Abstract
Polyomaviruses produce latent and asymptomatic infections in many species, but productive and lytic infections are rare. In immunocompromised humans, polyomaviruses can cause tubulointerstitial nephritis, demyelination, or meningoencephalitis in the central nervous system and interstitial pneumonia. This report describes 2 Standardbred horses with tubular necrosis and tubulointerstitial nephritis associated with productive equine polyomavirus infection that resembles BK polyomavirus nephropathy in immunocompromised humans.
Polyomaviruses have been identified in humans, primates, laboratory rodents, cattle, wild mammals, and birds. In many species, latent infections are common, and disease from productive, lytic infections is not seen in immunocompetent individuals. In immunocompromised hosts, however, polyomavirus infections can be lytic or oncogenic. 6 Immunosuppressed humans latently infected with BK polyomavirus can develop BK polyomavirus nephropathy (BKN) with tubular epithelial cell necrosis, intranuclear viral inclusion bodies, and variable inflammation. 8 Recently, an equine polyomavirus has been identified, but no lesions associated with this virus have been described. 10 This report describes 2 Standardbred horses with a polyomavirus-associated nephritis resembling BKN with 1 horse exhibiting widespread viral distribution and myocarditis.
Case 1
A 2-year-old, female Standardbred horse from a racing training facility presented after several months of recurrent upper respiratory tract infections, anemia, mandibular lymph node abscesses, and relapsing fevers. The Coggins test, culture, polymerase chain reaction (PCR) for Streptococcus equi subspecies equi, and PCR tests for tick-borne agents were negative. During 6 weeks of hospitalization, the horse developed an immune-mediated hemolytic anemia (IMHA) and suspected vasculitis. In addition to antibiotics for the respiratory tract infection, the horse was treated with immunosuppressive doses of intravenous (IV) dexamethasone (40 mg/d with dose tapering as packed cell volume stabilized). Ultimately, the horse developed severe pneumonia, collapsed, and was euthanized.
A chemistry panel from the day prior to euthanasia showed potassium, serum urea nitrogen, and creatinine values within reference ranges. Immunoglobulin (Ig) levels were evaluated during hospitalization and at the time of euthanasia by radial immunodiffusion. IgG levels were within the reference range at both points. IgM and IgA levels were low during the peak of dexamethasone therapy, but both levels were within the reference range at euthanasia. Lymphocyte counts were low during dexamethasone treatment, but subtype distributions as assessed by flow cytometry were unremarkable.
Autopsy identified a severe pleuropneumonia with mixed bacteria that was interpreted as the cause of the animal’s collapse. Other lesions included rhinitis, mycotic gastritis, bilateral aural papillomas, and numerous erythrocyte and hemosiderin-laden macrophages in the liver, spleen, and lungs (supporting the clinical diagnosis of IMHA).
Subtle gross lesions in the kidney included patchy congestion and accentuated medullary rays, appearing as up to 0.15-cm-wide, gray streaks through the medullae and cortices. Histologically, marked injury to many distal tubules and collecting ducts was present with tubular dilation and enlarged and frequently sloughed, necrotic epithelial cells with large, basophilic, intranuclear inclusions (Fig. 1). Tubular injury was associated with interstitial edema and small to moderate numbers of neutrophils, macrophages, lymphocytes, and plasma cells. Interstitial fibrosis was minimal. Glomeruli infrequently exhibited mild, segmental hypercellularity with a few neutrophils and hemosiderin-laden macrophages and small foci of karyorrhectic debris. Rare cells in the mesangium contained smudgy, karyomegalic nuclei similar to the tubular inclusions (Fig. 2).
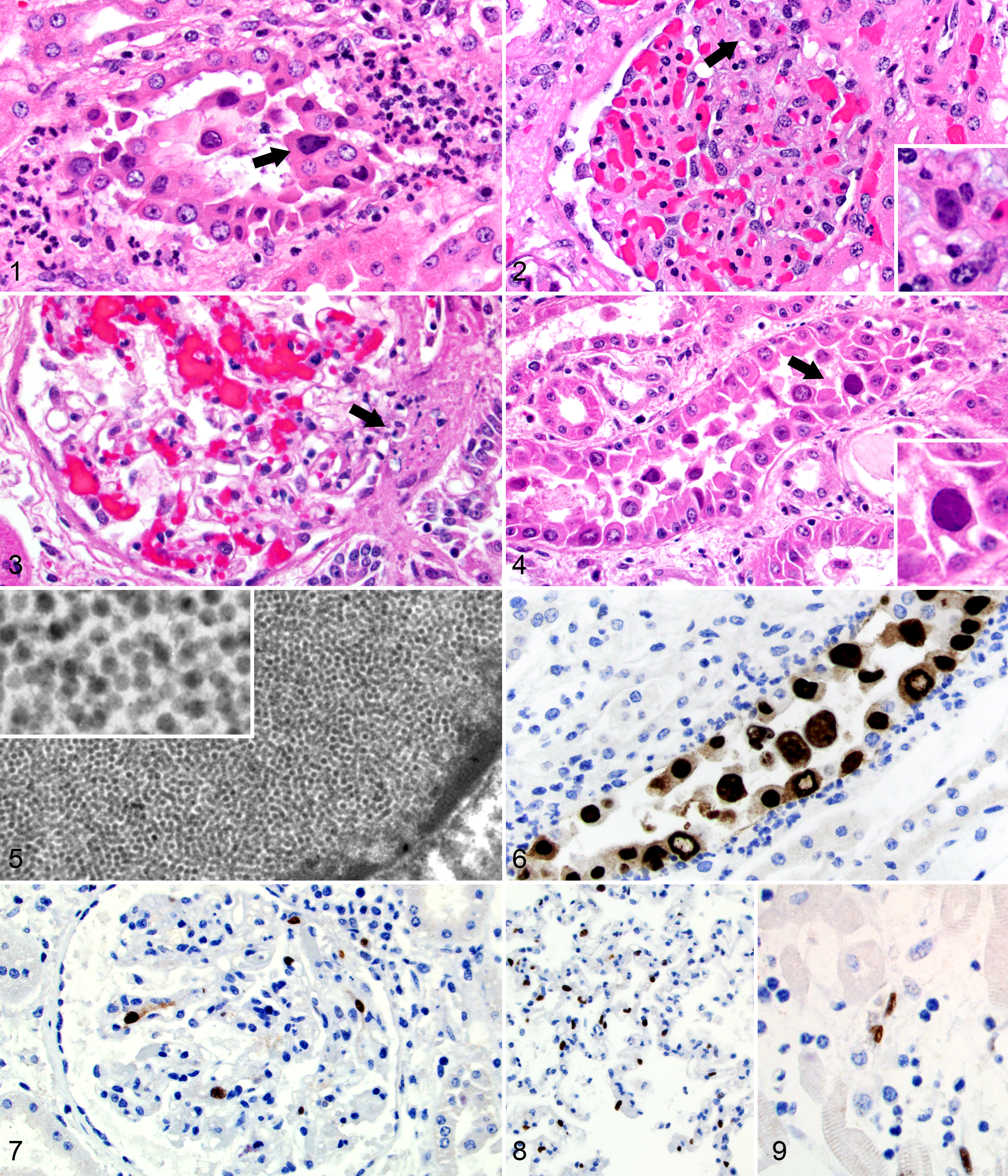
Kidney; horse No. 1. A collecting duct surrounded by neutrophils contains sloughed epithelial cells with some containing enlarged nuclei with intranuclear viral inclusion bodies (arrow). Hematoxylin and eosin (HE).
Case 2
A year later, a 2-year-old Standardbred gelding presented for lethargy, anorexia, fever, coughing, and mucopurulent nasal discharge. The horse was trained on the same track as horse No. 1; however, this horse did not arrive until at least 5 months after the death of horse No. 1, and the 2 horses had different trainers and were housed in different barns. The horse was diagnosed with bronchopneumonia due to Streptococcus equi subspecies zooepidemicus and hospitalized for 5 days. The horse was polyuric and polydipsic, and despite IV fluid therapy, the horse was mildly azotemic (serum urea nitrogen varied from 28–30 mg/dL with a reference range of 7–25 mg/dL; creatinine varied from 1.8–2.2 mg/dL with a reference range of 1–1.7 mg/dL). Immune-mediated vasculitis was suspected due to dependent edema. The horse was thrombocytopenic (52 × 103 platelets/μL) at presentation with mild improvement (83 × 103 platelets/μL) following prednisolone administration at immunosuppressive doses but subsequently worsened (33 × 103 platelets/μL). Prolonged bleeding followed transtracheal wash, and hematuria was noted at presentation. Intracavitary hemorrhage was suspected due to tachycardia and a decline in PCV (decreased from 31% to 18%) and plasma protein (decreased from 6 to 4.9 g/dL) over a 2-day period. A coagulation panel from 2 days prior showed only mildly prolonged prothrombin times (16.1seconds, reference range of 6.9–13.4seconds), but this panel was not repeated. Echocardiography revealed decreased cardiac contractility (fractional shortening 30%) and mild pericardial effusion, but troponin I levels were below the detectable limit (<0.2 ng/mL). On the fifth day of hospitalization, the horse collapsed and was euthanized.
Gross lesions suggestive of vascular injury and thrombocytopenia included petechiae and ecchymoses on serosal surfaces, 4 L of hemorrhagic abdominal effusion, hematomas in the splenic mesentery, diffuse splenic infarction, 4 L of serosanguinous thoracic effusion, and 2 L of straw-colored, watery pericardial effusion. The bronchopneumonia was confirmed, along with diffuse pulmonary edema with mild alveolar histiocytosis and increased numbers of circulating leukocytes. Aural papillomas were present bilaterally.
The capsular and cut surfaces of the renal cortices had numerous, pinpoint, red foci. Histologically, karyorrhectic debris often obscured the glomerular vascular poles (Fig. 3) and less frequently segments of glomerular tufts. Small interstitial aggregates of lymphocytes and macrophages were often clustered at the glomerular vascular poles. Glomerular intranuclear inclusions were not observed. Tubulointerstitial lesions, including the presence of intranuclear inclusions in collecting duct and distal tubular epithelial cells, were similar to horse No. 1 with milder inflammation (Fig. 4).
A moderate, multifocal myocarditis was present with edema, small amounts of karyorrhectic debris, mild infiltrates of lymphocytes and macrophages with fewer neutrophils, infrequent hemorrhage, and infrequent, enlarged, smudgy nuclei in hypertrophied stromal cells and capillary endothelial cells. Lesional myofibers were often atrophic or vacuolated with rare areas of extensive necrosis. Elsewhere, the myocardium contained broad bands of interstitial fibrosis.
Throughout the gastrointestinal (GI) tract, small arteries, capillaries, and small veins were frequently lined by hypertrophied endothelial cells with prominent nuclei and were cuffed by a few lymphocytes and rare neutrophils, but neither definitive intranuclear inclusions nor vasculitis was captured.
Virus Confirmation
Transmission electron microscopy (TEM) of the renal inclusions in both cases revealed myriad, 40- to 50-nm, icosahedral, nonenveloped viral particles consistent with a productive polyomavirus infection (Fig. 5). Two glomeruli were evaluated from horse No. 1, and no viral particles were observed. A wrinkled capillary loop in one tuft contained subendothelial electron-dense material of uncertain etiology and significance. Deposits suggestive of immune complex deposition were not observed in other loops to explain the glomerulonephritis seen histologically.
Immunohistochemistry (IHC) directed against the VP1 capsid protein of BK polyomavirus was performed on formalin-fixed, paraffin-embedded (FFPE) tissue sections of kidney from horse No. 1 with a mouse monoclonal antibody (noncommercial antibody, kind gift of Nickeleit laboratory). Strong viral antigen expression was identified in epithelial cell nuclei in injured collecting ducts and distal tubules and extracellularly in cast material (as expected for a capsid protein). Approximately 40% of glomerular tufts contained 1 to 3 karyomegalic cells with nuclear immunoreactivity. The identity of these cells was not clear.
IHC directed against SV40 large T antigen using a mouse monoclonal antibody (1:400 dilution, clone 416; Calbiochem, San Diego, CA) was performed on FFPE sections of kidney, brain, lymph node, lung, adrenal glands, skin, and GI tract from horse No. 1. In kidney sections, expression was similar to that seen with the capsid-directed antibody (Fig. 6) with the exception of no extracellular staining in casts (as expected for an early regulatory protein). In the intestines, rare, individual, vessel-associated cells in the lamina propria and endothelial cells in the submucosa exhibited strong, nuclear immunoreactivity. No viral antigen was detected in other tissues. The same IHC protocol was performed on sections of kidney, brain, lymph node, lung, heart, adrenal glands, skin, and GI tract in horse No. 2. In the kidney, lesional epithelial cells stained similarly to those in horse No. 1. Seventy to 80% of glomeruli contained at least 1 cell with antigen expression (Fig. 7). Numerous cells in alveolar septa (Fig. 8), myocardial interstitium (Fig. 9), and adrenal epithelial cells in the zona intermedia and deep zona glomerulosa exhibited strong, nuclear antigen expression. Viral antigen expression was seen in the nuclei of endothelial or other vessel-associated cells in the kidney, heart, lung, GI tract, liver, lymph node, adrenal gland, and brain.
PCR for herpesviruses and equine adenovirus-1 was performed as described previously with negative results. 4,14 Samples of formalin-fixed kidney from both cases were submitted to the Pennsylvania Animal Diagnostic Laboratory System for IHC directed against equine herpesvirus-1 and equine arteritis virus. Viral antigen was not observed in the kidneys of either horse.
A previously described, nested, broad-spectrum PCR that targets a ∼250-bp portion of the VP1 gene of polyomaviruses was performed. 7 A 212-bp sequence of the VP1 gene located close to the 3′ end of the gene was derived and sequenced. To allow for sequencing of the entire VP1 gene, PCR primers, VP1 MSU-f and VP1 MSU-r, were designed from this initial sequence and used with published degenerate polyomavirus primers, VP3-1f forward and T-2r reverse, 7 located upstream and downstream, respectively, of the VP1 gene region. Primer pair VP3-1f, 5′-CTCCAGGAGGTGCAMABCAAMG-3′, and VP1 MSU-r, 5′-GGTTCTTTACCCACCTCTTACGA-3′, was used to amplify a ∼1-kb product encompassing the remaining 5′ portion of the VP1 gene (∼700 bp) and a portion of the upstream gene, VP3. Primer pair VP1 MSU-f, 5′-TCGTAAGAGGTGGGTAAAGAACC-3′, and T-2r, 5′-AAAGGTCCAGTTAATAGTGGNAARAC-3′, was used to amplify a ∼1-kb product containing the remaining 3′ end of the VP1 gene (∼100 bp) and a portion of the T-Ag gene downstream. Products were purified and sequenced. Sequence assembly, open reading frame search, and multiple alignment of nucleotide sequences were performed. The entire coding sequence (1065 bp) of the VP1 gene of the equine polyomavirus was derived and compared with the VP1 sequences of 15 polyomaviruses from the GenBank database (Fig. 10). The highest percent identities were with bat and mouse polyomaviruses, at 61.1% and 62.7%, respectively.
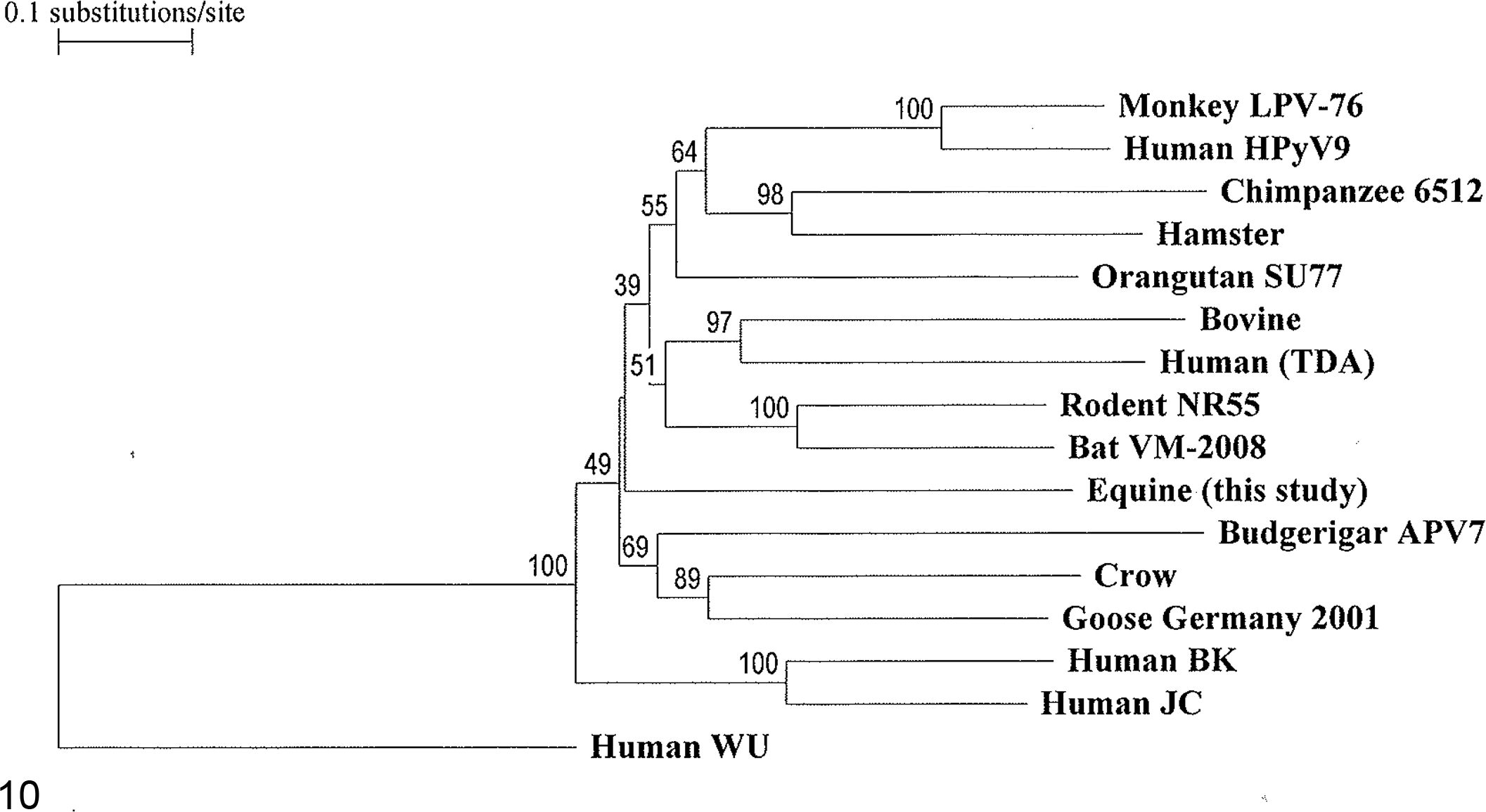
Phylogenetic tree based on the full coding sequence of the polyomavirus VP1 gene, showing the relationship of the newly identified equine polyomavirus to 15 previously reported polyomaviruses. A polyomavirus is designated with the species of origin and a virus or strain name when available in the GenBank database. The GenBank accession numbers are as follows: AB588640, Monkey LPV-76; HQ696595, Human HPyV9; HQ385748, Chimpanzee 6512; AJ006015, Hamster; FN356903, Orangutan SU77; NC_001442, Bovine; NC_014361, Human (TDA: trichodysplasia associated); AB588640, Rodent NR55; FJ188392, Bat VM-2008; AB453165, Budgerigar APV7; NC_007922, Crow; NC_004800, Goose Germany 2001; NC_001538, Human BK; AF281625, Human JC; and EF444549, Human WU, the outgroup sequence.
Discussion
The horses presented here developed tubulointerstitial renal lesions with intraepithelial nuclear inclusions consistent with a polyomavirus etiology. Although Koch’s postulates remain to be fulfilled, the restriction of epithelial injury to the distal tubules and collecting ducts and colocalization of viral cytopathic changes support a causative role for the virus. Epithelial injury at this location would be an atypical pattern for ischemic or most toxic insults. Ultrastructural identification of characteristic viral particles, molecular sequencing, and immunohistochemical labeling confirmed the presence of a polyomavirus. Additional IHC and PCR helped to exclude the contribution of other equine viruses. The deduced VP1 AA sequence of this virus was found to be identical to that of the equine polyomavirus sequenced by Renshaw et al. 10
The tubulointerstitial lesions in these horses bear a striking resemblance to BKN in humans 8 as well as its nonhuman primate counterparts caused by Simian virus 40 12 and cynomolgus polyomavirus. 13 Despite dramatic histologic lesions, the only evidence of renal impairment in either horse was mildly increased serum urea nitrogen and creatinine in horse No. 2. Had these horses survived longer, it is possible that renal failure would have eventually developed. Both horses had a mild, focal, segmental, glomerulonephritis with IHC evidence of the virus in the nuclei of many glomeruli. Although a causative role of the virus in inducing the glomerulonephritis is possible, only glomeruli in horse No. 1 showed histologic nuclear changes suggestive of viropathic effects. As neither viral particles nor definitive immune deposits were identified ultrastructurally within the 2 glomeruli examined from horse No. 1, the etiology of the glomerular changes remains unknown. In most cases of BKN, glomeruli are spared. 8 Infection of parietal epithelial cells and rarely podocytes has been documented in BKN, but infection of mesangial cells or endothelial cells within the glomerular tuft, as seen here, has not been described. 2
The difference in extent of extrarenal involvement between the 2 cases is striking. In horse No. 1, viral antigen expression was restricted to the kidney and rare cells within the GI tract. In contrast, horse No. 2 showed nuclear immunoreactivity in the majority of organs examined, including the heart, lung, GI tract, liver, adrenal gland, and brain, with antigen-expressing nuclei usually belonging to endothelial or other vessel wall–associated cells. In the heart, viropathic nuclear changes were associated with myocarditis. This cardiac lesion may have contributed to the animal’s collapse in combination with marked, intracavitary hemorrhage. Although multiorgan involvement is atypical in BKN, it is not unprecedented. Non–urinary tract lesions associated with BK polyomavirus include a fatal vasculopathy involving the heart, skeletal muscle, and esophagus; 9 interstitial pneumonia; 3,11 meningoencephalitis; 1 and retinitis. 1,5 The vascular, cardiac, and pulmonary lesions are most intriguing as these were major sites of immunoreactivity in horse No. 2.
Although infection and latency with BK polyomavirus are widespread in healthy humans, disease is only seen in patients with prolonged immunosuppression and typically in the context of renal transplantation or AIDS. 8 The prevalence of polyomavirus infection in the equine population is unknown and warrants future investigation. The role of an impaired immune response in the development of the lesions in these horses is also uncertain. In horse No. 1, 5 weeks of immunosuppressive corticosteroid therapy may have predisposed it to viral reactivation; however, in horse No. 2, corticosteroids were administered for a much shorter interval (days vs weeks). The possibility of an underlying immune deficiency in these horses was postulated but unproven.
These findings provide a pathologic entity associated with the recently reported equine polyomavirus. Productive polyomavirus infection should be considered in cases of tubulointerstitial nephritis in horses.
Footnotes
Acknowledgements
We are grateful to Sandra Horton, Laura Shewmon, and the rest of the histology laboratory staff at North Carolina State University College of Veterinary Medicine for their technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
