Abstract
Molecular localization techniques remain important diagnostic and research tools for the pathologist evaluating nonhuman primate tissues. In situ hybridization and immunohistochemistry protocols have been developed for many important pathogens of nonhuman primates, including RNA and DNA viruses, prions, and bacterial, protozoal, and fungal pathogens. Such techniques will remain critical in defining the impact and relevance of novel agents on animal health and disease. A comparative pathology perspective often provides valuable insight to the best strategy for reagent development and can also facilitate interpretation of molecular localization patterns. Such a perspective is grounded in a firm understanding of microbe-host pathobiology. This review summarizes current molecular localization protocols used in the diagnosis of selected primate infectious diseases.
In laboratory-bred nonhuman primates (NHPs), infectious agents remain problematic and may jeopardize animal health, confound research, and/or pose a risk to personnel. 5,83,84 This is in contrast to many other species used in research in which pathogens are well characterized and controlled. Novel infectious agents continue to be recognized in zoological collections. Moreover, they are an important source of morbidity and mortality in wild primate populations. 8,57,96 The clinical consequence of infection with naturally occurring pathogens may vary from asymptomatic to severe disease, and it is important to define the full spectrum of agents within a population of animals to understand their respective effects on animal health and research.
Microbial surveillance programs to monitor animals and define infectious disease patterns have been described for facilities that house NHPs. 51 There are many elements to such microbial quality control programs, but at a minimum periodic physical examination, serological assessments, fecal examination, and intradermal tuberculin purified protein derivative (PPD) skin testing have been recommended. A vigorous diagnostic approach in animals with clinical disease, including necropsy and histologic evaluation of animals at the time of death, has demonstrated value. Molecular localization of infectious agents in tissue samples remains an important diagnostic tool for the pathologist evaluating tissues from NHPs in a diagnostic setting and in the implementation of disease surveillance programs. Many localization protocols have the added benefit of often allowing evaluation of formalin-fixed, paraffin-embedded (FFPE) samples when other biological samples are not available. Moreover, immunohistochemical and immunofluorescence assays may yield substantially faster results than traditional diagnostic techniques requiring culture and microbial identification.
New molecular techniques have now made it possible to more easily recognize novel infectious agents in primate populations, and these can now be incorporated into disease surveillance programs. 28 These approaches, based on next-generation sequencing (NGS) technology and bioinformatics analysis of generated data, are enhanced when coupled with histological assessment and molecular localization of the pathogen in tissue sections. It is anticipated that the number of previously uncharacterized agents recognized in NHPs will rapidly expand as use of this technology increases, and integration of the results with anatomic evaluation will be critical in determining the role of novel pathogens with disease causation. In many cases, traditional microbial isolation and cultivation with experimental inoculation and fulfillment of Koch’s postulates will not be feasible. In such instances, a suspected relationship between the presence of the putative etiologic agent and a specific disease entity can be strengthened by co-localization of the pathogen anatomically to tissues with morphologic evidence of injury and inflammatory responses. 28
Molecular Assay Characteristics and Techniques
Sensitivity and Specificity of Molecular Localization Tools
As diagnostic tools, molecular localization techniques will vary in their sensitivity and specificity. The sensitivity of the approach will depend in part on tissue pathogen burden. Consequently, the appropriate approach may vary depending on whether one is attempting to diagnose infection or disease caused by a pathogen. For example, while detection of lymphocryptovirus (LCV)–induced lymphoma in simian AIDS by immunohistochemistry for Epstein-Barr nuclear antigen 2 (EBNA2) has high sensitivity, it will lack sensitivity in detecting asymptomatic infection in immunologically normal animals. 14 This is likely due to low viral replication and pathogen burden associated with viral latency in the latter. Thus, use of molecular localization assays in a diagnostic approach must be integrated with an understanding of the biology of the host and pathogen itself. In many cases, a gold standard by which to gauge sensitivity of an assay will not be available. Qualitative or quantitative assessment of tissue pathogen burden by culture or molecular techniques may be correlated with staining patterns to estimate the relative sensitivity of molecular localization techniques.
Acquisition of known positive control tissue can be challenging, particularly for novel infectious agents with unknown tissue tropisms and disease effects. If such agents can be propagated in vitro, staining of infected and noninfected cell lines may be of assistance in assessing sensitivity and specificity of a given reagent. This approach may be particularly useful when screening antibodies for their potential reactivity, but optimized conditions for FFPE solid tissue samples often differ from those developed for similarly processed or unfixed cell pellets. If an infectious agent cannot be propagated, identifying tissues with high pathogen burden by polymerase chain reaction (PCR) and acquisition of tissues from animals during primary infection or immunosuppression may be helpful. NGS approaches provide the added benefit that generated sequence data can be used to rapidly design in situ hybrization (ISH) probes and develop immunological tools through use of recombinant DNA vaccine technology without having to isolate and propagate the microbe. 75 In these instances, tissues from which sequence data are generated can serve as a positive control for molecular localization studies.
A number of techniques may be used to assess the specificity of molecular localization techniques. As with all immunohistochemistry protocols, use of isotype-matched irrelevant antibodies or preimmune serum controls is essential. Sense and antisense probes should be used in conjunction for ISH protocols. For monoclonal antibodies with a known epitope or polyclonal antibodies raised against peptides, competitive binding experiments to abrogate staining may be of some value in assessing the selectivity of new reagents. In addition, tissues from animals known to be free of an infectious agent are critical negative controls. In the final assessment, there is no single standard control that can be used to assess assay sensitivity and specificity. Depending on previous experience and current knowledge of host-pathogen biology, multiple controls will likely be needed.
Cross-Reactivity of Molecular Localization Tools
Because of conservation of gene function between closely related microbes, immunologically based detection methods such as immunohistochemistry may lack the specificity to distinguish between closely related pathogens. This conservation of gene function and pathogen epitopes may be of value as antibodies raised against some human pathogens may be cross-reactive with the corresponding primate pathogens. For example, because of the close phylogenetic relationship between Epstein-Barr virus and rhesus LCV genotypes, some of the commercially available reagents to the former may work in detecting the NHP virus. 14 However, this cross-reactivity or lack of specificity may be a liability when distinguishing closely related viruses. For example, commercially available antibodies to herpesvirus simplex 1 (HSV1) cross-react with other simplex viruses in the Alphaherpesvirinae subfamily, including B virus and herpesvirus tamarinus (HVT). 5 Distinguishing among these viruses may have important implications in defining the source and management of the infection. If a more definitive diagnostic answer is required, immunohistochemical localization should be coupled with viral isolation and identification or molecular tools such as PCR and in situ hybridization, which can distinguish between closely related pathogens.
Target Selection and Protocol Development
Target selection is a critical element in designing molecular localization techniques for pathogen detection and should be based on a firm understanding of host-pathogen biology. If assays have been developed for related pathogens in other species, an initial assessment should be made as to whether these reagents are likely to be cross-reactive. Cross-reactivity will be dependent on the degree of homology between agents and the nature of the reagent itself. In the absence of suitable cross-reactive reagents, a number of other factors should be considered. These may include the stage of the pathogen to be detected, anticipated transcript or protein expression levels, epitope shielding and native confirmation, availability of recombinant proteins as immunogens, and whether the agent can be isolated and cultivated. While technically more difficult than immunohistochemistry, in situ hybridization may offer advantages in localization of novel agents when existing reagents and knowledge of host-pathogen biology is limited.
Viral Localization
Adenoviridae
Simian Adenoviruses
Adenoviruses are widespread in NHP populations and are commonly associated with infections of the respiratory and gastrointestinal tract. A recent NGS approach identified several novel adenoviruses in a closed rhesus macaque colony, and additional work will likely reveal tremendous genetic heterogeneity within this group of viruses. 28 Adenoviruses may cause disease in both immunologically normal and immunocompromised animals. In normal animals, primary infection is usually unrecognized or self-limiting in nature and results in animals being latently infected for prolonged periods or life. 95 Rarely, more severe or fatal forms may occur sporadically or produce epizootics of high mortality. 16,20 In immunocompromised animals, more severe forms may be recognized, including gastroenteritis, hepatitis, nephritis, or pancreatitis, particularly in neonates. 55,61 In the gastrointestinal tract, the pattern is often highly segmental, limited to restricted regions or anatomic structures in a viral genotype-specific fashion. In these cases, viral infection is observed in epithelial cells and can be associated with marked cytomegaly and the presence of intranuclear inclusion bodies, which vary in size and characteristics based on adenovirus genotype (Figs. 1–4). A mixed inflammatory cell infiltrate that can be primarily mononuclear or neutrophilic may be observed, although in cases of severe immunodeficiency, the inflammatory reaction may be minimal. On hematoxylin and eosin (HE) evaluation, the characteristic inclusions may be deeply basophilic, completely filling the enlarged nucleus (Fig. 2), or more difficult to recognize as small well-defined eosinophilic inclusions (Fig. 3). In some tissues, such as pancreas or liver, viral infection may produce a chronic-active inflammatory response with considerable pancreatic acinar cell necrosis and fibrosis present in a multifocal or locally extensive pattern.
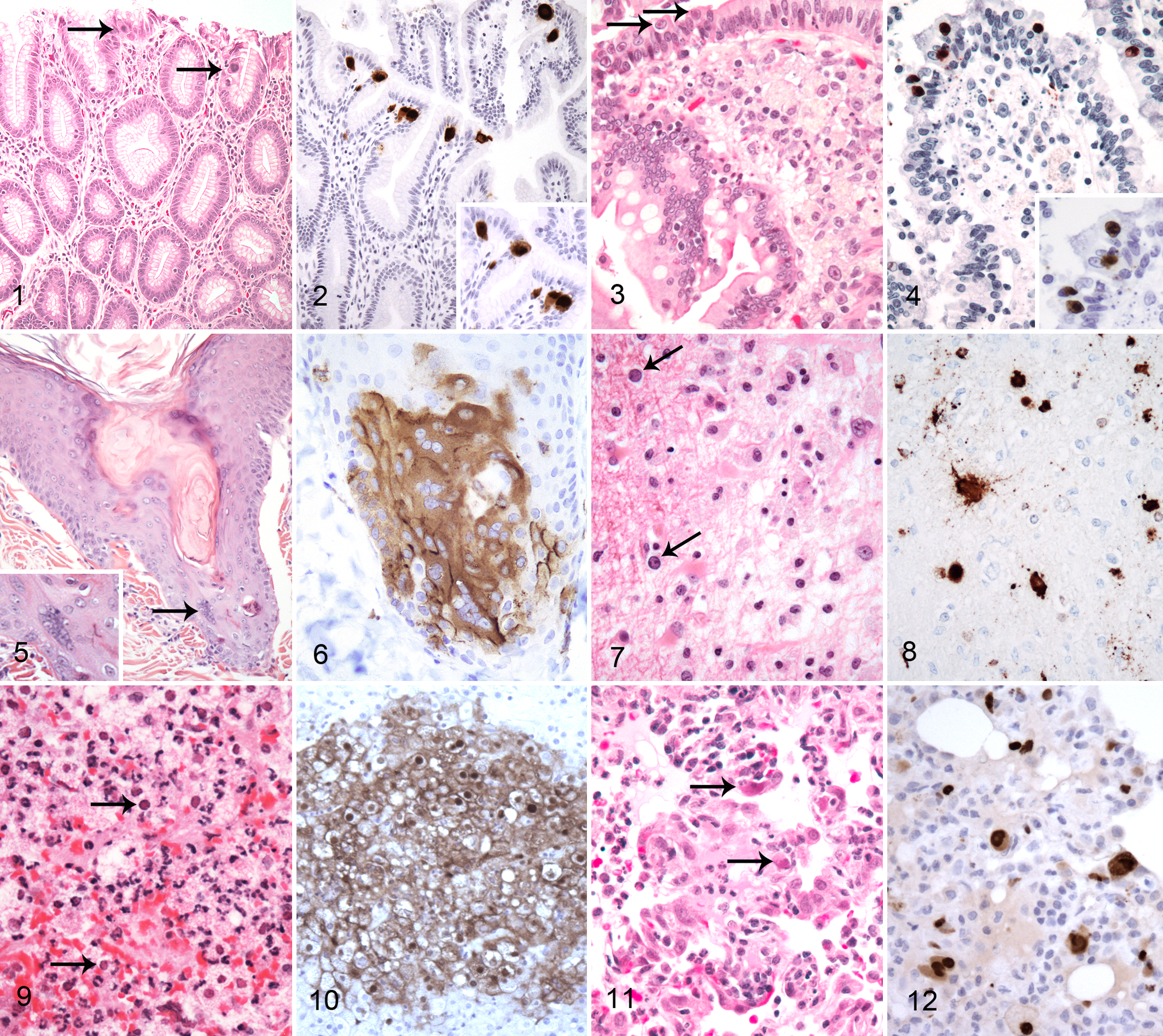
Differential diagnosis may include other viral infections associated with intranuclear inclusions, such as cytomegalovirus (CMV) and simian virus 40 (SV40), and immunohistochemistry may be used to distinguish these. A commercially available antibody directed against viral antigen Adeno3 appears to recognize all known serotypes of adenoviruses known to infect NHP tested to date, including those novel agents identified by NGS (Fig. 2). 28 While this reagent will help distinguish adenovirus infection from other viral agents, it will not differentiate among the different adenovirus strains. Additional virological methods such as viral isolation or PCR and sequencing may be required if further definition is required.
Paramyxoviridae
The Paramyxoviridae family contains a number of important pathogens of NHPs, including members of the morbillivirus, paramyxovirus, and pneumovirus genera.
Measles Virus
Measles virus is a morbillivirus within the Paramyxoviridae family and infects both humans and NHPs. 95 Despite an effective vaccine, measles virus remains common in some parts of the underdeveloped world and has made a resurgence in North America and Europe due to decreasing vaccination rates. Historically, many NHP colonies have been vaccinated, but in recent years, it has become difficult to source monovalent vaccine. In NHPs, the measles virus is transmitted from human contacts and may cause a severe epizootic disease characterized by a viral exanthema and bronchointerstitial pneumonia. Histologically, multinucleated viral syncytial cells with intracytoplasmic viral inclusions are evident in epithelium of the skin and respiratory tract (Fig. 5, arrow). Gastrointestinal involvement may occur and be most severe in New World primates (NWPs), where it may be accompanied by overt hemorrhage. Lymphoid depletion occurs and may be severe enough to allow the occurrence of opportunistic infections and impair immune responses that are typically used to diagnose other infectious diseases such as tuberculosis. 17
Viral isolation and serology are valuable in diagnosing measles virus infection and may be augmented by molecular localization assays to demonstrate viral antigen in affected tissues. This technique has found use diagnostically and as a research tool to understand viral pathogenesis. A commercially available antibody against the measles matrix protein demonstrates antigen in syncytial, epithelial, and immune cells (Fig. 6). 17,72,73
Influenza Virus
Influenza viruses cause frequent seasonal epidemics and less frequent pandemics worldwide and are a significant cause of mortality, particularly in the elderly or those with preexisting pulmonary dysfunction. Natural infection of NHPs has been infrequently described but likely occurs subclinically via close contact with infected humans. While infrequently associated with disease, NHPs have served as an important model to study the pathogenesis of influenza viruses, particularly those associated with virulent or pandemic potential in man. 79,80,82,97 The disease following experimental inoculation is dependent on the virulence of the strain used and may vary from a mild to severe bronchopneumonia. In chronic cases, septal fibrosis and type II pneumocyte hyperplasia have been described as well as multifocal squamous metaplasia of bronchiolar lining epithelium.
Immunohistochemistry has been developed using a monoclonal anti–influenza A nucleoprotein antibody in conjunction with an avidin-biotin complex (ABC) immunostain technique. Following experimental inoculation with the avian H5N1 virus, this technique demonstrated a granular staining pattern with greatest intensity in the nucleus and lesser intensity in the cytoplasm of bronchiolar epithelial cells. 79,80 The cytoplasmic immunoreactivity can show polarization to the apical membrane. Macrophages and mononuclear cells were also positive in regions with pneumonitis. A rabbit polyclonal antibody directed against an influenza A nucleoprotein produced similar findings following inoculation with H2N3 and H2N2 strains. 82 Immunohistochemistry could be considered a diagnostic tool in suspected natural cases but should be coupled with molecular analysis or viral isolation due to cross-reactivity of antibodies across multiple strains.
Polyomaviridae
Simian Virus 40
SV40 is a member of the Polyomaviridae and commonly infects several macaque species. 89 Serological surveys indicate frequent asymptomatic infection of juvenile macaques with the majority of animals acquiring infection by 2 years of age. While viral persistence is suspected, many elements of viral-host epizoology remain poorly described. This is somewhat surprising as SV40 has been used extensively in the laboratory as a tool to understand viral replication and life cycle strategies. While infection of immunocompetent individuals is asymptomatic, disease may occur during immunodeficiency and result in characteristic pulmonary, renal, or central nervous system (CNS) pathology. In the CNS, SV40 replication is associated with demyelination, gliosis, and the presence of poorly defined basophilic intranuclear viral inclusions and recapitulates features of progressive multifocal leukoencephalopathy (PML) in immunocompromised people (Fig. 7). 33,91 Similar inclusions are evident in SV40 nephritis and interstitial pneumonitis. These lesions arise independently of one another and are usually not all observed in the same animals.
The clinical presentation and light morphologic features of SV40 infection are usually sufficient to make a diagnosis, which can be confirmed by immunohistochemistry using a commercially available antibody against the SV40 large T antigen or to VP1 (Fig. 8). 33 In more subtle lesions, immunohistochemistry (IHC) may be of particular value as this technique will often reveal far more infected cells than appreciated by routine histological analysis. In addition, immunohistochemistry is useful in confirming the etiology of atypical SV40 lesions such as those observed in the lung, liver, or gastrointestinal tract. 47
Herpesviridae
The Herpesviridae family contains a number of important viral pathogens that can infect NHPs and can be subdivided into Alphaherpesvirinae, Betaherpesvirinae, and the Gammaherpesvirinae based on biologic and genetic features. Molecular localization can play an important role in the differential diagnosis of these infections.
Alphaherpesvirinae
The simplex viruses and varicella viruses are 2 genera within the Alphaherpesvirinae subfamily that cause disease in primates. HSV1 is the prototype simplex virus with which other NHP simplex viruses share biological properties. These viruses infect their natural hosts by the mucosal route, where they establish initial rounds of infection. Retrograde axonal transport eventually leads to lifelong viral latency in sensory ganglia, and this represents the site of viral persistence. While generally causing mild disease in the natural host, viral cross-species transmission to a nonadapted susceptible host may lead to severe disseminated disease as in the case of HSV1 transmission to NWPs or B virus (BV) transmission to a variety of species, including humans.
Simplex virus infections are characterized by vesiculation at oral, conjunctival, and genital mucosal surfaces, and cutaneous involvement is usually limited to the adjacent skin. In disseminated disease, as is observed following cross-species transmission, involvement of the CNS, liver, and lymphoid tissue is common. Occasionally, disseminated disease occurs without the characteristic mucosal lesions. Microscopically, extensive necrosis often with a minimal inflammatory reaction is observed along with multinucleated viral syncytia with intranuclear inclusion bodies (Fig. 9). A commercially available rabbit polyclonal antibody against HSV1 is cross-reactive among primate simplex viruses and can detect HSV1, BV, and HVT (Fig. 10). 90 The antibody reacts with all the major glycoproteins present in the viral envelope and at least 1 core protein. It is useful in confirming simplex virus infection but will not distinguish among the different viruses (unpublished data). Viral identification may be critical in assigning zoonotic risk and epizoological factors associated with infection. Molecular tools or viral isolation are required if definitive identification is needed. In the absence of lesions identified by light microscopy, the sensitivity of immunohistochemistry to detect infection is likely low.
Considerably less is known about the natural biology of simian varicella virus (SVV) infection, but this virus remains a significant pathogen of Old World primates and may cause sporadic or epizootic diseases of high morbidity and mortality. 26 Morphologically, the disease is characterized by a vesicular viral exanthema that may involve both haired skin and mucosal surfaces. In severe cases, the lesions often appear hemorrhagic due to viral infection of dermal or mucosal vessels. The viral infection may disseminate to multiple organs, including the gastrointestinal tract, liver, lymphoid tissues, and lungs. In the gastrointestinal tract, marked hemorrhage may be observed and rapidly lead to death.
As with other members of the Alphaherpesvirinae, multinucleated syncytial cells with intranuclear viral inclusion are evident. In general, the clinical presentation supported by viral isolation or molecular assays will enable a definitive diagnosis. Immunohistochemistry has been used to define viral distribution following experimental inoculation, but reagents are not commercially available. Rabbit polyclonal antisera against SVV Orf63 detected immunoreactivity in the cytoplasm of cells in the epidermis and trigeminal ganglion of animals following experimental inoculation. 63 This approach could be considered in cases in which fresh or frozen tissue is not available to assist with diagnosis. The commercially available HSV1 antibody is not cross-reactive with SVV epitopes and can be used to rule out simplex virus infections in the case of vesicular mucositis or dermatitis.
Cytomegaloviruses
CMVs are members of the Betaherpesvirinae and are known to infect a variety of nonhuman primates in a species-specific fashion. In immunologically normal animals, infection is common but overt clinical disease rare. In the context of immunodeficiency or immunosuppression, viral replication may increase and pathology may be observed in a number of tissues. Pneumonitis, encephalomyelitis, gastroenteritis, orchitis, neuritis, and vasculitis have all been described. 6,95 On routine HE evaluation, neutrophilic inflammation is commonly observed accompanied by cytomegaly and the presence of intracytoplasmic and intranuclear inclusion bodies (Fig. 11). While these morphologic findings are sufficiently characteristic for a diagnosis, they may be subtle in some cases, and immunohistochemistry is of value in increasing diagnostic sensitivity.
An antibody directed against the early intermediate protein of rhesus CMV has been developed and detects macaque CMVs (Fig. 12). 3 Positive signal is observed in many more infected cells than can be appreciated by routine stains, including stromal, endothelial, and inflammatory cells such as macrophages and neutrophils.
Lymphocryptoviruses
Lymphocryptoviruses are members of the Gammaherpesvirinae that induce frequent lifelong infections of many NHP species. As with other Herpesviridae, infection is common and overt disease rare. Serological surveys indicate that most macaques in a captive setting are infected as maternal immunity wanes by 1 to 1.5 years of age. Disease may occur in immunocompromised individuals, resulting in an epithelial proliferative disorder termed
Several tools developed for the detection of the human LCV virus (Epstein-Barr virus) are available and cross-react with the macaque LCVs detecting different viral stages. 54 A commercially available monoclonal antibody against EBNA2 detects latently infected cells and an antibody against BZLF1 detects lytically infected cells. While technically more challenging, ISH probes for Epstein-Barr encoded RNAs (EBERS) have also been developed and detect both latently and lytically infected cells. Some differences in the relative sensitivity of these approaches have been described for LCV-associated lesions, and generally a combination approach using antibodies that detect both latently and lytically infected cells is preferred. While these tools work well for rhesus and cynomolgus macaque LCVs, they do not appear to cross-react with the more distantly related NWP LCVs.
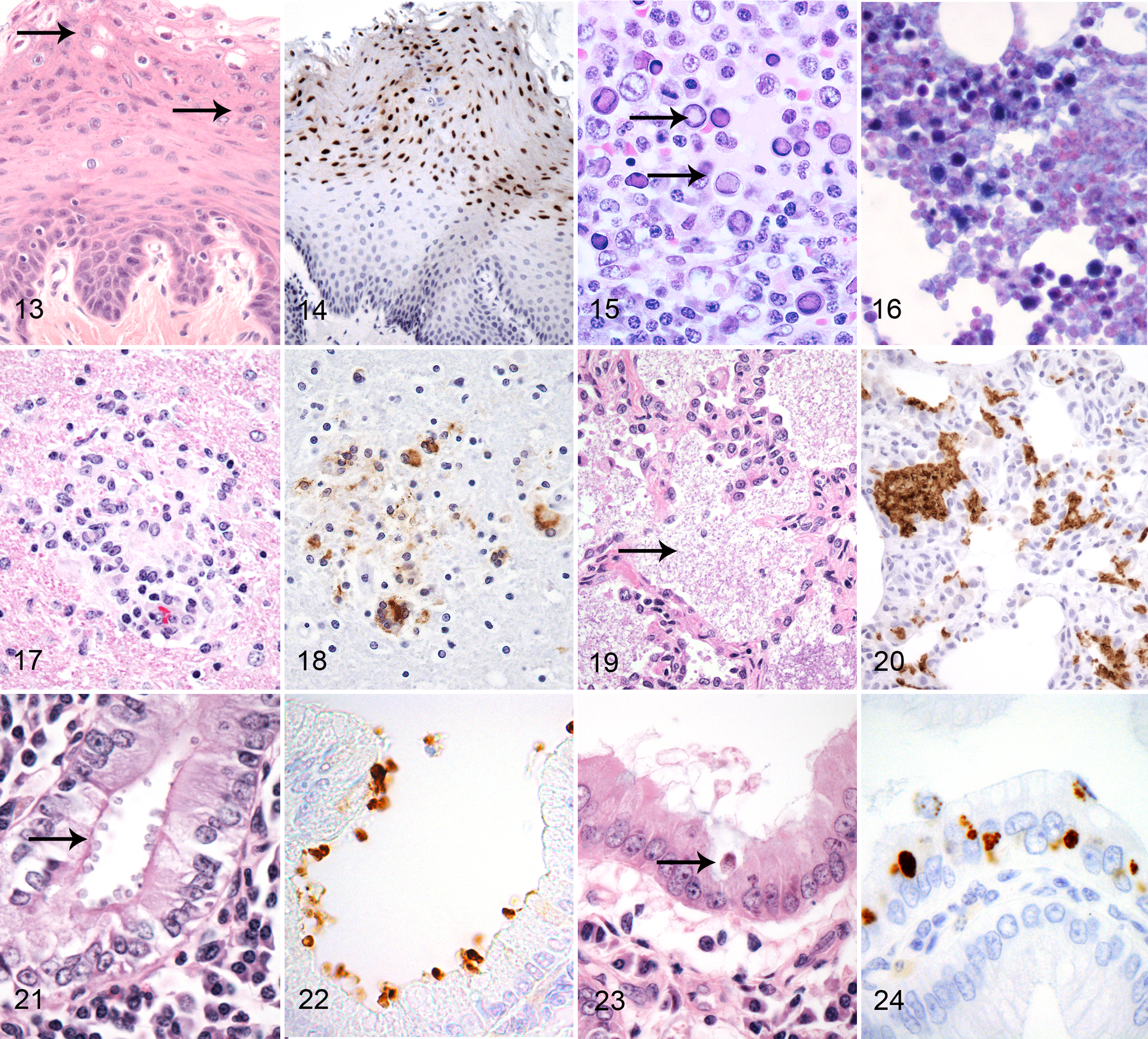
Leukoplakia is an epithelial proliferative disorder observed in immunocompromised rhesus macaques most frequently involving the oral and esophageal mucosa and less frequently haired skin of the face and genitals. 7 Microscopically, it is characterized by raised plaques of epithelial cells consisting of hypertrophy and hyperplasia of the stratum spinosum accompanied by hydropic degeneration. Intranuclear inclusions may be evident in some cells (Fig. 14) and are more easily detected by IHC. While usually asymptomatic in macaques, this lesion represents definitive morphologic evidence of immunodeficiency.
Several lymphoproliferative disorders have been recognized in macaque species in association with LCV infection, including malignant lymphomas in simian immunodeficiency virus (SIV)–infected macaques, posttransplantation lymphoproliferative disease (PTLD), and epitheliotropic T-cell lymphomas. 11,59,62,87 Molecular localization tools will generally reveal large numbers of latently and lytically infected cells in these lesions often associated with abundant noninfected CD4-, CD8-, and CD68-positive cells likely representing a host immune response to viral and potentially tumor antigens. While immunohistochemistry may not be required to make a diagnosis of lymphoma or PTLD, the observation of large numbers of LCV-infected cells supports a role for the viral infection in disease etiology and is consistent with underlying immunodysfunction.
Rhadinoviruses
Rhadinoviruses (RVs) are a second grouping within the Gammaherpesvirinae known to infect NHPs and have been divided into the RV1 and RV2 groupings based on biological behavior and phylogenetic relationship. 98 The RV1 grouping contains rhesus rhadinovirus (RRV) and the related macaque viruses and has been found to be widespread in captive macaque colonies. A monoclonal antibody against a viral interleukin 6 (IL-6) homolog has been developed and used to define pathogen distribution following experimental inoculation. 71 RRV produces a characteristic lymphadenopathy following experimental inoculation and has recently been associated with LCV-negative lymphomas in SIV-infected macaques. 11,21,52
Retroperitoneal fibromatosis herpesvirus (RFHV) is a second rhadinovirus found within the RV2 grouping and is closely related to the human Kaposi sarcoma herpes virus or human herpesvirus 8 (HHV8).
81,93
RFHV was first detected in archived tissues from animals with a mesenchymal proliferative disorder termed
Papillomaviridae
Papillomaviruses
Papillomaviruses (PVs) are a diverse group of viruses that infect a variety of vertebrates in a species- and tissue-specific fashion. Eighteen distinct genera have been described. Alpha-, beta- and gamma-papillomavirus genotypes are the most important in NHPs.
37
While initial infection by PVs occurs in the stratum basale of squamous epithelium, virion production occurs in mature keratinocytes (termed
Antibodies Used in the Molecular Localization of Infectious Agents of Nonhuman Primates.
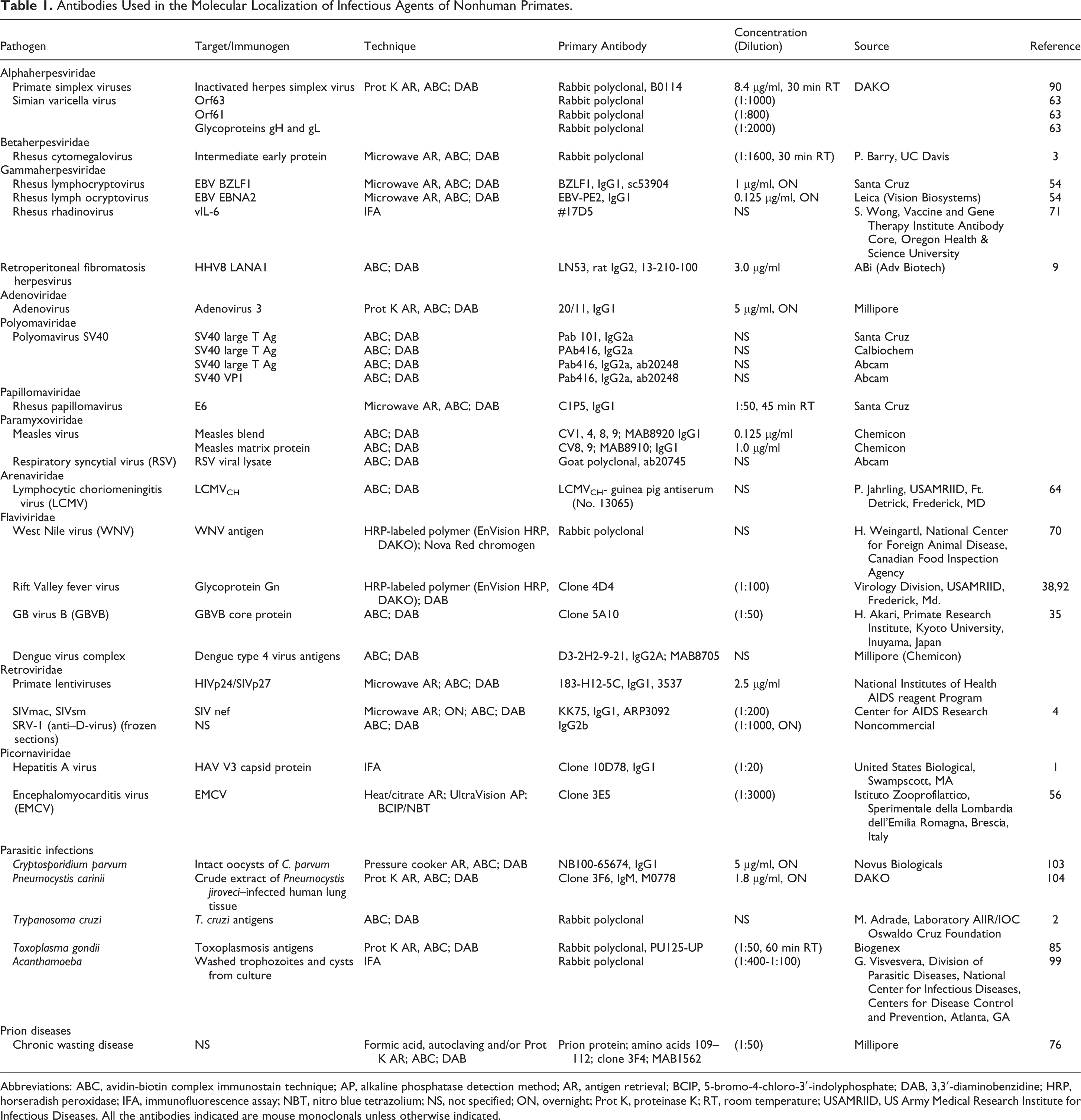
Abbreviations: ABC, avidin-biotin complex immunostain technique; AP, alkaline phosphatase detection method; AR, antigen retrieval; BCIP, 5-bromo-4-chloro-3′-indolyphosphate; DAB, 3,3′-diaminobenzidine; HRP, horseradish peroxidase; IFA, immunofluorescence assay; NBT, nitro blue tetrazolium; NS, not specified; ON, overnight; Prot K, proteinase K; RT, room temperature; USAMRIID, US Army Medical Research Institute for Infectious Diseases. All the antibodies indicated are mouse monoclonals unless otherwise indicated.
Since not all papillomas/fibropapillomas and carcinomas are caused by PVs, molecular localization tools can be used to define a viral etiology in clinical specimens. A number of antibodies have been developed to detect PV antigens in FFPE tissues, including rabbit polyclonal antibodies directed against bovine papillomavirus antigens and monoclonal antibodies against human PVs. Several of these antibodies were tested against a large panel of cervical and vaginal intraepithelial neoplasias from cynomolgus macaques 102 and cervical lesions in rhesus macaques. 30 The rabbit polyclonal antibody directed at bovine papillomavirus detected antigen in most infected tissues, while a monoclonal antibody directed against human papillomavirus E6 oncogene detected positive cells in cervical dysplasia and carcinoma lesions in rhesus. However, no single antibody detected all infected tissues, and definitive diagnosis of PV in lesions suspected of infection may require that multiple antibodies be tested.
Parvoviridae
Simian Parvoviruses
Simian and rhesus parvoviruses are erythroviruses related to human B19 virus. 68 The epizoology of these viruses in macaque populations has been poorly studied, but infection has been linked to severe anemias in animals with viral- or chemotherapeutic-induced immunosuppression. Accurate antemortem diagnosis is difficult as both viremic antibody-negative and nonviremic antibody-positive states have been recognized. In anemic animals, dyserythropoiesis along with intranuclear viral inclusions in bone marrow has been described (Fig. 15). In some cases, inclusions are accompanied by erythroid precursors with bizarre nuclear features, including nuclear blebbing and lobulation. No diagnostic antibodies have been described for immunohistochemistry, but both electron microscopy and in situ hybridization have been used to confirm the diagnosis. In situ hybridization often reveals far more infected cells than can be appreciated by routine stains (Fig. 16). 68,69
Arenaviridae
Lymphocytic Choriomeningitis Virus
Lymphocytic choriomeningitis virus (LCMV) is the etiologic agent of callitrichid hepatitis and has been shown to be transmitted to callitrichids through ingestion of infected rodents. 65 Macaques have also been shown to be susceptible to experimental inoculation. In common marmosets, hepatic involvement predominates with variable dissemination to multiple organs, including the spleen, lymph nodes, central nervous system, gastrointestinal tract, and adrenal glands. In the liver, multifocal random hepatic necrosis is observed with formation of Councilman-like (apoptotic) bodies and accompanied by inflammatory cell infiltrates. 64 Hemorrhage is often evident along serosal surfaces.
Immunohistochemistry performed with polyclonal guinea pig antisera raised specifically against an LCMV callitrichid isolate demonstrated reactivity in necrotic hepatocytes as well as some Kupffer cells and biliary epithelium of common marmosets. 64 In lymphoid tissue, antigen was detected in macrophages and fibroblastic reticular cells but not lymphocytes, suggesting that lymphoid depletion occurs through a bystander effect rather than direct infection of lymphocytes. Differences in LCMV distribution patterns have been observed dependent on the NWP species examined.
Flaviviridae
West Nile Virus
West Nile virus (WNV) is an arbovirus in the Flaviviridae family, which has recently been introduced to North America, resulting in a far-ranging epizootic of corvid birds. Culex mosquitoes are the principal vector of WNV, and infection of both birds and mammals may be clinically unapparent, produce a mild febrile illness, or result in severe CNS involvement and death. Serological surveys of outdoor housed primates in the southern United States have demonstrated that infection of macaques is common if exposed to the vector but that disease is rare.
77,78
Nonetheless, experimental inoculation of cynomolgus macaques can produce encephalitis, and fatal disease has been observed in a Barbary macaque (
Immunohistochemical techniques have been developed for several other members of the Flaviviridae, including Rift Valley fever virus and GB virus B.
35,92
GB virus B is a member of the
Retroviridae
Simian Immunodeficiency Virus
Simian immunodeficiency viruses (SIVs) are primate lentiviruses found in many species of NHPs throughout sub-Saharan Africa. 32 First recognized in rhesus macaques following inadvertent and unrecognized cross-species transmission from sooty mangabeys, SIV infection of macaques has been developed as an important animal model to study HIV pathogenesis and prevention. 18,19 While infection of the African natural hosts is largely nonpathogenic, cross-species transmission to Asian macaques leads to progressive loss of CD4 T cells and an acquired immunodeficiency syndrome, which recapitulates many aspects of human immunodeficiency virus infection of people.
Viral localization studies have been used to examine various aspects of the model and have been based on in situ hybridization and immunohistochemistry techniques. 45,105–107 Because of the relatively small size of the SIV genome, many in situ hybridization protocols have used plasmids spanning the length of the virus and have produced detailed information on viral localization throughout the course of primary and chronic infection. Morphologic staining patterns with in situ hybridization may vary depending on the stage of infection. Early in primary infection, a localized extracellular pattern may be observed in lymphoid tissue related to dendritic and fibroblastic reticular cell trapping of virions. 45 This is accompanied by staining of macrophages, lymphocytes, and dendritic cells with an intranuclear signal and less abundant cytoplasmic staining. As infection progresses, the extracellular pattern is lost, and there is often a decrease in the total number of infected cells identified. Later multinucleated syncytial cells will be positive and are associated with tissue-specific disease entities such as encephalitis (Fig. 17), pneumonitis, and lymphadenitis.
Several monoclonal antibodies have also been developed and can localize SIV antigens in FFPE tissues. An IgG1 monoclonal antibody (183-H12-5C) detects the gag protein of HIV-1 and HIV-2 (p24) and SIV (p27) and thus has broad reactivity with primate lentiviruses (Fig. 18).
Simian Retrovirus
Type D simian retroviruses (SRVs) were once common in domestically bred rhesus macaques and the most frequent cause of acquired immunodeficiency in this species.
31,41–43,86
Affected animals were viremic but variably seropositive, often developing progressive wasting, diarrhea, and opportunistic infections. Clinically, the disease overlaps with the condition caused by natural or experimental infection of rhesus macaques with SIVmac, but it differs in several important respects. SRV has a broader tissue tropism than SIVs and will infect T and B cells, macrophages, and epithelial cells. SRV viremic animals can develop opportunistic infections similar to that observed with SIV infection, including CMV and adenovirus. In addition, outbreaks of
Immunohistochemistry has been used to study the pathogenesis SRV infection and demonstrated infection of immune and epithelial cells. Infection of the CNS was limited to cells of the choroid plexus and found to be subclinical without an intrathecal immune response. 41
Picornaviridae
Encephalomyocarditis Virus
Encephalomyocarditis virus (EMCV) is a member of the Picornaviridae in the
Diagnosis can be difficult as morphologic features may be nondescript. Viral isolation or molecular techniques may be diagnostic if suitable tissue is available. Immunohistochemistry using FFPE tissue and a murine monoclonal antibody has been described and demonstrated EMCV antigen primarily within cardiomyocytes and less frequently within macrophages. 56 Localization of viral antigen to cardiomyocytes suggests that a direct cytotoxic effect of EMCV on these cells coupled with the host’s immune response leads to the observed fulminant cardiac failure.
Hepatitis A Virus
Hepatitis A virus (HAV) is a member of the Picornaviridae family/
Considerably less is known concerning the natural history of HAV infection of wild and captive populations of NHPs. Unrecognized HAV infection in macaques can result in elevations in LFTs that can confound or compromise experimental work. Diagnosis has largely relied on demonstrating recent seroconversion in animals. While a number of facilities require vaccination with an HAV vaccine licensed for use in humans, the efficacy of this vaccine in protecting animals against simian strains of HAV has not been rigorously evaluated and may complicate use of serology for diagnostic purposes. Immunohistochemistry on liver biopsy or necropsy material could be explored as a diagnostic tool in suspected cases with a history of vaccination or when serum samples are not available. 1
Poxviridae
Orthopoxvirus
The
The disease process in marmosets was recapitulated following experimental inoculation of animals, and viral antigen could be detected by immunohistochemistry using a human antivaccinia immunoglobulin. 60 This technique demonstrated viral antigen in the cytoplasm of epithelial cells, monocyte/macrophages, and endothelial cells and highlighted intracytoplasmic viral inclusion (Guarnieri) bodies. Similar techniques have been used to examine the distribution of monkeypox and smallpox viral distribution following experimental inoculation of macaques. 39,67 A rabbit polyclonal antibody directed against vaccinia virus demonstrated monkeypox viral antigen within necrotic foci surrounding bronchi and bronchioles. 67
Bacterial Localization
Helicobacter
A number of
Recently, in situ hybridization probes have been developed that can differentiate
Francisella tularensis
Tularemia is a zoonotic infectious disease caused by the Gram-negative coccobacillus
Rodents and lagomorphs are susceptible and the main reservoir hosts. In humans, ulceroglandular, glandular, oculoglandular, pharyngeal, typhoidal, and pneumonic forms have been recognized. Similar clinical entities have been described in a variety of neotropical primates as well as Old World monkeys. 23,27,74 Typically, acute multifocal hepatic necrosis with granulomatous inflammation in the lungs, spleen, lymph nodes, or gastrointestinal tract is observed at necropsy.
Culture and PCR have been used diagnostically, and special stains such as acid-fast and Giemsa stains may highlight the organism microscopically. Recently, immunohistochemistry was used on FFPE tissues from an epizootic in rhesus macaques. 58 Bacterial antigen was commonly identified in oropharyngeal lesions and in acute pulmonary perivascular lesions. It was often absent in more chronic lesions characterized by granulomatous inflammation.
Fungal Localization
Pneumocystis
Protozoan Localization
Cryptosporidium
Cryptosporidia are apicomplexa protozoal parasites that can infect a variety of vertebrates, including nonhuman primates. Cryptosporidiosis appears to be a common self-limiting infection of juvenile animals but may cause more severe disease in immunocompromised or stressed hosts. It often produces a necrotizing and proliferative gastroenteritis in which small 2.0- to 4.0-μm diameter basophilic bodies can be recognized along the luminal border of epithelial cells (Fig. 21). 6 The infection may extend into the liver, resulting in a sclerosing cholangiohepatitis with neutrophilic infiltrates and concentric fibrosis surrounding inter- and intralobular bile ducts. The biliary epithelium often appears attenuated and the number of organisms relatively sparse. Less commonly, the organism can disseminate to distant mucosal sites, including the conjunctiva, trachea, and vagina. 103
A commercially available antibody developed against intact oocysts of
Trypanosoma cruzi
Chagas disease or American trypanosomiasis is a zoonotic illness caused by the kinetoplastid protozoan
Toxoplasma gondii
Toxoplasmosis is caused by the apicomplexan parasite
Immunohistochemistry has been used to identify tachyzoites in an SIV-infected macaque with naturally occurring toxoplasmosis.
85
This technique may increase diagnosis sensitivity and help distinguish
Acanthamoeba
Free-living pathogenic amoebae belonging to the genera
Leishmania
Microsporidian Localization
Enterocytozoon bieneusi
Prion Localization
Transmissible spongiform encephalopathies are neurodegenerative diseases initiated by exposure to prion proteins and have been recognized in a number of mammalian species. Important prion-induced disease entities include bovine spongiform encephalopathy in cattle, scrapie in sheep, Creutzfeldt-Jakob disease in humans, and chronic wasting disease in cervids. While spongiform encephalopathy has not been recognized as a naturally occurring disease in NHPs, several NHP species have been shown to be susceptible to experimental inoculation with prion proteins by the intravenous and oral route and develop characteristic lesions within the CNS. Immunohistochemical protocols have been developed to localize prion proteins in such tissue. 76
Conclusion
Molecular localization techniques are an important diagnostic and research tool for the pathologist evaluating nonhuman primate tissues. This review, which summarizes current protocols and relevant host-microbial biology used in the diagnosis of viral, bacterial, parasitic, and prion-associated diseases, should aid the pathologist in defining the impact and relevance of selected agents on animal health and disease.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
