Abstract
Six captive maras (Dolichotis patagonum) were found to have cysts consistent with Besnoitia in the lungs by light microscopy and electron microscopy. Three were juveniles that died with no premonitory signs during a 17-month period. The most prominent finding consisted of severe, subacute, diffuse interstitial pneumonia with syncytia and Besnoitia cysts. The severity of pneumonia correlated with the number of cysts and the presence of lytic cysts, often surrounded by chronic granulomatous inflammation. Disseminated infection was observed in one of these maras. The three other maras died of unrelated conditions and had a few Besnoitia cysts in their lungs with mild or no inflammation associated. This is the first report of besnoitiasis in maras and of its association with fatal interstitial pneumonia in any species. Although other agents may have caused the interstitial pneumonia described here, it is possible that zoites released from lytic cysts were involved in the development of this lesion.
Keywords
Besnoitiasis is a protozoal disease caused by coccidia of the genus Besnoitia (family Sarcocystidae), characterized by an indirect life cycle with a definitive, carnivore host and an intermediate host (horses, ruminants, opossums, rodents, rabbits, and lizards). 1,5,6,8,9,11 This report documents for the first time besnoitiasis in maras (Dolichotis patagonum), which are large digitigrade rodents in the family Caviidae indigenous to Argentina. 10
Three captive-born, juvenile maras died unexpectedly with no premonitory signs in 1999–2000 at Africam Safari (Puebla, México). During this period, the mara colony consisted of 37 animals, with 28 births and 14 deaths (seven adults, four juveniles, and three neonates). Maras were provided daily with water ad libitum and a diet composed of orange, carrots, chopped alfalfa, and rabbit pellets. The enclosure was 120 m2, fenced with a 1.20-m-high wire mesh and bamboo on one side, and provided with a 15-m2 shelter. The substrate was sandy.
Mara No. 1 was a 2.9-kg, 2.5-month-old female of a triplet that died on June 1999, and mara No. 2 was a 1.7-kg, 1.3-month-old female of a duplet that died on September 1999; both animals died during the rainfall season and had been born to the same dam 4 months apart. Mara No. 3 was a 2-kg, 1.5-month-old female that died on December 2000. At the time of their death, there were four, five, and seven other juvenile maras, respectively; none of them or adult maras showed any sign of disease.
Gross findings consisted of good body condition (3/3), abundant froth in the tracheal lumen (3/3) that oozed from the nares in mara No. 1, diffusely reddened and edematous lungs that failed to collapse (3/3), multiple, tiny, whitish nodules on serosal and cut surfaces of the lungs (2/3) (Fig. 1) and visceral pericardium, diaphragmatic pleura and hepatic and renal capsule (mara No. 3), mild or severe bilateral serosanguineous pleural effusion (2/3), and reddish urine (mara No. 3; postmortem urinalysis revealed marked proteinuria and hematuria). Aerobic culture of the lung from mara No. 3 yielded Yersinia sp. Microscopically, mara Nos. 1–3 had severe, subacute interstitial pneumonia (Figs. 2–6) characterized by marked fibrin exudation into alveolar spaces; type II pneumocyte hyperplasia; interstitial infiltration of inflammatory cells (mostly macrophages); the presence of macrophages, lymphocytes, and syncytia within alveoli; and alveolar edema and hemorrhage. These lesions were less prominent and multifocal to coalescing instead of diffuse in mara No. 2. There were intralesional protozoal cysts up to 0.35 mm in diameter (most numerous in mara Nos. 1 and 3) with myriads of crescent-shaped bradyzoites in the cytoplasm of host cells that were hypertrophied, occasionally multinucleate, and surrounded by a thick eosinophilic capsule (Fig. 3). In mara Nos. 1 and 3, numerous cysts were surrounded by rings of inflammatory cells, mainly lymphocytes and macrophages; this lesion was most prominent around lytic cysts, which were invaded by inflammatory cells and fibroblasts (Figs. 3, 4). Mara No. 3 had similar cysts in the kidneys, associated with occasional large foci of granulomatous interstitial nephritis around lytic cysts and marked diffuse tubular reabsorption of proteins. The myocardium and skeletal muscle of mara No. 3 had similar cysts often surrounded by rings of lymphocytes and macrophages, fewer neutrophils, and occasional multinucleate giant cells. The pericardial, pleural, and peritoneal nodules described grossly in mara No. 3 corresponded to similar cysts with no or minimal surrounding inflammation. The esophagus, uterus, and lymph nodes of mara No. 3 had fewer cysts without surrounding inflammation. Lymph nodes had lymphoid depletion (2/3) or hyperplasia and histiocytosis with occasional syncytia (mara No. 1).
Lung; mara No. 3. Note the presence of multiple, tiny white nodules on the visceral pleura and the cut section. Formalin-fixed tissue. Bar = 5 mm.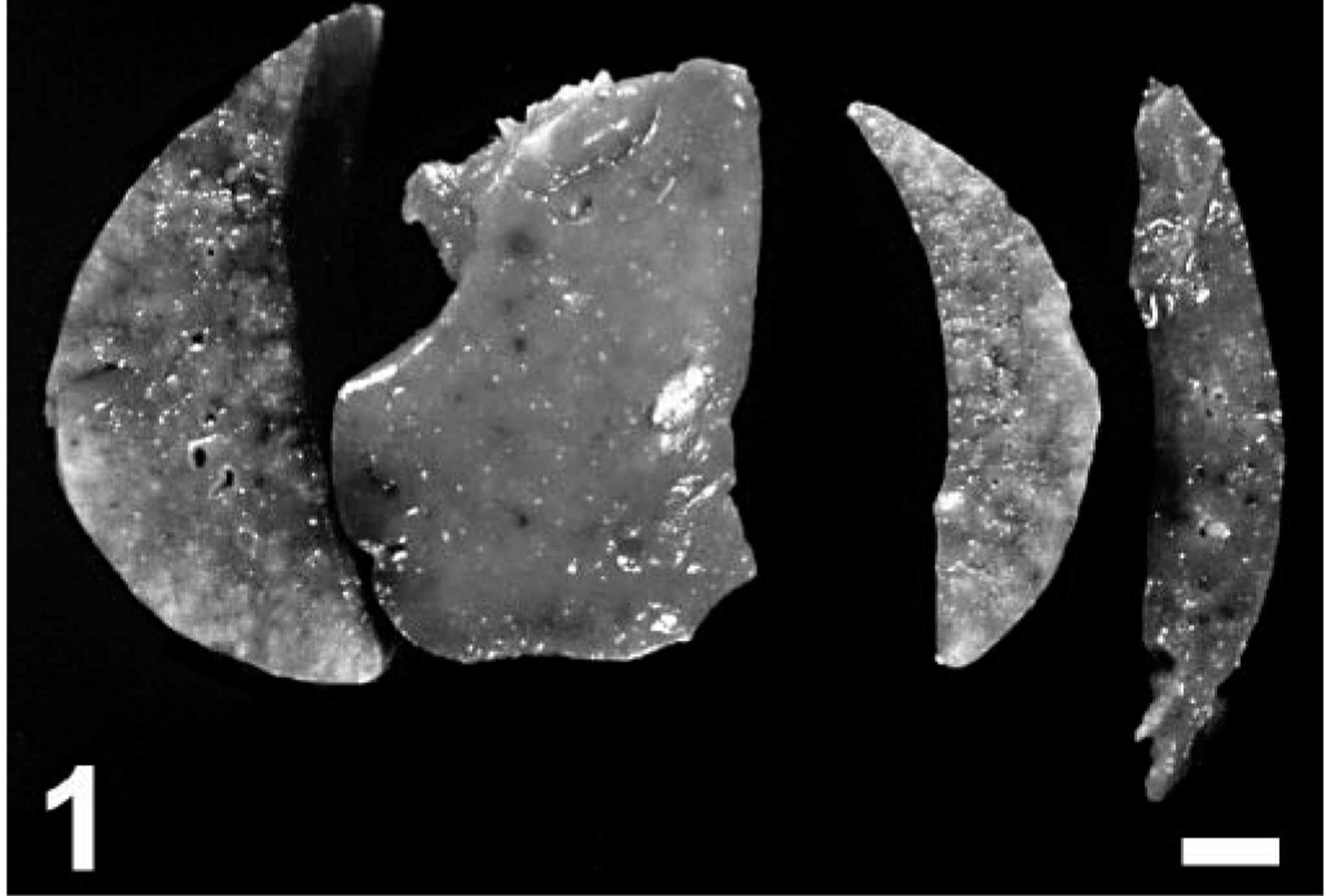
Lung; mara No. 3. Note severe, diffuse interstitial pneumonia with intralesional protozoal cysts (arrows). Hematoxylin and eosin. Bar = 335 µm.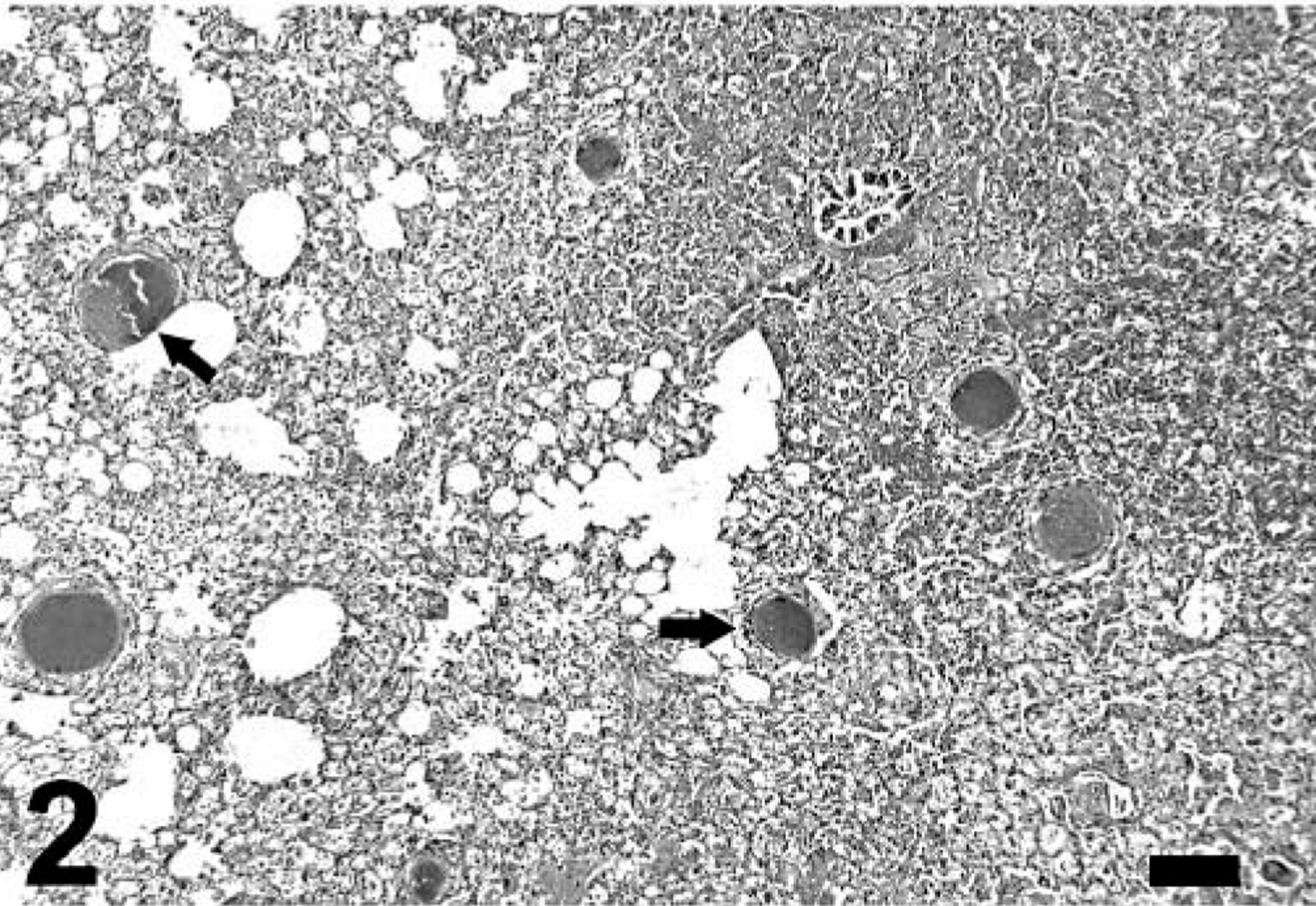
Lung; mara No. 3. Note severe interstitial pneumonia with two intralesional protozoal cysts; the smaller (long thin arrow) has numerous bradyzoites, and the host cell is hypertrophied, multinucleate, and surrounded by a thick capsule. The larger (short thick arrow) is lytic and surrounded and invaded by numerous inflammatory cells. Hematoxylin and eosin. Bar = 140 µm.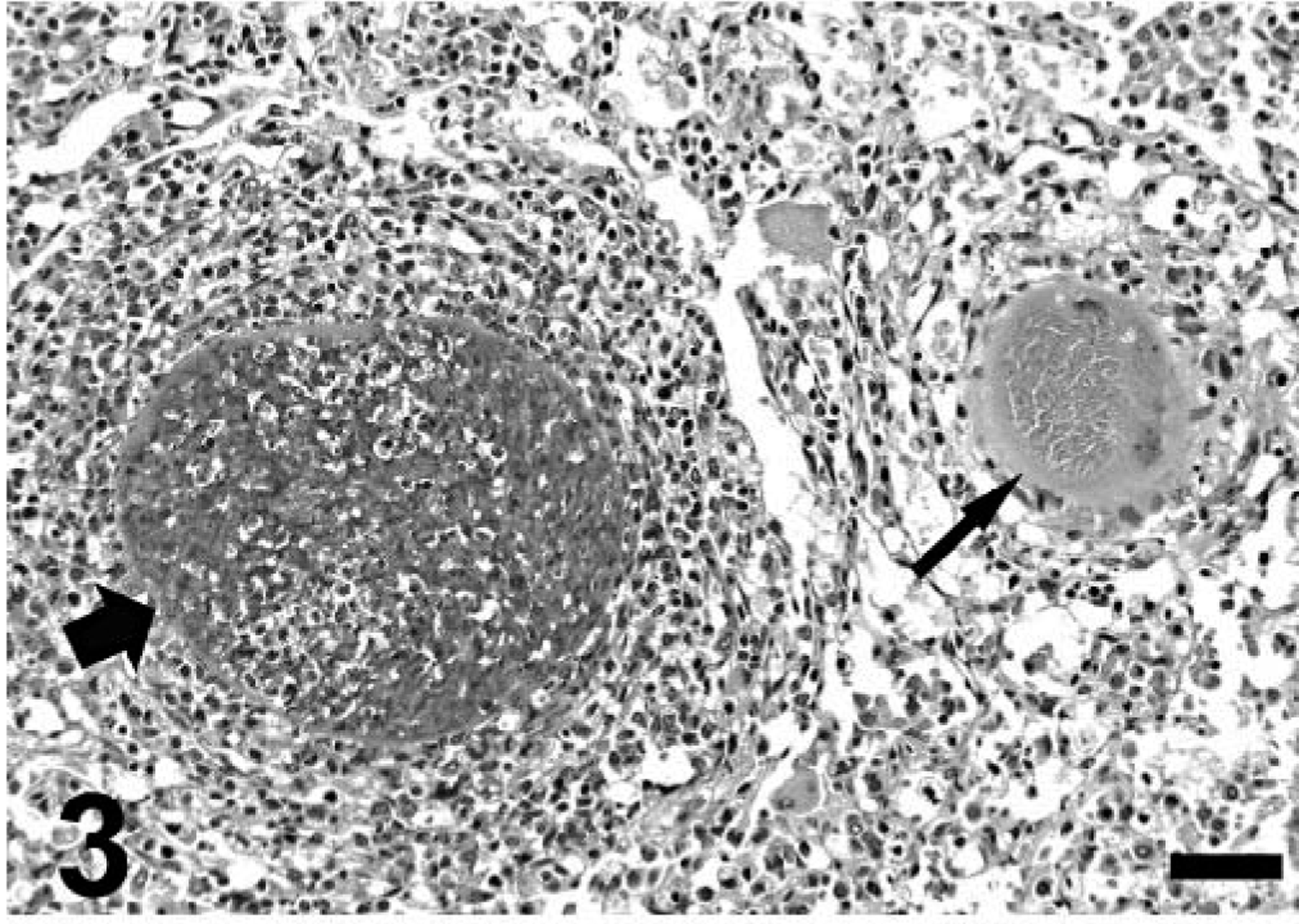
Lung; mara No. 3. Higher magnification of the lytic cyst depicted in Fig. 3. Hematoxylin and eosin. Bar = 95 µm.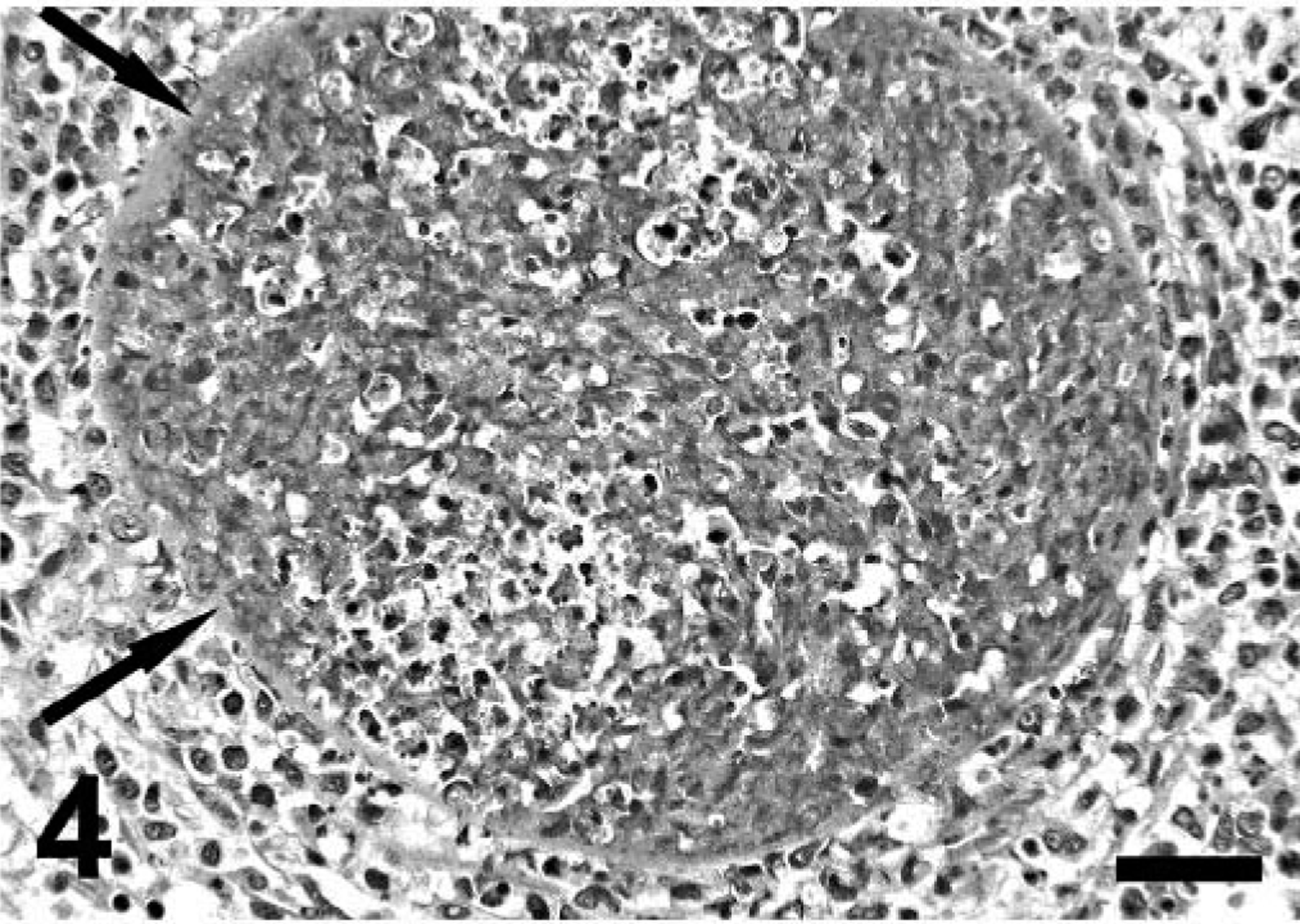
Lung; mara No. 3. Severe interstitial pneumonia with numerous macrophages and two syncytia in alveolar spaces. Hematoxylin and eosin. Bar = 75 µm.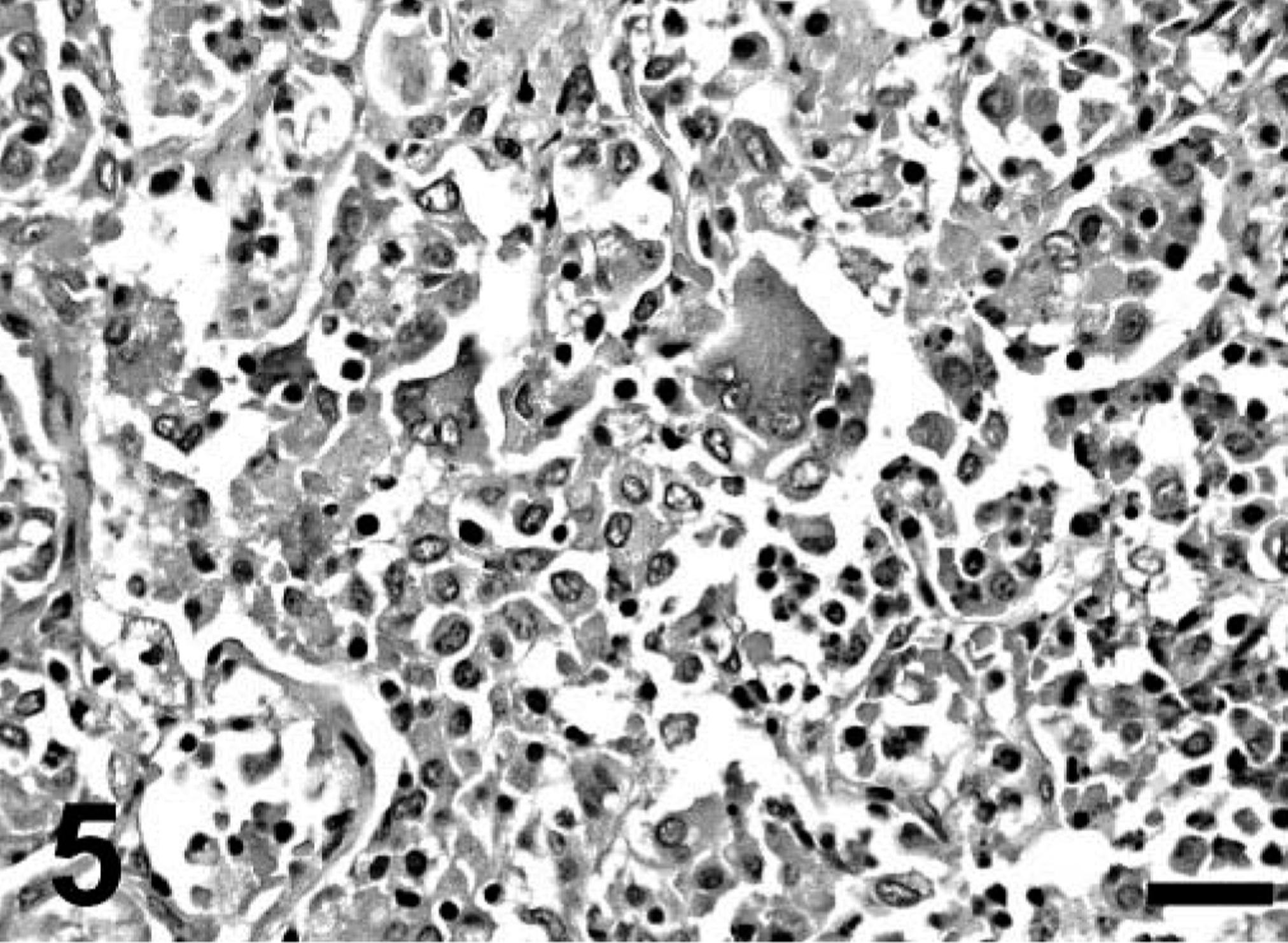
Lung; mara No. 3. Severe interstitial pneumonia with prominent fibrin deposits within alveolar spaces. Hematoxylin and eosin. Bar = 75 µm.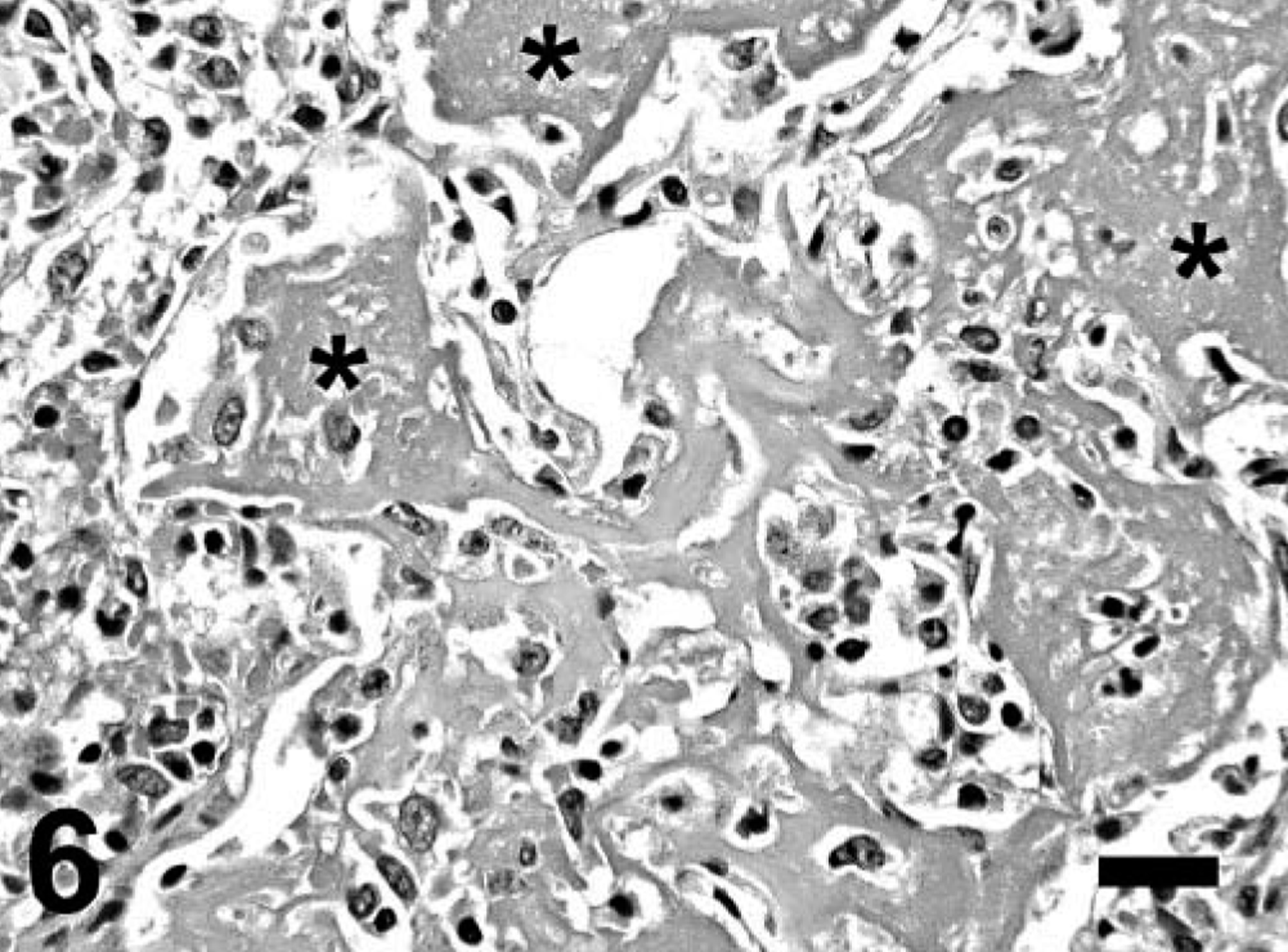
A retrospective review of all mara necropsy cases at Africam Safari revealed that three maras, two juveniles (mara Nos. 4 and 5) and an adult (mara No. 6), that died of unrelated causes had occasional or low numbers of similar cysts in the lungs with minimal to moderate inflammation surrounding them. The severity of these lesions correlated with the presence of lytic and mineralized cysts. Mara No. 4 also had cysts in the pancreas and peripancreatic adipose tissue.
The lung of mara No. 1 was selected for electron microscopy (EM). The cysts consisted of myriads of bradyzoites surrounded by a parasitophorous vacuole in the host cell cytoplasm (Fig. 7). Bradyzoites were crescent shaped and had a conoid, numerous micronemes, a few rophtries that extended well posterior to the nucleus, a uniform, electron-dense granular material, and numerous large granules with a fine granular, electron-dense or moderately dense substance, usually located posterior to the nucleus (Figs. 8, 9). Transverse sections through the apical pole of bradyzoites revealed 22 subpellicular microtubules beneath their outer membrane (Fig. 9). The host cell was hypertrophied and multinucleate and had projections of its cell membrane into the outer cyst wall, composed of collagen fibers and electron-dense granules (Fig. 7). No viral particles were seen.
Lung; mara No. 1. Note numerous bradyzoites within a parasitophorous vacuole (PV) in the host cell cytoplasm (HC) that has interdigitations into the outer cyst wall (CW). Note the presence of numerous electron-dense granules in the outer cyst wall. Uranyl acetate and lead citrate. Bar = 2.8 µm. Inset: higher magnification of the collagenous cyst wall (CW) with a few electron-dense granules (Gr), and host cell (HC). Uranyl acetate and lead citrate. Bar = 505 nm.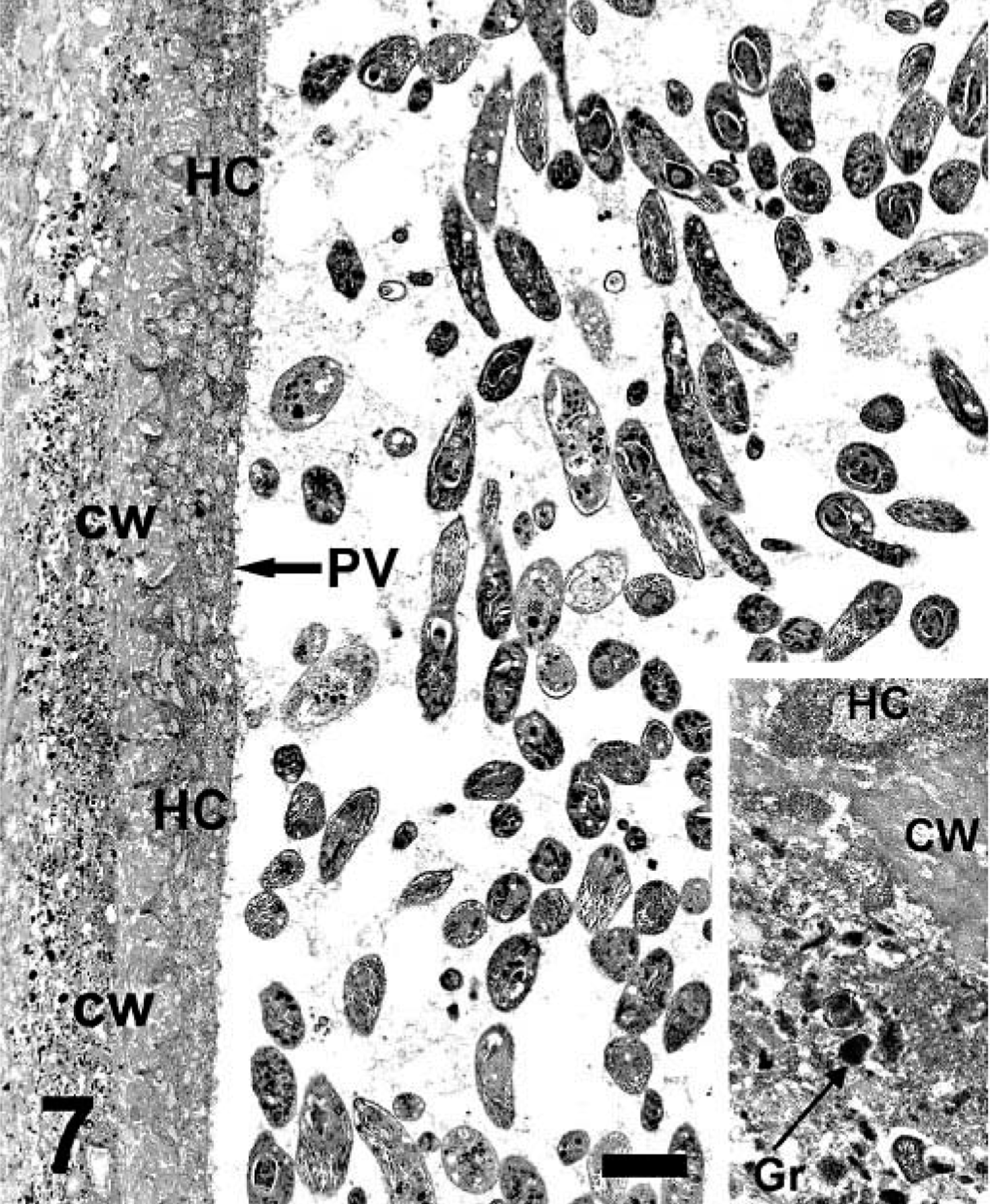
Lung; mara No. 1. Note the bradyzoite organelles, including the nucleus (Nu), numerous micronemes (M), a rophtry (R), and large cytoplasmic granules (G). Uranyl acetate and lead citrate. Bar = 0.5 µm.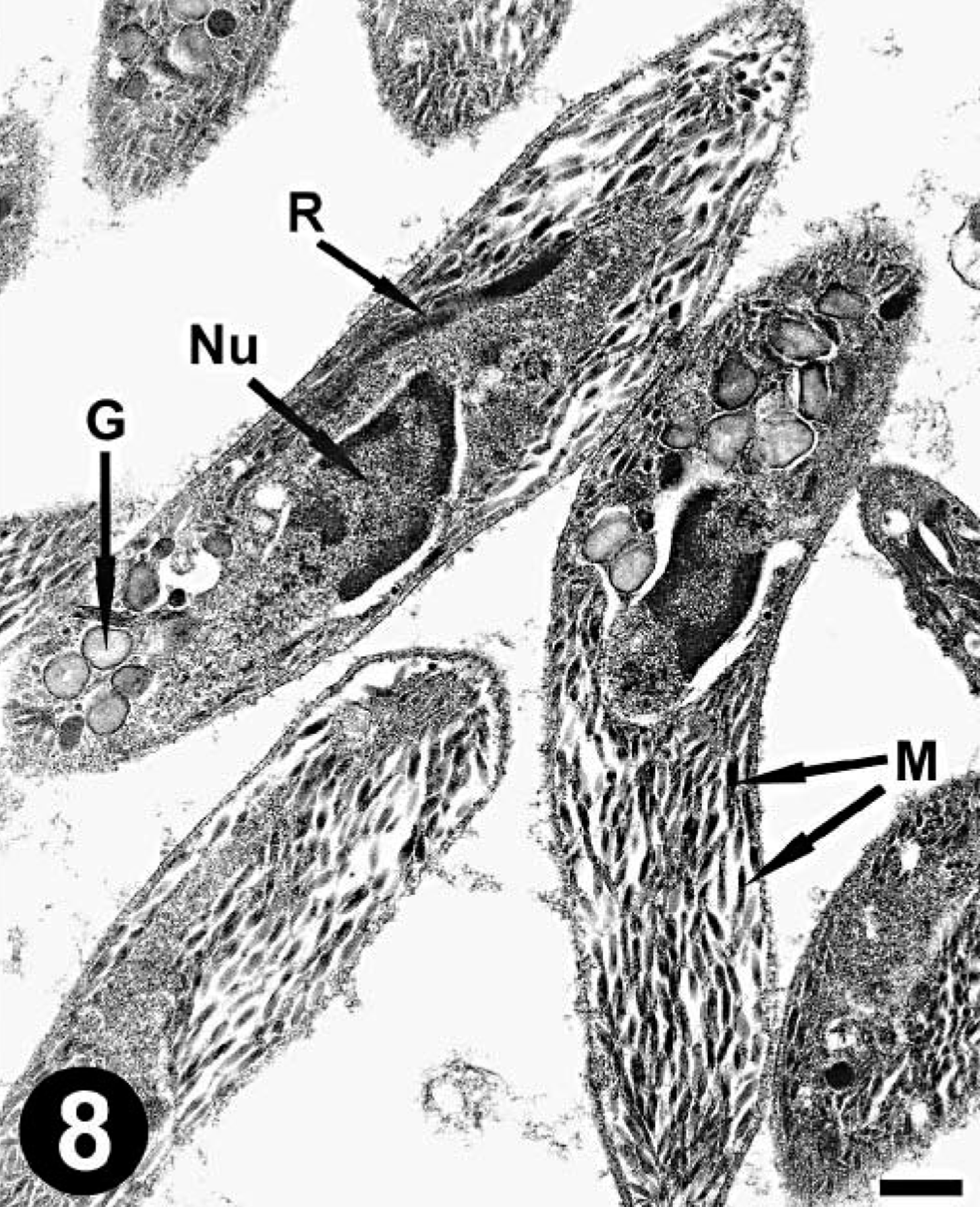
Lung; mara No. 1. Note the numerous large micronemes (M) and conoid (C) in a longitudinal section of a bradyzoite. Uranyl acetate and lead citrate. Bar = 138 nm. Inset: note the 22 subpellicular microtubules (SM) and rophtries (R) in this cross section through the conoid (C) of a bradyzoite. Uranyl acetate and lead citrate. Bar = 102 nm.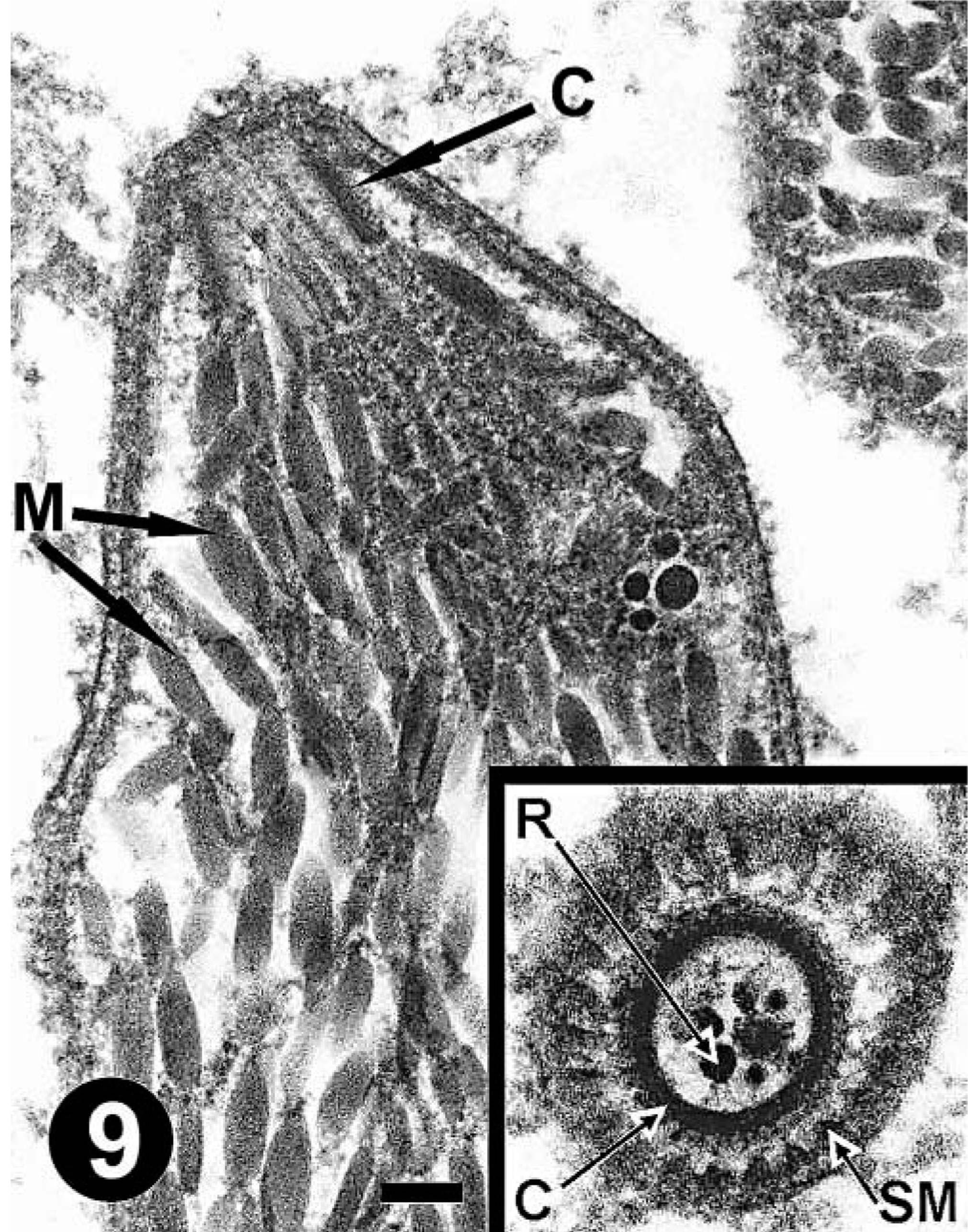
The pathologic and ultrastructural findings are consistent with pulmonary besnoitiasis (disseminated in mara No. 3). By light microscopy and EM, Besnoitia cysts can be differentiated from other tissue cyst–forming coccidia in the family Sarcocystidae; 1,7 specifically, bradyzoites are surrounded by a parasitophorous vacuole within the cytoplasm of a hypertrophied and multinucleate fibroblast surrounded by a thick capsule composed of collagen and electron-dense granules.
Besnoitiasis was associated with severe interstitial pneumonia in mara Nos. 1–3. To the authors' knowledge, this is the first report of besnoitiasis in maras and its association with fatal interstitial pneumonia in any animal species. Besnoitia cysts usually cause mild inflammation but can be surrounded by lymphocytic, plasmacytic, or granulomatous infiltrates. 5,7 Pulmonary besnoitiasis with minimal or no inflammation surrounding cysts has been reported in rabbits. 8 The severity of interstitial pneumonia in these maras correlated with the number of cysts and the presence of lytic cysts, which were often surrounded by a prominent granulomatous inflammatory response. Ruptured Besnoitia cysts can cause necrotizing and granulomatous inflammation. 8 Host immune response against Besnoitia cysts may have been detrimental in the three fatal cases described here. Zoites released from lytic cysts surrounded by preexisting chronic granulomatous inflammation may have been involved in the development of fatal interstitial pneumonia. Toxoplasma gondii tachyzoites can cause interstitial pneumonia. 3 Another possibility is that these maras were infected concurrently with an agent capable of inducing interstitial pneumonia and not detected with our diagnostic techniques. Viral cultures of lung were not carried out in any case, and an underlying viral infection cannot be ruled out though no viral particles were seen on EM in mara No. 1. The Yersinia sp. isolate from the lungs of mara No. 3 was interpreted as contamination because the pathologic findings did not resemble those of yersiniosis. Interstitial pneumonia was the cause of death in mara Nos. 1 and 3 and likely the major contributing factor for death in mara No. 2. The three other maras in this report died of unrelated conditions and had fewer Besnoitia cysts with mild inflammation surrounding them, and this may indicate resistance due to low infective dose or effective immunity (or both).
Although studies are needed to demonstrate an etiologic role of Besnoitia in the fatal pneumonia described here, besnoitiasis should be included in the differential diagnosis of pneumonia in juvenile maras. Although besnoitiasis in maras may be mostly pulmonary, it can be disseminated and even associated with inflammation in multiple tissues. Besnoitiasis usually causes cutaneous, genital, nasopharyngeal, or disseminated infections (or all) in intermediate host species. 1,4,5,7
Cats are the definitive hosts of B. darlingi and B. wallacei; 3 however, the role of cats and other carnivores in the life cycle of other Besnoitia species is unclear. 2,9 The definitive host and Besnoitia species involved in these cases are unknown; cats trapped in the mara enclosure surroundings have been negative for fecal shedding of Besnoitia oocysts. However, the sandy substrate of the mara exhibit and the easy access of stray cats to it may have favored contamination of mara's food and water with cat feces. Wild carnivores at the park may have also served as definitive hosts by fecal shedding of oocysts. In addition, transmission by hematophagous insects may occur in ungulates. 1,5
Footnotes
Acknowledgements
This study was funded by the Departamento de Investigación, Conservación y Alcance (DICA) at Africam Safari (Puebla, México).
