Abstract
To validate the use of the tissue microarray (TMA) method for immunophenotyping of ferret lymphomas, a TMA was constructed containing duplicate 1-mm cores sampled from 112 paraffinembedded lymphoma tissue specimens obtained from 43 ferret lymphoma cases. Immunohistochemical (IHC) expression of CD3, CD79α, and Ki-67 (MIB-1) was determined by TMA and whole mount (WM) staining of each individual case for result comparison. There was a high correlation between CD79α and CD3 results comparing ferret TMA and WM sections (kappa statistic 0.71–0.73 for single-core TMA and 0.79–0.95 for duplicate-core TMA) and between continuous data from Ki-67 staining of ferret TMA sections and WM sections (concordance correlation coefficients 0.77 for single cores and 0.87 for duplicate cores). Subsequently, a panel of commercially available antibodies was applied to the TMA for the analysis of expression in ferret lymphomas. The results of this study confirmed previously published results suggesting specific cross-reactivity of the applied IHC markers (CD3, CD79α, Ki67) with ferret lymphoma tissue. Other IHC markers (CD45Ro, bcl2, bcl10, MUM1, CD30, vimentin) were also expressed in subsets of the included ferret lymphomas. Further studies are necessary to determine the usefulness of these markers for diagnostic and prognostic evaluation of ferret lymphomas. In conclusion, the TMA technology was useful for rapid and accurate analysis of protein expression in large archival cohorts of ferret lymphoma cases.
Lymphoma is the third most common neoplasm and the most common malignancy in domestic ferrets. Currently published classifications of lymphoma in this species (and subsequent treatment recommendations) are mainly based on the morphology of the neoplastic cells although several criteria have been proposed, including cellular morphology,4,7,9–11 age of onset,2,5 organs affected,15,18 and T- or B-cell lineage.3,5 Lymphoma immunophenotyping is uncommonly performed in ferrets, although a limited number of papers exist that have determined B- or T-cell lineages in a small number of cases.3,5
Increased understanding and recognition of animal neoplasia demands improved techniques for animal tumor diagnosis and classification. Recently a World Health Organization (WHO) international histologic classification of lymphomas in domestic animals was published,20 providing a comprehensive classification mirroring the WHO classification of human hematopoietic tumors,13 which defines neoplasm categories according to the principle that the classification should be based on the real entities of diseases and should contain the morphology, genetic alterations, clinical features, and immunophenotypes, rather than simply emphasize the morphologic presentations. Analysis of molecular cell markers will be of increasing importance, both for animal cancer diagnosis and as a tool for defining animal models for human cancers.
Tissue microarray (TMA) is a recently described tissue-conserving high-throughput technology that makes it possible to collect archived paraffin-embedded tissues from large cohorts in one or more new paraffin blocks.14 The tissue samples are then available for staining with various techniques, including immunophenotyping and in situ hybridization, to analyze gene expression in a highly efficient manner. The increased efficiency enables the use of a wider array of tissue markers than previously possible, and large numbers of different molecules can be evaluated for their possible role in solid tumor development and/or progression or in disease characterization and prediction of patient outcome.6 Because of the small size of the tissue cores obtained by TMA (0.6 or 1 mm), studies comparing the efficacy of the technique compared to whole mount (WM) preparations are required. Considering the general paucity of ferret studies in the literature, such studies are all the more relevant.
The objectives of this study were to investigate the usefulness of TMA technology to perform large-scale retrospective cohort studies of ferret malignant lymphomas by rapid phenotypic profiling of large numbers of archival formalin-fixed, paraffin-embedded lymphoma samples and to identify possible markers for the classification of ferret lymphomas using the TMA technology.
Materials and Methods
Archival, routinely stored, formalin-fixed and paraffin-embedded tissue specimens (
Ferret lymphoma cases(




WHO = World Health Organization, A = peripheral T-cell lymphoma, B = cutaneous T-cell lymphoma, C = T-cell lymphoblastic lymphoma, D = T-cell chronic lymphocytic lymphoma, E = anaplastic large T-cell lymphoma, F = diffuse large B-cell lymphoma, G = Burkitts-type lymphoma, H = lymphoplasmacytic B-cell lymphoma, I = T-cell rich B-cell lymphoma, J = B-cell lymphoblastic lymphoma.
TMA construction and sectioning
Four-micrometer sections were cut from donor blocks, stained routinely with hematoxylin and eosin, and submitted for morphologic review. Representative tumor areas were identified and marked on each slide. From the corresponding area on the donor blocks, 2 tissue cylinders of 1-mm diameter were cored using a manual tissue microarrayer (Beecher Instruments, Silver Spring, MD). Cores were arranged in a recipient paraffin block with 0.4-mm distance between adjacent cores (see Figures 1 and 2), using the technique described by Kononen et al.14 Paraplast X-TRA paraffin (PELCO International, Redding, CA) was used for the recipient block. The top right corner of the array was marked by 2 cores of liver tissue. Tissues known to express the antigens under study (human diffuse large B-cell lymphoma, chronic lymphatic B-cell lymphoma, T-cell lymphoma) and normal ferret tissues (liver, lymph node, and spleen) were included in the TMA as baseline controls. Four-micrometer TMA sections were routinely cut on microtome and transferred to Superfrost plus slides (Menzel-Glaser, Braunschweig, Germany) using a waterbath (45°C).

Tissue microarray recipient block containing 1-mm cores punched from formalin-fixed, paraffin-embedded ferret tissue specimens. The TMA includes 240 cores; 240–300 cores is the maximum number of 1-mm cores that can be contained in a TMA block of this size without risking the stability of the block.

Lymphoma; ferret. Immunohistochemical demonstration of Ki-67 in 1-mm TMA cores showing varying positive staining with a brown positive reaction product. Monoclonal mouse antihuman Ki-67, MIB1, EnVision+ system (Dako, Glostrup, Denmark), counterstained in hematoxylin.
Immunostaining
TMA sections were submitted to a panel of antibodies (all primary antibodies applied to TMA sections are listed in Table 2). In parallel, WM sections from a subset of 53 donor blocks (representing 43 lymphoma cases and 10 control tissue specimens) were submitted to immunostaining with 3 antibodies (CD3 [F7.2.38], CD79α [HM57], and Ki-67 [MIB-1]).
2. Clone, manufacturer, antigen retrieval procedure, optimal dilution, and visualization system for each of the primary antibodies applied to ferret lymphomas.∗
Mab = monoclonal antibody, Pab = polyclonal antibody.
Source: 1 = Dakocytomation, Glostrup, Denmark; 2 = Novocastra, Newcastle upon Tyne, United Kingdom; 3 = BD Pharmingen Europe, Oxford, UK; 4 = Rocky Mountain Laboratory, Hamilton MT.
Antigen retrieval procedure: HIER = heat-induced epitope retrieval in microwave oven for 6 minutes at 750 W and 15 minutes at 350 W (unless otherwise stated) in TEG = TEG buffer pH 9.0 (10 mM tris-base and 0.5 mM EGTA), citrate = citrate buffer pH 6.0, or proteinase = Proteinase K (Dako, Glostrup, Denmark).
Visualization systems: EnVision = EnVision+ System-HRP (DAB), Dako, Glostrup, Denmark; ABC = avidin–biotin complex, Dako, Glostrup, Denmark; NS = no specific staining.
Sections were deparaffinized in xylene (20 minutes) and rehydrated in graded alcohol. Sections were incubated with primary antibody in optimal dilutions. Clone, manufacturer, antigen retrieval procedure, optimal dilution, and visualization system are presented for each of the primary antibodies applied to ferret lymphomas in Table 2. Incubation procedures were performed in a moist chamber at room temperature. Each incubation step was followed by 3 washes for 3 minutes in Tris Buffered Saline (TBS) buffer. Optimal dilution was determined by titration on whole sections of ferret control tissue. After washing with TBS, buffer sections were incubated for 30 minutes with Envision + System-HRP (DAKO, Glostrup, Denmark), followed by incubation with DAB+ chromogen (DAKO). All sections were counterstained with Mayer's hematoxylin (Merck, Darmstadt, Germany). The sections were then rewashed in water, dehydrated by graded alcohol, and mounted with Pertex mounting medium (Histolab Products, Göteborg, Sweden). Each stain was accompanied by a positive reference control. Lung tissue from mink diagnosed with acute Aleutian Disease viral infection was used as control for immunohistochemical (IHC) staining for Aleutian Disease virus. The diagnoses were confirmed by histopathology and antibody detection by countercurrent immune electrophoresis.19 In negative controls TBS buffer replaced antibody. Immunoreactivities to Ki-67 were classified as continuous data (undetectable levels or 0% to homogeneous staining or 100%). The cut-off values used in this study were defined as high proliferative Ki-67 index if >50% tumor nuclei stained, intermediate if 20–50% tumor nuclei stained, and low if less than 20% stained. For other antibodies, IHC staining was interpreted as positive when at least weak to moderate staining was seen in more than 20% of the neoplastic cells.
For validation of the TMA technology, TMA sections containing duplicate core biopsies and WM sections from tumor blocks were stained with CD3, CD79α, and Ki-67 antibodies (see Figures 2, 3, and 4). Immunohistochemical staining results were read in a blinded manner and later compared to one another. Single readings from each core were obtained and evaluated as 2 separate experiments. Two different statistical analyses were performed for validation of the TMA technique. The construction of 2 TMAs containing either 1 or 2 cores per specimen was simulated by assembling 1 tissue array with 2 cores per specimen and comparing the accuracy analysis based on either 1 or 2 cores to WM staining. For single-core analysis we excluded cases in which cores were lost during processing of the array section. For both single- and double-core analysis we calculated the number of cases lost from analysis because of tissue loss or inconclusive data. In one-core analysis all assessable cores were included as individual cases and compared with the corresponding WM section. For duplicate core analysis, 2 cores were used to obtain a single value and the one with the highest reading defined the case.
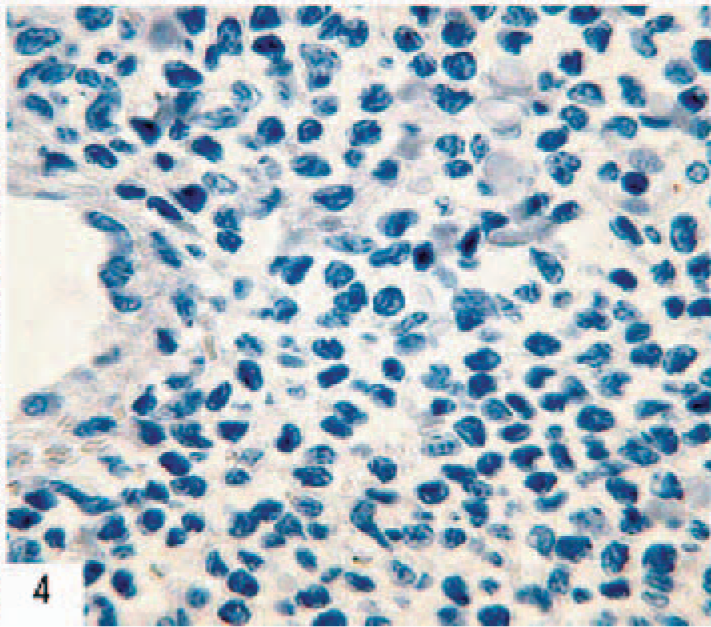
Skin; ferret. Immunohistochemical demonstration of the B-cell marker CD79α showing negative staining. Monoclonal mouse antihuman CD79α, HM57, EnVision+ system (Dako, Glostrup, Denmark), counterstained in hematoxylin.
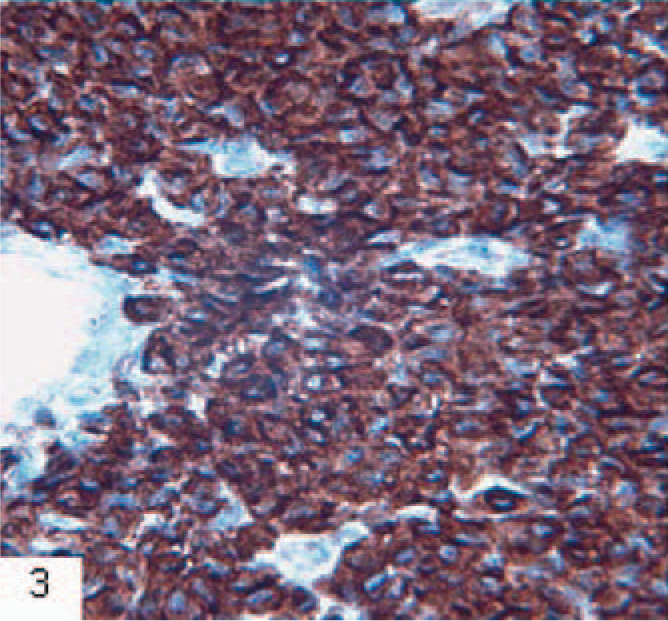
Skin; ferret. Immunohistochemical demonstration of the T-cell marker CD3 in a cutaneous T-cell lymphoma showing positive staining with a brown positive reaction product. Monoclonal mouse anti human CD3, F7.2.38, EnVision+ system (Dako, Glostrup, Denmark), counterstained in hematoxylin.
The association between WM and TMA results was studied using specific agreement and kappa statistics for dichotomous data1 and Lin's concordance correlation16 for continuous data. Kappa and concordance correlation values >0.7 were considered to express a strong association between the 2 methods. All statistical analyses were performed using STATA 7.0 (Stata Corporation, College Station, TX).
Results
Validation
Tissue microarray and WM sections from a subset of 53 donor blocks (representing each of the 43 lymphoma cases included in this study and 10 control tissue specimens) were immunostained with 3 antibodies (CD3 [F7.2.38], CD79α [HM57], and Ki-67 [MIB-1]). Results based on 1 and 2 TMA cores were compared with WM staining results (Table 3). The TMA contained 2 cores from each of 43 ferret lymphomas and 10 normal ferret tissues. Ninety-nine of the106 cores examined were assessable. The remaining 7 cores were missing from the TMA or could not be evaluated for other reasons. There was a strong concordance between the Ki-67 staining results comparing WM evaluation with single-core TMA analysis (Lin's concordance coefficient: 0.77,
3. Immunohistochemical staining results obtained from duplicate 1-mm TMA cores representing 43 ferret lymphoma specimens and 10 control tissue specimens (normal lymphoid tissue) compared with whole sections from the same specimens. Staining was performed with commercially available monoclonal antibodies toward human CD3 and CD79α (DakoCytomation, Glostrup, Danmark). Specimens were interpreted as positive for CD3 or CD79α when at least weak to moderate staining were seen in >20% of the neoplasticô cells.
Immunohistochemical readings from single cores were compared to cumulative values from 2 cores simulating the situation of tissue arrays constructed with only 1 core or 2 cores per specimen, which were then compared to the WM sections. The 2 methods (WM and TMA) showed a strong statistical association for all 3 antibodies when applied to ferret lymphoma tissue: CD79α (kappa value, 0.95;
Immunophenotyping
Based on immunostaining of a TMA containing duplicate tissue cores from 112 biopsy specimens obtained from 43 ferret lymphoma cases, 62.8% cases were categorized as T-cell type based on immunophenotyping, and the majority were found to have an intermediate (16.3%) or a high (51.2%) proliferative index (based on Ki-67 expression). The average age, sex distribution, and organ distribution for each lymphoma entity is presented in Table 1. In a large proportion of T-cell (48.1%) and B-cell lymphomas (43.8%) the lymphoma involved more than 1 organ. In the case of multicentric lymphomas, the agreement between IHC results obtained from different organ samples included from the same animal appeared to be close to 100%, although in a few cases biopsies from individual organs with lymphoma involvement appeared to show lower reactivity with the antibodies. In both T- and B-cell lymphomas, varying numbers of reactive T cells (CD3 positive) were identified.
Tissue microarray sections were subjected to IHC staining with a panel of markers. The primary antibodies CD3 (F7.2.38, PS1 and polyclonal), CD79α (HM57), CD45RO (UCHL1), CD30 (Ber-H2), bcl-2 (124), bcl10 (151), MUM1 (MUM1), vimentin (V9), and Ki-67 (MIB-1) stained varying proportions of the ferret lymphomas (see Table 4). The remaining primary antibodies, CD79α (JCB117), CD20 (L26), fCD4 (1F6), CD5 (54B4), CD8 (4B11), CD21 (1F8), P53 (DO-7 and 1F8), Bcl6 (PG-B6), and Cyklin-D1 (DCS-6), did not produce staining in ferret lymphomas or produced high levels of nonspecific staining.
4. Immunohistochemical staining results obtained from tissue microarray sections including duplicate tissue cores(1-mm diameter) from 43 ferret lymphoma specimens classified according to the World Health Organization classification of haematopoietic tumors (12).∗ Ki-67 staining was classified as low (<20%), intermediate (20–50%), or high (>50%). Immunoreactivities to other antibodies were interpreted as positive when staining was seen in more than 20% of the neoplastic cells.
WHO = World Health Organization, A = peripheral T-cell lymphoma, B = cutaneous T-cell lymphoma, C = T-cell lymphoblastic lymphoma, D = T-cell chronic lymfocytic lymphoma, E = anaplastic large T-cell lymphoma, F = diffuse large B-cell lymphoma, G = Burkitts-type lymphoma, H = lymphoplasmacytic B-cell lymphoma, I = T-cell rich B-cell lymphoma, J = B-cell lymphoblastic lymphoma.
Discussion
Out of 43 ferret lymphoma cases, the majority (
The markers CD3, CD79α, and Ki-67 were chosen for the validation study because of their relevance in lymphoma research and because they have been applied in published studies of ferret lymphomas.3,5,8,18 Immunohistochemical readings from single cores were compared to cumulative values from 2 cores simulating the situation of tissue arrays constructed with only 1 core or 2 cores per specimen. Both scenarios (single core and duplicate TMA) were evaluated for the number of cases lost from the analysis because of tissue loss or inconclusive data. Disagreements between duplicate cores from 1 specimen were uncommon for 2-core analysis. Nonconcordance (percent mismatches of evaluated cases) was the lowest for duplicate core versus single core for all stains. This demonstrates the importance of multiple cores to keep the number of lost cases as low and concordance rates as high as possible. Single core results generally showed lower correlation with WM results and the use of duplicate core TMA reduced the numbers of lost cases (in none of the evaluated stains in this study were both cores representing a case missing).
The 2 methods (WM and TMA) showed a strong statistical association. This result corresponds well with results of previous validation studies of human lymphomas.12,14,21
In this study, the CD79α clone HM57 was found to be the only B-cell marker, which successfully and repeatedly stained ferret B-cells. The monoclonal CD3 clone F7.2.38 was found to be the most sensitive and specific of the examined CD3 markers. The markers CD79α (JCB117), CD20 (L26), CD4 (1F6), CD5 (54B4), CD8 (4B11), CD21 (1F8), P53 (DO-7), Bcl6 (PG-B6), and Cyklin-D1 (DCS-6) did not stain ferret lymphomas and control tissue, indicating their lack of validity in the immunologic examination of ferret lymphomas. Also, there are no previous reports of successful application of these markers in ferret tissue.
The antibodies CD45 (LCA), CD45RO (UCHL1), CD30 (Ber-H2), Bcl2 (124), Bcl10 (151), MUM1 (MUM1), vimentin (V9), CD34 (QBEND10), p53 (CM-1), and Ki-67 (MIB-1) did stain variable proportions of the ferret lymphomas and could potentially be useful markers for diagnostic or prognostic evaluation of ferret lymphomas. Bcl2 (124), Bcl10 (151), and p53 (MH-1) did produce levels of staining in both normal lymphoid tissue and B- and T-cell lymphomas that could be interpreted as nonspecific staining. Further studies including whole section tissues would be necessary to evaluate the specificity of this staining.
In conclusion, the results of this study demonstrate a high reliability of tissue array–based immunohistopathologic stains of ferret lymphomas. The use of 2 1-mm-tissue cores is preferable to 1 core. The use of multiple cores keeps the number of lost cases as low and concordance rates as high as possible. Previous studies of human lymphomas support the need for core sampling redundancy, that is, the inclusion of duplicate cores from the same cases or the construction of duplicate TMA blocks.12,17 Duplicate core biopsies of 1-mm-diameter taken per specimen provided a reliable system for large-scale analysis of ferret cancer tissues on TMAs without compromising the efficiency of the array technology, and the availability of this tool should encourage veterinary pathology institutes to keep records and archives of valuable paraffin-embedded tissue samples. Several primary antibodies did stain variable proportions of the ferret lymphoma tissue and could potentially be useful markers for diagnostic or prognostic evaluation of ferret lymphomas, although further studies including whole section tissues would be necessary to evaluate the usefulness of these markers.
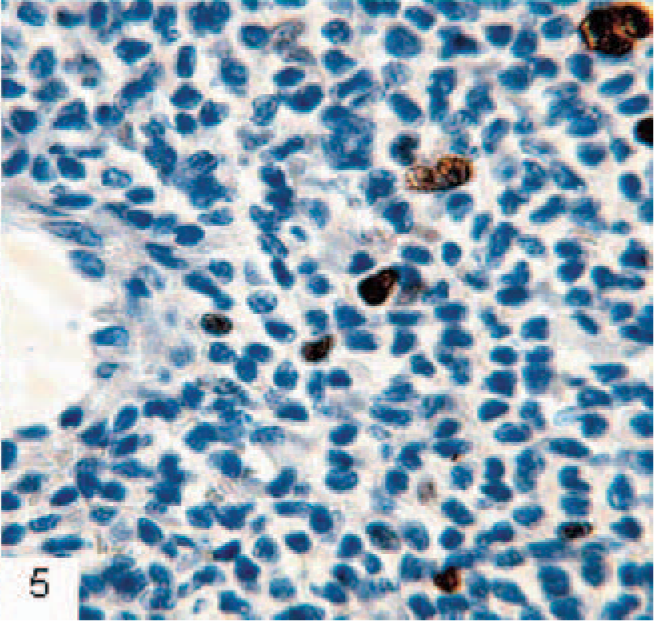
Skin; ferret. Immunohistochemical demonstration of the proliferation marker Ki67 show positive nuclear staining. The proliferation index is low. Monoclonal mouse antihuman Ki-67, MIB1, EnVision+ system (Dako, Glostrup, Denmark), counterstained in hematoxylin.
Footnotes
Acknowledgement
We thank Dako, Denmark, for sponsoring several primary antibodies applied in this study.
