Abstract
Ten of 12 red-bellied short-necked turtles from a single clutch presented at 9 months of age with multiple white to tan nodules on their feet. Histologically, the nodules were composed of large periarticular deposits of mineralized crystalline material that extended into the joint spaces of interphalangeal joints and was surrounded by granulomatous inflammation and fibrosis. Crystallographic analysis determined the material to be apatite (calcium phosphate hydroxide) consistent with the tumoral calcinosis form of hydroxyapatite deposition disease (HADD). HADD has previously been described in aquatic turtles and rarely lizards and must be differentiated from gout in reptiles. A cause for the tumoral calcinosis lesions in these turtles could not be determined; however, based on previous reports in this species, a species-specific predilection, in conjunction with unknown environmental factors, is suspected. The use of the terms HADD, pseudogout (calcium pyrophosphate crystal deposition disease), and calcinosis circumscripta has been inconsistent, creating confusion in the literature.
Keywords
Gout is the most common and best-characterized cause of nodular deposits of crystalline material in and around the joints of reptiles. Gouty tophi are composed of monosodium urate crystals surrounded by a granulomatous inflammatory response. 12 However, nodular periarticular deposits of crystalline material other than urates also occur in reptiles and clinically must be differentiated from gout. Variably termed pseudogout, false gout, and hydroxyapatite deposition disease, these reports include 2 juvenile red-bellied short-necked turtles (Emydura albertisii [now subglobosa]), 24 a red-eared slider turtle (Trachemys scripta elegans), 6 a painted turtle (Chrysemys picta), 3 clutches of newly hatched turtles of unspecified species, 27 and a Uromastyx sp lizard. 5 Crystallographic analysis of the deposits in some of these cases has shown the material to be calcium phosphate in the form of hydroxyapatite (Ca10(PO4)6(OH)2), 5,6,24 the predominant mineral found in bone. The term hydroxyapatite deposition disease (HADD) has been suggested 24 to differentiate them from pseudogout caused by deposition of calcium pyrophosphate dihydrate (Ca2P2O7 .2H2O) in articular tissues, as described in humans, nonhuman primates, and dogs. 16,17,23,27
In humans, the term tumoral calcinosis is commonly used to refer to single or multiple, nodular, periarticular deposits of hydroxyapatite, 1,11,14 whereas the alternative term calcinosis circumscripta is frequently used in the veterinary literature. 7,22,23,27 This report describes a tumoral form of hydroxyapatite deposition disease seen in 10 related juvenile red-bellied short-necked turtles (Emydura subglobosa) from a zoological collection. The clinical, cytologic, histologic, and crystallographic features of the deposits in these turtles are described. Despite investigation into husbandry and heredity, as well as complete necropsies on all euthanized turtles, a cause for this condition could not be determined.
Materials and Methods
Twelve juvenile red-bellied short-necked turtles (E. subglobosa) were included in this study. Table 1 summarizes the clinical presentation and diagnostic testing. The turtles represented a single clutch from which 10 presented at 9 months of age with multiple nodules on their feet (case Nos. 1–10). Six females, 3 males, and 1 turtle of undetermined sex were affected. The turtles were housed in 2 adjacent tanks; several additional tanks that shared the same water and contained related turtles and turtles of other species were unaffected. Lesions were not observed in turtles from additional clutches from the same parents, either before or after the affected clutch. No differences in the handling of the clutch of eggs or the diet or husbandry for this group of hatchlings were reported. Initially, physical examinations and radiographs were performed on 4 affected turtles, 2 from each tank, and biopsies, cytologic preparations, and bacterial cultures of material expressed from lanced nodules were performed on 2 of these turtles. No improvement was seen after 3 weeks of treatment with ceftazidime (20 mg/kg intramuscular/subcutaneous every 3 days; Hospira Worldwide, Lake Forest, IL). After their initial appearance, the nodules remained static for up to 4 to 10 months until the turtles were humanely euthanized due to the failure of these lesions to resolve or as the result of ulcerative shell disease. In total, 10 of the clutchmates, 8 with and 2 without foot nodules, developed sloughing and erosions of the skin of the shell, sometimes with bone exposure, that were variably treated with topical medications and ceftazidime injections. At the time of euthanasia, blood was collected from 5 turtles (case Nos. 6–10), and urine was collected from 2 turtles (case Nos. 8 and 9) with foot nodules. Blood and urine were also collected from 1 unaffected clutchmate (case No. 11) euthanized due to shell lesions and from another related turtle from a different clutch (control) that had shell lesions but no foot nodules. Complete blood counts (CBC), serum chemistries, and urinalyses were performed in house on collected blood and urine samples. One turtle from the clutch (case No. 12) was alive at the end of the study after no foot lesions developed and minimal shell lesions resolved.
Clinical information of the study population.
aPooled.
Necropsies were performed on all euthanized turtles (case Nos. 1–11), and brain, eye, heart, lung, gastrointestinal tract, liver, kidneys, spleen, urinary bladder, muscle, and bone, as well as entire feet, were fixed in 10% neutral buffered formalin for histologic examination. Formalin-fixed tissues were routinely processed and embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin. Selected sections were stained with von Kossa for mineral and Ziehl-Neelsen or Goodpasture’s Gram stain for microorganisms. Some sections of feet containing nodules were demineralized for approximately 30 minutes to 24 hours prior to processing using RDO Gold decalcifier (Apex Engineering Products Corporation, Aurora, IL). Non-demineralized sections from 5 turtles (case Nos. 1, 2, 3, 5, 7), as well as partially demineralized sections from 4 of these (case Nos. 1, 3, 5, 7), were stained with nonaqueous alcoholic eosin stain (NAES) as described in Shidham et al, 20 except that 0.5% alcoholic eosin Y was prepared from a 2% aqueous solution of eosin Y stock using 95% reagent-grade ethanol and slides were cover-slipped with Surgipath Micromount mounting medium (Leica Microsystems, Richmond, IL).
Prior to formalin fixation, material was excised from foot nodules of 6 affected turtles and kept at room temperature (4 samples, case Nos. 6–9) or frozen at –70°C (2 samples, case Nos. 3 and 4). The 4 samples kept at room temperature were pooled, the 2 frozen samples were thawed, and all 3 were analyzed for identification and semi-quantification of components by polarized light microscopy with the oil immersion method and by infrared spectroscopy (IR) as previously described. 25
Results
Clinical Findings
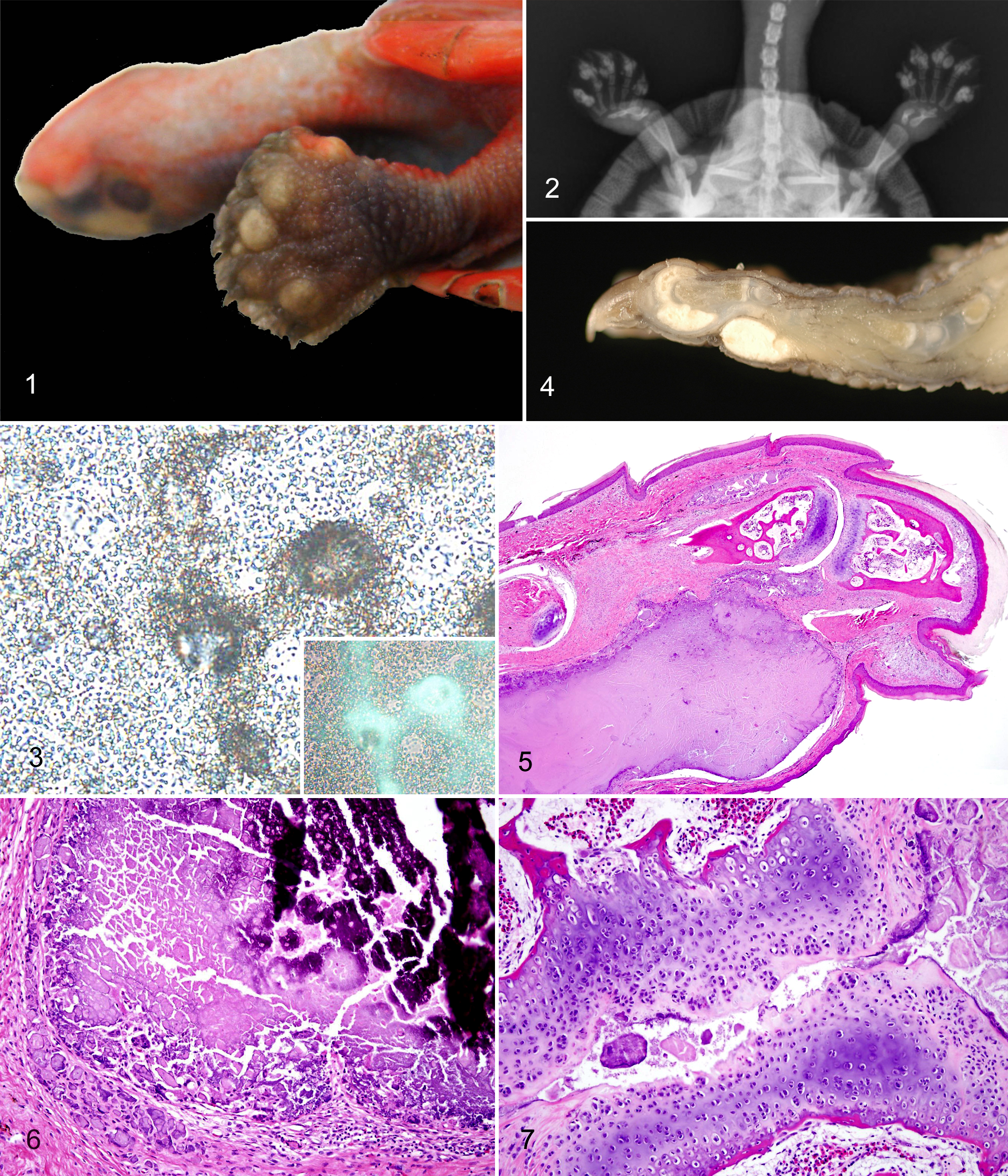
In the 10 affected turtles, multifocal to coalescing, smooth, nonulcerated, pale tan, firm, 1- to 4-mm diameter nodules were present on multiple feet on both the dorsal and palmar or plantar surfaces predominantly in the digits (Fig. 1). Coalescing mineral density deposits corresponding to these nodules were seen in radiographs (Fig. 2). When examined with a compound microscope, unstained cytologic preparations of material from the nodules demonstrated finely granular to coarsely clumped, acellular, refractile material with weak, glowing white birefringence when examined under polarized light (Fig. 3). Wright-Giemsa–stained smears showed only larger clumps of refractile, blue to purple material and lacked birefringence under polarized light. Bacterial cultures yielded small numbers of Pseudomonas aeruginosa and Acinetobacter sp from one sample and small numbers of Stenotrophomonas maltophilia from the other. These were interpreted as contaminants. No consistent abnormalities were detected on CBC, serum chemistry analyses, and urinalyses from the affected or unaffected turtles compared with International Species Information System aggregated values. 8 Selected serum chemistry values are presented in Table 2. Although average serum calcium and phosphorus levels (and the corresponding products of these values) were slightly higher in the 5 affected turtles tested compared with the 2 unaffected turtles, these differences were not statistically significant (Student’s t-test, P > .05), and the values were within references ranges.
Selected serum chemistry data from turtles with and without foot nodules.
ISIS, International Species Information System.
Necropsy, Histopathology, and Special Stains
At postmortem examination, the articular and periarticular tissues of the digits contained well-demarcated accumulations of white, soft, pasty to chalky material (Fig. 4). Histologically, foot nodules expanded dermal and periarticular connective tissues and were composed of multiple irregular, nodular accumulations of variably mineralized (depending on degree of decalcification), granular to amorphous, basophilic to eosinophilic, non-birefringent material (Fig. 5). This material was surrounded by a thin rim of fibrous tissue and mixed with variable numbers of epithelioid macrophages and multinucleated giant cells and less frequently lymphocytes, plasma cells, and heterophils (Fig. 6). The material was also sometimes present within and expanding interphalangeal joint spaces, and the articular cartilage of these joints was variably degenerate or necrotic, fibrillated, eroded, or lost with remodeling of adjacent bone (Fig. 7). In one joint examined, a deposit disrupted the articular cartilage and extended into the subchondral bone. Rarely, the deposited material was present in small areas of epidermal necrosis and loss and in overlying crusts, suggestive of transepidermal elimination (Fig. 8).
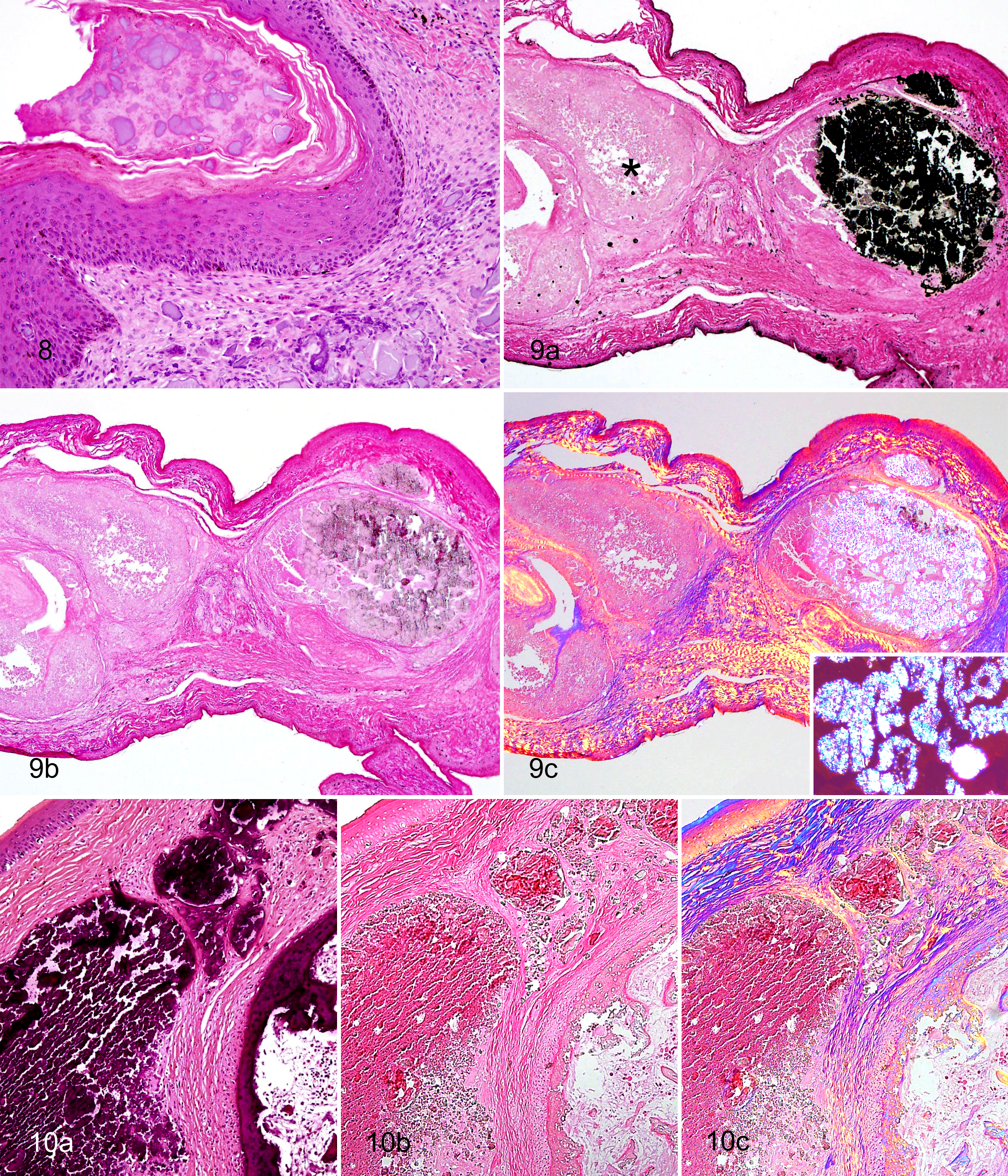
Gram and Ziehl-Neelsen stains for bacteria were negative. In non-demineralized sections, the material stained black with von Kossa stain, confirming the mineral content (Fig. 9a). With the NAES, non-demineralized sections of foot nodules were characterized by clusters of clear, rounded, irregularly shaped, refractile but non-birefringent crystalline structures mixed with amorphous eosinophilic material (Fig. 10). In partially demineralized sections (approximately 30–60 minutes in decalcifier), the material lacked the refractile crystalline structures except in incompletely demineralized areas (Fig. 9b). In these areas, refractile crystals were finer, sharper, and more irregular than in non-demineralized sections and were strongly birefringent under polarized light (Fig. 9c). This effect was seen in case Nos. 5 and 7 between different nodules and in case No. 3 between 2 opposing sections from the same nodule that were treated identically except that one section was placed in decalcifier for 30 minutes prior to processing.
Shell lesions were noted in all but 2 of the turtles with foot lesions as well as the 2 turtles without them. The lesions involved both carapace and plastron and ranged from acute ulcerations with bone exposure to chronic skin-covered defects in the bone to raised plaques. Histologically, shell lesions varied in chronicity from acute necroulcerative dermatitis and osteomyelitis with intralesional bacteria to chronic bone loss with fibrosis, re-epithelialization, and hyperkeratosis. There were no other consistent lesions seen histologically, although many of the turtles had mild heterophilic or lymphoplasmacytic inflammatory infiltrates in various tissues, including the intestine (case Nos. 2, 6, 8–11), lung (case Nos. 1, 3, 7), and kidney (case Nos. 4 and 7). Three turtles (case Nos. 8, 9, 10) had focal areas of mineralization of the corneal epithelial basement membrane and small intestinal wall or serosal vasculature.
Crystallographic Analysis
All samples of the material submitted for crystallographic analysis were composed of at least 90% apatite (calcium phosphate hydroxide, Ca5(PO4)3OH) and up to 10% calcium carbonate (CaCO3) when evaluated by IR. Observation under the polarized light microscope showed the content of the crystalline form of calcium carbonate was <1%, suggesting that most of the calcium carbonate was amorphous or was included in the apatite phase, which can be called carbonated (hydroxyl)apatite.
Discussion
In this group of red-bellied short-necked turtles, multicentric nodules limited to the feet of 10 of 12 juvenile clutchmates were determined to be deposits of predominantly calcium phosphate (in the form of apatite) by cytologic, histopathologic, and crystallographic evaluation. Therefore, the diagnosis of a tumoral calcinosis form of HADD was made. The initial differential diagnoses for the foot nodules included abscesses or granulomas, articular gout, pseudogout, and HADD, but the cytologic and histopathologic findings were not consistent with infectious causes or gout. Although gouty tophi can mineralize and be radiodense, when examined in unstained cytologic preparations under polarized light, the monosodium urate crystals of gout are needle-like and strongly birefringent in contrast to the granular, weakly birefringent material observed in these cases. 18,19 In histologic tissue sections, urate deposits are usually lost during routine processing, leaving pale, radiating, wispy outlines, unlike the coarse clumps of mineralized material seen with calcium phosphate deposits.
Differentiation of pseudogout and HADD is difficult using routine cytology and histology. The calcium pyrophosphate dihydrate (CPPD) crystals of pseudogout are described as rhomboid to rod shaped and birefringent, and these properties can be retained in histologic sections. Although birefringence in histologic sections differentiates CPPD crystals from coarse granular, non-birefringent deposits of hydroxyapatite, their birefringence is often dulled or lost, especially in demineralized sections. 9,19,20 To help overcome this issue, the NAES method has been proposed to allow differentiation of CPPD crystals and hydroxyapatite crystals in tissue section. 20 In humans, CPPD crystals show birefringence with this method, whereas hydroxyapatite crystals do not. In sections of the turtle nodules stained with the NAES method, there was a clear difference in the birefringence of the crystalline material between sections depending on whether tissue was placed in a decalcifier prior to processing. Non-demineralized sections lacked birefringence, whereas partially demineralized sections had areas of strong birefringence corresponding to areas where some mineral remained on hematoxylin and eosin–stained slides. This is important because, although decalcification did not affect the staining properties of CPPD crystals in humans, none of the tumoral calcinosis sections were demineralized in that study, so its effect on the birefringence of hydroxyapatite deposits was not assessed. 20 This potential effect of a decalcifier on the birefringence of hydroxyapatite deposits could have significance for at least 1 previous report of pseudogout in a painted turtle that relied on birefringence seen with the NAES method for the diagnosis of deposited material as calcium pyrophosphate. 3
Based on histopathologic appearance and composition, the nodules in these turtles were consistent with tumoral calcinosis (calcinosis circumscripta), which represents a form of hydroxyapatite deposition disease. 1,11,21 Tumoral calcinosis in humans can be primary (familial and nonfamilial) or secondary to other diseases. The primary familial forms are inherited as autosomal recessive disorders and present as single or multiple periarticular calcified masses in juveniles and young adults. 2,11,21 This form is associated with elevated serum phosphorus and/or vitamin D levels as a result of mutations that cause decreased renal excretion of phosphate and increased vitamin D synthesis. 10,11 The lesions in this type therefore represent a form of metastatic mineralization. 23 A possibly familial form of HADD not associated with elevated calcium or phosphorus has also been described in Great Dane dogs, 26 and sporadic tumoral calcinosis (or calcinosis circumscripta) in dogs shows breed predilections and has been seen in related individuals, suggesting a hereditary component. 13,23 The nonfamilial primary form of tumoral calcinosis in humans is similar to most cases of calcinosis circumscripta described in dogs. The lesions are generally solitary, occur in young individuals, and can be associated with known or suspected trauma (ie, dystrophic mineralization) or be idiopathic. 7,11,22,23 Bilateral multicentric calcinosis circumscripta lesions were described in the feet of a cynomolgus macaque and attributed to prior tissue damage. 15 Finally, the secondary form of tumoral calcinosis is associated with other diseases, such as renal failure, primary hyperparathyroidism, hypervitaminosis D, connective tissue disorders, and diseases causing bone lysis (eg, infection or neoplasia). 4,11,21 The mineralization associated with these diseases can therefore be metastatic or dystrophic. Calcinosis circumscripta, particularly in the foot pads, has been associated with chronic renal failure in dogs. 7,22
The cause of this condition in these related turtles was not determined, but several aspects of their presentation showed similarities to known and hypothesized causes in humans and other animals. A species predilection is suggested by a previous report of this condition in the same turtle species. 24 The relationship between the 2 turtles in that report was unknown. Although all the affected turtles in this study were siblings from the same clutch, and the clinical presentation of multiple lesions in juvenile animals was suggestive of a primary familial form, a solely hereditary condition in these turtles is unlikely. Previous and subsequent clutches from the same parents were unaffected, and serum phosphate levels from 5 affected individuals were not significantly elevated compared with 2 unaffected turtles or published reference values. Although the lesions could be due to metastatic mineralization, only 1 of the turtles (case No. 9) in this study and 1 of 2 in a previous report 24 had a high calcium × phosphorus product (ie, >60) at the time of euthanasia. Mineralization in other soft tissue sites—namely, blood vessels and cornea—was seen in several turtles but was minimal. No other cause of calcium, phosphorus, or vitamin D imbalances was found in the course of this investigation. There was no evidence on serum chemistries or histopathology of renal disease in any of the turtles in this study. In previous reported cases, improper diet or husbandry deficiencies were primary considerations, 6,24 but no known changes had been made in the care of these turtles to account for the appearance of this condition in this group. However, because of the small number of cases and controls in this study, additional testing, preferably around the time of development of the nodules, would be required to rule out the possibility of a role for a transient hyperphosphatemia or hypervitaminosis D in this condition. Vitamin D levels were not tested in these turtles and could be a direction of future investigation.
The possibility of a dystrophic mineralization process in these turtles was also considered, although evidence was lacking. The turtles in this study had no known history of foot trauma, and enclosure substrates were reportedly unchanged from previous use by unaffected individuals. Many of the turtles developed shell lesions that were likely due to sepsis or trauma, but these lesions were not noted in any of the turtles until 4 months after the onset of the foot nodules and were therefore difficult to relate to the foot nodule development. Moreover, 2 turtles with foot nodules had no shell lesions, whereas 2 with shell lesions did not develop foot nodules. In conclusion, this species seems predisposed to developing the tumoral calcinosis form of HADD, but the underlying factors remain undetermined.
Footnotes
Acknowledgements
We thank Yvonne Cates for all histopathology and special stains, April Gorow and Karen Good for necropsy assistance, Dr Rebecca Kagan for diagnostic work on an early case, and Dr Allan Pessier for many helpful comments on the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
