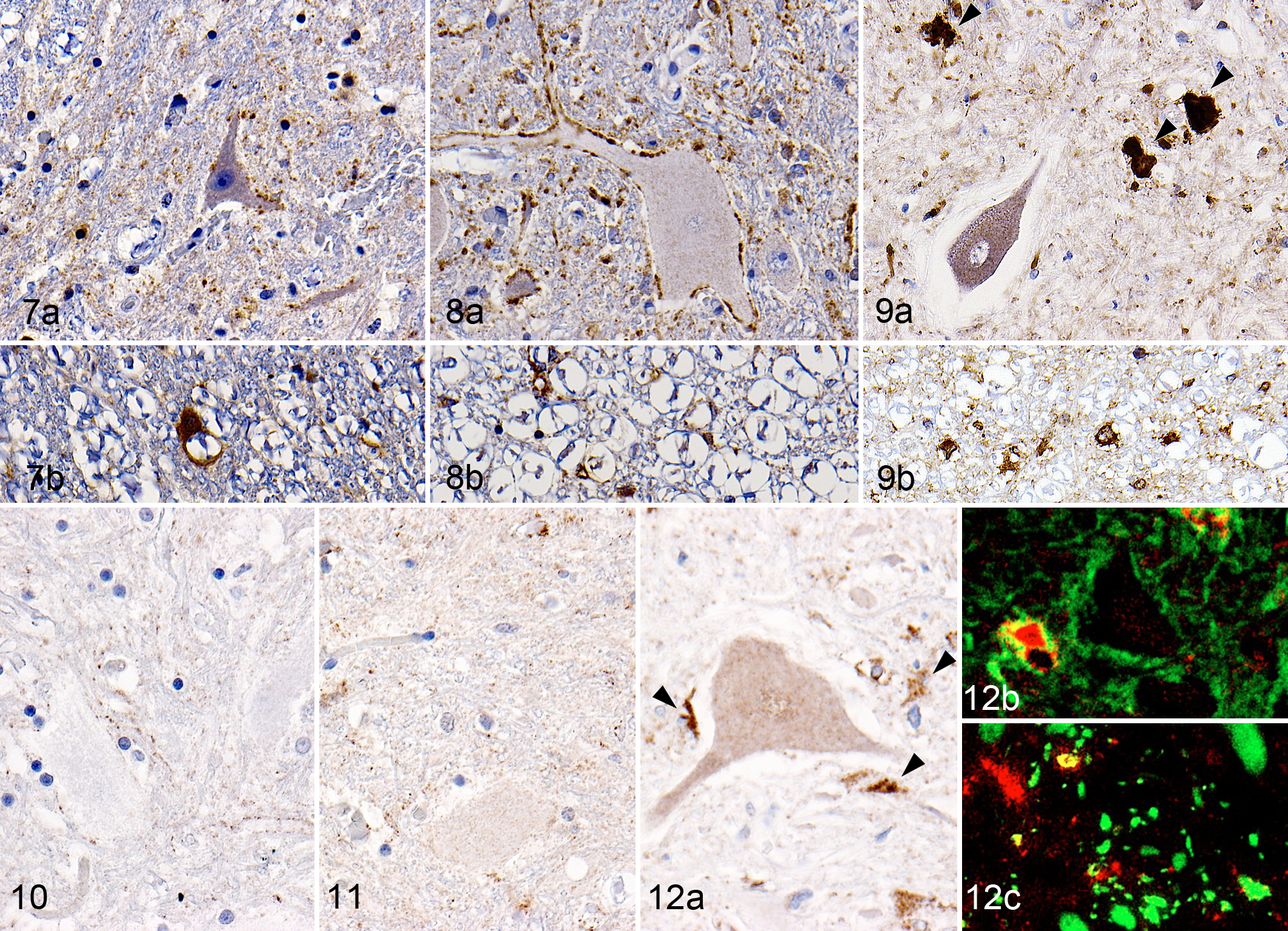
In all the dogs, p62-positive granules were observed in the neuropil mainly in the gray matter (Figs. 7a, 8a, 9a), and p62-positive cells were frequently found in the white matter (Figs. 7b, 8b, 9b). In addition, p62-positive foci were often observed in the neuropil of DM dogs (Fig. 9a). The p62-positive foci in the neuropil did not colocalize with βIII-tubulin, GFAP, HLA-DR, CNPase, or pNF (data not shown). NBR1-positive granules were observed in the neuropil of all the dogs (Figs. 10–12), whereas strong immunoreactivity was detected around ventral neurons in all the DM dogs (Fig. 12) and occasionally in non-DM PWC dog No. 10 (data not shown). The result of double immunofluorescence revealed that most of the NBR1-positive cells around ventral neurons were positive for GFAP (Fig. 12b) and partially positive for βIII-tubulin (Fig. 12c).
Control, spinal cord, T5: (a) ventral horn and (b) ventral funiculi. Beagle dog No. 2. Granular expression of p62/A170/SQSTM1 (p62) was observed in the neuropil. Immunohistochemistry (IHC) for p62.
Western blotting was performed for Beclin1, Atg16 L, p62, and LC3 in both the gray and the white matter of the spinal cord. The expression level of Beclin1 in the gray matter was significantly lower in non-DM PWC dogs and relatively low in DM dogs compared with that of Beagle dogs (Fig. 13). The Atg16 L expression level was relatively low in both the gray and the white matter of DM dogs compared with that in the other 2 groups, and a significant difference in the gray matter was observed between non-DM PWC and DM dogs (Fig. 14). The expression level of p62 was relatively high in both the gray and the white matter of DM dogs compared with that of the other groups, but there were no significant differences (Fig. 15). There were no differences in the LC3-II:LC3-I ratio in the gray matter among the 3 groups, whereas the ratio was significantly higher in the white matter of DM dogs than the other 2 groups (Fig. 16). Correlation coefficient between protein expression levels of each molecule and age of examined dogs were also examined (Table 1). Atg16 L expression levels in gray and white matter showed moderate negative correlation to age of dogs, while p62 expression levels in gray matter and LC3-II/LC3-I ratio in gray and white matter were moderately correlated to age of dogs. When DM dogs were excluded from the analysis, Beclin1 levels in white matter were positively correlated to age, while Atg16 L in white matter and p62 in GM were negatively correlated to age. Expression levels of Beclin1 in GM and p62 in WM showed no significant correlation to age of dogs in or without DM dogs.
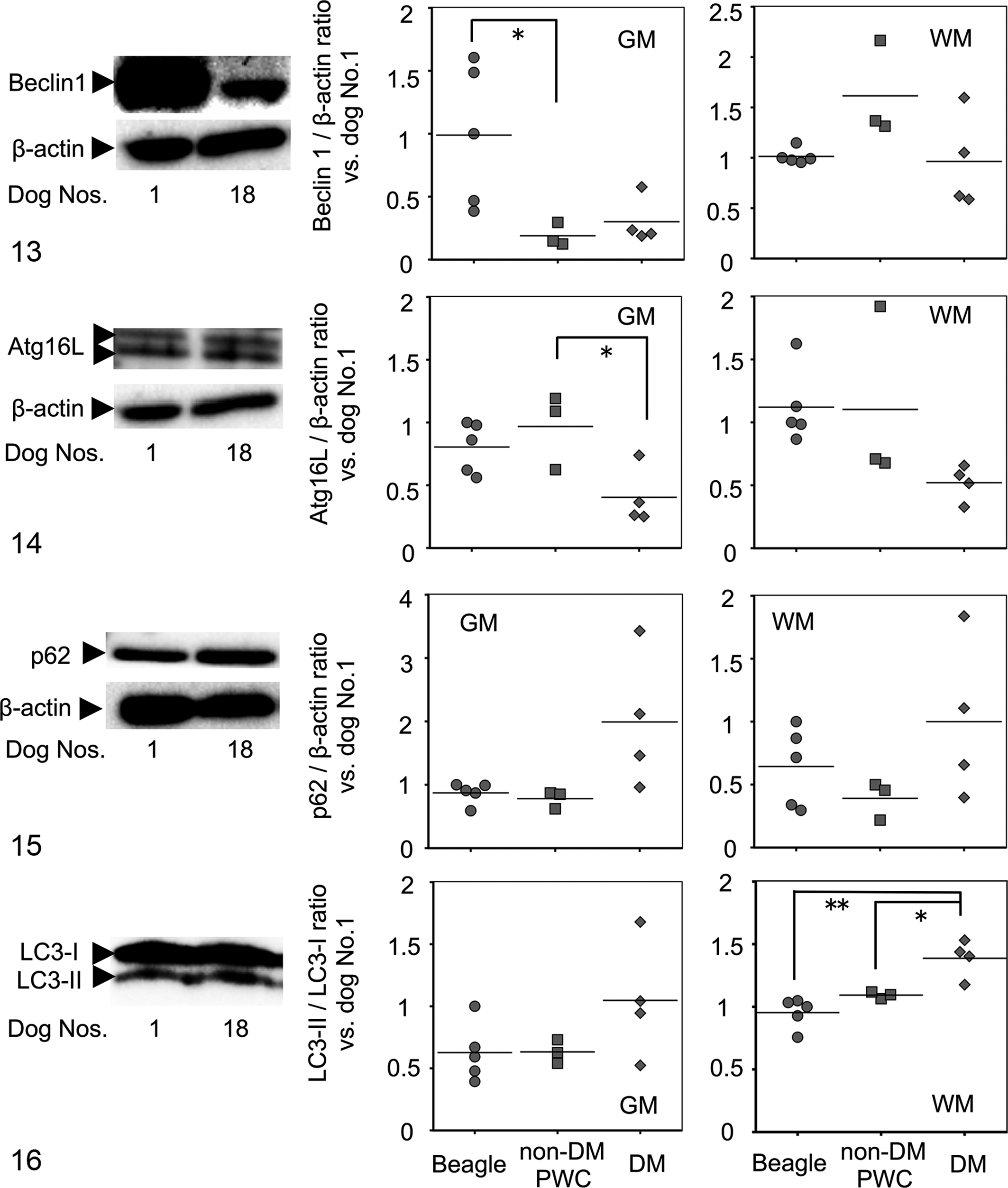
Representative bands and scatter plots displaying the Beclin1 protein expression levels analyzed by Western blotting. The pictures show bands obtained for Beclin1 protein (60 kDa) and for β-actin (42 kDa) of gray matter (GM) of Beagle dog No.1 and degenerative myelopathy (DM) dog No.18. Two boxes of scatter plots show the expression levels of Beclin1 in GM and white matter (WM). Dogs: Beagle (round), non-DM Pembroke Welsh corgi (PWC; square), and DM (rhombic). The expression levels were significantly lower in GM of non-DM PWC dogs than Beagle dogs, whereas no significant differences were observed among the 3 groups in expression levels in WM. Intensity of Beclin1 protein was quantified and standardized with reference to that of β-actin. The dots represent the values in the presence of dog No. 1 as a standard (1.0). *
P < .05.
Pearson Product-Moment Correlation Coefficient Between Protein Expression Levels by Western Blotting and Age of Dogs.
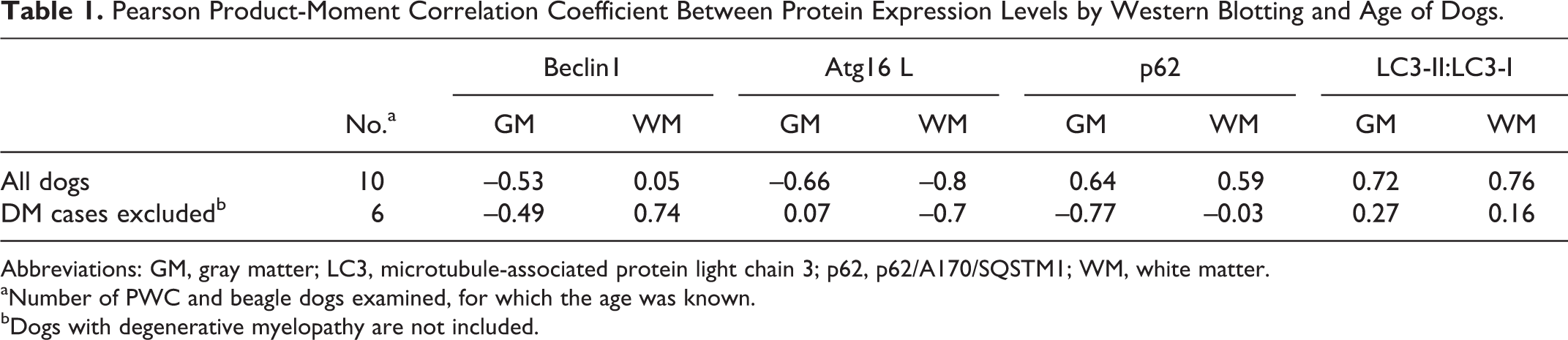
Abbreviations: GM, gray matter; LC3, microtubule-associated protein light chain 3; p62, p62/A170/SQSTM1; WM, white matter.
aNumber of PWC and beagle dogs examined, for which the age was known.
bDogs with degenerative myelopathy are not included.
Discussion
Although there have been no reports concerning the distribution of LC3, p62, and NBR1 proteins in normal canine CNS tissue, the expression pattern of these molecules observed in all the examined dogs in the present study may reflect the steady-state expression of these molecules. The reduction of LC3-positive neurons found in the lumbar cord of DM dogs is presumably due to ventral neuron loss. In fact, the ratio of LC3-positive neurons did not differ among the 3 groups in the cervical, thoracic, and lumbar cords. However, although synapse loss was demonstrated in DM dogs in our previous study, 18 neurites with punctate LC3 immunoreactivity were significantly increased in DM dogs in all the spinal regions examined in the present study. Based on these observations taken together with the absence of an intraneuronal ubiquitin-positive structure in DM dogs, 17,18 aggregation of denatured proteins is likely to be localized in the neurites, not only in the neuronal somata. The punctate LC3-positive structures did not colocalize with lysosome marker LAMP2, indicating the deficiency of autophagosome-lysosome fusion, resulting in the accumulation of autophagosomes and autophagy stress, no matter whether positive puncta indicated LC3-I or LC3-II.
In the present study, we could not define which cell types are responsible for formation of p62-positive foci, unfortunately. To our knowledge, p62 is a cytoplasmic protein, not an extracellular one, so the p62-positive foci may exist in cellular processes (such as neurites) that form the neuropil. This unique expression pattern of p62 as large foci in the neuropil observed in DM dogs in the present study resembles that of ubiquitin. 17 The selective autophagy process involving p62 is thought to be a supplemental pathway to degrade abundant ubiquitinated proteins that are beyond the ability of the ubiquitin-proteasome system. Deficiency of this pathway would result in aggregation and/or accumulation of ubiquitin and p62 8 enough to form the observed p62-positive foci. Given this hypothesis, p62 expression in the spinal cords of DM dogs suggests the dysfunction of selective autophagy. Yet, a previous study using SOD1G93A transgenic mice (a common rodent ALS model) indicated that p62 binds to mutant SOD1 protein and to LC3 without ubiquitin, 7 suggesting the existence of a ubiquitin-independent process for p62 aggregation. As such, although we could not see a significant increase of p62 protein expression levels by Western blot analysis, there might be deficiency of the p62-related selective basal autophagy pathway in the spinal cords of DM dogs, with or without ubiquitin involvement, resulting in the formation of p62-positive foci.
NBR1, known as another adaptor protein, is reported to have a different distribution and function from p62 in neurodegenerative diseases. 16,20 Although there have been no reports on the NBR1 expression pattern in ALS patients or ALS mouse models, the distributions of NBR1 and p62 expression were found to differ in DM dogs in the present study. In addition, the majority of the NBR1-positive granules were localized in reactive astrocytes, especially around the motor neurons (Table 2). These findings indicate that proliferated reactive astrocytes may play important roles in the basal or possibly inducible autophagy pathway involving increased NBR1 expression, while formation of the p62-positive foci possibly resulted from deficiency of p62-related basal autophagy in the spinal cords of DM dogs.
Comparison of Pathologic Features of Human Amyotrophic Lateral Sclerosis, Canine Degenerative Myelopathy, and SOD1 Transgenic Mice.

aResults of the present study.
The results of Western blotting indicate that proteins involved in the formation of the isolation membrane, such as Beclin1 or Atg16 L, were relatively downregulated in the spinal cords of DM dogs. If non-DM PWC dogs were at the preonset stage of DM, the decreased Beclin1 expression in the gray matter possibly inhibited the basal autophagy pathway before disease onset. However, the level of Atg16 L decreased in DM dogs compared with that in non-DM PWC dogs; this change seems to have happened after the onset of disease. A high expression level of p62 in the spinal white and gray matter was observed in several DM dogs. This may reflect the p62 accumulation mentioned above through disease progression. The LC3-II:LC3-I ratio did not differ among the 3 groups in the gray matter, indicating that the neuritic accumulation of LC3 puncta was not due to the accelerated transformation of LC3-I to LC3-II. These 2 types of LC3 could not be distinguished in immunohistochemical examination. Considering the inhibition of the isolation membrane formation and the absence of LAMP2/LC3 colocalization (autolysosomes), the deficiency of flux of autophagosomes is likely to be the cause of increased formation of p62-positive foci and LC3-positive puncta. The increased LC3-II:LC3-I ratio observed in the white matter of DM dogs may involve reactive upregulation of basal autophagy to severe myelin/axonal degeneration as a major event of the DM pathology. Although we have realized that the SOD1 mutation is not the exclusive factor in the pathogenesis of DM, as reported recently in Japan, 3 the PWC breed may have additional factors associated with the development of the disease. Considering the results that the non-DM PWC dogs have significantly increased and/or altered expression patterns of autophagic proteins compared with Beagle dogs, altered expression patterns or levels could be a cause of disease, not only a consequence.
In this study, we divided the spinal samples of dogs into 3 groups, regardless of age or sex. Because of the difficulty of sample collection, we could not add any other age-matched Beagle or PWC dogs without spinal lesions. Based on the fact that morphologic features such as formation of p62-positive foci were observed in all DM dogs and not in any other dogs, the findings suggest that these changes were induced by the disease process. In addition, the correlation coefficient analysis revealed that the protein expression levels of molecules with significant difference among 3 groups were not necessarily correlated to the age of the dogs, including or not including DM dogs. However, the average ages of the 3 groups were significantly different, and it is known that the autophagy system could be affected by aging; so, the possibility that the differences in the expression levels of the 3 groups were caused by aging could not be completely denied, and a larger study would be required to clarify this issue.
In conclusion, deficiency of autophagy function, especially autophagosome degradation—as indicated by the decrease of Atg16 L and Beclin1—is likely to be localized in neurites and components of neuropil, resulting in the formation of p62-positive foci and LC3 puncta (ie, not colocalized with lysosome markers) and so may be involved in the pathogenesis of DM in PWC dogs. The distribution of autophagy-related molecules did not resemble that in ALS patients or mouse models of ALS, suggesting that the role of autophagy in DM in PWC dogs differs from that in these other 2 diseases (Table 2). In addition, the consequences of such pathologic events in DM seem to be neuronal cell death involving type II programmed cell death.
Footnotes
Acknowledgements
We are grateful to the owners of the examined dogs who participated in this research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
