Abstract
Tuberculosis (TB) develops in 5% to 10% of people infected with Mycobacterium tuberculosis (M.tb), but we do not understand how TB develops. CBA/J mice may model these events, as sick mice share features with TB patients, including weight loss, M.tb growth, extensive granulomatous infiltrates, neutrophils, necrosis, and fibrosis. Here, M.tb-infected CBA/J mice were categorized clinically: those with no signs or those with 10% weight loss to determine whether clinical state was associated with lung lesions. The type and distribution of infiltrates (granulomatous with lymphoid aggregates and scattered neutrophils) were similar in mice with weight loss and in mice with no signs. The amount of infiltration and neutrophil foci were higher in mice with weight loss than in mice with no clinical signs. Necrosis and fibrosis were only identified in mice that lost weight. Our results suggest that CBA/J mice may be useful to determine if and how neutrophils contribute to TB disease progression in mouse models.
Intact TH1 immunity is essential for resistance to Mycobacterium tuberculosis (M.tb) in animal models and in humans. 9,16,18,26,27,38 However, pulmonary tuberculosis (TB), which is the most common form of TB and is responsible for M.tb transmission, does not appear to be due to severe immune defects. 22 How pulmonary TB develops is not fully understood but is important to investigate. 11 Some contributing mechanisms have been identified such as the sst1 locus, 34 the matrix metalloproteinase-1, 13 interleukin-10, 2 and neutrophils or neutrophil-like cells. 24,30,33 We use CBA/J mice to model pulmonary TB because these mice have no known immunological defects, are relatively susceptible to M.tb infection, and maintain long-term stable M.tb burdens. 2,3,36 Yet, the manifestation of TB disease in CBA/J mice has not been completely described. Here, we show microscopic features of TB disease in CBA/J mice and in M.tb-infected CBA/J mice that have no clinical signs. This may provide another method to investigate the transition between controlled M.tb infection and TB disease.
Specific-pathogen-free, 6- to 8-week-old, female CBA/J mice from Charles River Laboratories (Wilmington, MA) were maintained in ventilated cages within biosafety level 3 (BSL3) facilities at The Ohio State University (Columbus, OH) and provided with sterile food and water ad libitum. All protocols were approved by The Ohio State University’s Institutional Laboratory Animal Care and Use Committee (IACUC). Mice were infected with M.tb by aerosol exposure and weighed weekly throughout M.tb infection, as previously described. 3 Mice were euthanized when all of the following IACUC removal criteria were met: weight loss of 20%, unthrifty hair coat, social isolation, and tachypnea or dyspnea. In some experiments, mice were euthanized when 10% of their peak body weight was lost and there were no other clinical signs. The M.tb pulmonary burden was calculated by colony-forming units as previously described 3 or lungs were fixed in 10% neutral buffered formalin for hematoxylin and eosin (HE), Masson’s trichrome, Von Kossa, or modified Ziehl-Neelsen acid-fast staining. Slides were evaluated by a board-certified veterinary pathologist (G. Beamer) and scanned at 40× using an Aperio ScanScope (Aperio, Vista, CA). Digital images were captured and analyzed using an Aperio ImageScope. For morphometry, 4 sections 100 μm apart from 2 lung lobes per mouse were analyzed. The percentage of lung infiltration (“granuloma fraction”) was calculated as previously described. 32 Foci of neutrophils containing 5 or more multilobulated nuclei associated with foamy macrophages were identified at 20× magnification and counted. Foci of lymphocytes containing 5 or more cells were identified at 10× or 20× magnification and counted. Statistical analyses were performed using 1-way analysis of variance (ANOVA) and Tukey’s posttest or the Student’s t-test (Prism 6; GraphPad Software, San Diego, CA). Statistical significance was defined as *P < .05, **P < .01, and ***P < .001.
Features of Pulmonary TB Disease in CBA/J Mice
Weight loss is an established feature of TB disease in other mouse strains. 6,20,30 Here, CBA/J mice infected with a low-dose aerosol exposure of M.tb were clinically monitored weekly and euthanized when all of the IACUC removal criteria of weight loss, unthrifty hair coat, social isolation, and tachypnea and/or dyspnea were met. Mice euthanized due to removal criteria weighed 19.57% less than the lower standard deviation [SD] of M.tb-infected mice that had no signs of disease. Weight loss occurred over a period of 38 ± 16.5 days. M.tb-infected mice that had no signs of disease gained weight at the same rate as noninfected, age- and sex-matched mice housed under identical BSL3 conditions 3 and unpublished data. The lungs of sick mice were consolidated (Fig. 1) or contained necrosis (Fig. 2) and harbored significantly more M.tb bacilli (Fig. 3) as compared with mice that had no signs of disease and gained weight during the same infection period. Two main patterns were observed in the lungs of all 11 sick mice evaluated: coalescing sheets of epithelioid and foamy macrophages (Figs. 4, 5), lymphoid aggregates (Figs. 4, 6), rare neutrophils (not shown), and occasional multinucleated giant cells (not shown). This pattern has been described as pneumonic 22 to distinguish the diffuse granulomatous alveolar infiltrate from discrete granulomas. The pneumonic distribution is common among M.tb-infected mice. 1,8,21,34 Necrosis, neutrophils, and bronchiolar exudate characterized the second pattern. Necrotic foci contained basophilic nuclear debris (not shown) or lightly eosinophilic macrophage “ghosts” consistent with caseation necrosis 25,29 (Fig. 7). Necrosis has been shown in other immune-competent mice (C3H/HeB/FeJ, 34 B6D2F1 hybrids 31,32 ) and in immune-deficient mice. 10 This contrasts with C57BL/6 or BALB/c mice, which develop progressive nonnecrotic granulomatous infiltrates and alveolar fibrosis. 5,17,35,37 Neutrophils were abundant at the periphery of necrotic regions (Fig. 8) and in the centers of small foci of foamy macrophages (Fig. 9). Bronchioles with intraluminal necrotic cellular debris, macrophages, neutrophils, and mucus were also identified in sick CBA/J mice (not shown), similar to other mice with TB disease. 32,35 Very rare bronchiolar epithelial erosion was observed (not shown). Fibrotic, cell-poor granulomas associated with healed primary M.tb infection in humans 25 were absent in CBA/J mice, and that absence is consistent across mouse strains. 1,8,21,34
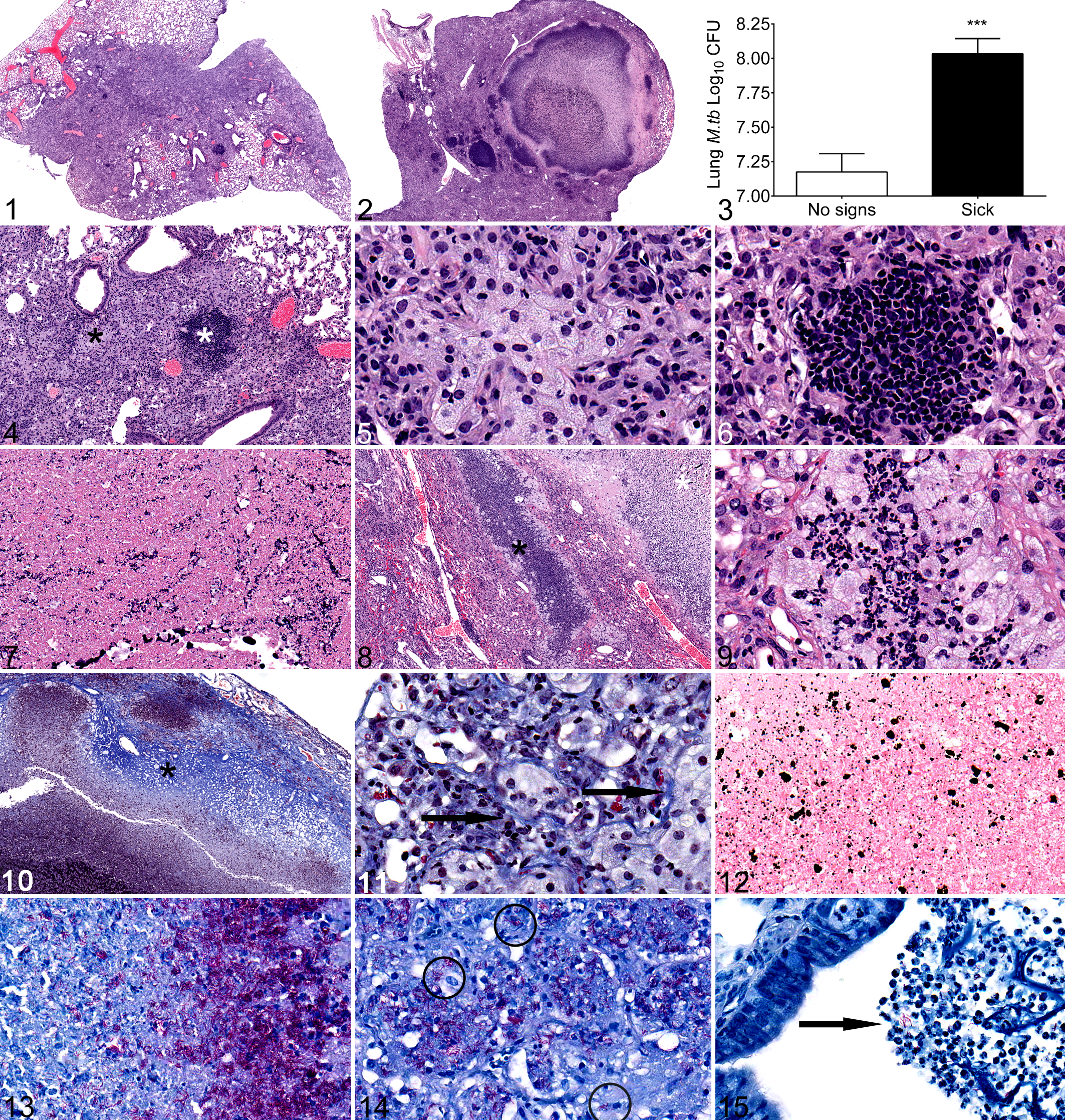
Collagen, Mineral, and M.tb Bacilli in CBA/J Lungs With TB Disease
In noninfected lung, collagen was present within the peribronchiolar and perivascular adventitia and absent from the alveolar septae as expected (not shown). In CBA/J mice euthanized due to IACUC removal criteria, collagen formed at the periphery of necrotic foci (Fig. 10), similar to mice genetically prone to M.tb-induced pulmonary necrosis 34 and to the necrotic granulomas described in humans. 25 Collagen in alveolar septae (black arrows, Fig. 11) was not associated with necrosis and has been attributed to chronicity of M.tb-infection. 8,12,32,35 Mineralized foci formed in necrotic regions (Fig. 12) but not in noninfected lung (not shown) or in pneumonic granulomatous infiltrates (not shown). As expected, acid-fast M.tb bacilli were abundant in sick CBA/J mice. M.tb bacilli were extracellular within necrotic foci (Fig. 13), intracellular within macrophages (Fig. 14), and in airway lumens (Fig. 15).
Association Between Clinical State and Lung Lesions in M.tb-Infected CBA/J Mice
Describing the features of pulmonary TB in mice that met IACUC removal criteria was useful because it has not been previously performed, and the features are similar to other M.tb-susceptible mice. 34 However, evaluation of end-stage TB in sick mice does not necessarily provide insight into disease pathogenesis or progression. To begin to understand the progression from controlled infection to pulmonary TB, M.tb-infected CBA/J mice were clinically monitored. Body weight was used because weight loss was the first clinical indicator of TB disease in mice, and it manifest well before the other IACUC removal criteria (unthrifty hair coat, social isolation, tachypnea and/or dyspnea). Weighing mice was also minimally invasive and required no specialized equipment. Here, mice were euthanized when 10% weight loss was reached; no other signs of disease were observed at this level of weight loss during M.tb infection. Age- and sex-matched noninfected mice maintained in the same BSL3 housing conditions were controls. Cohorts of noninfected mice and M.tb-infected mice that had no clinical signs were euthanized on the same day. Weight charts over time for individual mice are shown in Fig. 16 (noninfected), Fig. 17 (10% weight loss), and Fig. 18 (no signs). Noninfected mice and M.tb-infected mice with no clinical signs gained weight at the same rate (not shown) and body weights at the time of euthanasia were not significantly different (not shown). Thus, weight gain in M.tb-infected mice may be a surrogate marker of controlled infection.
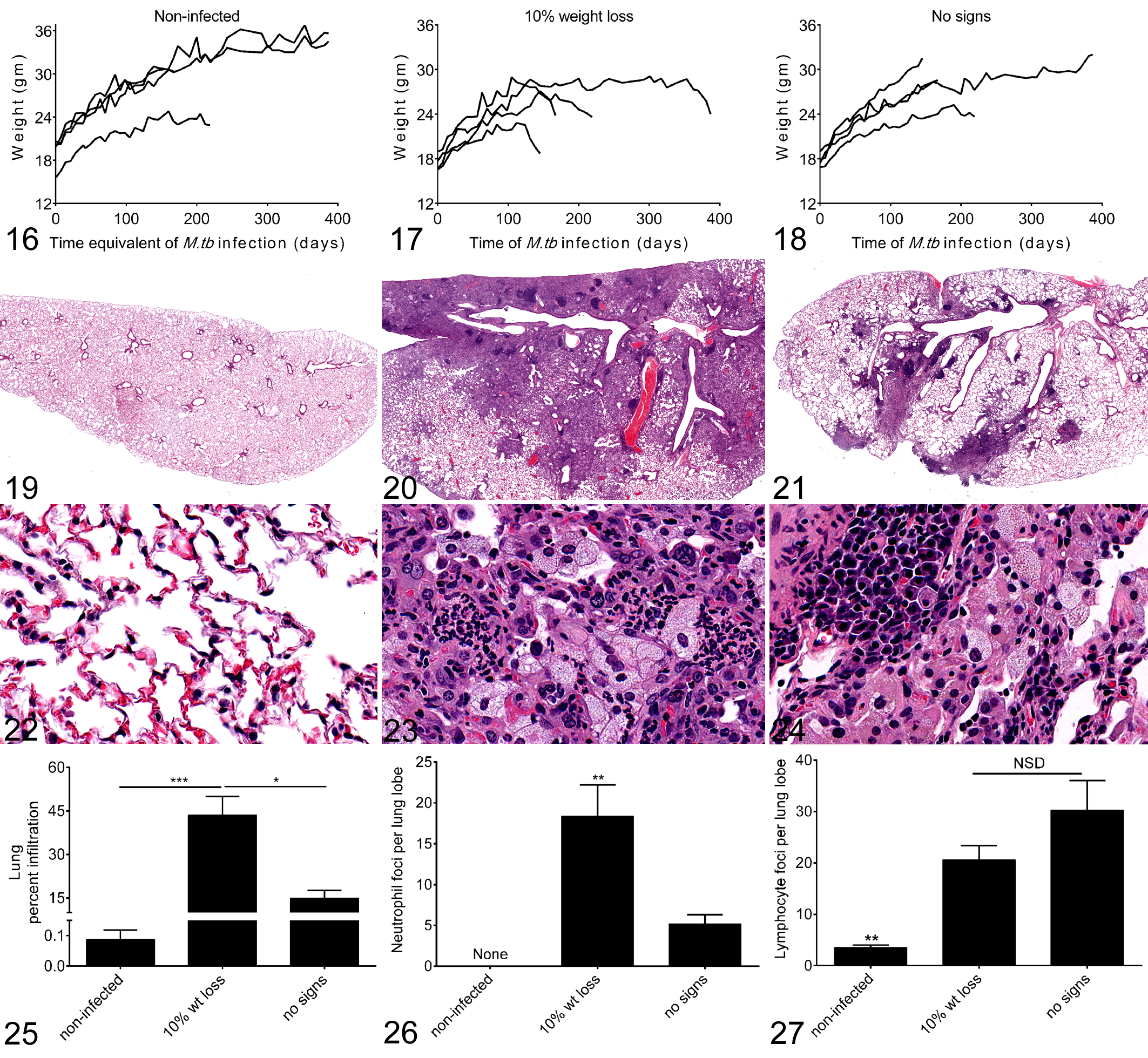
We asked whether there were differences in the lung lesions of M.tb-infected mice with 10% weight loss and M.tb-infected mice that had no clinical signs. Lung infiltration by pneumonic granulomatous inflammation was significantly larger in mice with 10% weight loss as compared with mice with no clinical signs (Fig. 25). The histomorphological analysis captured bronchiole-associated lymphoid tissue (BALT), not granulomatous inflammation, in the lungs of noninfected mice that were normal by light microscopy (Figs. 19, 20). Total lung areas for the 3 groups were equivalent (not shown). The lungs from mice with 10% weight loss contained significantly more neutrophil foci associated with foamy macrophages than the lungs from mice with no clinical signs (Figs. 23, 26). Lymphocytic foci were prominent in the lungs of mice with no clinical signs (Fig. 24), but the difference was not statistically significant as compared with mice with 10% weight loss (Fig. 27). Only 1 mouse with 10% weight loss had a single necrotic granuloma (not shown). In contrast, no necrosis was present in M.tb-infected mice with no clinical signs. Together these results suggest that neutrophils infiltrate small foci of macrophages and may contribute to TB disease in CBA/J mice.
Overall, pulmonary TB in CBA/J mice shares some similarities to pulmonary TB in humans 23 and to other M.tb-susceptible mice 34 : extensive granulomatous infiltration, necrosis, fibrosis, and bronchiolar intraluminal exudate. Pulmonary cavity formation, which is an important feature of pulmonary TB in humans, 19,25 was not consistent in CBA/J mice with TB, perhaps due to insufficient lung mass to support cavitation. Here, by assigning mice to clinical categories, our results indicate that 10% weight loss may be a surrogate marker for TB disease in mice. This level is reliably recognized and occurs before other signs of disease develop. Weight gain at the same rate as noninfected controls in M.tb-infected mice may be a marker of controlled M.tb infection. Histopathological comparison of mice that lost weight and those without signs demonstrates that neutrophils may contribute to, or be a marker of, TB disease progression in CBA/J mice. Neutrophils can be numerous in pulmonary TB patients and are specifically associated with TB disease in humans. 7,15,29 Neutrophils, neutrophil precursors, and/or neutrophil-like cells also cause M.tb susceptibility and/or TB progression in some other M.tb-susceptible mouse strains, 4,14,24,30 suggesting a common disease phenotype. With this growing interest in understanding the protective and detrimental roles for neutrophils in M.tb infection and TB disease, 28 CBA/J mice may provide another useful model in which to understand the role(s) for neutrophils in TB disease progression.
Footnotes
Acknowledgements
We thank Alan Fletchner, Anne Saulsbery, and Shelly Haramia in The Ohio State University’s College of Veterinary Medicine, Department of Veterinary Biosciences.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support was provided by the NIH NIAID K08AI071111 (G. Beamer) and R01AI064522 (J. Turner).
