Abstract
A 1-year-old female Boer goat was presented for necropsy following spontaneous death and history of acute recumbency, nystagmus, and sialorrhea. A swollen area was grossly observed in the brainstem at the level of the pons. On cut surface, the right cerebellar peduncles were expanded by a focal, pale, poorly demarcated, slightly depressed, and soft area of malacia. Microscopically, this area contained diffuse edema and necrosis, with microabscesses, neuronal necrosis, neuronophagia, axonal spheroids, vasculitis, and perivascular accumulations of lymphocytes, plasma cells, macrophages, and neutrophils. The diagnosis was based on the morphologic findings, fluorescent antibody test results, and special staining.
History, Clinical Findings, and Gross Changes
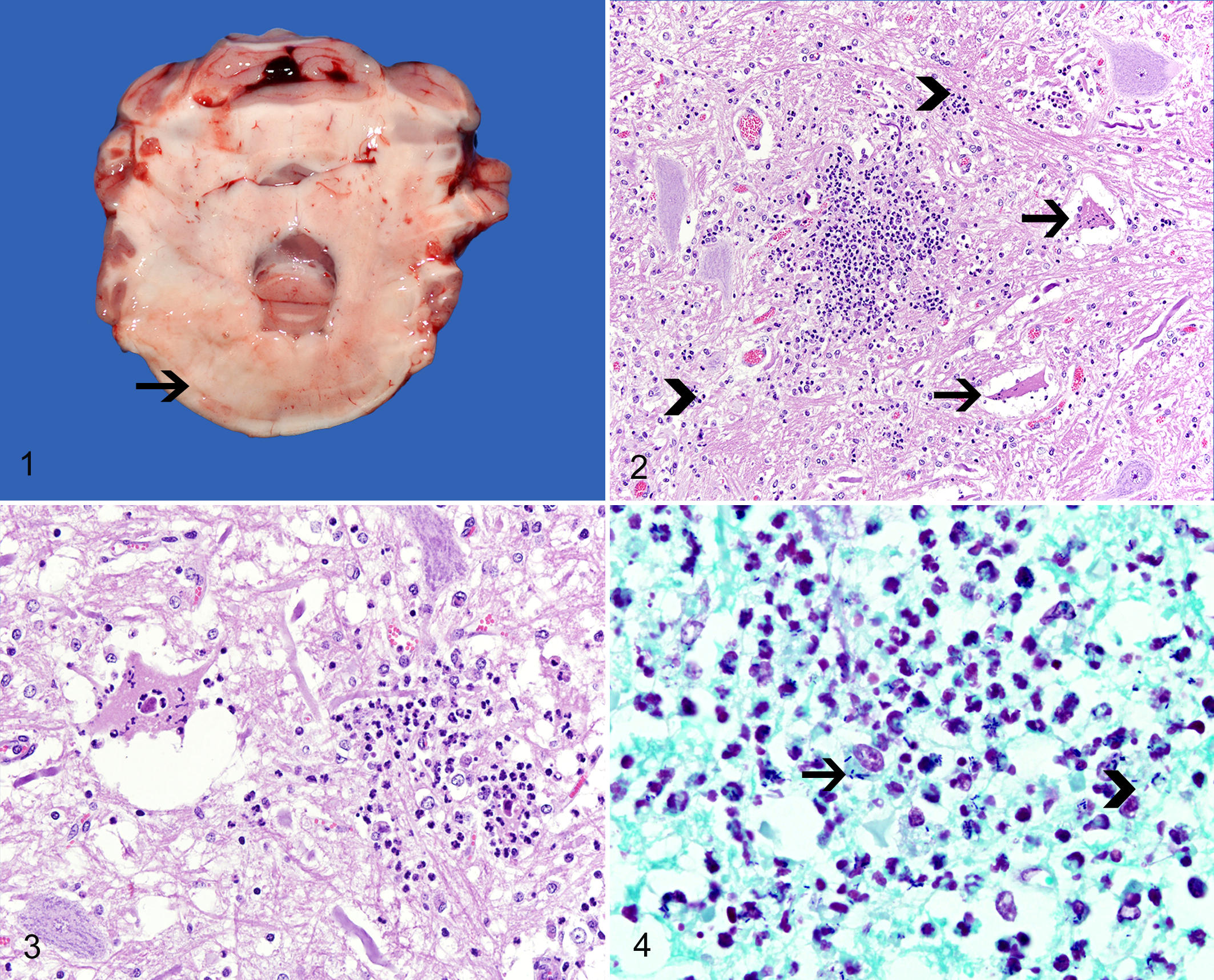
A 1-year-old female Boer goat was referred to a private clinic with history of acute recumbency in March 2012. The goat was part of a herd of 50 goats in northern Georgia and had been fed exclusively pelleted food and hay. Physical examination revealed nystagmus and sialorrhea. The clinical signs worsened, and the goat died 24 hours after the referral. The carcass was submitted for necropsy. The goat was in good nutritional condition, with abundant subcutaneous and abdominal fat deposits. Gross findings were restricted to the brainstem and consisted of a small area of swelling at the level of the pons. On cut surface, the right cerebellar peduncles were expanded by a focal, pale, poorly demarcated, slightly depressed, and soft area of malacia (Fig. 1). Gross inspection of the external ears, ear canals, tympanic bullae, and pituitary gland did not reveal any evidence of inflammation or neoplasia. Fresh samples of brainstem were submitted to fluorescent antibody test (FAT) for rabies virus and Listeria monocytogenes.
Differential Diagnoses
The presence of nystagmus and sialorrhea was suggestive of a lesion in the brainstem or adjacent cranial nerves. 3 Based on these findings, the main differential diagnoses during necropsy included rabies, listeriosis, cerebral abscess, otitis media and/or interna with possible extension to the brainstem, and pituitary abscess. 6 Neoplasia was also considered but deemed unlikely in a young goat. 5
Microscopic and Laboratory Findings
Microscopic findings were restricted to the brainstem (pons and cerebellar peduncles). Diffusely throughout the edematous gray and white matter, there were large numbers of neutrophils that formed multifocal, well-demarcated microabscesses (Fig. 2). These neutrophils were admixed with large numbers of macrophages occasionally containing foamy cytoplasm (gitter cells). Scattered throughout were multiple neurons with deeply eosinophilic and shrunken cytoplasm and pyknotic nuclei (neuronal necrosis) that occasionally were surrounded or infiltrated by neutrophils (neuronophagia) (Fig. 3). Transversal sections of axons with well-defined, swollen, eosinophilic, and round cytoplasm (spheroids) were also seen. Occasionally, capillary walls were expanded by fibrin and a few neutrophils, with luminal occlusion by fibrin thrombi (vasculitis and thrombosis). Multifocally, perivascular spaces in the brainstem and leptomeninges were distended by moderate to large numbers of lymphocytes, plasma cells, macrophages, and neutrophils. FAT performed in fresh samples of brain was negative for rabies virus and positive for L. monocytogenes. Replicate sections of brainstem stained with Lillie Twort special stain revealed numerous intralesional coccobacilli occurring free in the neuroparenchyma or within the cytoplasm of inflammatory cells and neurons (Fig. 4).
Diagnosis
Based on the gross, microscopic and ancillary diagnostic tests, the morphologic diagnosis in this case was focally extensive, severe necrosuppurative rhombencephalitis and meningitis, with microabscesses, vasculitis, and intralesional Gram-positive coccobacilli (confirmed as L. monocytogenes by FAT).
Discussion
The gross and microscopic findings in the brain of this goat are consistent with listeriosis. 1,5 –12 The diagnosis was confirmed by the positive FAT for L. monocytogenes and by the visualization of intralesional Gram-positive coccobacilli. Other ancillary tests that may be used to confirm infection by L. monocytogenes include immunohistochemistry, polymerase chain reaction (PCR), and bacterial culture. 2,4 Immunohistochemistry and PCR are useful in the diagnosis of listeriosis, especially when the antigen load is not sufficient to be detected by FAT or Gram stains and also in retrospective studies using archived, paraffin-embedded tissues. 2,4 Bacterial culture is difficult and requires special procedures, which makes it less reliable for diagnostic purposes. 5
Listeriosis is an infectious and zoonotic disease caused by Listeria spp. It affects several animal species and has been associated with 3 main classical and distinct manifestations: (1) septicemic disease with widespread abscesses in aborted fetuses and neonatal ruminants, horses, pigs, birds, and rabbits; (2) abortive disease in ruminants; and (3) neurological disease occurring mainly in adult ruminants. 5 Only 2 species of Listeria have been known to be pathogenic—namely, L. monocytogenes and Listeria ivanovii. 9 Infection by L. monocytogenes has been associated mainly with abortion and neurologic disease in ruminants, 9 whereas L. ivanovii has been incriminated in cases of abortion in ruminants. 9
Neurologic disease is the most common manifestation of listeriosis in ruminants, and it can occur as isolated cases or small outbreaks, with no age or sex predisposition. 6 Outbreaks of neurologic listeriosis tend to occur during the winter or early spring 1,10 –12 and are usually associated with consumption of silage by the affected animals. 1,12 Twenty-one cases of caprine listerial meningoencephalitis were diagnosed at The University of Georgia’s Department of Pathology between 2005 and 2012. Most of the affected goats developed clinical disease during the winter (12 cases), followed by spring (5 cases), summer (3 cases), and fall (1 case). It is presumed that the highly unfavorable climatic conditions during the cold or rainy season significantly contribute to the production of poor-quality silage with a high pH, which favors bacterial growth and therefore facilitates infection of susceptible animals. 1,12 The increased incidence of outbreaks of listerial meningoencephalitis during the winter may be also attributed to increased animal density, direct contact, and stress under which susceptible animals are submitted during this period of inhospitable climatic conditions. 10,12 However, in some parts of the world such as Australia and South America, outbreaks usually occur during spring and summer and may affect animals that had no contact with silage. 7,8 In these areas, neurologic listeriosis has been described in animals consuming pasture, hay, and a variety of grains or grain by-products. 7 Under these circumstances, the infection is assumed to be acquired from the environment since Listeria spp are ubiquitous organisms and have been isolated from soil, foodstuffs, and feces or milk of normal animals. 5,9 Although the goat of this report developed disease during the winter, no silage had been ever offered to the animals in the herd, confirming that other foodstuffs or the environment can also be the source of infection in cases of listeriosis in ruminants.
A wide range of clinical manifestations may occur in animals affected by neurologic listeriosis. 6 However, similar to the goat of this report, the most consistent clinical signs usually reflect the unilateral or bilateral lesions present in the brainstem, cranial nerves, and less often cerebellum. 5,6 The most commonly affected cranial nerves include the trigeminal, facial, vestibulocochlear, and glossopharyngeal nerves. 5,6 Clinical disease has an acute onset and rapid progression, which is more evident in small ruminants. 6 Clinical signs associated with lesions in the brainstem and vestibular system may include depression, head pressing, head tilt, and circling. Trigeminal nerve involvement may manifest as loss of sensation in the face with paralysis of masticatory muscles and poor jaw tone; facial nerve damage may cause ear, palpebral, and lip drop and loss of menace response; glossopharyngeal nerve dysfunction may cause dysphagia and excessive salivation. 5,6 Other clinical signs may be nonspecific and include anorexia, fever, dyspnea, and tachycardia. 6
Gross changes in the brain of animals with neurologic listeriosis are rare and, when present, consist of unilateral or bilateral areas of softening in the brainstem or spinal cord. 5 Microscopic findings are also restricted to the brainstem, cerebellum, or adjacent nerves. 5 This particular distribution is a consequence of the mechanism of bacterial invasion that occurs by ascending migration through the cranial nerves after infection in the oral, nasal, or conjunctival mucosae. 5 The classic presentation will be characterized by multifocal microabscesses extending from the cranial portions of the cervical spinal cord to the pons, midbrain, and thalamus. 5,6 Lesions contain marked glial reaction that may be associated with variable numbers of foamy macrophages (gitter cells) and extensive areas of edema and necrosis. Other findings often include perivascular accumulation of lymphocytes, plasma cells, macrophages, and neutrophils in the Virchow-Robin spaces and also in the leptomeningeal vessels, as well as inflammation in the cranial nerves. 5,6
Treatment of neurologic listeriosis is usually not effective unless instituted early after the onset of clinical signs. Antibiotics such as penicillin and oxytetracycline are used most frequently in cases of listeriosis, and mixed results have been accomplished. 6 Prevention of disease is based on reduction of exposure to the agent in the feed and environment, as well as on the prevention of stressful situations or concurrent diseases that may increase susceptibility to infection by L. monocytogenes. 6
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
