Abstract
The role of mast cells (MCs) in allergic reactions and parasitic infections is well established. Their involvement in host immune response against bacterial and viral infections is reported. In this study, investigation is made to determine if MCs are associated with Canine parvovirus-2 (CPV-2)–induced enteritis with crypt abscess (ECA). Mast cell count (MCC) was made on toluidine blue–stained intestinal sections from a total of 34 dogs. These included 16 dogs exhibiting ECA positive for CPV-2 and negative for Canine distemper virus and Canine coronavirus by immunohistochemistry and fluorescent antibody test, 12 dogs with inflammatory bowel disease (IBD), and 6 non-ECA/non-IBD (control) dogs. The average total MCC per high-power field in ECA (40.8 ± 2.2) and IBD (24.7 ± 2.1) was significantly higher (P < .05) than in the control (3.4 ± 0.6). Although not significant (P > .05), MCC was also higher in ECA than in IBD. The present study for the first time has documented significantly increased MCs in CPV-2–associated ECA as was previously reported for IBD, showing that MCs may also play an important role in CPV-2–associated ECA. Further studies involving more CPV-infected dogs are recommended to substantiate the findings.
Keywords
Mast cells (MCs) are crucial elements in several physiological and immunological functions of the gastrointestinal tract and provide membrane mediators as well as cytokines in allergic and inflammatory diseases. 5,20 Apart from their well-established role in allergic reactions, MCs are suggested as essential for immediate pathogen containment and clearance with long-term host responses to pathogens by modulating draining lymph nodes, influencing cell trafficking to the draining lymph nodes, and promoting development of adaptive immunity to pathogens. 1 Mast cells act not only as the first line of defense against parasitic and bacterial infections but also for certain viral challenges through programs tailored for antiviral defense. 18 However, MC responses to viruses have hardly been examined. 1 Their role is reported in the pathogenesis of viral myocarditis; 10 Newcastle disease virus–induced proventriculus mucosal injury; 19 Dengue virus (DENV) infection in rodent, monkey, and humans; 18 infection with HIV that directly infects human MCs; and against double-stranded RNA viruses recognized by Toll-like receptors. 2 Mast cells not only resist DENV infection themselves but also appear to enhance the recruitment of natural killer and natural killer T cells to facilitate viral clearance. 18
Sufficient information on the role of MCs in the infectious process in veterinary medicine, particularly in canine gastrointestinal inflammation, is not available. Involvement of MCs in canine inflammatory bowel disease (IBD) and lymphoplasmacytic and eosinophilic enteritis has been previously reported. 12,14 IBD refers to a group of idiopathic, chronic, gastrointestinal disorders, characterized by infiltration of the gastrointestinal tract by inflammatory cells (lymphocytes, plasma cells, eosinophils, and neutrophils) that generally are confined to the mucosae. 12 In dogs, enteritis with disrupted and ectatic crypts filled with necrotic cellular debris (enteritis with crypt abscess, ECA) are mainly described in Canine parvovirus-2 (CPV-2) infections 4 and recently also reported in Canine coronavirus (CCV) infection. 7 In this study, an investigation was made to determine the association of MCs in CPV-2–induced ECA in comparison with control dogs and dogs with IBD.
Mast cell count (MCC) in CPV-2–associated ECA (n = 16) was made and compared with MCC in intestines of dogs with IBD (n = 12) and dogs with no microscopic and clinical evidence of enteritis (control, n = 6). In all groups, sections of duodenum, jejunum, and ileum were examined. Sections of large intestine were not included. The specimens from dogs with ECA were collected at necropsy. Transmural biopsies from clinically suspected IBD and microscopically diagnosed as lymphoplasmacytic enteritis (n = 7) and eosinophilic enteritis (n = 5) 6,8 with no microscopic evidence of other infections were included in IBD. Intestines collected at necropsy from 6 dogs that died due to multiple body traumas (2 dogs, one with gunshot wound), hydrocephalus (3 dogs, one also with granulomatous meningitis of unknown etiology), and a dog with cardiomyopathy were used as control. The age of dogs with CPV-2–associated ECA varied from ≤13 weeks (n = 11) to 5 months (n = 1) and young dogs of unknown age (n = 4). The age of the dogs with IBD varied from 2 to 12 years (average, 6.25 years) and that of the control dogs varied from ≤6 weeks (n = 3) and 3 to 11 months (n = 3).
The specimens were fixed in neutral-buffered 10% formalin solution, processed for routine histology, sectioned at 5 μm, stained with hematoxylin-eosin (HE) and toluidine blue stains, and examined by light microscopy. MCs in the lamina propria mucosa (LP) and the remaining intestinal walls (the submucosae and tunica muscularis, SM) were counted on toluidine blue–stained sections in 6 high-power fields (HPF/400×) in each section. Immunohistochemistry (IHC) against CPV-2 (Athens Diagnostic Laboratory, Athens, GA) and Canine distemper virus (CDV; Tifton Veterinary Diagnostic and Investigational Laboratory [TVDIL], Tifton, GA) and fluorescent antibody (FA) test against CPV, CCV, and CDV were employed (TVDIL) to determine the underlying cause of ECA. The MCs were further stained for tryptase immunoreactivity (TVDIL) to determine if the toluidine blue–stained mast cells were also tryptase positive.
For IHC, tissues were cut at 4- to 5-μm sections and subjected to CPV and CDV immunoreactivity using the streptavidin-biotin method following manufacturer’s instructions. Briefly, the sections were incubated for 1 hour with monoclonal mouse anti-CPV (MCA2064; AbD Serotec, Oxford, UK) at 1:300 dilution using Dako Antibody Diluent (S0809; Dako, Carpinteria, CA) for CPV and with mouse monoclonal primary antibody against CDV (CDV-NP; VMRD, Pullman, WA) at 1:50 dilution for CDV. A biotinylated horse anti–mouse IgG (rat adsorbed, BA-2001; Vector Labs, Burlingame, CA) for CPV and anti–mouse immunoglobulin (Dako) for CDV were used as secondary antibodies. The sections were then incubated in Dako’s LSAB 2 streptavidin conjugated to horseradish peroxidase in phosphate-buffered saline (K1016; Dako) for CPV and streptavidin-conjugated alkaline phosphate for CDV. 3,3′-Diaminobenzidine (DAB; K3466; Dako) for CPV and Bio-Red-Fast Red Chromogen (Biopath Laboratories, Oklahoma City, OK) for CDV were used as substrate-chromogens. Positive control sections of canine small intestine and brain were used for CPV and CDV, respectively. Negative controls were made by substituting the primary antibodies with Universal Negative Control (N1698; Dako) for CPV and Tris-buffered saline (TBS) for CDV.
For tryptase IHC, the sections were incubated for 2 hours with primary antibody against mouse-antihuman tryptase (AbD Serotec, Raleigh, NC) at 1:50 dilutions and later treated with secondary biotinylated anti–mouse immunoglobulins (Dako) in TBS for 20 minutes at room temperature. The sections were washed with TBS, incubated in streptavidin-conjugated horseradish peroxidase (Dako) in phosphate-buffered saline for 20 minutes at room temperature, and later treated with DAB (Dako). Negative control sections were made by substituting TBS for the primary antibody. Finally, in all cases of IHC, the tissue sections were counterstained with Gill’s Hematoxylin (Gills III-Formula; Surgipath, Richmond, IL).
MCC in each group was calculated as mean ± standard error. Analysis of variance was used to analyze the data. Least significant difference was used to separate the data. The results were considered statistically significant if P values were <.05.
Significantly higher (P < .05) average total MCC was recorded in ECA (40.8 ± 2.2) and IBD (24.7 ± 2.1) than in the controls (3.4 ± 0.6) (Table 1). Increased numbers of toluidine blue–stained MCs in IBD and ECA are shown in Figs. 2 and 3, respectively, compared with the control group (Fig. 1). The MCs showed strong immunostaining for tryptase. Although not significant, total MCC was also higher in ECA than in IBD. The MCC was higher in the LP than in the SM in ECA and IBD, but not in the control, where slightly higher MCC was evident in the SM than in the LP. There was a significant difference (P < .05) between MCC in the LP (54.3 ± 2.7) and in the SM (27.2 ± 1.7) in ECA, but not between MCC in the LP and SM in both IBD (P = .385) and the control (P = .554). The respective total MCC in ECA (40.8) and IBD (24.7) was about 12 and 7 times higher than that in the controls (3.4). Intestinal sections from the 16 dogs with ECA were positive for CPV-2 by IHC (Fig. 4) and FA. The sections were negative for CDV and CCV.
Average MCC/HPF ± Standard Error and the P Values in Dogs With Enteritis (ECA and IBD) and the Control Dogs (CN).

CN, control; IBD, inflammatory bowel disease; ECA, enteritis with crypt abscess; HPF, high-power field; LP, lamina propria mucosa; MCC, mast cell count; SM, submucosae and tunica muscularis. Different superscript letters in the same column indicate statistically significant differences (P < .05).
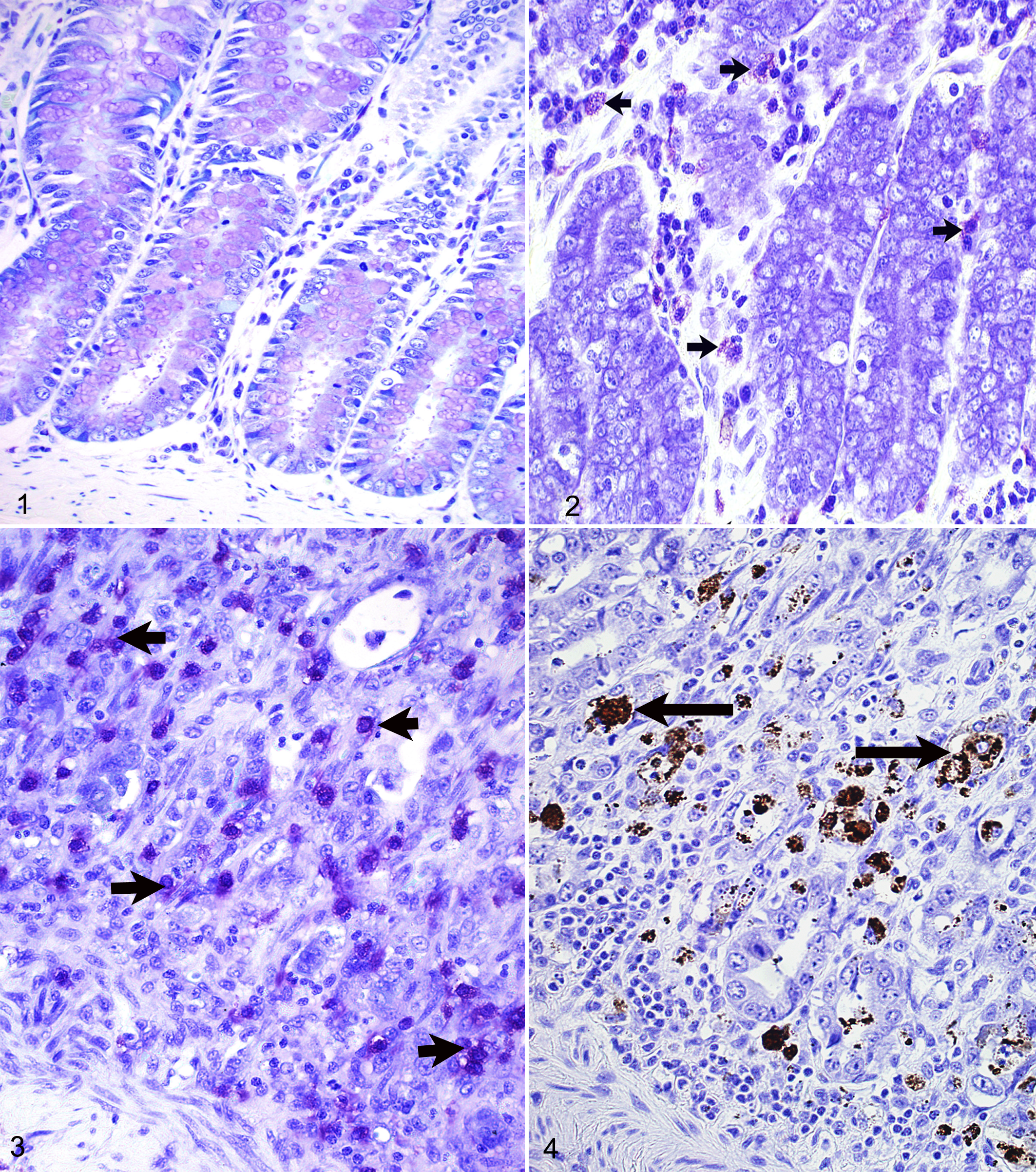
Lesser numbers of MCs were observed in areas where other inflammatory cells were abundant than in areas with mild inflammation, where more MCs were counted in 2 or more different sections per case. MCs were not seen on toluidine blue–stained sections in 1 case of CPV-associated ECA (9-week-old), although large numbers of plasma cells, lymphocytes, and macrophages were evident on HE-stained sections.
Increased MCC in the LP in ECA and IBD indicates inflammation-induced proliferative response of MCs. MCs are crucial for optimal immune response during infection and mobilize various immune cells to the site of infection. 1 Preventing MC degranulation may cause a reduction in the infiltration of inflammatory cells. 19 The higher MCC in the LP of ECA (54.3 ± 2.7), where severe lesions such as disruption of the crypts were encountered, than that of IBD (29.9 ± 2.3), where minimal tissue damage was evident, further indicates that increased MCs in the LP may be associated with severity of lesions. In the control, however, more MCs (P = .554) were counted in the SM (4.4 ± 0.8) than in the LP (2.5 ± 0.2). This most likely is because more normal resident MCs are present in connective tissue around blood vessels and nerve plexus in SM and may not be seen in increased numbers in noninflamed LP.
Lesser numbers of MCs counted in areas with abundant other inflammatory cells suggest that MCs may decrease in number after an initial phase of mobilizing other inflammatory cells to the site of infection as previously described. 1 A decrease in MCC with increasing histologic grade in different stages of the disease is reported in canine IBD. 14 In human systemic mastocytosis, decreased mucosal MCC with increased histamine content in gastric tissue was associated with a negative feedback, in which MCs were reduced due to high tissue histamine levels derived from increased neoplastic MCs. 17 Detailed investigation is required to determine if similar negative feedback or other mechanisms underlie the decrease in MCC in areas where other inflammatory cells were abundant.
This study indicates that MCC is significantly increased in CPV-2–associated ECA. In dogs, ECA is reported in association with CPV-2 4,11 and CCV. 7 Recently, CDV was detected by polymerase chain reaction (PCR) in a case of canine ECA. 13 In this study, all dogs with ECA positive for CPV-2 were negative for CDV and CCV. The immunoreactivity varied from section to section along the intestinal segments, as observed in some cases in which multiple sections from the same animal stained on one slide, some sections were positive for CPV-2 by IHC, and others were negative. Therefore, ECA negative for CPV by IHC could be true or false negative. Marked intestinal crypt necrosis with complete loss of parvovirus-infected cells and mucosal collapse is suggested to decrease the diagnostic sensitivity of FA, IHC, and PCR. 16
The significantly higher (P < .05) total MCC in canine IBD (24.7 ± 2.1) than in the controls (3.4 ± 0.6) concurs with the previous report, which indicates that MCs play an important role in IBD in dogs 14,15 and humans. 3,9 The mechanism through which MCs play a role in the pathogenesis of IBD and ECA is not known.
In summary, MCC was significantly increased in dogs with CPV-2–associated ECA and IBD than in control dogs. MCs are not readily visible in routine HE stain, particularly in areas where other inflammatory cells are numerous. This leads to an easy overlook of MCs undermining their possible role in inflammatory processes in HE-stained sections unless special metachromatic stains such as toluidine blue or IHC for mast cell tryptase are used. This study for the first time documented increased MCC in CPV-2–induced ECA, suggesting their possible role in the pathology of virus-induced canine enteritis as is in IBD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
