Abstract
The bacterium Listeria monocytogenes (Lm) is widely distributed in the environment as a saprophyte, but may turn into a lethal intracellular pathogen upon ingestion. Invasive infections occur in numerous species worldwide, but most commonly in humans and farmed ruminants, and manifest as distinct forms. Of those, neuroinfection is remarkably threatening due to its high mortality. Lm is widely studied not only as a pathogen but also as an essential model for intracellular infections and host-pathogen interactions. Many aspects of its ecology and pathogenesis, however, remain unclear and are rarely addressed in its natural hosts. This review highlights the heterogeneity and adaptability of Lm by summarizing its association with the environment, farm animals, and disease. It also provides current knowledge on key features of the pathology and (molecular) pathogenesis of various listeriosis forms in naturally susceptible species with a special focus on ruminants and on the neuroinvasive form of the disease. Moreover, knowledge gaps on pathomechanisms of listerial infections and relevant unexplored topics in Lm pathogenesis research are highlighted.
Keywords
Listeria monocytogenes (Lm) is a globally distributed bacterial pathogen with zoonotic potential, able to cause disease (generally termed “listeriosis”) in a wide variety of domestic and wild mammalian and non-mammalian species, 94,203 including humans, cattle, sheep, goats, dogs, cats, horses, pigs, South American camelids, farmed deer, and poultry. 125,180,306 Species other than humans and ruminants, however, are only sporadically affected. As an animal pathogen, Lm is a cause of concern not only in terms of public health and food safety but also as a significant cause of economic losses when livestock and their offspring are affected.
First described in the 1920s in a human patient, 83 then a few years later in laboratory animals 202,232 and in the following decade in ruminants, 125 the pathogenic nature of Lm has long been recognized. However, the bacterium has been perceived as an important and deadly human foodborne pathogen only following major outbreaks in the 1980s, even though its oral infection route was known decades prior in farm animals. 60,260,261 Since then, surveillance in various countries has continually ranked Lm among the most fatal foodborne pathogens despite the low prevalence, as the mortality rate in listeriosis is high. 85,260 In spite of rigorous, albeit unharmonized, food safety regulations adopted by different countries, 90,287,288 major listeriosis outbreaks continue to occur worldwide today 134,280 with an increasing incidence rate reported in many countries, 45,91,120 causing a noticeable burden on global public health. 60
Lm is an intensely studied pathogen that has been used for decades as a model for bacterial cell invasion, adaptation to and subversion of the host-cell molecular machinery, 136,233 and for innate and adaptive immune responses toward bacterial infection. 219,276 Noticeable features of Lm are its high resilience and versatility allowing it to reside as a saprophyte in various environmental habitats 296 and its rapid switch into a dangerous opportunistic and intracellular pathogen once it is in contact with the host. 103,281 Intriguingly, the bacterium causes various disease manifestations in susceptible hosts, 91,250 of which central nervous system (CNS) infection, known as neurolisteriosis, is particularly threatening and occurs frequently in ruminants. 63,198,309
Despite the advances in our understanding of Lm infection at the cellular level, its environmental and farm life cycle as well as organ targeting mechanisms during listeriosis, in particular CNS infection, remain to be fully deciphered. This review aims to summarize the current knowledge and to map research gaps regarding Lm epidemiology and infection in ruminants with a focus on its peculiar neuroinvasive phenotype.
Lm, a Heterogeneous and Multi-Skilled Bacterium
Lm is a member of the genus Listeria, which currently comprises 21 recognized species of Gram-positive nonsporulating coccobacillary bacteria. 68,171,207,214,241 Among them, Lm is by far the most relevant member in terms of virulence, although Listeria ivanovii is also regularly associated with disease, but exclusively in domestic ruminants. Other species (L. innocua, L. seeligeri, L. grayi, among others) are only sporadically identified in human and animal infections. 6,131,226,243,244,250,252,274,297 Lm is a particularly versatile bacterium being facultative intracellular, facultative anaerobic, and notably osmo- and halotolerant, able to replicate in media containing up to 10% NaCl. Furthermore, it grows in a wide range of temperatures (between 1 °C and 45 °C) and pH (5–9). 51,95 This remarkable resistance to environmental stressors allows the bacterium to survive in a wide variety of ecological habitats and within the host. 294
Lm Strain Heterogeneity
Epidemiological and experimental studies suggest that the variability in environmental distribution, virulence, and clinical manifestations between different hosts are linked to genetic heterogeneity of Lm. 17,190,192,199,213,221 However, the underlying bacterial determinants and mechanisms driving the variability and niche adaptation of Lm are not yet clear, and the investigation of such determinants is currently one focus of research. 66 Characterization of bacterial subtypes and their association with particular niches and virulence has been attempted through various techniques. 54,177 Classically, serotyping methods based on specific antisera allowed for the distinction of Lm into 13 serotypes. 271 Despite its low discriminatory power, 54,177,266 serotyping has been employed for decades as the standard subtyping technique in epidemiological investigations and provided first evidence that Lm subtypes are differentially distributed between environment and clinical disease. 213 Among the 13 serovars, 1/2a, 1/2b, and 4b are the most commonly identified in human and animal clinical isolates, with a noticeable preponderance of serotype 4b in major listeriosis outbreaks 157,266,278 and ruminant neurolisteriosis cases. 155,164,251,308 All 3 serotypes, apart from being implicated in disease, were additionally isolated from food, food processing and farm environments, and animal feces. 26,89,102,152,211,234,277 More recently, molecular typing methods such as pulsed field gel electrophoresis (PFGE), 36,106,123 multilocus sequence typing (MLST), 114 and whole-genome sequencing (WGS) 161 (reviewed in Datta and Burall 54 and Datta et al 55 ) have been employed to link clinical, food, and environmental isolates in epidemiological investigations during outbreaks (PFGE), to study genetic relatedness in Lm populations (MLST), or both (WGS). Phylogenetic analyses performed with MLST and WGS identified 4 distinct lineages (I–IV), further subdivided into clonal complexes (CCs) and sequence types (STs), or sublineages (SLs) and core genome MLST types (CTs), respectively. Lm clusters into 2 major lineages (I, II) that are frequently isolated from diverse sources, and 2 minor lineages (III, IV) that are only sporadically isolated from animal infections. 213 Of the major lineages, lineage I is the genetically most homogeneous and overrepresented in human clinical isolates and ruminant neurolisteriosis cases. 79,192,213,221,251 In contrast, lineage II is genetically heterogeneous and includes predominantly food- and environment-associated strains, but to a lesser extent also strains associated with clinical disease in humans and ruminants. 151,213,234
Prevalence of Hypervirulent Versus Hypovirulent Lm Clonal Complexes in Disease
Various CCs from lineage I belonging to serotype 4b (such as CC1, CC2, CC4, and CC6) are significantly linked to clinical cases in humans and have been shown to be hypervirulent in experimental models. Additionally, they are better adapted to host colonization than clones overrepresented in food and the environment (such as CC9 and CC121). 190,192,199,312 In ruminants, lineage I and, in particular, CC1 and CC4 are significantly overrepresented in clinical isolates and notably in isolates from neurolisteriosis when compared with other clinical listeriosis syndromes such as abortion, mastitis, or gastroenteritis. However, other CCs from lineage I (CC2, CC217, CC6, CC191, CC59) and lineage II (CC7, CC11, CC14, CC37, CC204, CC412) are also regularly isolated from diseased animals and their environment. In contrast, other strains, such as CC9, are predominantly detected in food processing facilities and the environment (Fig. 1). 22,79,220,221,277 However, the cause for the epidemiologically evident predominance of a limited number of subtypes in clinical cases of both humans and ruminants is not exactly understood. Host-associated hypervirulent clones have been shown to possess conserved virulence genes, 17,149,192,199 whereas environmental clones may harbor inactivating mutations of such genes, making them hypovirulent and potentially accounting for their sporadic association with disease. Expression of more stress resistance genes, such as benzalkonium chloride resistance genes in CC9 and CC121 strains, together with negative regulation of transcriptional factors for virulence genes may favor their environmental fitness. 149,190 –192,253,279 Yet, many hypovirulent clones express virulence genes crucial for host invasion and may cause severe disease in specific circumstances, for example, in immunosuppressed patients. 157,313
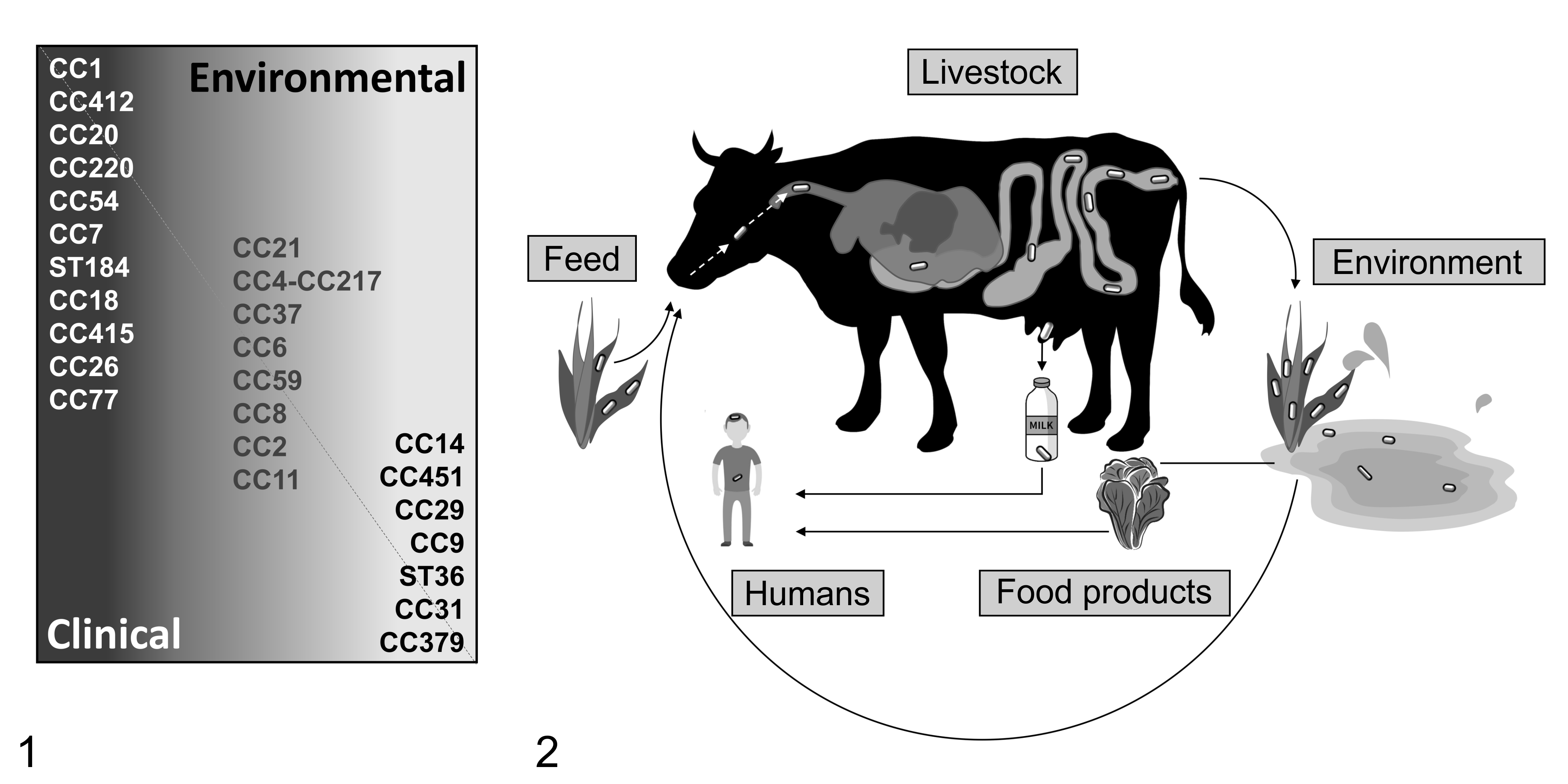
Prevalent clonal complexes (CC) and farm-host cycle of Listeria monocytogenes (Lm).
Global Distribution of Major Lm Clonal Complexes
Hypervirulent strains (eg, CC1) that are overrepresented in clinical isolates from major outbreaks appear to be distributed worldwide 17,43 and, notably, often appear to share a similar distribution between humans and ruminants. 79,190,192,221 Major Lm CCs have also been shown to spread globally over time, causing historically relevant outbreaks. Moreover, within a given geographic region, predominant lineages might shift over time. 199 For instance, hypervirulent (eg, CC6) 17,192 and hypovirulent (eg, CC9, CC121) 17,190 clones are emerging in the 21st century in various continents. This is speculated to occur due to genetic diversification, possibly as a result of fitness adaptation or global dispersion through human travel, animal, or food trade. 17,199 CC1 constitutes a notable example in these regards, as it is speculated to have spread globally from North America through cattle trade. 200 Given the high ability of Lm to adapt to disparate environments and hosts, as well as its relevance for public and animal health worldwide, it is important to map and understand the distribution and pathogenic potential of frequently isolated strains to make surveillance and control more efficient. Moreover, given the potential role of ruminants as carriers of strains pathogenic for humans, further comparative genomic studies are essential to identify strains causing disease in both species, as well as to understand their distinctive features of virulence and inter-host transmission.
Reservoirs of Lm in the Environment
Lm is considered to be a ubiquitously distributed bacterium, remarkably adaptable to a wide range of natural and anthropized habitats (such as agricultural and food-associated ones), but knowledge of its environmental dynamics and ecology is only fragmentary. 204,258,298 Importantly, the dynamics of Lm transmission between the natural environment and ruminant and human hosts remain unclear. In nature, Lm has been isolated from various sources, including soil, water, and vegetation, as well as from feces of numerous wild birds and mammals. 150,204,296,305,314 Bacteria can be generally isolated from natural sources in low numbers and with low prevalence. However, the presence of animals and moisture (in the form of bodies of water and precipitation) has been proposed to favor growth and dispersion of Lm in the environment. 147,176,258 Interestingly, serotypes prevalent in human and ruminant infections (specifically 1/2a and 4b) were commonly isolated from wild birds, red deer, wild boars, and black bears, suggesting that wild animals might constitute a reservoir for pathogenic strains in the natural environment. 138,222,304,314 Invertebrates (eg, slugs) and free-living protozoans have also been shown to be capable of supporting bacterial growth, indicating their potential role as environmental vectors or reservoirs. 117,296 Amoebae, in particular, have been speculated to constitute a niche for Lm, as they have been shown to phagocytose and host bacteria, 169,183,316 while other studies reported them to be bactericidal (reviewed in Schuppler 267 ). It has therefore been speculated that key Lm virulence genes have emerged during its co-evolution alongside environmental phagocytic unicellular eukaryotes to enhance its survival upon predation. 267 Given the similarities of amoebas with macrophages, such an adaptation to amoeba could potentially function as a “training ground” for macrophage infection, explaining the ability of Lm to invade and survive inside cells of higher vertebrates. 183 The ability to successfully adopt a saprophytic lifestyle in the environment and switch to an opportunistic pathogen in the host by reprogramming its gene expression raises serious concerns regarding food safety and animal health. Considering that no definitive reservoir host has been identified in nature, the possibilities of intervention and prevention of its spread into the agriculture and farm industry remain currently limited.
Epidemiology of Lm in the Farm Environment
How ruminants become exposed to Lm in the farm environment is not exactly understood. Contamination of feed, water, or pasture appears to be the most likely route through which Lm is transmitted to the livestock host (Fig. 2), 101,204 but conditions enabling enrichment of the pathogen in these sources have not been fully elucidated. The Lm infectious dose remains currently undefined, estimated to be as low as 104 CFU (colony-forming unit) in susceptible humans and as high as 109 CFU for healthy individuals. Nevertheless, it is believed that infection generally requires high bacterial numbers or repeated consumption of food sources contaminated with low bacterial levels. 32,96,235 Therefore, sources that allow high bacterial replication are also most likely to be involved in Lm infection of ruminants.
Sources of Farm Contamination
Interestingly, moist soil, decaying plant matter, and bodies of water, although shown to provide an appropriate environment for bacterial survival, do not favor extensive bacterial growth. Moreover, it remains unclear whether they are environmental reservoirs without the presence of shedding animals. 148,176,258 Fecal shedding from wild animals and bacterial persistence in invertebrates and protozoal carriers, as previously discussed, may promote bacterial introduction into the agricultural environment, 189,273,282,291 but a direct link between strains isolated from the pristine natural environment, farm environment, and ruminant host remains to be shown.
Fecal-Oral Lm Enrichment Cycles in the Farm Environment?
Persistent in-farm transmission cycles and outbreaks have been increasingly linked to a contaminated animal environment, 38,80,197,204 possibly indicating a role for ruminants themselves in sustaining bacterial persistence in their environment. Fecal shedding may facilitate bacterial contamination and persistence in the farm ecosystem. Agricultural fertilizers based on sewage sludge and manure if left untreated can provide means for significant contamination of pastures or crops. Sheep manure has been the source for the major coleslaw-associated outbreak in 1981 during which the foodborne origin of human Lm infection was discovered. 59,111,261
Lm shedding has been observed in healthy ruminants, 89,141,204,286 and cattle have been reported to shed Lm more frequently and at higher bacterial titers than small ruminants, suggesting a potential role for cattle as significant reservoirs for Lm in the context of an animal-farm cycle of transmission. 80,89,204,205 Various studies also indicate that farm management practices may contribute to bacterial contamination of and persistence in the farm environment. 38,141,197,205 These observations point to the relevance of a fecal-oral route in the maintenance of Lm in the agricultural-farm environment, through fecal contamination of plant-derived food or feed (Fig. 2).
However, animal-farm transmission dynamics and their relevance remain currently rather unclear, 298 hampering the possibilities of adopting effective measures to prevent animal infection. The presence of unidentified asymptomatic ruminant shedders has the potential to significantly contribute to bacterial spread into the food industry via fecal contamination of milk or meat (Fig. 2). 38,100,135,141 The numerous outbreaks linked to contaminated fresh produce and ready to eat foods 37,110 highlight the importance of following a farm-to-fork strategy by restricting bacterial contamination at the farm/farm animal level, in order to ultimately prevent foodborne illness in consumers through the application of a One Health approach.
Silage: The Culprit?
In ruminants, poorly acidified silage has long been implicated as the main source of bacterial contamination, in which Lm is indeed able to replicate abundantly. 99,124,291 Ruminants fed with high quantities of silage have been reported to excrete bacteria in their feces more frequently and in higher numbers, and to develop clinical disease more frequently. 14,100,181,205,309 Silage feeding during hibernal indoor housing has also been linked to the seasonality of clinical listeriosis cases in ruminants in northern hemispheres increasing during winter and peaking in spring. 47,118,172,193,286 However, different epidemiological studies failed to link silage feeding to listeriosis outbreaks, challenging the common perspective that silage constitutes the exclusive source of infection. 33,155,187,289,308 Moreover, studies in the southern hemisphere frequently report listeriosis cases in ruminants unrelated to silage feeding and occurring during the warmest months of the year or during the transition from rainy to dry season. 237,247,248,256 As listeriosis cases can occur all year round in both hemispheres and with different diets, it appears likely that additional factors contribute to infection.
Predisposing Factors for Lm Infection
While in humans clinical listeriosis has a clear predilection for defined risk groups collectively known as YOPI (young, old, pregnant, immunosuppressed), 45,104,119,278 predisposing factors in farmed ruminants have yet to be clearly identified. Concomitant stressing factors, such as late pregnancy or overcrowding, have been proposed to constitute predisposing factors for listeriosis, 51,125,180,205,306 but the extent and mechanisms through which they contribute to disease onset have not been systematically explored. Season and spoiled wet feed due to climatic changes, particularly heavy rains, together with stressing factors including overcrowding and inadequate animal management have also been speculated to increase the incidence of listeriosis. 196,245,289,301
Molecular Mechanisms of Lm Intracellular Lifestyle
The pathogenic potential of Lm relies on the intracellular survival and replication of this microbe. For the intracellular life cycle of Lm (Fig. 3), PrfA (positive regulatory factor A), the transcriptional factor initiating the transcriptional switch from the saprophytic (extra-host) to the intra-host infectious stage, is essential. The expression of PrfA itself is thermo-regulated and becomes efficient at mammalian body temperature (37 °C). 292 PrfA induces the transcription of virulence factors located on the crucial Listeria pathogenicity island (LIPI-1) (eg, hly [LLO], actA, plcA, plcB, and mlp), and additionally virulence factors outside LIPI-1 (eg, inlA, inlB, inlC, and hpt) that altogether are essential for the intracellular infection cycle. 58,165,290
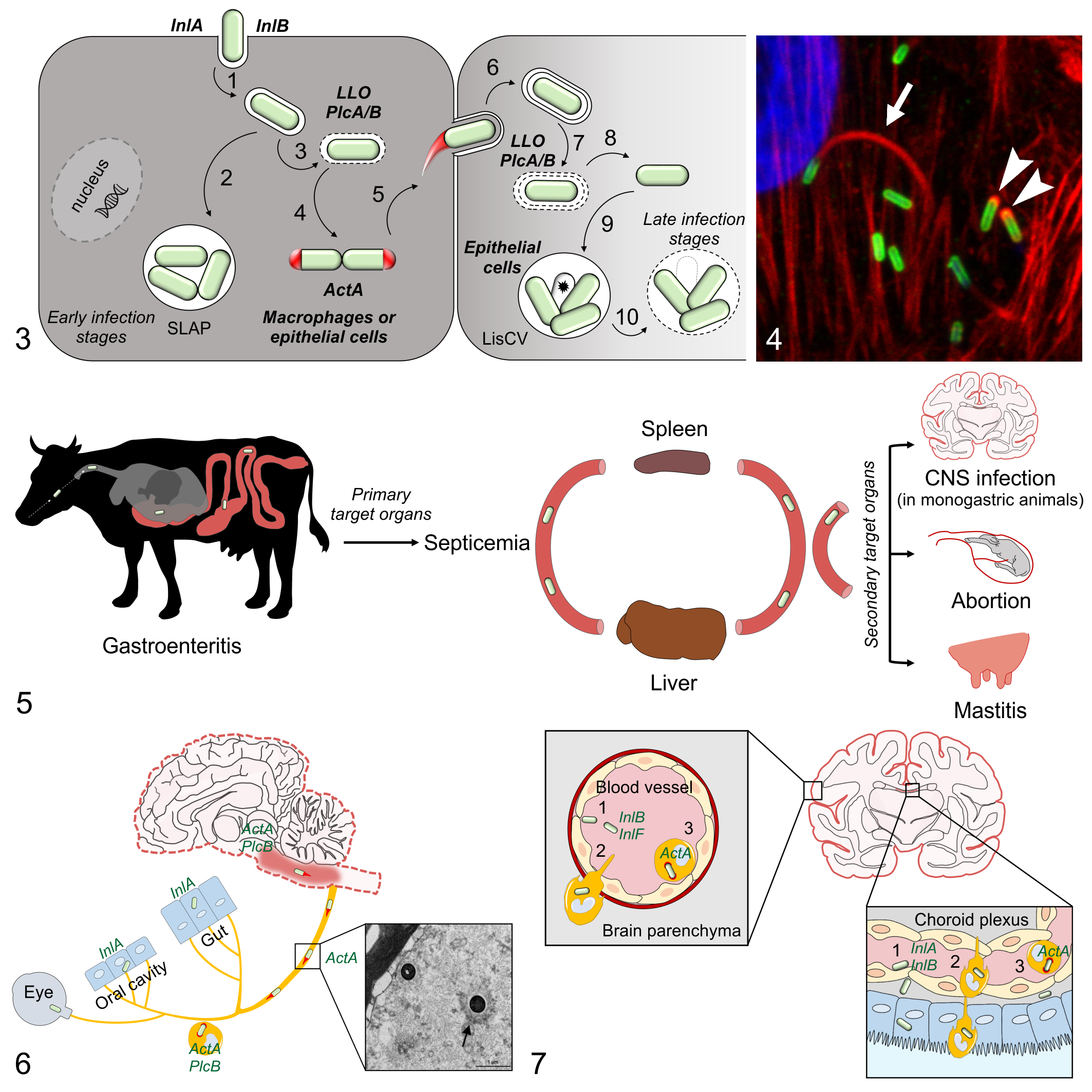
Lm intracellular lifestyle and host infection pathways.
While Lm enters phagocytes via phagocytosis, it initiates internalization in non-phagocytic cells through a process called receptor-mediated endocytosis, 19 primarily via 2 virulence factors, internalin A (InlA) and B (InlB). 70,108 Both internalins bind to eukaryotic cell membrane receptors, InlA to E-cadherin and InlB to Met, gC1QR, and proteoglycans, respectively. 19
Following internalization, Lm is temporarily confined to a primary vacuole, from which it escapes prior to phagolysosomal fusion via membranous pores that are formed by listeriolysin O (LLO). This process is facilitated by the 2 phospholipases PlcA (phospholipase A) and PlcB (phospholipase B). Vacuolar escape enables Lm to avoid phagosomal degradation, which is essential for Lm virulence, as shown by the strong attenuation of mutants in which LLO is deleted. Once in the cytosol, the bacterium starts rapidly replicating using nutrients acquired from the host cell. Actin assembly-inducing protein (ActA) promotes intracellular bacterial motility and cell-to-cell spread by hijacking and polymerizing actin from the host cytoskeleton. 159 Polymerized actin can be identified as “actin clouds,” surrounding Lm, or as polar filaments, termed “actin tails,” which enable Lm movement within the cytosol (Fig. 4). By propelling forward, Lm may arrive into a neighbor cell within a double-membrane vacuole (named a secondary vacuole) and reinitiates a new cycle escaping the vacuole. The dogma of Lm’s canonical intracytosolic infection cycle was recently challenged by several studies that discovered various intravacuolar infection stages associated with bacterial survival and persistence in different phagocytic and non-phagocytic cell types. 21,163,225 It has been shown in phagocytes 21 and more recently in epithelial cells 225 that bacteria-secreting reduced amounts of LLO at early stages of infection remain entrapped in large vacuolar compartments named spacious Listeria-containing phagosomes (SLAPs), where they can replicate at a slow pace. 21 Another mechanism of intravacuolar persistence associated with slow replication (Listeria-containing vacuoles, LisCV) occurs subsequently to downregulation of bacterial ActA expression in the cytosol during long-term infections (2–3 days post-infection) of epithelial cells. 163
The previously mentioned virulence factors are also essential for host infection, as knock-out mutants for key virulence genes (eg, prfA, hly, actA, internalins-encoding genes) 20,31,191,215,253 are almost completely attenuated in vivo. However, it remains open for exploration whether they are involved in different invasion routes and organotropism of Lm. The vast arsenal of additional genes in the Lm genome provides appealing candidates to potentially explain the versatility of Lm to exploit different entry routes and reach various organs. However, the impact on host infection of most genes is currently unknown, and the function of others that have been recently associated with enhanced virulence (eg, LIPI-4) 192 requires further investigation.
Listeriosis: Clinical Disease, Pathology, and Pathogenesis
Listeriosis occurs most commonly upon oral infection, but can rarely result from local bacterial implantation at body surfaces (keratoconjunctivitis, dermatitis) or from ascending infection of the genital tract. 125,236,262,294 Following oral infection, Lm may colonize the gastrointestinal (GI) tract and may either be shed from subclinical carriers or cause self-limiting enteritis, although Lm frequently crosses the GI barrier and causes invasive disease. Current research indicates that bacterial crossing may happen at the intestinal or oral cavity level, depending on the host and clinical form. The main forms of invasive listeriosis include septicemia, fetomaternal/perinatal infection, and CNS disease (Fig. 5), which tend to occur separately and are rarely concomitant in affected individuals or in the same herd or flock. 180,181 The reason why different listeriosis forms seldom overlap remains unknown. As the neurologic manifestation of the disease (neurolisteriosis) is especially relevant in ruminants, it will be discussed in this review more extensively than other listeriosis forms.
Enteric Listeriosis
Lm can colonize the GI tract following ingestion via food or feed, but underlying pathomechanisms are yet to be fully elucidated. Survival in the inhospitable GI environment requires resistance against gastric and biliary acids, which is provided by a complex interplay of genes that are mainly coordinated by the stress-responsive sigma factor SigB (σB) and PrfA, among others (reviewed in Davis et al 56 , and Gahan and Hill 107 ). Additionally, Lm needs to escape from the control mechanisms of the commensal microbial community.
Molecular pathogenesis of Lm enteritis in mouse models
In orally infected mice, enteric colonization is facilitated by the bacteriocin listeriolysin S (LLS), expressed by a subset of lineage I strains. 240 Whether luminal colonization is sufficient to cause isolated enteritis or whether enteritis requires invasion and/or crossing of intestinal epithelium is not fully clear. Similarly, bacterial factors involved in enteritis remain to be identified. Factors enhancing or restricting bacterial invasion of the intestinal barrier are not entirely known. Experimental models provide evidence that Lm translocates the intestinal epithelium (via interaction of the major Lm internalins, InlA and InlB, with their respective cell receptors) without causing significant intestinal inflammation and damage to the intestinal barrier. 173,283 InlA and InlB bind to their cell receptors with variable affinity in different species (reviewed in D’Orazio 52 , Drolia and Bhunia 81 , and Hoelzer et al 142 ), with ruminants and humans allowing for both InlA- and InlB-mediated cell entrance. On the other hand, mice are quite resistant to GI crossing due to a single amino-acid polymorphism in their E-cadherin, which impairs its affinity for InlA. 172 In this species, Lm was shown to primarily enter M cells residing in Peyer’s patches by either InlB-mediated endocytosis or macropinocytosis. 44 In mice expressing “humanized” E-cadherin, bacterial InlA binds luminally expressed E-cadherin on intestinal goblet cells, allowing for bacterial crossing of the intestinal epithelium by exocytosis into the lamina propria at the villus level. 206 It has been proposed that Lm additionally accesses luminal E-cadherin exposed at villus tips during epithelial renewal. 224 Last, LAP (Listeria adhesion protein) was shown to cause the opening of cellular junctions and bacterial crossing of the intestinal barrier upon binding to its receptor Hsp60 on intestinal epithelial cells in mice, independently of InlA/E-cadherin interaction. 82 Intestinal crossing into the lamina propria is relatively silent in terms of inflammation, 283 while bacterial entry into the Peyer’s patches triggers a strong inflammatory response 173 and proliferation of intestinal epithelium with resulting loss of goblet cells and decrease of the mucus layer thickness. 64
Enteritis in natural hosts
Mechanisms of bacterial interaction with the ruminant GI tract, on the other hand, are completely unknown. Clinically associated strains, however, often display marked lysozyme resistance together with enhanced invasiveness. 22,302 It has therefore been speculated that these strains might better survive abomasal passage and are more efficient in invading conjunctival and caruncular epithelial tissues.
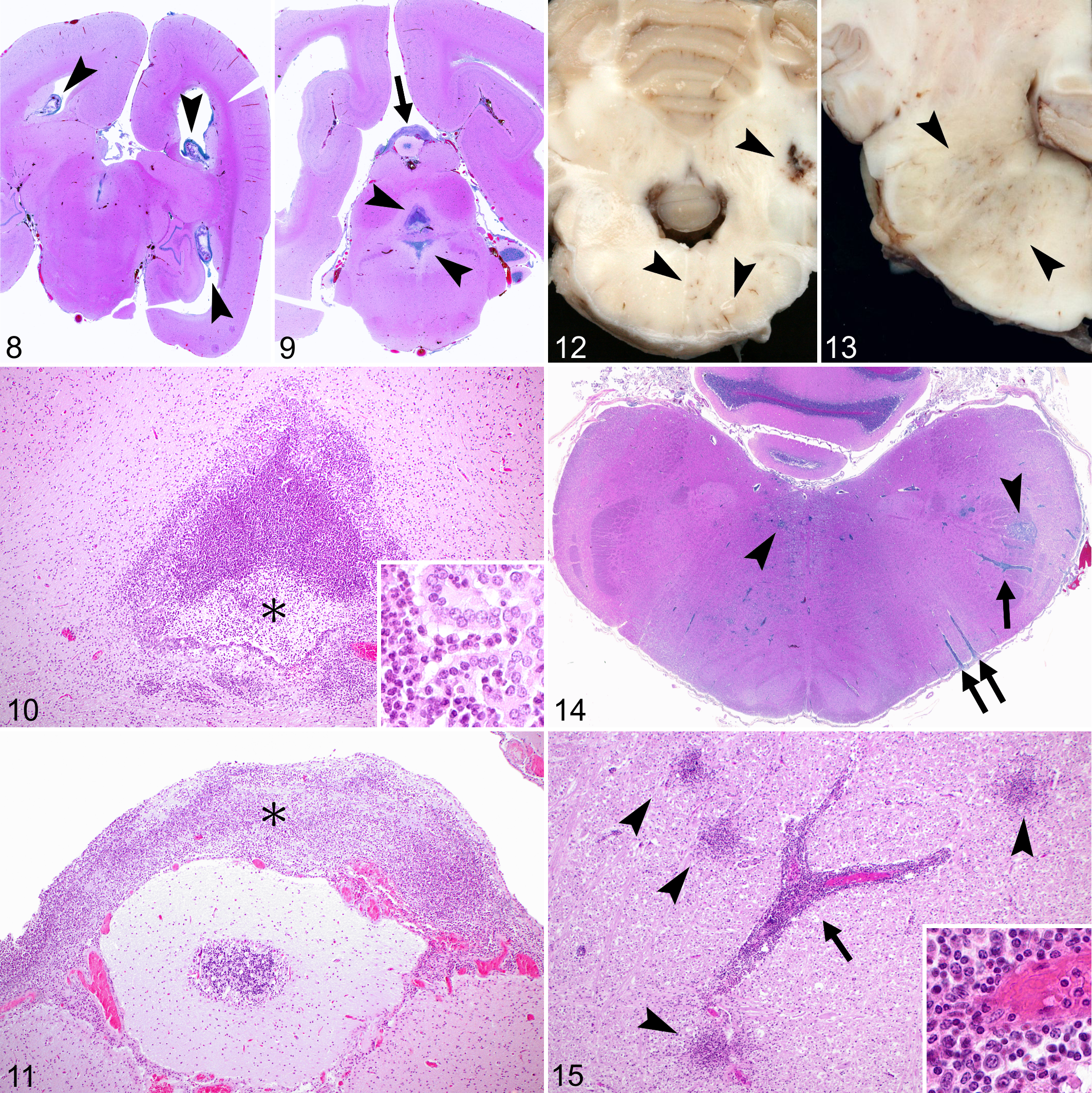
In ruminants, the enteric location of Lm is most commonly associated with prolonged fecal shedding in asymptomatic animals. However, acute enteric listeriosis has been reported in sheep and cattle of different age. 47,93,94,109,317 Affected animals may show lethargy, anorexia, hyperthermia, and diarrhea. Clinical enteric listeriosis is associated with abomasitis and enteritis consisting of multifocal neutrophilic infiltrations, which are strikingly centered on the muscularis mucosae where bacteria reside inside myocytes. 47,93,94,109 The cause and mechanisms of muscular targeting are not known. Associated fibrinosuppurative mesenteric lymphadenitis with intralesional bacteria further supports bacterial crossing of the GI barrier during enteritis. 93,94,109 Moreover, small parenchymal pyogranulomas, mononuclear periportal infiltration, or foci of coagulative necrosis were observed in the liver following enteritis. 93,94,109 Orally infected sheep, however, carried bacteria in the spleen, liver, and lymphoid organs in the absence of clinical signs, 317 indicating that Lm intestinal infection and translocation to visceral organs may occur asymptomatically. As asymptomatic fecal shedders are relatively common and overt disease is infrequently identified, clinically evident enteric listeriosis in ruminants seems to constitute a fairly exceptional event.
In humans, GI colonization can also be asymptomatic with bacterial shedding in feces. 129,201 Alternatively, it can lead to self-limiting gastroenteritis with acute clinical manifestation characterized by fever, diarrhea, and arthromyalgia. These signs can be prodromic to invasive infection in predisposed patients. 212 Pathological data, unlike in ruminants, are lacking, possibly due to the self-limiting nature of the disease. Therefore, cellular targets of Lm in human gastroenteritis remain unknown.
Listerial Septicemia
Following intestinal breaching, Lm enters a bacteremic phase in which it spreads hematogenously to the viscera, mainly liver and spleen. 125,186,317 It is assumed that hepatic and splenic infection can be either temporary in asymptomatic animals or provide a replication niche for further septicemic spread if the bacterium overcomes local innate immunity. 262,317 In the latter case, systemic spread and colonization of other organs, notably placenta and brain, can occur (Fig. 5). 294 Pathomechanisms of listerial septicemia are largely unknown. Recent evidence in experimental models suggests that hypervirulent strains are apt to thrive for a longer time in spleen and liver of infected mice, thus increasing the chance of secondary bacteremia with infection of target organs, 295 which might explain the propensity of such strains to cause invasive disease.
Septicemic listeriosis is best known in humans, affecting about one third of patients with invasive disease. It is potentially associated with fatal complications such as disseminated intravascular coagulation and multi-organ failure. 67,262 Focal infections after septicemic events include myocarditis and valvular endocarditis, hepatitis and cholecystitis, splenic abscessation, peritonitis, osteomyelitis, arthritis, pneumonia, and endophthalmitis. 67,260,262 In non-ruminant mammals, septicemic listeriosis occurs more frequently than other forms of the disease. In ruminants, septicemia occurs mainly in perinatal or juvenile infections and manifests with hyperthermia, anorexia, and diarrhea. 125,180,181,306 Although clinically evident septicemia is uncommon in adult ruminants, placentitis often occurs as a frequent sequela of systemic bacteremia.
Fetomaternal Listeriosis
Lm manifests a particular tropism for the pregnant uterus, which is rapidly colonized. 30,125,180,294,306 In ruminants, fetal infection can develop from transplacental hematogenous transmission and inhalation of contaminated amniotic fluid, usually leading to stillbirths, while in humans it has also been proposed to develop from ascending infection from the maternal lower reproductive tract. 180,236,306 It is unknown if the latter pathway also occurs in ruminants. Mechanisms of listerial invasion of the pregnant uterus are not yet fully understood. 293 Studies have commonly relied on in vitro models of placental trophoblasts, placental explants, or in vivo infection of pregnant animals (most commonly rodents). 39,182,236 These studies identified a variable role for InlA- and InlB-mediated cell entrance, depending on species-specific permissiveness, and a role for LLO and ActA in placental replication and placental-fetal spread, respectively. 39,182 Epidemiological data support InlA’s role in placental invasion of pregnant women, as clinical isolates from abortions invariably express nontruncated InlA. 149 Additionally, InlP, a recently identified Lm virulence factor, appears to be essential for placental invasion in pregnant rodents, and its deletion significantly attenuates bacterial growth in human placental organ cultures. 98 InlP interacts with the cytosolic protein Afadin, involved in cell-cell junctions, on the basal face of polarized epithelial layers, promoting bacterial transcytosis through the formation of actin-rich protrusions. 97 The role of the previously mentioned virulence factors in ruminants has not been characterized, as in vitro models of ruminant placental infection are only recently being developed. 22,249,253,254 As hypervirulent strains (CC1, CC4-CC217, CC6, 221 CC14, 277 CC59 22 ) are associated with abortions, knowledge on pathomechanisms of fetoplacental infection in ruminants might benefit from investigation of such strains in these in vitro systems.
Abortion in natural hosts
Although experimental inoculation of Lm in pregnant ruminants has been shown to be capable of causing abortion regardless of the gestational stage, 125,142 it is not known whether and how frequent early embryonic death occurs in invasive infection. 236 Abortions in ruminants typically ensue during the third trimester of pregnancy, either sporadically or as outbreaks. 125,181,306 Infection at early third trimester may cause fetal death and placental retention with minor maternal sequelae, while near term infection potentially causes serious complications for pregnant dams including dystocia, severe metritis, and septicemia. 30,180,259,306 The affected placenta shows multifocal cotyledonary necrosis and exudative intercotyledonary placentitis, while aborted fetuses are usually autolytic and manifest miliary foci of necrosis in various organs, especially in liver and spleen, and severe necrotizing enteritis. 180,259 Fetal lesions are indicative of oral infection, as they reflect the distribution observed in listerial septicemia. Perinatal listeriosis in animals typically develops within 2 weeks from birth, following in utero infection, and usually manifests with multiple necrotic foci in the liver, spleen, and other organs. 137,180,272,306 Occasionally, it can manifest as neurolisteriosis characterized by fibrinosuppurative meningoencephalitis with vasculitis, thrombosis, perivascular cuffs, and microabscesses, the latter predominating in the brainstem. 137,272 Of note, L. ivanovii can also be responsible for fetomaternal infection with a similar clinicopathological presentation in ruminants, albeit less frequently than Lm, 6,274 but has not been associated with neurolisteriosis.
In humans, maternal infection commonly presents with mild and unspecific signs of malaise, chorioamnionitis and preterm delivery, miscarriage, stillbirths, or fetal death. 185,236 Neonatal listeriosis results in septicemia and encephalitis in infants, with typical widespread multifocal granulomas (granulomatosis infantiseptica) occasionally seen in early-onset cases. 185,236
Listerial Mastitis
Mastitis caused by Lm has been reported exclusively in ruminants, ranging from subclinical chronic interstitial inflammation to severe suppurative inflammation. 27,118,284,311 These infections are thought to arise hematogenously or through local invasion via the teat canal. It is currently not known whether it also occurs in other species, and molecular mechanisms underlying mastitis and Lm interaction with the mammary epithelium remain completely unexplored. Infected animals respond poorly to treatment. In latent infections transient bacterial excretion in milk may occur for prolonged periods. 118,307 Subclinical bacterial shedding in milk has been reported in cows, ewes, and goats in both natural and experimental infections, and its association with contaminated milk products has been proposed in numerous studies as a source for human infection. 2,105,145,220,231,265,284 Hypervirulent strains belonging to CC2, CC4, and CC11, in particular, were found to be significantly associated with subclinical mastitis in dairy cattle. 220 Hypervirulent CC1 strains, however, were isolated most commonly from dairy products and were more efficient in colonizing the GI tract in infected mice. 190 Hence, further studies on mastitis- and milk-associated clones are required to clarify whether dairy products are possible relevant vectors between “farm” and “fork.”
Cutaneous and Ocular Listeriosis
Cutaneous and ocular listeriosis are rare forms and occur following direct bacterial implantation in the absence of enteric infection and bacteremia. Cutaneous listeriosis has been observed in humans exposed to infected abortive material from ruminants and presents as mostly self-limiting papulo-pustular dermatitis, cellulitis, or skin abscesses. It may also occur in immunosuppressed and elderly patients unrelated to any contact with animal sources. Moreover, Lm has been sporadically isolated from “pox-like” skin lesions in pigs and from a dog with papulo-pustular dermatitis. 125,178,194,230
Listerial keratoconjunctivitis and uveitis is rather common in ruminants (with a reported farm prevalence of up to 8.6%), 87,88 especially in cattle, as outbreaks or sporadic cases. 87,167,170 Sporadic cases have also been rarely reported in horses. 92,246 The infection most likely arises from direct conjunctival implantation of contaminated material during feeding and is strongly associated with big bale silage and ring feeding (hence the name “silage eye”). 88 Listerial keratoconjunctivitis is only rarely described in immunocompetent human patients, most frequently following exposure to farm environment, suggesting silage or infected animals as the origin for the infection. 143 Barely anything is known about pathogenetic mechanisms of listerial invasion into ocular tissues. Interestingly, a frequent involvement of the oculomotor nucleus in ruminant neurolisteriosis cases suggests that conjunctival tissue might provide a bacterial port of entry in neurolisteriosis, 209 although experimental conjunctival instillation of Lm in various animal species could only rarely produce encephalitis. 125
Neurolisteriosis
CNS infection has been reported in many species and is a significant problem in humans and domestic ruminants. Despite its low incidence, averaging 1 to 11 cases/million persons annually, listeriosis accounts for the highest fatality rate among all food acquired illnesses in humans. 60,195,275 Neurolisteriosis, which occurs in up to 79% of non-perinatal and 19% of perinatal cases, respectively, considerably contributes to mortality, being associated with fatality rates of 17% to 30% in spite of antimicrobial treatment. 29,40,60,203,275 Unfortunately, similar data for ruminant listeriosis are unavailable, as large-scale surveillance studies are currently lacking. Moreover, in contrast to humans, animal listeriosis is not included among notifiable diseases in reporting systems of many countries, which severely hampers the possibility of accurately estimating its incidence.
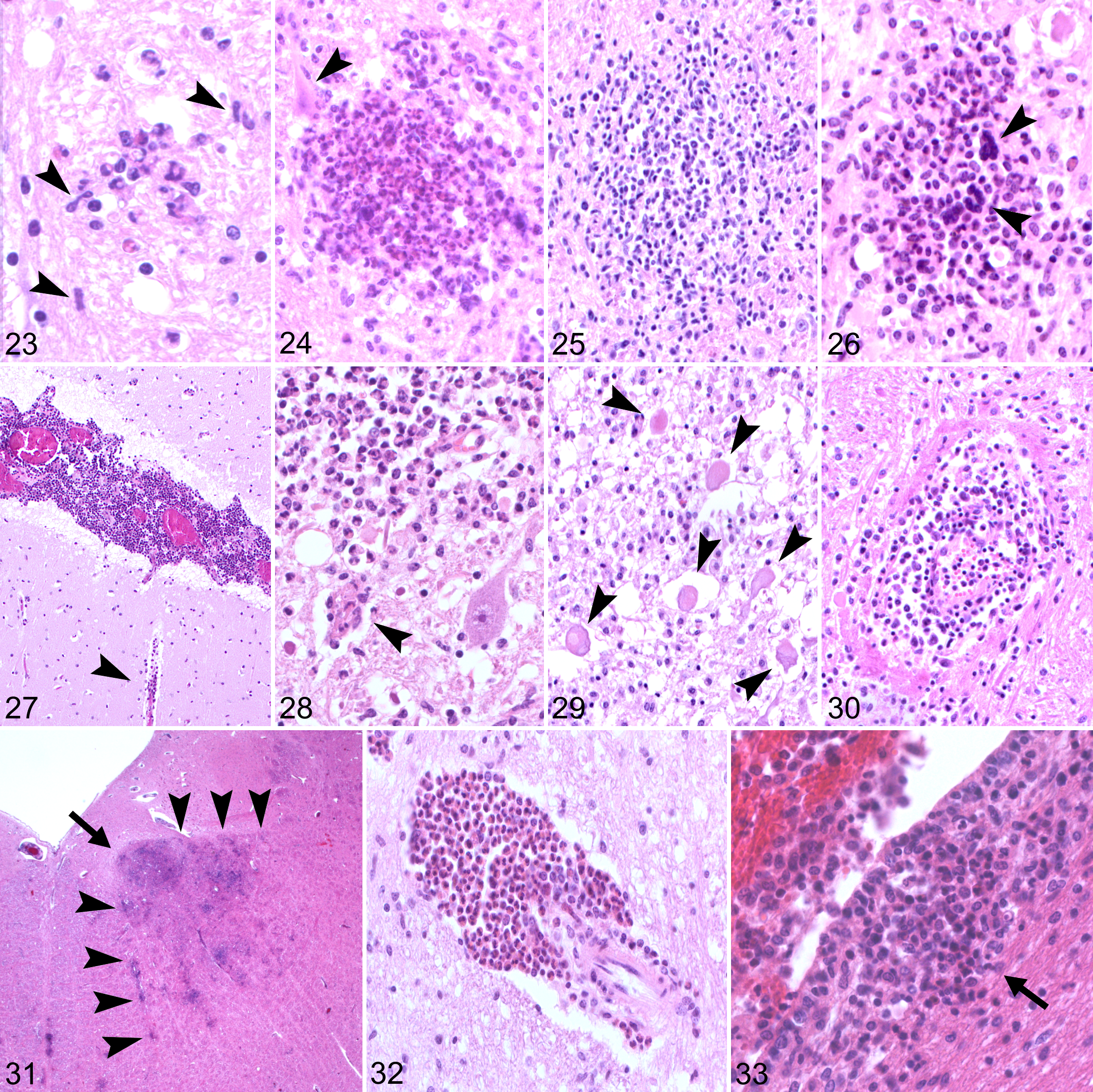
Neurolisteriosis manifests in pathologically distinct forms, which is clearly indicative of different neuroinvasive pathomechanisms among species (Figs. 6, 7). In humans and monogastric animals 3 forms can be distinguished, of which meningitis/meningoencephalitis is the most frequent manifestation (Figs. 8 –11), while brain abscessation and brainstem encephalitis (rhombencephalitis) occur less commonly. 15,125,306 By contrast, rhombencephalitis is clearly the most common phenotype in ruminants (Figs. 12 –39). 210 As the pathogenesis of the various neurolisteriosis forms is largely reflected in the pathological phenotype, the pathology will be discussed prior to the pathogenesis.
Neurolisteriosis: principal patterns of meningitis and rhombencephalitis.
Schematic topography of microabscess as investigated in 41 ruminants (cattle, goats, sheep) with neurolisteriosis (from Henke et al
139
). Brain areas are color-coded: red indicates areas affected by microabscesses in >50% of animals, orange indicates areas affected in 25% to 50% of animals, and yellow areas affected in <25% animals, respectively.
Listeriosis, brain, ruminants. Histopathological features of rhombencephalitis. Hematoxylin and eosin (HE).
Listeriosis, medulla oblongata, sheep. Lm association with lesions in rhombencephalitis.
Rhombencephalitis: Incidence and Clinical Disease
Initially described as “circling disease” in sheep, 116 rhombencephalitis occurs worldwide in farmed small ruminants and cattle, and accounts for the vast majority of invasive clinical infections caused by Lm in these species. 63,198,309 Data from numerous retrospective studies and surveys of transmissible spongiform encephalopathies (TSE) rank neurolisteriosis among the most frequent neurological diseases affecting ruminants. The prevalence among CNS diseases ranges between 8% and 35.8%, 3,115,146,174,193,208,282 and small ruminants appear to be more susceptible than cattle. The disease occurs either as single or multiple cases, including outbreaks (most frequently seen in sheep and goats), in a herd or flock. 125,126,180,198,289,309 There appears to be no particular sex-, breed-, or age-related predisposition. Some authors speculate that most cases occur concomitantly with tooth loss and eruption, 14,126,198 while other studies describe the highest prevalence of rhombencephalitis cases beyond teething. 208,209
Clinical manifestations of rhombencephalitis in ruminants
In wild, farmed, and other domesticated ruminant species, CNS infection follows the pattern of rhombencephalitis in adult animals and septicemic episodes in young animals. 50,125,137,237,306 Clinical signs of rhombencephalitis are similar in all ruminant species and commonly appear unrelated to stressful conditions or any other clinical sign. 198,210,289,309 Several studies claim a prolonged incubation period lasting between 2 and 6 weeks. However, neurological signs generally manifest acutely and progressing rapidly, more so in small ruminants than in cattle. Fever can be present during early phases of infection but is not constant. 5,30,125,180,181 Typically, rhombencephalitis signs in ruminants manifest as unilateral or, less frequently, bilateral brainstem and cranial nerve deficits. 28,30,180,198,268 Unilateral facial and tongue paralysis are common and result in ipsilateral drooping of ear, eyelid, lip, and muzzle, and hypersalivation, anorexia, and dehydration, respectively. Deficits of the oculomotor, facial, and trigeminal nerves can lead to loss of reflexes, strabismus, secondary exposure keratitis, and rarely blindness. Vestibular signs, consisting of head tilt and nystagmus, appear ipsilaterally, if the brainstem is involved, or contralaterally to the side of the lesion if cerebellar peduncles are affected. The topography of the brainstem and cranial nerve lesions (V–XII) is usually responsible for the variability in clinical manifestations (Table 1). Delayed proprioception is common, superficial sensitivity is generally reduced, and spinal reflexes can be weak or absent. Small ruminants generally display more severe signs than cattle. If able to stand, they are ataxic and can manifest circling movements (hence the name “circling disease”). In later stages of disease, they often show depression and recumbency, and death can occur within 48 hours. On the other hand, cattle show milder clinical signs (typically head tilt) and tend to succumb later to the disease. Fatality rates are high in spite of treatment, especially if not instituted early. 30,125,198,309 Rarely, myelitis without brainstem involvement has been reported in sheep, with variable clinical signs ranging from limb weakness to quadriplegia and death. 112,270 Cerebrospinal fluid (CSF) features can be quite variable, showing either mononuclear or neutrophilic pleocytosis, and Lm isolation from CSF fails in up to 90% of cases. 35,229,269
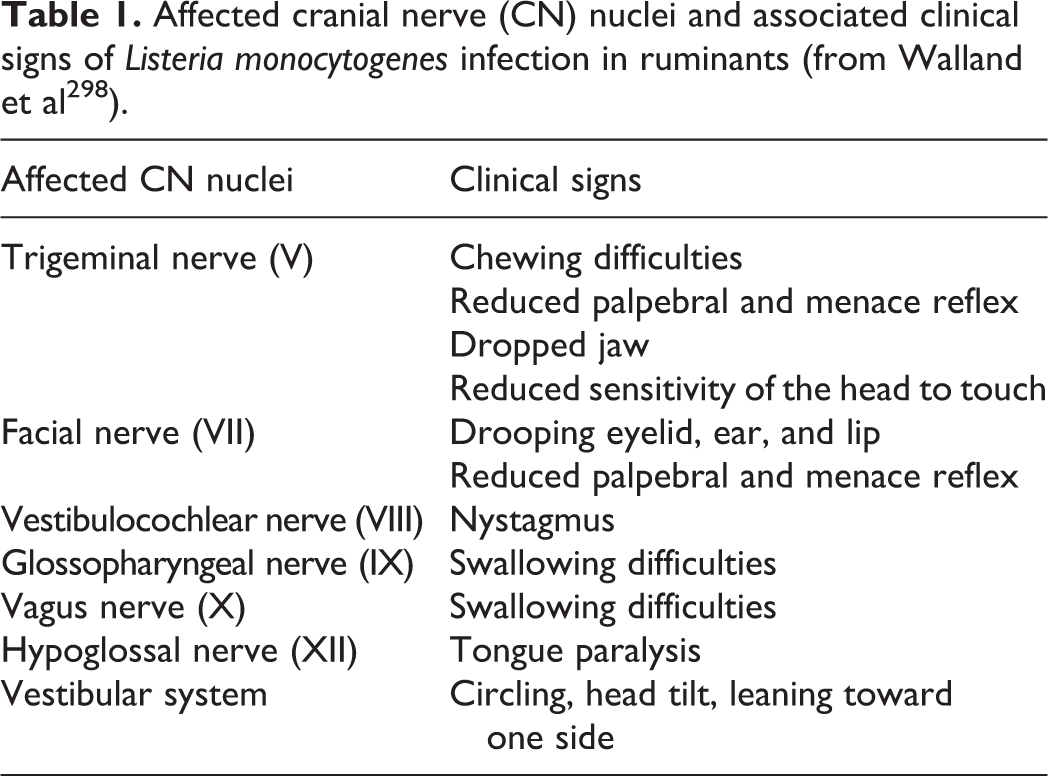
Affected cranial nerve (CN) nuclei and associated clinical signs of Listeria monocytogenes infection in ruminants (from Walland et al 298 ).
Rhombencephalitis in non-ruminant species
In non-ruminant animals, the brainstem can be targeted in meningoencephalomyelitis during septicemia, 125,306 and rhombencephalitis without generalized brain involvement has only been reported in a cat 242 and a horse. 255
Rhombencephalitis is uncommon in humans, accounting for only 1% to 24% of all neurolisteriosis manifestations. 12,15,203,285 Interestingly, several studies report that similar to ruminants and in contrast to meningitis/meningoencephalitis, the vast majority of rhombencephalitis cases occur in individuals without underlying clinical conditions. Clinical signs tend to present in a peculiar biphasic fashion, with prodromic unspecific signs including fever, malaise, headache, nausea, and vomiting lasting up to 2 weeks, followed by rapidly appearing neurological signs indicative of brainstem involvement, unilateral facial palsy being the most frequently recognized. 12,15,285 Such a biphasic pattern has not been reported in ruminant rhombencephalitis.
Neuropathology of Rhombencephalitis
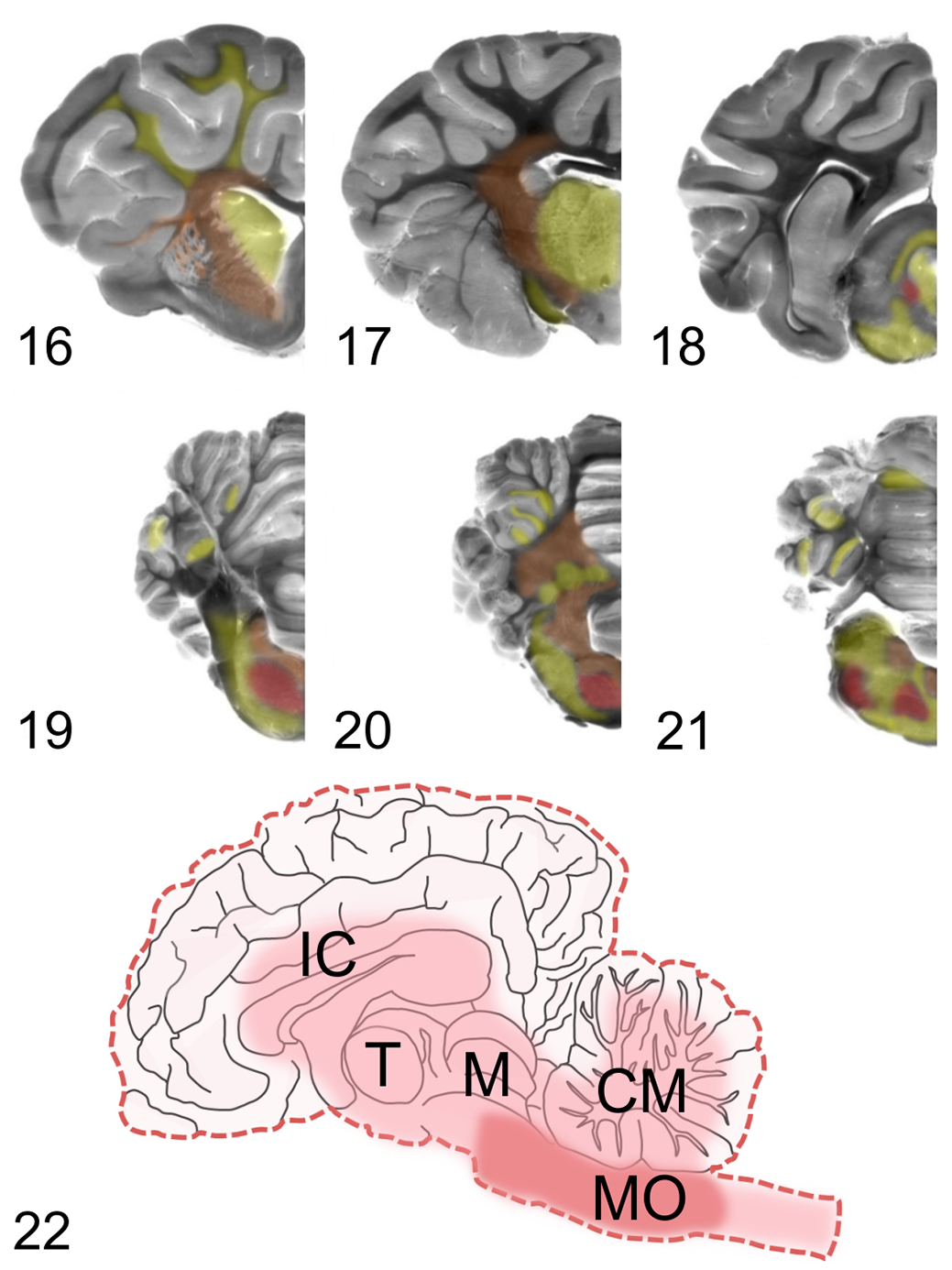
The pathology of rhombencephalitis is rather peculiar, particularly regarding the topography and nature of its inflammatory lesions. The brainstem is specifically targeted, which is quite unusual for encephalitis. Moreover, cardinal lesions consist of a combination of suppurative/granulomatous foci (so-called microabscesses) alongside mononuclear perivascular cuffs (Fig. 15), which is fairly uncommon for bacterial encephalitis, but consistent with an intracellular microorganism. Lesions in ruminant rhombencephalitis are generally quite massive. They can span from the medulla oblongata and pons to regions localized caudally (cervical spinal cord) and rostrally (cerebellum, midbrain, thalamus, exceptionally hippocampus, basal nuclei, and cerebral cortex) in the affected animal. 5,42,49,209,216 Notably, lesions appear to be continuous along the neuraxis, with generally higher severity and chronicity in the pontomedullary area than in other brain regions (Figs. 16 –22). 42,209
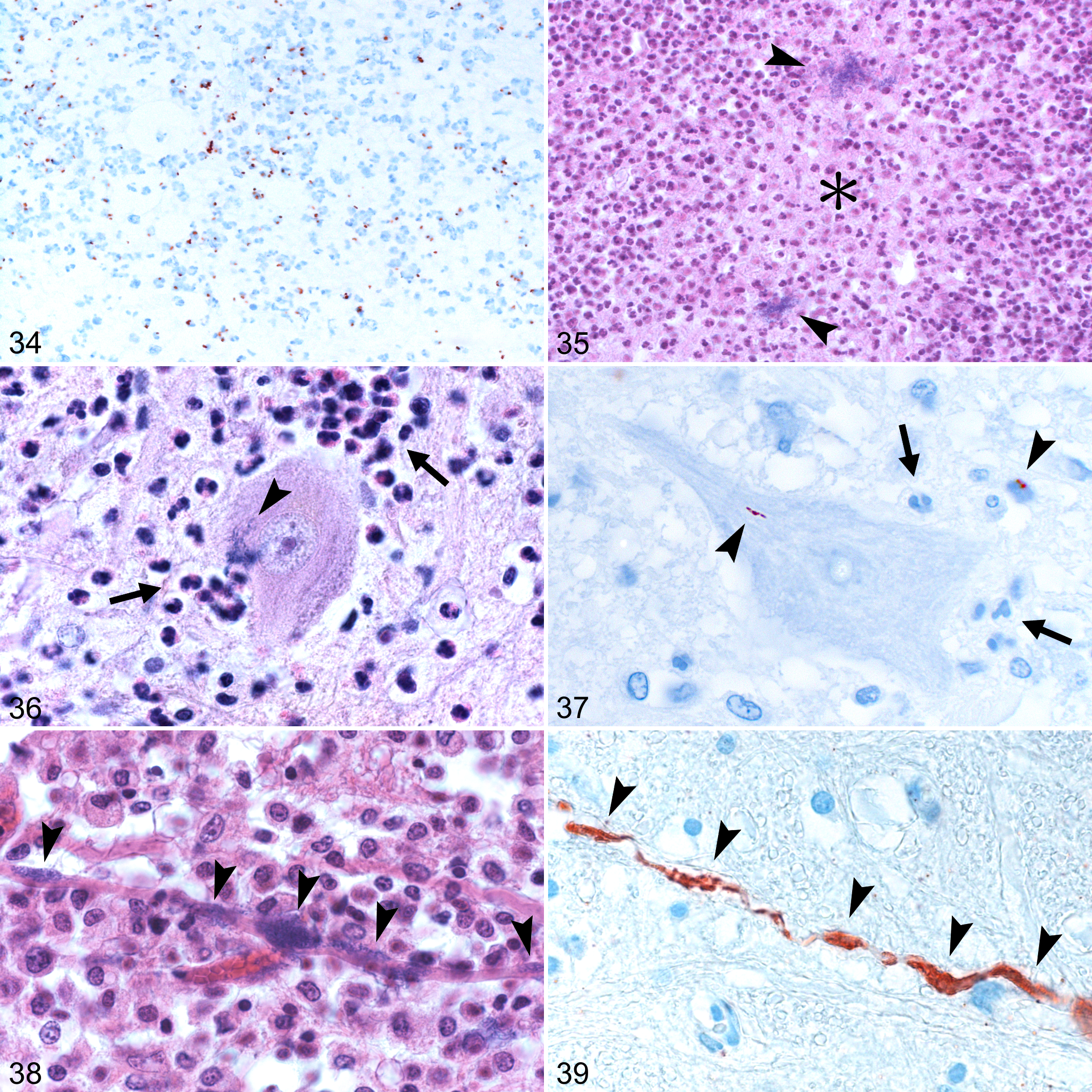
Microabscesses can range from small, delineated lesions to large coalescing areas, occurring both in gray and white matter. Frequently, they manifest a peculiar pattern in which they appear to follow the direction of axonal fibers or single cranial nerve nuclei, and rarely lesions affecting both a cranial nerve and its fibers can be observed (Fig. 31). In the brainstem, microabscesses frequently affect the trigeminal spinal, facial, hypoglossal, and oculomotor nuclei and their tracts. 139,209 The composition of microabscesses varies with chronicity: early lesions appear as small agglomerations of neutrophils and microglia (Fig. 23), progressing to almost purely neutrophilic microabscesses (Fig. 24) followed by a mixture of neutrophils and macrophages (Fig. 25), and finally by chronic granulomatous lesions where macrophages predominate and in which multinucleated giant cells may be occasionally seen (Fig. 26). 39,46 Large coalescing microabscesses may have an extensive central necrotic core (Fig. 35), appearing similar to an abscess but devoid of a fibrous capsule, and can progress to frank malacia with infiltration of numerous gitter cells. 42,49 Associated features include prominent perivascular cuffs predominantly composed of mononuclear cells, gliosis, focal edema, and vascular damage characterized by fibrinoid necrosis of the vessel wall, perivascular exudation of proteinaceous fluid, and hemorrhages (Fig. 30). 209 Axonal spheroids are frequently located in proximity to microabscesses (Fig. 29), but it is unclear whether axonal destruction occurs due to direct bacterial axonal damage or as collateral damage due to local secretion of toxic/inflammatory mediators. Neuronal necrosis is also seen in the neighborhood of microabscesses (Fig. 28), and it is unknown whether it is elicited by intraneuronal Lm, by neutrophils attacking neurons, or by some other mechanism. Meningitis is also frequently present, often as an extension of local perivascular cuffs or microabscesses (Fig. 27), but mild meningeal lymphocytic infiltrates may occur in sites distant from parenchymal lesions. 209 Moreover, cranial nerve ganglioneuritis, especially of the trigeminal nerve, can often be seen ipsilaterally to the parenchymal lesions. 4,14,42,216 Rarely, ependymitis is observed when microabscesses breach the ependymal barrier (Fig. 33). 209
Bacteria in varying numbers are generally associated with phagocytes in microabscesses (Fig. 34) but can also be seen extracellularly, occasionally forming colonies (Fig. 35) and inside neurons, axons, and neuropil, particularly during early stages (Figs. 36 –39). 139,209,216
Perivascular cuffs and microabscesses tend to be more severe in small ruminants, in which a neutrophilic component of the latter predominates (Figs. 24, 35), which is compatible with the more fulminant clinical course of the disease in these species. 209 Microabscesses containing predominantly macrophages are instead more common in cattle, in which multinucleated giant cells and perivascular eosinophils can be occasionally seen (Figs. 26, 32). 209
Despite the inflammation being severe on microscopic examination, gross pathology can be unrewarding, as macroscopic lesions are frequently absent or subtle on cut section. In a number of cases, areas of malacia, presenting as brownish discoloration with or without tissue loss, and multifocal hemorrhages, can be observed (Figs. 12, 13). Rarely, frank abscessation can occur. In contrast, severe inflammation was found to be consistently detected on magnetic resonance imaging (MRI) scans of infected small ruminants, in which the MRI lesion distribution was rather specific for neurolisteriosis. 239 However, despite the diagnostic value of MRI for in vivo diagnosis in ruminants, associated costs impede its wide use in farm animals.
The overall pattern and distribution of rhombencephalitic lesions in human patients closely resemble ruminant rhombencephalitis, indicating similar pathogenetic mechanisms in both hosts. 11,210
Pathogenesis of Rhombencephalitis
Route of invasion
The mechanisms through which Lm selectively targets the brainstem in naturally infected hosts, especially ruminants, have been the object of speculation for decades. Very little is known about the pathogenesis of rhombencephalitis, partially due to the lack of adequate experimental animal models, although several rhombencephalitis models have been proposed over the years. 5,8,7,23,154,273 Nevertheless, pathological specimens provide strong evidence that the infection route differs from septicemic listeriosis and meningitis/meningoencephalitis. Some authors have suggested a hematogenous origin for the infection, given the distribution of the inflammatory infiltrate and the association of early microglial foci with bacteria in the vicinity of parenchymal microvasculature. 49 The nature, distribution, and evolution of lesions in rhombencephalitis, however, is highly suggestive of a localized Lm centripetal intraaxonal spread from peripheral sites to the brainstem (Fig. 6). 5,11,14,43,209,216,303 Lesions are characterized by brainstem targeting with a distinct pattern of involvement of cranial nerve nuclei and their root fibers, 11,209 a feature that would not be expected from a hematogenous invasion route during septicemia, which generally causes random lesions throughout the whole brain. Additionally, dating of lesions reveals a consistent pattern with most chronic lesions in the brainstem and more acute lesions in the rostral brain, indicating that the infectious process originates in the brainstem and subsequently spreads to remote areas. Marked widespread meningitis and ependymitis, typically seen in blood-borne neurolisteriosis (Figs. 8 –11), is lacking in rhombencephalitis (Fig. 14). Moreover, cranial nerve ganglioneuritis, especially of the trigeminal nerve, frequently occurs ipsilaterally to the CNS lesion and is often more chronic, which is not suggestive of hematogenous spread. 5,14,42,216 Bacteria may be observed unilaterally in cranial nerve axons and ganglia of naturally infected animals and humans, as well as their intra-encephalic fibers and nuclei, once again suggesting localized instead of hematogenous spread. 11,42,209,216 Taken together, the absence of concomitant systemic infection, lack of generalized brain and cranial nerve involvement, and the presence of bacteria in intact axons strongly indicates that the infection does not arise hematogenously or spread peripherally to the nerves from infectious foci within the brain. 5,14,42,216 Experimental modeling of rhombencephalitis in animals has been attempted through various routes, 210 but could only be replicated via feeding of bacteria-soaked abrasive foodstuffs or bacterial injection in the lip, conjunctival sac, dental pulp, or snout in small ruminants and mice, indicating that Lm can access nerve endings after breaching of mucocutaneous barriers. 5,10,14,154 Additionally, mice injected with Lm in the triceps surae, or sciatic nerve developed ascending unilateral myelitis unless nerve resection was performed, further indicating that bacteria can efficiently spread to the CNS via intra-axonal migration. 10,154
Mechanisms of Lm entrance into nerves
Although it is rather clear that bacteria can access the brainstem via nerve fibers, it remains uncertain how Lm enters the nerve fibers themselves and whether invasion of the neuraxonal compartment involves direct interaction with neuronal receptors or receptor-independent cell-to-cell spread. While intraneuronal and intra-axonal bacteria can be observed in natural cases, 41,42,139,184,209 Lm does not appear to be particularly neurotropic in vitro. Infection of ruminant and rodent dissociated brain cell cultures and organotypic brain slices indicates that neurons are not heavily targeted in contrast to other cells (notably, microglia) and are variably infected depending on their anatomical origin and culturing methods. 69,71,133,227,228 An InlA-dependent mechanism of invasion of cranial nerves has been proposed in ruminants, in which Lm may initially invade E-cadherin expressing oral epithelium or myelinating Schwann cells and subsequently spread to neighboring axons. 184 Phagocytes have also been hypothesized to provide a source for axonal infection by ActA-dependent bacterial cell-to-cell spread. 71 Moreover, experimental infections in mice suggested a role for PlcB-dependent spread from peripheral macrophages to the trigeminal nerve. 153,154 Whether the mechanisms mentioned above are also relevant for naturally occurring infection in natural hosts is yet unknown.
Mechanisms of intra-axonal and intra-encephalic spread
Actin polymerizing bacteria have been observed inside axons of pathological specimens and ActA-dependent intraneuronal migration has been reported to occur in cultured bovine neuronal cells, indicating that actin-based motility plays a role in Lm intra-axonal spread. 139,216 This is further supported by the observation of severely impaired neuroinvasion of actA-deletion mutants in the murine intranasal infection model. 218 This study also showed that LLO and, partially, ActA are required for efficient breaching of the olfactory epithelium prior to the neuronal invasion, indicating their possible role in peripheral barrier breakdown, brain invasion, and spread.
Experimental and pathological evidence suggests that, after entry into the brain, Lm efficiently disseminates within the brain. Topographical distribution of microabscesses in naturally infected ruminants suggests that intra-encephalic bacteria spread between anatomically connected brain structures via white matter fiber tracts. 139 Intra-axonal migration is further supported by bacterial association with axons in naturally infected cases and ruminant organotypic brain slices. 133,209 ActA-mediated actin polymerization has been observed in neurons in vitro and in situ, 139 suggesting its participation in intra-encephalic spread, but its role in vivo has only been partially explored. 218 PlcB-mediated cell-to-cell spread has also been implicated in intra-encephalic spread, as mice intracerebrally infected with a plcB-deletion mutant showed prolonged survival, lower bacterial load, and delayed intra-encephalic spread in comparison to a inlA/B-deletion mutant that retained a virulence comparable to the wild-type. 263 These findings indicate that PlcB is relevant for neurovirulence in vivo, while InlA and InlB do not play a role during the intra-encephalic stage.
Phagocytes as local bacterial amplifiers?
Additional factors involved in intra-encephalic Lm dissemination are currently unknown, but a role for phagocytes has been proposed based on observations in natural infections. 139 Neutrophils and, less frequently, macrophages were shown to access the axonal space and phagocytose intra-axonal bacteria, while a high bacterial load was found inside adaxonal microabscesses, as previously reported in other studies. 62,139,209 It was therefore suggested that Lm could replicate locally in microabscess-associated phagocytes and subsequently reenter the neuraxonal compartment, further propagating the infection. This view is in line with studies in mice and ruminants showing that the innate immune response is inefficient in providing sterilizing immunity in listeriosis, and resolution of infection appears to be dependent on the adaptive immune response. 18,62,168,257 The ability of Lm to survive inside phagocytic cells further supports this view, 57,158 but the protective role of the immune response or its impact in ruminant neurolisteriosis remain severely underinvestigated in comparison to murine experimental models in which the immune responses toward Lm have been extensively dissected. 53,189,219 Overall, additional studies are required to unravel the role of phagocytes in bacterial persistence and potential intra-encephalic spread.
Bacterial strains in rhombencephalitis
Further relevant yet unclear aspects of ruminant neurolisteriosis are the bacterial dose required for establishing infection and potential predisposing factors. 210 Despite common bacterial exposure, as indicated by widespread/high prevalence of LLO antibodies in the bovine population, 24 and frequent asymptomatic fecal shedding, 89,204,286 only a limited number of animals develop brain disease. It has been suggested that in such cases immune responses mounted against orally acquired bacteria are inefficient in preventing neuroinvasion. 180 Inefficiency of preexisting immunity might be partially dependent on the bacterial strain, but its involvement in neuroinvasion in ruminants remains unknown. Interestingly, immunization with a homologous strain induced a protective brain response in mice, 264 while outbreak-isolated strains displayed neuroinvasiveness regardless of preexisting immunity toward a reference strain. 114 The high prevalence of hypervirulent and hyperinvasive CCs, particularly CC1, in ruminant rhombencephalitis cases could be in line with such observations. 13,79,132,221,251 Although genomic comparisons identified putative CC-specific neurovirulence factors, 4,13 their role in hypervirulence was not confirmed. 122,254 Thus, strain-related Lm neurovirulence factors in ruminants require further exploration.
Brain Abscessation
Rarely, Lm can cause single or multiple cerebral and cerebellar abscesses in human patients, especially if immunocompromised. 1,46,48,61,84,203 However, they are not reported to occur with a similar pattern in ruminants. The distribution of brain abscesses strongly indicates hematogenous entry of Lm into the brain. However, abscesses in the deep white matter may be aligned along connected white matter fiber tracts suggesting that following hematogenous invasion Lm may enter and spread within axons from original foci of infection. 25,144 This pattern parallels that seen in rhombencephalitis cases in ruminants, 139 indicating that, no matter how Lm enters the brain, it can spread within the brain along axonal pathways and that similar mechanisms of intra-encephalic Lm spread are shared between rhombencephalitis and brain abscesses.
Meningitis/Meningoencephalitis: Clinical Disease and Pathology
The most frequent neurolisteriosis manifestation in humans is meningitis/meningoencephalitis, which predominantly occurs in predisposed individuals (especially elderly and immunosuppressed patients) and is the consequence of hematogenous spread to meninges and brain during bacteremia. 29,40,203,275 Clinical signs appear between 1 and 14 days following contaminated food consumption 9,121 and variably include fever, headache, neck stiffness, altered sensation, seizures, and focal neurological signs. 29,40,203 Severe long-term neurologic impairment persists after recovery and treatment in up to 60% of surviving patients, which can manifest altered consciousness, sensorimotor dysfunctions, or rarely cranial nerve palsies. 29,40,61,203 Bacterial isolation from hematologic and CSF samples yields positive results in about 60% and 40% of cases, respectively, which is in contrast to the low rate of bacterial isolation in rhombencephalitis. 29,40,203
Lesions mainly consist of mild to severe multifocal or diffuse meningeal inflammation with focal cortical infiltration, diffuse ventriculitis with ependymal erosion and focal periventricular infiltration, meningeal and parenchymal vasculitis, thrombosis, and small parenchymal perivascular abscesses. 86 The inflammatory infiltrate is mostly composed of monocytes/macrophages and neutrophils, with frequent efferocytosis of phagocytes operated by macrophages, while bacteria can be seen intra- and extracellularly in the meninges, parenchymal abscesses, and ependyma. 86 Neurolisteriosis in monogastric mammals and birds is rarely encountered and, like in humans, prevalently manifests as meningoencephalomyelitis in the context of septicemia (Figs. 8 –11), especially in juvenile animals or in association with concurrent predisposing conditions in adults. 51,125,242,306 On the other hand, meningoencephalitis is extremely rare in ruminants and is usually observed during perinatal septicemia, in which patterns of hematogenous neurolisteriosis and rhombencephalitis are concurrently featured, 137,272 possibly suggesting simultaneous hematogenous and ascending brain invasion.
Pathogenesis of Meningitis/Meningoencephalitis
Little is known about the pathomechanisms involved in blood-borne neurolisteriosis in naturally susceptible hosts and studies on Lm brain infection in monogastric animals other than rodents are virtually nonexistent. Surveys on human neurolisteriosis are rare, 8,29,217,223 generally include small patient cohorts and bacterial typing has been rarely performed. 161,175 Therefore, neuroinvasive strains remain poorly characterized.
Bacterial strains in meningitis/meningoencephalitis
Lineage I hypervirulent clonal complexes (CC1, CC2, CC4, CC6) were shown to be significantly associated with fetomaternal and, notably, CNS infection, possibly indicating their proclivity toward efficient breaching of placental and neural barriers. 160,192 Experimental in vivo infections correlated the novel LIPI-4 gene-cluster (encoding a putative cellobiose-family phosphotransferase system) in CC4 strains to such enhanced invasiveness, but underlying mechanisms are yet unknown. 192 Interestingly, other frequently isolated neurovirulent CCs (eg, CC1) do not possess LIPI-4, indicating that factors specifically involved in neuroinvasiveness remain to be discovered or might differ among hypervirulent strains. Emerging CC6 strains were increasingly found in cases of meningitis with poorer prognosis, 160,161 possibly due to a plasmid conferring resistance to antibiotics and disinfectants. 166 However, they were less neurovirulent than CC1 strains in mice infected intracisternally and their neuroinvasive mechanisms were not assessed. 162 Thus, further studies on listerial meningitis isolates are needed to characterize strain-specific factors involved in neuroinvasiveness and neurovirulence.
Route of invasion in meningitis/meningoencephalitis
Most of the knowledge concerning mechanisms of Lm brain invasion was derived from mouse infection models, in which artificial routes of inoculation (intravenous, intracerebral, intracisternal) are often used to bypass the GI phase. 16,108,155,181,244,252 Despite proving useful for circumventing the species-specific barrier posed by the lack of affinity of InlA for murine E-cadherin, 172 such approaches poorly translate to infection dynamics observed in natural hosts. Despite these caveats, pathological and experimental findings indicate that CNS invasion in blood-borne listeriosis is achieved through breaching of the blood-brain barrier (BBB) or blood-cerebrospinal fluid barrier (B-CSFB; Fig. 7). Direct infection of brain endothelium or choroid plexus epithelial cells from bloodstream bacteria has been proposed as a likely route of hematogenous brain invasion following bacteremia. 65,77 A second mechanism for Lm CNS entry is the so-called “Trojan horse” model, in which bacteria are transported to the brain as cargo in infected circulating phagocytes, efficiently sheltered from extracellular factors. 65,73,77
Direct invasion of the BBB/B-CSFB by extracellular bacteria
Direct bacterial invasion of brain endothelium is supported by clinical and pathological findings. Lm is frequently isolated from the blood of patients with meningitis, 29,40,203 and bacteria have been identified in the cytosol and adhering to the vascular side of cerebral endothelial cells in meningoencephalitis cases. 158 Moreover, bacteremia appears to be a requirement in murine experimental brain invasion, especially following sublethal inocula, in which bacteria are initially cleared from the bloodstream, proliferate in the liver, and then undergo a subsequent secondary wave of hematogenous spread following proliferation in liver and spleen. This can provide means for bacterial invasion of brain endothelium and choroid plexus epithelial cells, with subsequent translocation across the BBB/B-CSFB. 16,72,156 Experimental findings also indicate that Lm is capable of efficiently infecting human brain microvascular endothelial cells (HBMECs) in vitro. 127,128 Mechanisms responsible for such invasion are still controversial, as some authors speculate that an InlB-dependent internalization is required, 127,128 while others could not find any evidence for an InlA or InlB role in bacterial entrance into endothelial cells. 310 Recently, InlF has been shown to be involved in the invasion of various cells (including a human brain endothelial cell line) by interacting with surface-expressed vimentin and, concurrently, InlF deletion mutants were deficient in their ability to colonize the brain of intravenously infected mice. 113 Mechanisms underlying this interaction, however, remain unknown. As an alternative route to BBB invasion, Lm has also been proposed to be capable of invading the B-CSFB in vivo, 238 a process that was shown to require both InlA and InlB in vitro. 130 Another bacterial surface protein, the autolysin IspC expressed by serotype 4b strains, has been shown to promote bacterial attachment and invasion of choroid plexus epithelial cells and is involved in virulence factor regulation, positively contributing to brain invasion in intravenously infected mice. 300
In addition to the previously mentioned ones, other Lm virulence factors can enhance brain invasion in the context of hematogenous infection. However, their precise mechanisms and interaction underlying brain invasion and their relevance in CNS infection of naturally susceptible hosts remain unexplored. The bacterial surface protein Vip, expressed in lineage I and II strains, has been shown to bind host cell gp96 and appears to be relevant for cell and brain invasion. 34 A gene encoding a putative leucine-rich-repeat-containing protein (LMOh7858_0369) from a serotype 4b strain was also found to be relevant for systemic and brain invasion in an intravascular sepsis mouse model. 315
In spite of such observations, the importance of free bacteria in the bloodstream in establishing cerebral invasion has been called into question in the context of natural infection, as antibodies present in normal adult human serum strongly inhibit HBMECs invasion. 140 Moreover, mice systemically infused with gentamicin (an antibiotic that poorly diffuses intracellularly but rapidly inhibits or kills extracellular bacteria) show a bacterial brain burden similar to untreated mice, indicating that most bacteria that enter the brain are intracellular. 76,188 Thus, additional mechanisms involving infected phagocytes sheltering and transporting intracellular Lm to the brain have been speculated to play a pivotal role in hematogenous neurolisteriosis.
Invasion via infected phagocytes (“Trojan horse” model)
The “Trojan horse” model of listerial brain invasion claims that phagocytes containing intracellular bacteria enter the bloodstream from peripheral sites of infection and carry intracellular Lm to or across the BBB and B-CSFB. 77 Numerous in vivo studies have shown that peripherally infected monocytes can allow Lm survival following phagocytosis and greatly enhance bacterial brain invasion. 72,75,156,188 It is currently unclear whether Lm invades the BBB/B-CSFB by spreading to the endothelium from luminally adhering phagocytes or whether bacterial escape follows phagocyte transmigration across brain barriers. Nevertheless, cell-to-cell spread appears to be essential for bacterial dissemination through this infection route. Macrophages in human neurolisteriosis cases can host cytosolic Lm displaying actin polymerization, 158 indicating that bacteria have the potential to undergo intercellular spread following phagocytosis. Similar findings have also been reported in mice, in which Lm was shown to polymerize actin inside monocytes adhering to brain endothelial vessels. 188 In vitro studies, in which bacteria were able to efficiently spread from infected macrophages to HBMECs and rat spinal neurons in a PlcB- and ActA-dependent manner, respectively, further support the idea that intracellular Lm can spread from infected phagocytes to cells of both vascular and neural compartments. 71,128
It is not precisely known how infected phagocytes are targeted to the brain. Peripheral infection with Lm has been proposed to enhance monocyte recruitment to the brain even before neuroinvasion occurs. Indeed, proinflammatory cytokines released in the plasma (especially IFN-γ) were shown to activate monocyte-recruiting inflammatory pathways in the brain prior to detectable cerebral infection. 74,78 Subcutaneous infection in mice also resulted in upregulation of adhesion molecules (ICAM-1, P-selectin) in cerebral endothelium days before brain infection was detected, 179 but it is not known whether the process is involved in enhanced influx of infected monocytes and adhesion to the cerebral vasculature. Moreover, it remains unknown whether bacterial spread from monocytes to brain endothelial cells could enhance the recruitment process. Lm invasion of HBMECs has been shown to induce upregulation of adhesion molecules (E-selectin, ICAM-1) and chemoattractant cytokines (IL-8, IL-15), 299,310 but it is unknown whether this process also occurs in vivo. Last, a significant upregulation of MCP-1 was shown to occur in infected mice’s brains concomitantly with influx of infected monocytes, 75 but it is unclear whether MCP-1 initiates monocyte recruitment. An explanation for monocyte predisposition in carrying intracellular Lm to the brain was recently proposed. 188 Lm may prolong the intravascular survival of infected monocytes via InlB-mediated selective block of CD8+ T-cell mediated killing, thereby enhancing the chances of bacterial transmission to the brain. It is interesting to note that mononuclear phagocytes appear to play a central role in brain invasion independently of the neurolisteriosis form, despite being considered to be at least partially bactericidal. 57 Further studies focusing on their interaction with Lm in the context of neuroinfection, therefore, will prove crucial in establishing their definitive role in neuroinvasion and, possibly, unravel new mechanisms of listerial intraphagocytic persistence and cerebral spread.
Conclusion and Outlook
A clearer understanding of the pathogenesis of listeriosis and its molecular epidemiology at the interface between environment, ruminants, and humans will foster the development of efficient surveillance and control measures to prevent disease in ruminants and transmission to humans according to farm-to-fork and One Health concepts. However, despite the high death toll of listeriosis, the molecular pathogenesis of infection remains only partially known. This is particularly true for neurolisteriosis, the most devastating and pathomechanistically complex form of the disease. The striking versatility of Lm is reflected in its ability to swiftly switch from a saprophyte to an opportunistic pathogen that is able to survive in a wide range of host cells including members of the immune system and to reach the brain through different pathways. The investigation of mechanisms and factors involved in the various neuroinvasion routes and in intra-encephalic spread will contribute to the identification of new targets for neurolisteriosis therapy in affected species. In this context, ruminants might provide a valuable, yet so far underappreciated, animal model for the study of human infection.
Footnotes
Acknowledgements
We would like to thank all current and former lab members for their work and contribution to the understanding of ruminant neurolisteriosis. We also thank Dr Sohvi Blatter (Institute of Animal Pathology, Vetsuisse Faculty, University of Bern, Bern, Switzerland) for providing us with the case reported in Figures 8
to ![]() .
.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The writing of this review was supported by funding of the Charlotte und Ernst Frauchiger-Reyher-Stiftung.
