Abstract
In a collection of 6 young binturongs (Arctictis binturong), 3 presented with anorexia, lethargy, and footpad swelling diagnosed by surgical biopsy as calcinosis circumscripta. Despite supportive care over the next 4 years, affected binturongs had progressive weight loss, hyperphosphatemia, and hyperkalemia and developed large radiodense deposits in tissues adjacent to appendicular joints, thoracolumbar vertebrae, and the sternum. Two binturongs died and necropsies showed severe periarticular mineral deposition with fibrosis and granulomatous inflammation. Additionally, binturongs had mineralization of distal renal tubules, mineralization of the renal interstitium, and marked mineralization of the pulmonary interstitium and peribronchiolar smooth muscle. Foot biopsies from the surviving binturong were evaluated by spectroscopic microanalysis and were positive for hydroxyapatite, Ca10(PO4)6(OH)2. Periarticular and footpad mineralization in these binturongs is consistent with hydroxyapatite deposition disease, which is rarely described in humans and animals as a primary familial condition or a secondary condition often associated with renal injury.
Keywords
Binturongs (Arctictis binturong) are the largest member of the family Viverridae, weighing 9 to 20 kg. They are defined as a hypocarnivores, preferentially consuming high carbohydrate fruit with a <30% meat diet. Despite this high degree of frugivory, they have an unusually short gut lacking a functional cecum, 2 and they do not exhibit carbohydrate fermentation typical of frugivores. In their natural habitat, wild binturongs have a diet largely dependent on ripe figs (Ficus spp.). 8 Due to their unique diet and specific anatomy, management of binturongs in zoos requires special attention. In Japan, only 16 of 91 zoos manage binturongs (37 binturongs), and in North America there are approximately 70 binturongs in managed care in 2021. Literature on health management and disease is understandably sparse in this species. This report details the clinicopathological findings of severe periarticular and footpad mineralization in a group of young binturongs and discusses the possible causes.
Six binturongs (2 males, 4 females) were born under managed care in Thailand in a facility holding a total of 16 males and 30 females. The breeding information included only the sire and dam. Of the affected binturongs, case 1 was unrelated to other binturongs and cases 2 and 3 were full siblings. Of the healthy binturongs, animal 4 shared a sire with cases 2 and 3 and animals 5 and 6 were full siblings unrelated to the others. Binturongs were transported at 1 to 3 years of age to Ichihara Elephant Kingdom Zoological Park, Chiba, Japan, and were group-housed in an indoor-outdoor facility and individually fed mixed fruits with animal proteins (commercial cat food, chicken heads, quail, boiled eggs).
One year after arrival, a 2-year-old male (case 1) acutely developed anorexia with loss of 2.2 kg body weight (13% loss), and firm swelling of all footpads (clinical day 1). Footpad lesions consisted of dense granulation tissue with pockets of pasty white material (Fig. 1). Aerobic and anaerobic cultures were negative. Lesions were debrided and palliative therapy attempted with progression of clinical disease and lesions. Two females, case 2 (4 years old) and case 3 (3 years old), developed footpad swelling on days 78 and 218, respectively (Fig. 2), which was also unresponsive to treatment. Surgical biopsy of foot pads from case 1 (day 264) showed granulation tissue, ectopic calcification, and granulomatous inflammation including multinucleate giant cells, consistent with calcinosis circumscripta.
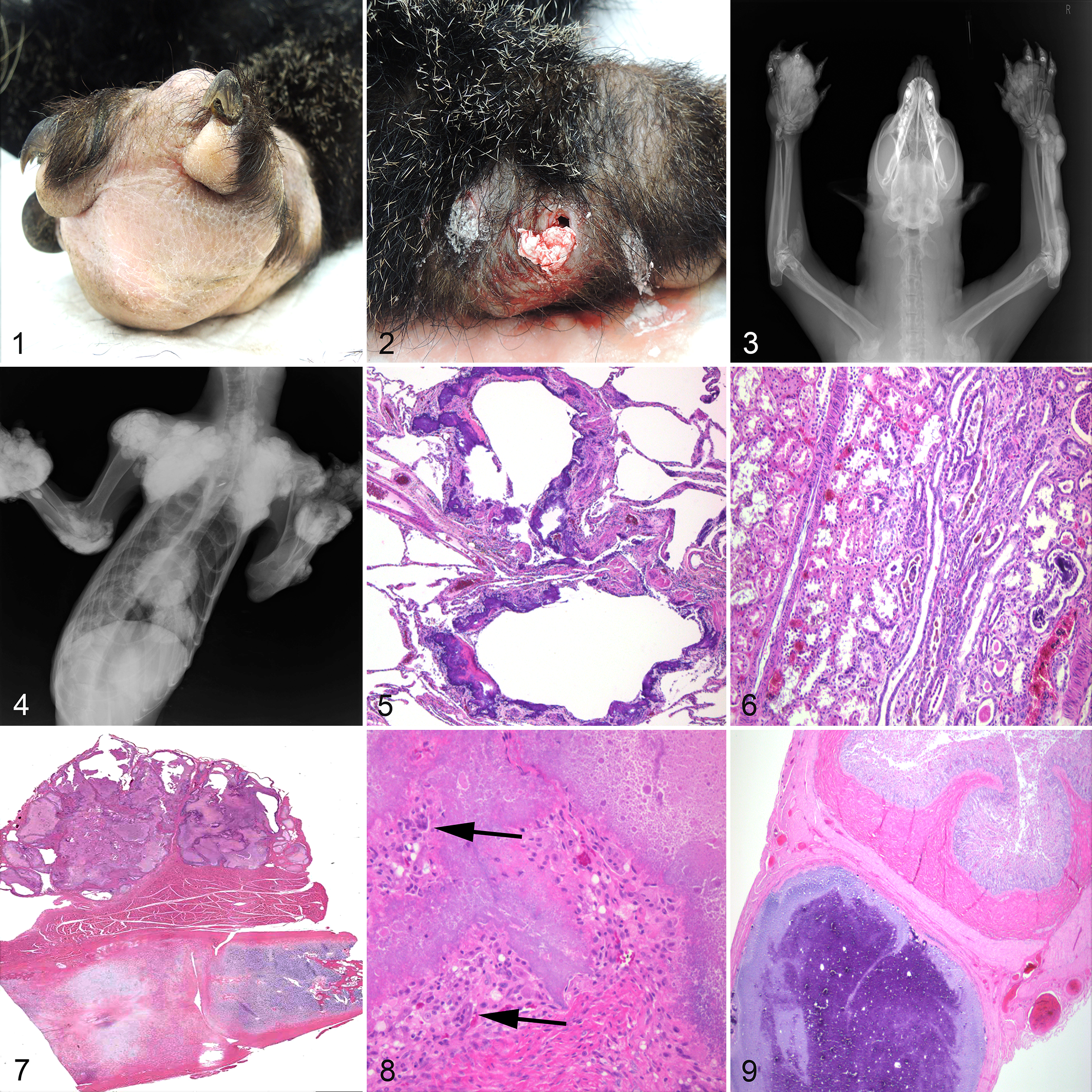
Hydroxyapatite deposition disease, binturongs.
Blood was collected for complete blood cell count and serum biochemistry from cases 1 to 3 in the first and last years of clinical disease and compared to blood from healthy animals 4 and 5 (3- and 4-year-old females) during the same time periods (Table 1). Affected binturongs had hyperphosphatemia (7.0–10.1 mg/dL, reference range 5.6 ± 1.9) 3 and hyperkalemia (5.8–8.1 mEq/L, reference range 4.8 ± 0.5) 3 with normal serum urea, creatinine, and calcium (9.5–12 mg/dL, reference range 10.0 ± 0.9) 3 throughout their disease compared to the healthy animals. The calcium X phosphorus product was consequently elevated in these binturongs (66.6–121.2, reference range ≤70), consistent with increased risk of ectopic mineralization. In the late disease stage, urine specific gravity from case 3 was 1.012, within the range of healthy animals 4 and 5 (1.022, 1.002). Serum 25-hydroxyvitamin D concentrations in cases 1 to 3 varied throughout disease from 82.9 to 204.5 ng/mL, compared to healthy animal 4 (55.8 ng/mL). Parathormone levels in cases 1 and 2 were similarly low (<2 and 5 pg/mL) compared to animal 4 (8 pg/mL), though normal serum levels of 25-hydroxyvitamin D and parathormone are not available for binturongs and this assay has not been validated in this species.
Blood Chemistry and Urinalysis of Affected (Cases 1–3) and Healthy (Cases 4 and 5) Binturongs (Arctictis binturong) in a Zoological Collection in Japan.
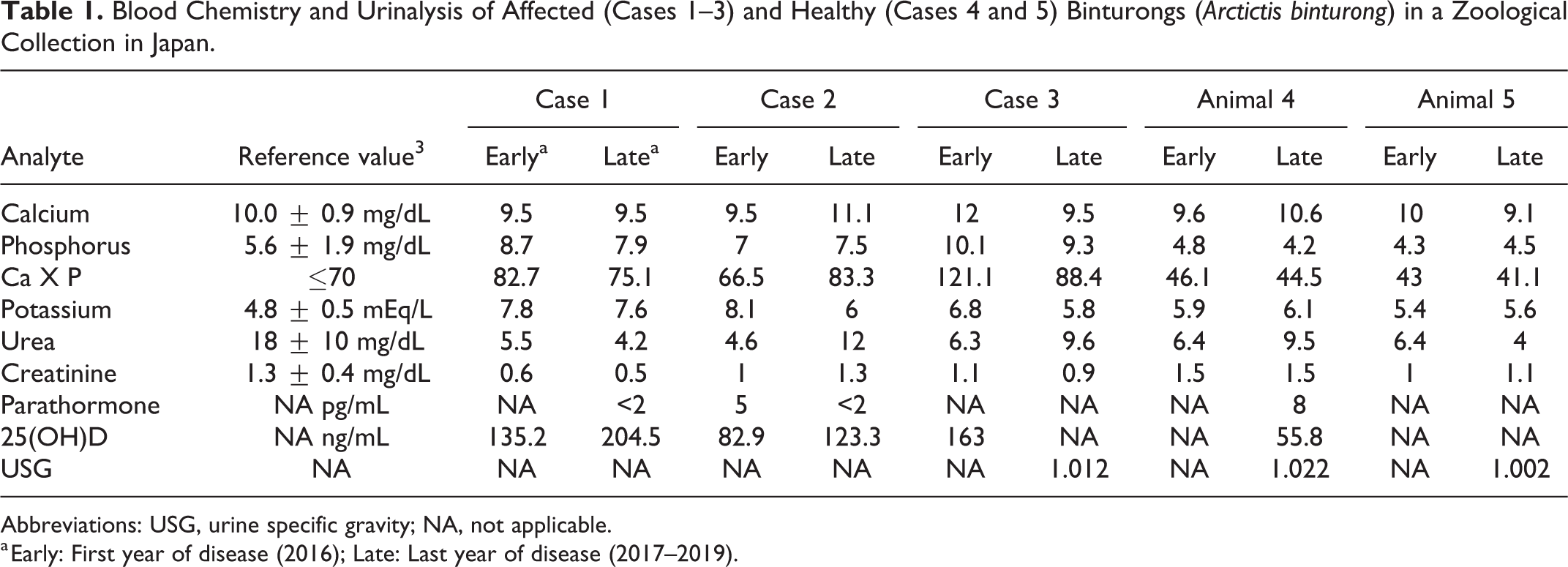
Abbreviations: USG, urine specific gravity; NA, not applicable.
a Early: First year of disease (2016); Late: Last year of disease (2017–2019).
Foot and body radiographs of animals 1 to 5 were performed periodically during disease. Cases 1 to 3 had progressive deposition of radiodense material expanding footpads and adjacent to all appendicular joints, particularly the phalanges, scapulohumeral and coxofemoral joints, dorsal thoracolumbar vertebrae, and sternum (Figs. 3, 4). No visceral mineralization was detected radiographically. Animals 4 and 5 remained normal. Animal 6 was clinically vibrant and was not additionally handled for bloodwork or radiographs during the study.
Cases 1 to 3 were supported throughout disease with a low-phosphorus feline renal diet (Prescription Diet k/d, Hill’s Pet Nutrition) and various fruits (bananas, pineapples, grapes, oranges), with palliative treatment including an oral phosphorus adsorbent lanthanum (7 mg/kg PO BID, FOSRENOL, Bayer Yakuhin, Ltd) and an oral anti-hyperkalemia drug, polystyrene sulfonate calcium (250 mg/kg PO BID, ARGAMATE, Astellas Pharma Inc). Cases 1 to 3 remained hyperphosphatemic (Table 1) with hyporexia, weight loss, lethargy, and progression of periarticular and footpad lesions over the next 3 to 4 years.
Case 2 died 3 years following clinical onset of disease (day 1085). Gross postmortem evaluation was not performed, and an immediate cause of death was not determined. Limited tissues were collected and processed for histopathology (lung, kidney, thyroid gland, parathyroid gland), showing severe ectopic mineralization in the pulmonary alveolar septa and bronchiolar smooth muscle (Fig. 5). Approximately 20% of the renal cortex and medulla was streaked with interstitial fibrosis and mineralization and entrapped tubules were moderately ectatic with mineralized eosinophilic granular material and cellular casts (Fig. 6). Collecting ducts throughout the cortical-medullary junction and medulla were commonly dilated with lamellar mineral. There was mild, segmental glomerulosclerosis with no inflammation. Thyroid glands and parathyroid glands were mildly atrophied.
Case 1 died 3.6 years after clinical onset (day 1343). Gross findings were severe emaciation and dehydration (presumed to be the cause of death), with severe periarticular mineral deposition of the cervical-thoracic vertebrae, sternum, and appendicular joints (shoulder, elbow, carpal, hip, stifle, tarsal joints, phalanges), consistent with the radiographic findings. The major viscera and sternum were fixed in 10% formalin and the sternum was additionally decalcified in 8% formic acid for 24 hours. No decalcification was necessary on visceral specimens, which were easily sectioned for processing. Select tissues were stained with von Kossa (lung, kidney), Fite’s acid fast, Gram, and Grocott’s methenamine silver (sternum) for identification of calcium and for the exclusion of microorganisms.
Sternal bone and articular cartilage were normal, but there were large extra-articular deposits of granular basophilic mineral with lakes of amorphous eosinophilic material. Mineral was non-birefringent, and was circumscribed by large numbers of epithelioid macrophages, multinucleated foreign body-type giant cells, and fibrosis with moderate numbers of lymphocytes and plasma cells (Figs. 7, 8). No microorganisms were detected with special stains. Ectopic mineralization of the lungs was similar to case 2. Mineralization of the kidneys was limited to prominent lamellar and globular deposits within the lumen of collecting ducts with mild interstitial fibrosis and mineralization, and mild glomerulosclerosis. A discrete encapsulated focus of mineral with granulomatous inflammation and fibrosis was within the serosa of the jejunum (Fig. 9). There was mild thyroid and parathyroid gland atrophy.
Surgical biopsies of footpad lesions from case 3 were submitted (day 1446) for mineral identification using X-ray fluorescence and Raman spectroscopic analysis. The spectra were consistent with hydroxyapatite Ca10(PO4)6(OH)2. This binturong was alive at the time of publication of this report, with little progression of lesions. Based on similar unique presentation of clinical disease in case 3, it is presumed that hydroxyapatite is the composition of periarticular mineral described in cases 1 and 2.
Periarticular deposition of hydroxyapatite, Ca10(PO4)6(OH)2, is termed tumoral calcinosis or hydroxyapatite deposition disease (HADD) in the human literature and is synonymous with calcinosis circumscripta, a term frequently used in veterinary medicine. HADD in humans is rare and often primary, presenting in young people as an inherited autosomal recessive condition with failed renal excretion of phosphorus and excessive vitamin D synthesis, resulting in hydroxyapatite deposition surrounding major joints (shoulder, elbows, hips)—a form of metastatic mineralization. 4 HADD is described rarely as an inherited condition in veterinary medicine, with possible reports in related red-bellied short-necked turtles (Emydura subglobosa) and great Dane dogs, for which the pathogenesis is unknown and lesions are not clearly metastatic nor dystrophic. 1,10 Secondary HADD is described in humans and animals with chronic renal disease associated with calcium-phosphorus imbalance and metastatic mineralization. 4 –6,9,10
HADD manifesting in footpads is described in domestic dogs 6 and zoo species, including the turtles noted above and in rock hyraxes (Procavia capensis), occurring as either primary or secondary deposition disease. 1,5,6 The pathogenesis of HADD localizing in the footpads is unknown, though hydroxyapatite deposition occurs under conditions of hyperphosphatemia (metastatic mineralization), preferentially in areas of repetitive microtrauma and hemorrhage, which may be most prominent on footpads and pressure points similar to major joints. HADD of the footpads may occur particularly in animals maintained on hard, abrasive substrates, suggesting a possible component of dystrophic mineralization. Deposited mineral elicits histiocytic inflammation that causes local proteolysis and collagenolysis, with bursa formation. Deposited mineral and chronic inflammation incite reactive fibroplasia, with characteristic histopathologic findings. 4
The cause of hyperphosphatemia and HADD in this group of young binturongs is not known. Primary renal insufficiency was not entirely excluded, though serum urea and creatinine of affected binturongs remained normal and comparable to healthy cohorts throughout the study period, urinalyses at these time points were mostly not available. Kidneys of cases 1 and 2 had soft tissue mineralization with limited primary degenerative lesions, possibly supporting a non-renal cause of electrolyte imbalance. Importantly, degenerative renal disease and renal neoplasia appear to be relatively common in binturongs in managed care. 7
As these binturongs developed hyperphosphatemia and HADD at a young age and within a narrow window of time, a possible toxic event is considered, including vitamin D toxicosis. Serum parathormone levels in cases 1 and 2 were low but comparable to healthy animal 4, and mild atrophy of the parathyroid was detected histologically in postmortem examination of affected binturongs, a paradoxical finding in the face of chronic hyperphosphatemia. Serum vitamin D levels of affected binturongs were significantly elevated compared to a single specimen from healthy animal 4. It is important to consider that the assays are not validated in this species and reference values are not available. No source of excessive dietary vitamin D and phosphorus were identified. Nevertheless, binturongs have specialized digestion and a unique natural diet is problematic to replicate. Primary dietary cause of hypervitaminosis D and/or hyperphosphatemia, with or without renal injury, is not excluded.
Early onset and progression of HADD may also support primary familial hyperphosphatemia and hypervitaminosis D. Two of the 3 affected binturongs were full siblings from a collection of approximately 46 binturongs, which did not report any previous incidence of this condition. Though more extensive genealogy was not available, binturongs are relatively rare in managed care, and some degree of inbreeding is possible. Further genetic analysis is of interest with this cohort.
In conclusion, HADD in these binturongs was characterized by acute onset of footpad swelling with slowly progressive anorexia and emaciation, lethargy, hyperphosphatemia, and hyperkalemia, with extensive mineralization of the footpads and in tissues adjacent to multiple joints, with mineralization of the kidneys and lungs. HADD in these binturongs may be a primary familial condition or secondary to dietary or renal causes. Careful evaluation of the diet, medications, and environmental treatments and enrichments for this species is suggested.
Footnotes
Acknowledgements
The authors would like to thank Dr T. Tamano (Matsubacho Animal Hospital) and Dr S. Matsunaga (Tokyo Dental College) for assisting with treatment and examination; Dr Brian Petroff and the ABQ Biopark for comparative information on vitamin D. The authors are grateful to Ms S. Sakamoto, the director of Ichihara Elephant Kingdom Zoological Park.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
