Abstract
Environmental stresses can alter immunoreactivity of biomarkers in stored tissue sections. The effect of temperature and lighting on 49 cellular or microbial antigens was evaluated in 4 serial paraffin sections, cut 12 months, 10 months, 8 months, 5 months, 3 months, 1 month, 3 days, and 1 day before immunohistochemistry. Slides were stored at room temperature (RT) in the dark, at 4°C in the dark, at RT under fluorescent light, or at RT with windowpane exposure to sunlight. Immunohistochemistry was performed simultaneously in an automated immunostainer. Immunoreactivity was compared with that in the corresponding 1-day-old section and scored as 4 (<10% reduction), 3 (10%–25% reduction), 2 (26%–60% reduction), 1(>60% reduction), or 0 (no reactivity). Any loss of immunoreactivity was proportional to the tissue section age and was least in sections stored in the dark. Immunoreactivity was only completely lost in light-exposed sections and as early as 1 month for CD45. Other markers with complete loss of immunoreactivity were bovine viral diarrhea virus, CD18 (only with fluorescent light), CD31, CD68, canine parvovirus, chromogranins, and thyroid transcription factor-1. Markers with complete loss after light exposure also had reduced immunoreactivity when stored in the dark, as early as day 3. Eight markers (Bartonella spp, CD11d, high molecular weight cytokeratins, feline coronavirus, GATA-4, insulin, p63, progesterone receptor) had minimal decrease in immunoreactivity, regardless of treatment. In conclusion, light-induced antigen decay (tissue section aging) is antigen dependent and could explain unexpectedly weak or negative immunohistochemical reactions in stored paraffin sections.
Keywords
Formalin-fixed, paraffin-embedded (FFPE) tissues are the standard archived specimen in pathology departments and are often the only materials available for immunohistochemistry and other morphologic techniques in hypothesis-driven studies. 14 With the advances in protein and nucleic acid recovery after formalin fixation, FFPE tissues have been used since the mid-1980s in biomarker discovery studies, with variable success, to improve prognostic and clinical outcome of certain diseases.* Immunohistochemical detection of biomarkers can be affected by multiple factors involved in tissue processing, 11 although most studies have focused on the effects of fixation. With formaldehyde fixatives, biomarker degradation is proportional to fixation duration but is also antigen dependent. 37,56,57 Tissue section storage is a common practice in many pathology laboratories. It improves efficiency, quality, and consistency of tissue sections, particularly when using valuable paraffin blocks such as those containing tissue microarrays. 18,33,48,55 Degradation of immunoreactivity (also called epitope instability or antigen decay) in stored paraffin sections of human tissues is well documented. † In contrast, antigenicity is preserved at least for several decades in stored paraffin blocks; if antigen degradation does occur, it is at a very slow rate. 4,6,8,12,25,50,52 Similarly, preservation of nucleic acids in paraffin blocks is good for at least several years. 16
Targeted therapies in humans use the immunoreactivity score of biomarkers, such as epidermal growth factor receptor (EGFR), to evaluate drug efficacy for individual patients. Tissue sections stored for prolonged periods have a lower score for EGFR than fresh-cut sections, which affects the use of immunoreactivity scores for therapeutic planning. The reduction in immunoreactivity with storage depends on the type of neoplasm, 1 the fixative used, and the antigen of interest. 2,55 For EGFR, antigen decay is noticeable after 6 to 9 months storage of FFPE tissue sections; 1 however, antigens can decay in just a few days depending on the environmental conditions (eg, temperature, humidity) and marker examined. 3,5,30,54 Loss of immunoreactivity due to tissue section storage can affect antigens located in different cellular compartments (nucleus, cytoplasm, cytoplasmic membrane) and is directly proportional to the duration of storage. 1,2,55
The cause of loss of antigenicity (antigen instability) in stored tissue sections is uncertain, but oxidation and environmental temperature have been implicated. 3,54,58 Recently, endogenous water in paraffin blocks due to inappropriate processing or environmental humidity has been implicated as a cause of antigen degradation in tissue sections. 59 Published methods to reduce antigen decay in stored tissue sections include storage in a vacuum, covering slides with a layer of paraffin, addition of wax-soluble antioxidants to paraffin, storage of sections in a nitrogen chamber, immersion of deparaffinized slides in stabilizing solutions at 4°C, and optimizing fixation and antigen retrieval methods. 5,6,10,21,22,50,51
In our laboratory, inconsistent immunoreactivity of certain biomarkers in stored paraffin sections prompted an examination of the antigenic stability of 49 biomarkers (infectious agents and cell antigens) used in diagnostic pathology in FFPE tissue sections stored at room temperature (RT) or 4°C in the dark, or at RT with exposure to sunlight or fluorescent light.
Methods
The effects of tissue section storage conditions on the immunohistochemical detection of 49 biomarkers in use in the Purdue University Animal Disease Diagnostic Laboratory were evaluated. The markers included infectious organisms and tissue/cell antigens (Table 1). Nine markers were mostly located in the nucleus, 28 in the cytoplasm, and 12 in the cytoplasmic membrane. The same paraffin block with tissues fixed in buffered formalin for 1 to 7 days was used for each marker throughout the study.
Effect of Tissue Section Storage on Immunoreactivity.
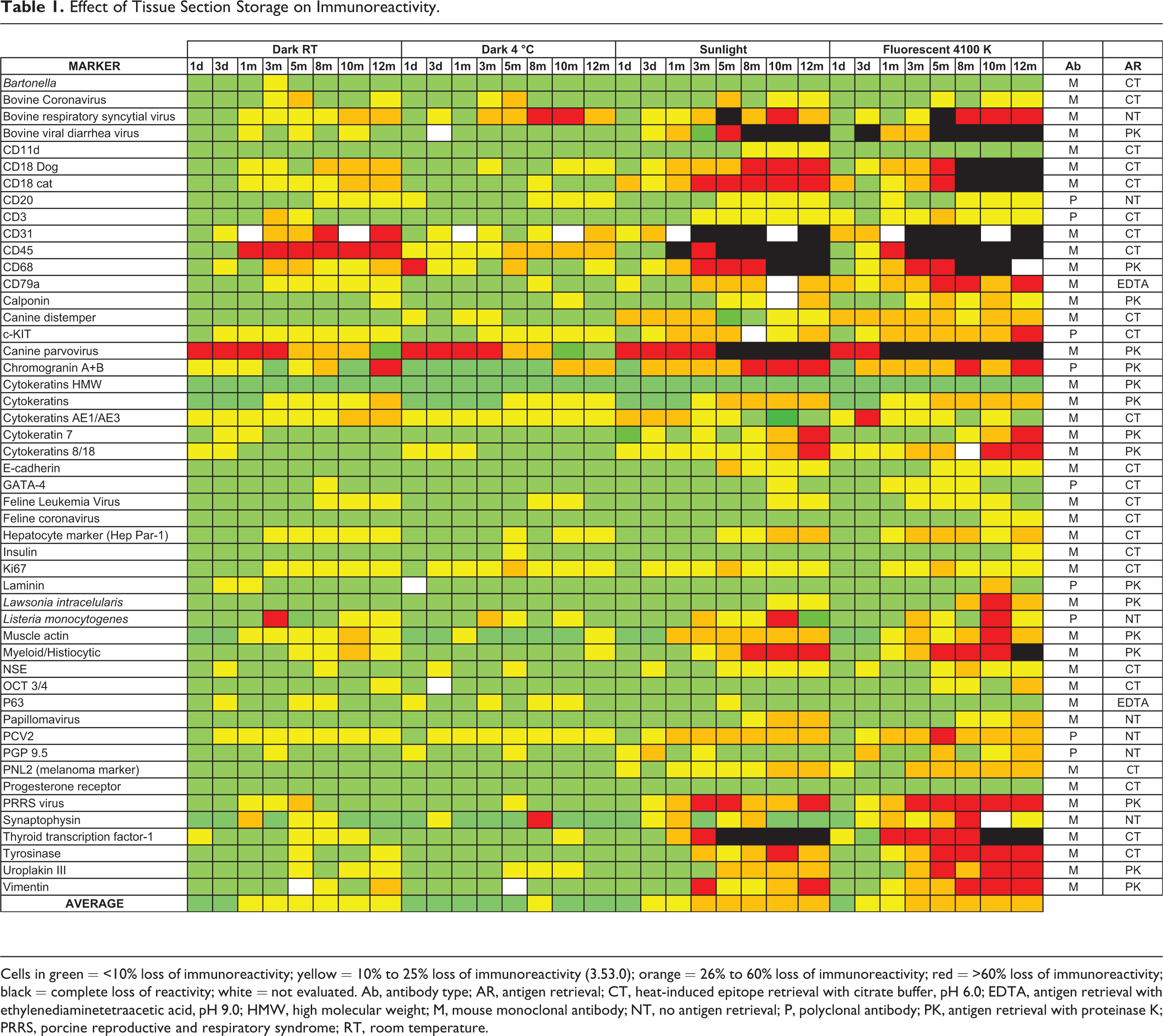
Cells in green = <10% loss of immunoreactivity; yellow = 10% to 25% loss of immunoreactivity (3.53.0); orange = 26% to 60% loss of immunoreactivity; red = >60% loss of immunoreactivity; black = complete loss of reactivity; white = not evaluated. Ab, antibody type; AR, antigen retrieval; CT, heat-induced epitope retrieval with citrate buffer, pH 6.0; EDTA, antigen retrieval with ethylenediaminetetraacetic acid, pH 9.0; HMW, high molecular weight; M, mouse monoclonal antibody; NT, no antigen retrieval; P, polyclonal antibody; PK, antigen retrieval with proteinase K; PRRS, porcine reproductive and respiratory syndrome; RT, room temperature.
Four sets of paraffin sections were produced for each marker, corresponding to the different storage conditions being evaluated (Fig. 1): (1) darkness (in a slide drawer) at RT (DARK RT), (2) darkness at 4°C in a sealed box (DARK 4°C), (3) exposure to sunlight (5200 K; SUN), and (4) exposure to fluorescent light (4100 K; FLUO). Slides exposed to sunlight through glass were placed in racks on a windowsill oriented to the west (6 months) and to the east (6 months); the daily sunlight exposure was not quantified. The slides exposed to fluorescent light were placed vertically in racks with the upper edge of the slides 5 cm from the fluorescent light bulbs. Slides were exposed to fluorescent light without interruption for the designated period. Tissue sections from the same paraffin block for each antigen examined were cut at 3 days, 1 week, and 1, 3, 5, 8, 10, and 12 months before the immunohistochemical procedure. A fresh section of each block was cut the day before (day 1) testing of each antigen. All tissue sections were placed in an oven and incubated at 56°C overnight the day before immunohistochemistry. Nine polyclonal antibodies and 40 monoclonal antibodies were used. Antigen retrieval with proteinase K was used for 16 antigens, heat-induced epitope retrieval (HIER) with citrate buffer for 24 antigens, HIER with EDTA buffer for 2 markers, and no antigen retrieval for 7 markers (Table 1). To minimize variation in immunoreactivity among sections, all sections corresponding to different time points of storage for each antigen were processed simultaneously 18 in an automated immunostainer (Dako, Carpinteria, CA) following routine diagnostic immunoperoxidase procedures in our laboratory. 19,32,38 –46

Three pathologists (J.D.W., M.A.M., J.A.R.-V.) reviewed all immunohistochemical preparations independently and scored the specific reactivity for each antigen. Immunoreactivity was scored against the day with the most intense labeling, which was generally day 1. The scoring system was based mostly on the number of positive cells: 0 (no reactivity), 1 (>60% reduction), 2 (26%–60% reduction), 3 (10%–25% reduction), and 4 (<10% reduction). In case of disagreement of more than 1 unit among the pathologists, the sections were reevaluated to reach a consensus. Otherwise, the final score was the mean of the 3 scores. Scoring was difficult with some sections subjected to fluorescent light exposure over 5 months due to tissue degradation that obscured the reaction and hindered accurate scoring, particularly with some of the infectious agents.
Results
Antigen degradation under the different storage conditions varied in severity among the biomarkers (Table 1). Any loss of immunoreactivity was proportional to tissue section age and was greater in sections exposed to light (SUN or FLUO) than those stored in the dark, with the exception of p63, in which sections stored in the dark (RT or 4°C) had a mild loss of immunoreactivity when compared with sections exposed to light (Fig. 3). When immunoreactivity scores were averaged for all the markers, SUN and FLUO sections had the highest loss of immunoreactivity, followed by sections stored in the dark at RT. Tissue sections stored in the dark at 4°C had the least loss of immunoreactivity. Immunoreactivity was eliminated completely only in light-exposed sections and was evident as early as 1 month of storage (for CD45). Other markers with complete loss of immunoreactivity by 5 months in either SUN- or FLUO-exposed sections were bovine viral diarrhea virus, CD18, CD31, CD68, canine parvovirus, and thyroid transcription factor-1. Canine and feline CD18 immunoreactivity was only abolished with FLUO treatment. Myeloid-histiocytic marker was not detected at the last time point (month 12) with FLUO treatment. Markers with complete loss of immunoreactivity after light exposure also had reduced immunoreactivity, albeit less severe, when stored in the dark, as early as day 3. Eight markers (Bartonella spp, CD11d, high molecular weight cytokeratins, feline coronavirus, GATA-4, insulin, p63, progesterone receptor) had minimal or no immunoreactivity loss during the study, regardless of treatment (Fig. 4).
When the effects of tissue section storage were compared by antibody type (monoclonal vs polyclonal), there was higher reduction of reactivity for antigens detected by polyclonal antibodies, particularly with FLUO treatment at 10 and 12 months. Sections stored in the dark (RT or 4°C) and incubated with polyclonal antibodies had slightly less antigen degradation.
Tissue sections exposed to light (SUN or FLUO) and treated with proteinase K as antigen retrieval had more pronounced loss of immunoreactivity than those subjected to other antigen retrieval methods or without antigen retrieval; this negative effect when using proteinase K was apparent after 1 month of storage and was severe by 10 months; FLUO sections were more affected than SUN sections. There were no appreciable differences among antigen retrieval methods for slides stored in the dark (RT or 4°C).
Antigen degradation due to tissue storage was somewhat dependent on the cellular location of biomarkers. Intranuclear antigens were more resistant to loss of immunoreactivity throughout the study even with exposure to light. Cytoplasmic membrane antigens were the most severely affected by tissue section storage, particularly those exposed to light (FLUO more severe than SUN), but also with sections stored in the dark at RT. Loss of immunoreactivity after several months of storage was also apparent with infectious disease markers in sections exposed to FLUO or SUN. Other storage methods had no apparent effect on immunoreactivity of infectious disease biomarkers.
Immunoreactivity among different tissues was examined for a few biomarkers (canine parvovirus, chromogranin A + B, high molecular cytokeratins, muscle actin, and synaptophysin). In general, the only biomarker for which antigen degradation varied among different tissues was chromogranin; pancreatic sections lacked immunoreactivity at the end of the study in slides exposed to light, whereas chromogranin was still detected, albeit very weakly, in the thyroid gland.
Baking slides at 56°C overnight (a standard procedure followed in our laboratory prior to immunohistochemistry) did not melt the paraffin in tissue sections (for all 49 biomarkers) exposed to SUN or FLUO light (Fig. 2). The lack of paraffin melting under high temperature was observed as early as 3 months of tissue section storage. In some cases, FLUO slides developed this change earlier than SUN slides. There was no correlation of this change with variation in the immunoreactivity among biomarkers. Paraffin sections stored in the dark did not become resistant to melting.
Discussion
Although it is well documented that various environmental conditions affect immunoreactivity in stored tissue sections, the ultimate mechanism of antigen degradation is unknown. Tissue section oxidation (chemical, thermal, or photonic) has been proposed to explain the loss of immunoreactivity. 3 This study confirms that light and temperature affect immunoreactivity of stored tissue sections for many biomarkers used in veterinary diagnostics. Whether antigen loss is the result of tissue oxidation or other chemical modifications in the tissue section could not be determined. However, in a previous study, adding antioxidant compounds to embedding paraffin did not block antigen degradation. 10 In our study, the antigen degradation was temperature and light dependent, with the most severe loss of immunoreactivity observed in tissue sections exposed to fluorescent light for more than 3 months, followed very closely by sections exposed to sunlight. However, loss of immunoreactivity was also dependent on the biomarker evaluated, as previously reported. 2,58 Furthermore, loss of immunoreactivity of a biomarker may depend on the environmental conditions. 3 Although antigen degradation is usually proportional to tissue section age, it can remain stable for several months, depending on the antigen. 54
Some results of this study differ from those in the literature. For instance, immunoreactivity of CD45 in stored sections at RT for several years was not affected in a previous study, 2 whereas in our study, CD45 tissue sections stored at RT in the dark (and less so at 4°C) had severe antigen degradation and complete loss of immunoreactivity in sections exposed to sunlight or fluorescent light for several months. Loss of reactivity for progesterone receptor (PR) has been reported in human tissue sections stored for 6 months. 13 In our study, loss of immunoreactivity for PR was not apparent under any of the testing conditions and time points evaluated. Multiple factors might explain such discrepancies, including the formulation of the fixative, tissue processing, temperature and other conditions of storage, antigen retrieval, tissue species, and antibody type. In other instances, our results were similar to those of others (eg, vimentin). 2 In a few cases (eg, canine parvovirus), paraffin sections on day 1 had lower intensity than those at later time points. The reason for that initial reduction of antigen detection was not determined.
Temperature is one of the contributing factors in antigen degradation of stored tissue sections. 3,18,21,28,31,35,54,58 Jacobs et al 18 also found reduced antigenicity in tissue sections stored at room temperature compared to those stored at 4°C. This loss of antigenicity was not only time and temperature dependent but also antigen and tissue/paraffin block dependent, as documented by the variable loss of reactivity of several biomarkers (p53, Bcl-2, estrogen receptor, and factor VIII–related antigen) among breast carcinomas. 18 Other authors have confirmed the antigen-dependent variation of immunoreactivity in stored tissue sections, even when stored at 4°C or –80°C. 15,28 In our study, temperature alone had little effect on immunoreactivity, with a slight reduction of immunoreactivity in sections stored in the dark at RT in comparison to storage in the dark at 4°C.
To our knowledge, only 1 study has focused on the effects of light on stored tissue sections. 3 Those authors observed loss of immunoreactivity in tissue sections exposed to ultraviolet light of 320 to 400 nm for many of the markers examined after as little as 3 days of exposure. Both cytoplasmic and nuclear markers were affected. In our study, loss of immunoreactivity occurred in sections exposed to fluorescent light (4100 K; 730 nm) or sunlight (5200 K; 580 nm), and it was proportional to the length of exposure. The blocking effect of UV light by the window glass was not evaluated in our study. In the study by Blind et al 3 and our studies, there was antigen decay for CK7 and c-kit; however, we did not observe loss of antigenicity for progesterone receptor and p63, which has been reported to occur under UV exposure conditions. 3
In this study, polyclonal antibodies incubated in tissue sections exposed to light were more sensitive to loss of immunoreactivity than were monoclonal antibodies. When comparing the effects of tissue storage on immunoreactivity of 20 monoclonal and 12 polyclonal antibodies, van den Broeck et al 54 found that both antibody types were similarly affected. Many of the antigens examined in this study were different from those examined by these authors, which might explain the observed differences.
Although originally, antigen degradation due to tissue section storage was observed with nuclear markers, 50 others have not reported major differences in loss of immunoreactivity based on the cellular location of antigens. 21,54 In this study, antigens located in the cytoplasmic membrane were more sensitive to antigen degradation when sections were exposed to light; nuclear antigens did not have marked loss of immunoreactivity even when sections were exposed to light for 12 months.
Several antigens (estrogen receptor, Ki67, cytokeratins, aquaporin 1) are very sensitive to loss of immunoreactivity under humid conditions; this loss can be observed as early as 3 days of storage. 10,59 Inadequate fixation or processing of samples promotes water retention in tissue blocks, which also negatively affects immunoreactivity. 59 Environmental humidity was not measured in the current study, but it was apparent that those samples exposed to daily environmental humidity (SUN and FLUO) had greater loss of immunoreactivity than those stored in the dark in a sealed box.
One unexpected finding in this study was the lack of paraffin melting after overnight baking slides at 56°C (Fig. 2). This happened only with slides exposed to light (sunlight or fluorescent light) for several months. Although antigenicity was preserved in many sections that lacked paraffin melting, it is uncertain if this could contribute to loss of antigenicity for some antigens. The cause for resistance of the paraffin to melting is unknown but could be the result of chemical changes upon exposure to light, increasing its melting point. Tissue damage was also noted in light-exposed tissue sections in the later time points of this study (10 and 12 months), which in some cases hindered evaluation of immunoreactivity. Therefore, the viability of the tissue sections, aside from preservation of antigenicity, is an important consideration for long-term storage of tissue sections. Although antigenicity may be maintained for some antigens, inadequate tissues for evaluation may become a more important limiting factor.
For most cellular markers, a modest loss of immunoreactivity may not have clinical significance, particularly if cut points for scoring the immunohistochemical reaction as positive have not been standardized in human or veterinary pathology. 13,28,47 However, in those instances in which the biomarker is present only in small quantities in a tissue or quantitative changes in the expression of the biomarker must be used for prognostic or therapeutic determinations, prolonged storage of tissue sections could produce a false-negative result. For those markers in which the percentage of positive cells or the intensity of the immune reaction is used to develop targeted therapies or in tumor grading, this loss may be important and unacceptable. 1,11
Modification of antigen retrieval or other immunohistochemical parameters (eg, prolonged incubation of the primary antibody) has been used to counter antigen degradation during tissue section storage. Mirlacher et al 28 suggested harsher antigen retrieval procedures to improve immunoreactivity. Standard antigen retrieval does not restore antigenicity in stored tissue sections; 54 however, more efficient antigen retrieval methods can restore antigen expression or reduce antigen degradation. 15,17,50,58 The effects of tissue section storage on antigen degradation can be partially or completely prevented or reversed if tissues are fixed optimally in formalin and/or subjected to an optimal antigen retrieval procedure or by increasing the concentration of the primary antibody. 21,22,31 In this study, we followed standard procedures developed in our laboratory for each biomarker and did not modify them when reduced immunoreactivity was observed. Antigens retrieved with proteinase K had more pronounced loss of immunoreactivity than those subjected to other antigen retrieval methods. Similarly, in previous studies, antigens retrieved with proteinase K tended to have reduced antigenicity following prolonged formalin fixation. 56,57 In combination, these results suggest that proteinase K digestion is a less reliable antigen retrieval methodology compared with HIER and therefore should be avoided whenever possible.
In summary, the results of this study of tissue section aging of formalin-fixed, paraffin-embedded tissues and 49 biomarkers used in veterinary diagnostics demonstrate that (1) tissue section storage has variable effects on immunoreactivity that are proportional to the duration of storage; (2) exposure of tissue sections to fluorescent light at room temperature reduced or abolished immunoreactivity of many biomarkers after several months and had the most deleterious effect of all storage conditions evaluated, followed by exposure to sunlight; (3) of the 4 storage methods, slides stored in the dark at 4°C had the least antigen degradation; and (4) the effect on immunoreactivity of stored tissue sections is not only temperature/light dependent but also antigen dependent.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Preliminary results from this study were presented as a poster at the 2011 annual meeting of the ACVP (abstract 144); December 3-7, 2011; Nashville, Tennessee.
*References 7, 9, 14, 23, 24, 27, 29, 34, 36, 49, 53.
†References 3, 5, 13, 18, 20, 26, 28, 30, 35, 54, 55, 59.
