Abstract
An 18-month-old Charolais cow developed depression and drooling and was submitted for necropsy after euthanasia. The cow was 1 of 50 moved between 2 farms approximately 5 days before the onset of clinical disease. Gross findings included swollen and hemorrhagic areas of malacia in the frontal, temporal, and parietal lobes of the cerebral cortex. Microscopically there was a necrotizing meningoencephalitis with intranuclear astrocytic and neuronal eosinophilic viral inclusions in the frontal, temporal, and parietal cerebral cortex as well as in the basal nuclei and thalamus. The gross and microscopic findings were consistent with necrotizing meningoencephalitis caused by bovine herpesvirus (BHV-1 or BHV-5), and the diagnosis was confirmed by detection of bovine herpesviral antigen on fresh samples of brain via fluorescent antibody test using a monoclonal antibody against BHV-1 glycoprotein C.
History, Clinical Findings, and Gross Changes
An 18-month-old Charolais cow with a 5-day history of depression and drooling was submitted for necropsy at the Laboratory of Veterinary Pathology (LVP), Universidade Federal de Santa Maria (UFSM), southern Brazil. The cow was 1 of 50 moved between 2 farms approximately 5 days before the onset of clinical disease. The owner reported that the clinical signs were severe and progressed rapidly. Because of the possibility of rabies, the referring veterinarian euthanatized the cow without completing a detailed physical examination and submitted the carcass for necropsy. The other animals did not show any clinical signs. Gross findings were restricted to the brain. The frontal, temporal, and parietal lobes of the telencephalic hemispheres were bilaterally swollen and soft, with flattening of gyri and extensive areas of leptomeningeal hemorrhages (Fig. 1). On cut surface, the frontal cerebral cortex contained extensive, dark brown and granular areas of hemorrhage and malacia (Fig. 2).
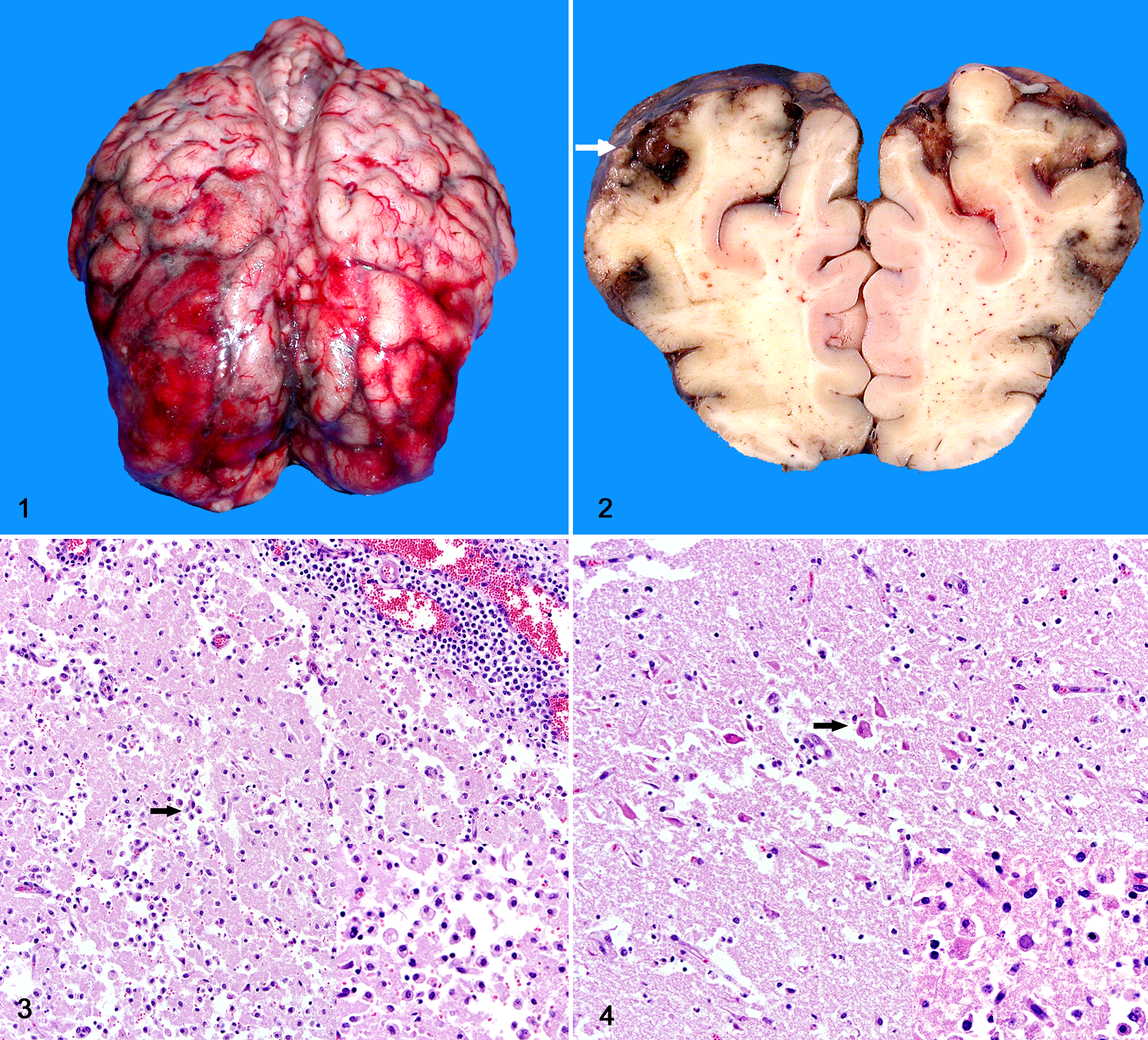
Brain; cow. The frontal, temporal, and parietal telencephalic lobes contain extensive, swollen, and soft areas with marked leptomeningeal hemorrhage.
Differential Diagnoses
Based on the geographic location and gross findings in this cow, the main differential diagnoses at the time of necropsy included bovine herpesvirus (BHV-1 or BHV-5) meningoencephalitis, polioencephalomalacia (PEM), and rabies. 7 Severe cases of PEM may rarely present with gross lesions that resemble those described for this cow. Although gross findings in cases of bovine rabies are usually absent, this viral infection is always considered to be a potential cause of neurological disease in Brazil until diagnostic confirmation is performed. 7
Microscopic and Laboratory Findings
Microscopic examination of the brain revealed extensive areas of necrotizing meningoencephalitis that were restricted to the frontal, temporal, and parietal cortex and less often to the basal nuclei and thalamus (Fig. 3 and 4). In these sites there were multifocal areas with loss of neuropil and cavitation, with accumulation of moderate to large numbers of foamy macrophages (gitter cells). The adjacent neuropil contained extensive areas of edema with astrogliosis and segmental areas of laminar neuronal necrosis where neurons exhibited hypereosinophilic and shrunken cytoplasm and pyknotic nuclei. Multifocally throughout the adjacent neuropil and neuroparenchyma, blood vessels had prominent endothelial cells and were surrounded by large numbers of lymphocytes and plasma cells that also extended to the leptomeningeal vessels. Occasional astrocytes and less often neurons contained intranuclear eosinophilic viral inclusions. Fluorescent antibody test (FAT) performed on fresh samples of brain were negative for rabies and positive for bovine herpesviral antigen using a monoclonal antibody against BHV-1 glycoprotein C.
Diagnosis
The diagnosis was focally extensive, bilateral necrotizing meningoencephalitis, with intranuclear eosinophilic astrocytic and neuronal viral inclusions due to BHV (1 or 5).
Discussion
The gross and microscopic findings in this cow are consistent with meningoencephalitis due to BHV-1 or BHV-5, and the diagnosis was confirmed by bovine herpesviral antigenic detection on fresh samples of brain via FAT. 2 , 7 , 8 The negative FAT for rabies and the distribution of the lesions in the brain of this cow allowed us to immediately rule out this viral infection. Bovine rabies usually causes a nonsuppurative meningoencephalitis that affects mainly the brainstem and cerebellum, and unlike what was observed in this cow, it spares the frontal areas of the brain. 7 Cases of PEM usually lead to extensive cortical neuronal necrosis but lack the severe inflammatory changes observed in this cow. 7
Necrotizing meningoencephalitis due to BHV affects mainly calves and young adults submitted to environmental or management-related stressors, including weaning, high concentration of cattle, transportation, and introduction of new animals into the herd.
2
,
7
This cow had been transported before the onset of clinical disease, which most likely provided close contact with other animals and might have facilitated infection or reactivation of a latent herpesviral infection.
5
,
6
BHV-1 and BHV-5 are genetically and antigenically related viruses that belong to the family
Necrotizing meningoencephalitis due to BHV has been described worldwide, particularly in South America. 2 , 7 – 9 The reason for the lower incidence of meningoencephalitis due to BHV in other parts of the world is unknown, but it has been proposed that widespread vaccination against BHV in North America and Europe protects susceptible animals and prevents clinical disease in these areas. 10 Viral transmission occurs via direct or indirect contact among susceptible individuals, with primary viral replication occurring in the ocular and oropharyngeal mucosal epithelium. 3 Following primary replication, viral particles reach the rostral portions of the brain and sensory ganglia via axonal retrograde transportation and direct invasion through the olfactory bulb and trigeminal nerves. 3 Viral invasion into the brain may result in secondary replication and neurological disease or subclinical infection and viral latency in the trigeminal ganglia and central nervous system. 5 Latently infected individuals will become an important source of virus to other susceptible cattle in the case of virus reactivation. 5 , 6 A hematogenous route of infection has been proposed by some authors, but it seems less likely due to the characteristic distribution of the lesions in the frontal areas of the brain, which support direct viral invasion through the olfactory bulb. 5 , 7
The clinical course in cases of meningoencephalitis due to BHV varies from 1 to 15 days, and affected cattle may develop a wide range of clinical signs such as severe depression, serous ocular and nasal discharge, grinding teeth, circling, blindness, incoordination, head pressing, nystagmus, recumbency, paddling, opisthotonus, and seizures. 2 , 7 , 8 The gross and microscopic findings reported in this cow are typical of those observed in cases of meningoencephalitis due to either BHV-1 or BHV-5. 2 , 7 , 8 The occurrence of intranuclear astrocytic or neuronal viral inclusions may vary among cases, but when present they are important in the presumptive diagnosis of bovine herpesviral infection. 7 In addition, we have observed cases of neurological disease by BHV with neither gross nor microscopic lesions despite the development of severe neurological disease in our diagnostic service. 7 , 8 These variations in the pathological presentation of neurological disease due to BHV are attributed to potential differences in the neurovirulence or to individual susceptibility of animals to viral infection. 1 , 8
Meningoencephalitis due to BHV-1 or BHV-5 shares many similarities regarding epidemiological and clinicopathological findings. 7 Although the history, clinical signs, and characteristic lesions in the brain may warrant a presumptive diagnosis of BHV infection in endemic areas, the diagnostic confirmation should be performed using FAT or viral isolation. 7 However, the differentiation between neurological infection caused specifically by BHV-1 or BHV-5 can only be achieved through the use of molecular ancillary tests, including a glycoprotein C–based polymerase chain reaction (PCR) performed on viral isolates from brain samples recovered from affected cattle. 9 Results obtained from studies using this method of differentiation in south Brazil showed that BHV-1 and BHV-5 may be interchangeably involved in very similar clinicopathological conditions that were previously attributed to a single strain (either BHV-1 or BHV-5). 7 , 9 Thus, the glycoprotein C–based PCR is a highly reliable test to be used in the viral identification and typification in cases of neurological, respiratory, or genital disease in cattle caused by BHV. 9 These tests are used only for research purposes in our diagnostic routine, and the confirmation of bovine herpesviral infection by either FAT or viral isolation is usually sufficient for confirmation of herpesviral infection (but not viral typification) in the diagnostic service.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
