Abstract
Cellular senescence is a cell cycle arrest in damaged or aged cells. Although this represents a critical mechanism of tumor suppression, persistence of senescent cells during aging induces chronic inflammation and tissue dysfunction through the adoption of the senescence-associated secretory phenotype (SASP). This has been shown to promote the progression of age-associated diseases such as Alzheimer’s disease, pulmonary fibrosis, and atherosclerosis. As the global population ages, the role of cellular senescence in disease is becoming a more critical area of research. In this review, mechanisms, biomarkers, and pathology of cellular senescence and SASP are described with a brief discussion of literature supporting a role for cellular senescence in veterinary diseases. Cell culture and mouse models used in senescence studies are also reviewed including the senescence-accelerated mouse—prone (SAMP), senescence pathway knockout mice (p53, p21 [CDKN1A], and p16 [CDKN2A]), and the more recently developed senolysis mice, which allow for direct visualization and elimination (or lysis) of senescent cells in live mice (p16-3MR and INK-ATTAC). These and other mouse models have demonstrated the importance of cellular senescence in embryogenesis and wound healing but have also identified a therapeutic benefit for targeting persistent senescent cells in age-associated diseases including neurodegeneration, diabetes, and cardiac fibrosis.
Keywords
Cellular senescence is a highly durable mode of cell cycle arrest that occurs in aged or DNA-damaged cells. 56,57,59,149 Following the induction of cellular senescence, senescent cells adopt the senescence-associated secretory phenotype (SASP) and produce a variety of factors including cytokines, chemokines, and matrix metalloproteinases. 49,75,109,151 SASP proteins reinforce cellular senescence programming and mediate multiple tissue effects. 1,32,52 During development and healing, tightly regulated induction of cellular senescence promotes tissue reorganization and repair. 39,100 Secreted SASP chemokines recruit inflammatory cells, such as macrophages, which help remove senescent cells and restore normal tissue functions. 21,49,100,101 However, in some cases, senescent cells are not effectively removed. 101,108,121 This may be caused by an imbalance of senescence induction and clearance due to aging, 66,118 immune dysfunction, 97,99,110 or an acute stressor such as radiation 31,87 or traumatic injury. 65,140 Regardless of the cause, senescent cells accumulate in aged humans and animals and continue to secrete proteins which promote chronic inflammation, disrupt tissue homeostasis, and induce organ dysfunction. 32,58,101,109,151 It is in this way that cellular senescence has been implicated as an important mechanism underlying age-associated degenerative disease (Fig. 1). For example, senescent cells have been implicated in the progression of atherosclerosis, 29,49 cystic fibrosis, 45 age-related cataracts, 155 and radiation-induced pulmonary fibrosis. 31 In addition, mouse models have identified a therapeutic benefit for targeting and removing these senescent cells in disease including rescue of neurocognitive function in tau-induced neurodegeneration, 19 improved glucose metabolism in a model of type 2 diabetes, 2,112 and attenuated myocardial hypertrophy and fibrosis in cardiac aging. 5,82 Finally, although cellular senescence inhibits tumor initiation by preventing proliferation of damaged cells, it can also promote tumor progression and contribute to the late effects of cancer therapy through SASP. 32,38,81 Consistent with this, in vivo clearance of senescent cells has reduced tumor recurrence and chemotherapy-associated side effects in a mouse model. 38 In this review, we discuss the current understanding of the mechanisms and functions of cellular senescence and outline several experimental models used to study cellular senescence and SASP in disease.
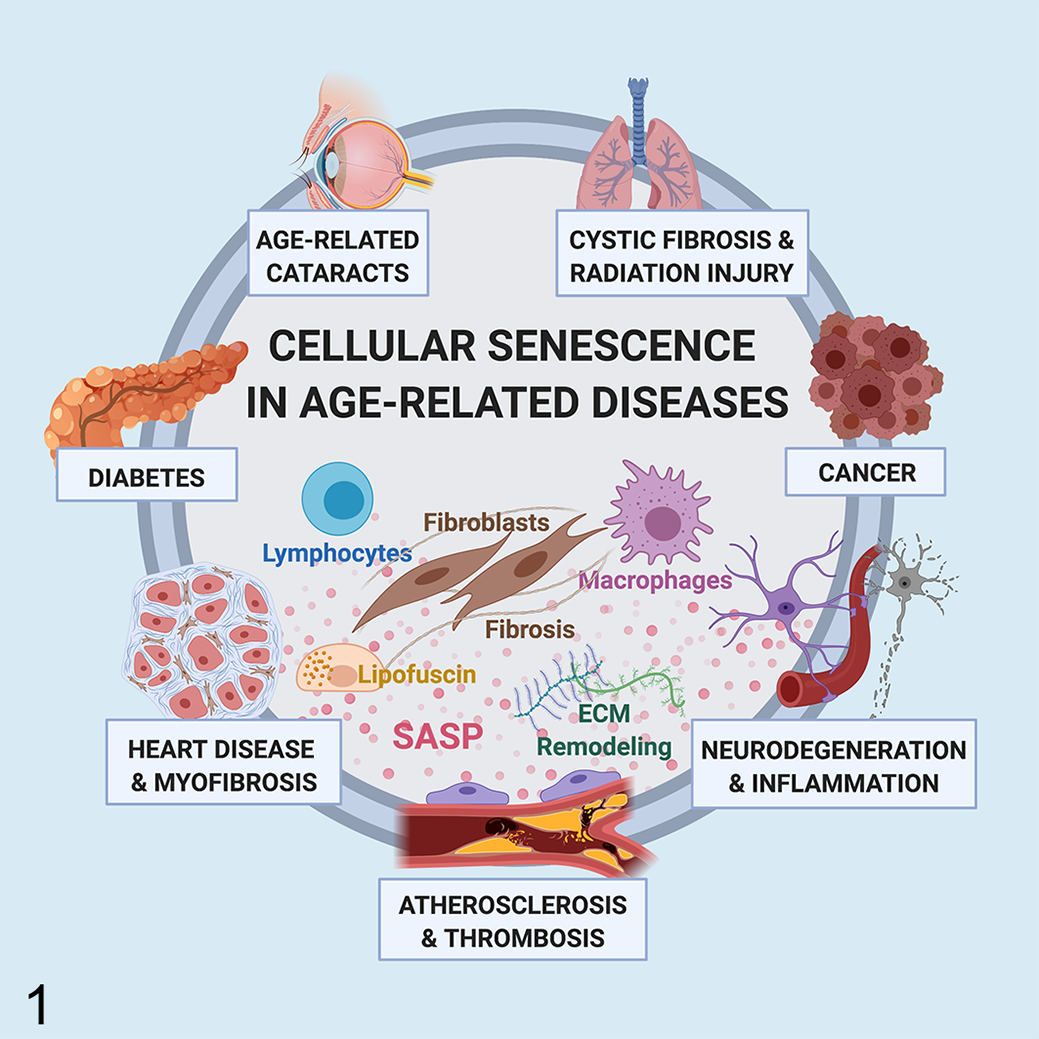
Cellular senescence in disease. Cellular senescence has been shown to promote or exacerbate age-associated diseases in humans through the induction of chronic inflammation and tissue dysfunction by the senescence-associated secretory phenotype (SASP). SASP proteins mediate an array of effects including mononuclear cell recruitment, fibroblast activation, and degradation of the extracellular matrix. Senescent cells, such as endothelial cells, astrocytes, and myocardiocytes, contribute to the progression of age-related diseases including atherosclerosis, neurodegeneration, and myocardial fibrosis, respectively. In addition, although cellular senescence inhibits tumor initiation by preventing the proliferation of damaged cells, persistence of senescent cells within the tumor microenvironment promotes tumor recurrence. Thus, cellular senescence and SASP are potential therapeutic targets in the treatment of disease in aged individuals.
Mechanisms of Cellular Senescence
The 2 major pathways of cellular senescence are replicative and stress-induced senescence (Fig. 2). 56,57,59,149 In replicative senescence, successive rounds of cellular replication and telomere shortening expose the chromosomal ends, which are sensed by the cell as endogenous DNA damage. 33,128 In contrast, internal or external stressors such as traumatic injury, 65,140 oxidative stress, 44,109,149 radiation, 31,109 or chemotherapy 38,109 cause cells to rapidly accumulate DNA damage and undergo stress-induced senescence. 59 Both pathways trigger the DNA damage response mediated by p53, p21, and p16. 33,54,59,138 This leads to inhibition of cyclin-dependent kinases resulting in cell cycle arrest and induction of cellular senescence. 59,132 Critically, once cells undergo senescence, they secrete numerous proteins as part of the senescence-associated secretory phenotype (SASP). 75,109,151 Secreted chemokines recruit macrophages that assist in removing these stressed cells. 49,100 Secretory factors have also been shown to induce senescence reprogramming in adjacent cells, often referred to as the “bystander effect.” 31,35,153 This is particularly well described in the lung where senescent pneumocytes produce secretory proteins that induce senescence in adjacent pneumocytes. 31 Additional studies in mice have found significant increases in senescent cell burden following injection of senescent cells, further underscoring the importance of SASP in reinforcing cellular senescence. 35,153 Finally, senescent cells can also affect neighboring cells of different cell types to induce a range of cellular states, including apoptosis or activation. 32 For example, senescent astrocytes can induce cell death in neuronal cells 10,19,83,143 while senescent pneumocytes activate fibroblasts resulting in pulmonary fibrosis. 31,125 These findings highlight the roles of cellular senescence in disease progression and identify cellular senescence as a potential therapeutic target in age-associated disease. 71,108,125
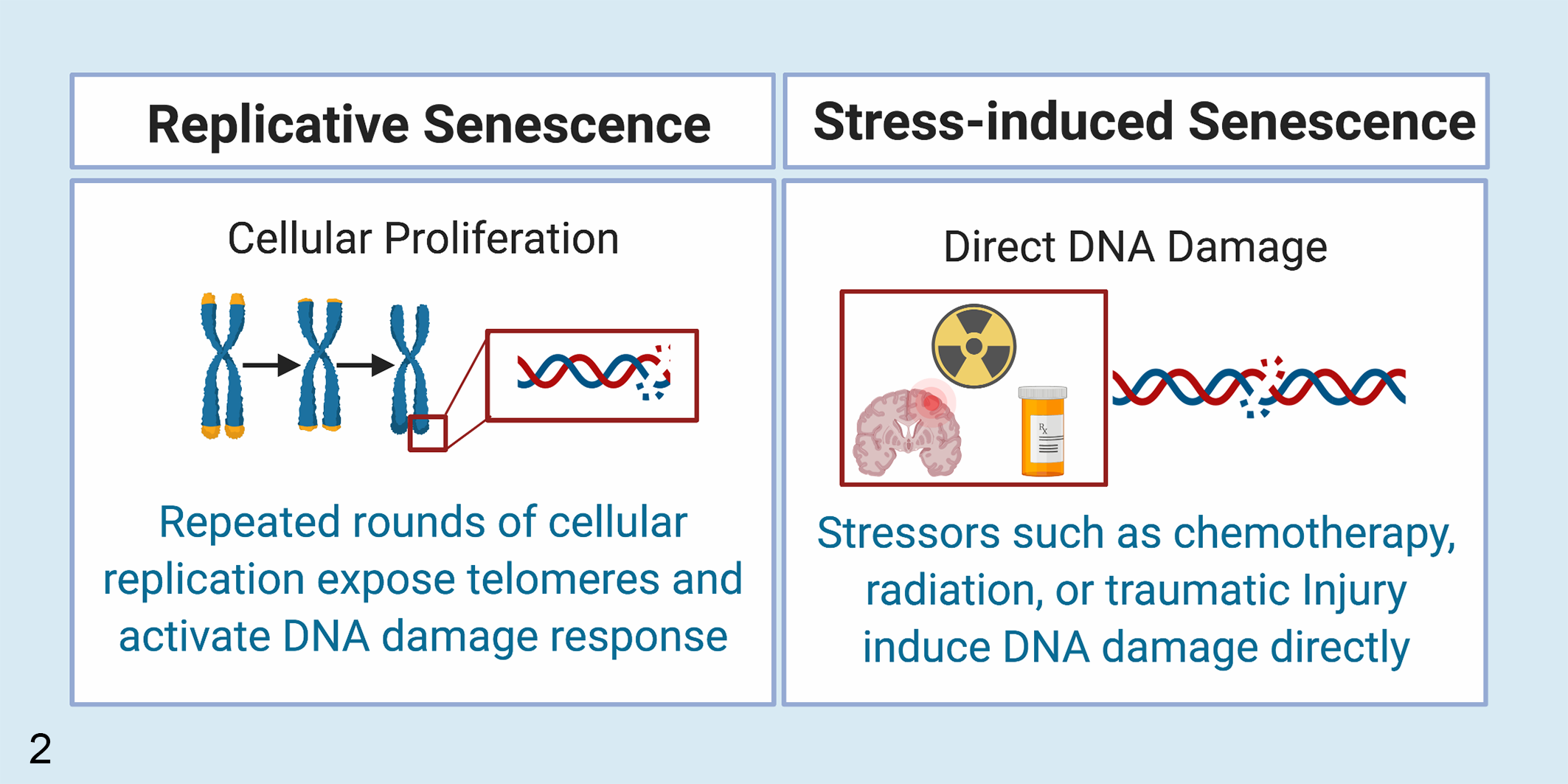
Mechanisms of cellular senescence. Cells undergo cellular senescence following successive rounds of replication leading to exposure of telomeric DNA and activation of the DNA repair pathway (replicative senescence). Cellular senescence may also be induced following the accumulation of non-telomeric DNA damage due to a variety of a stressors including radiation, chemotherapy, or traumatic injury (stress-induced or premature cellular senescence).
Senescent Cell Morphology and Biomarkers
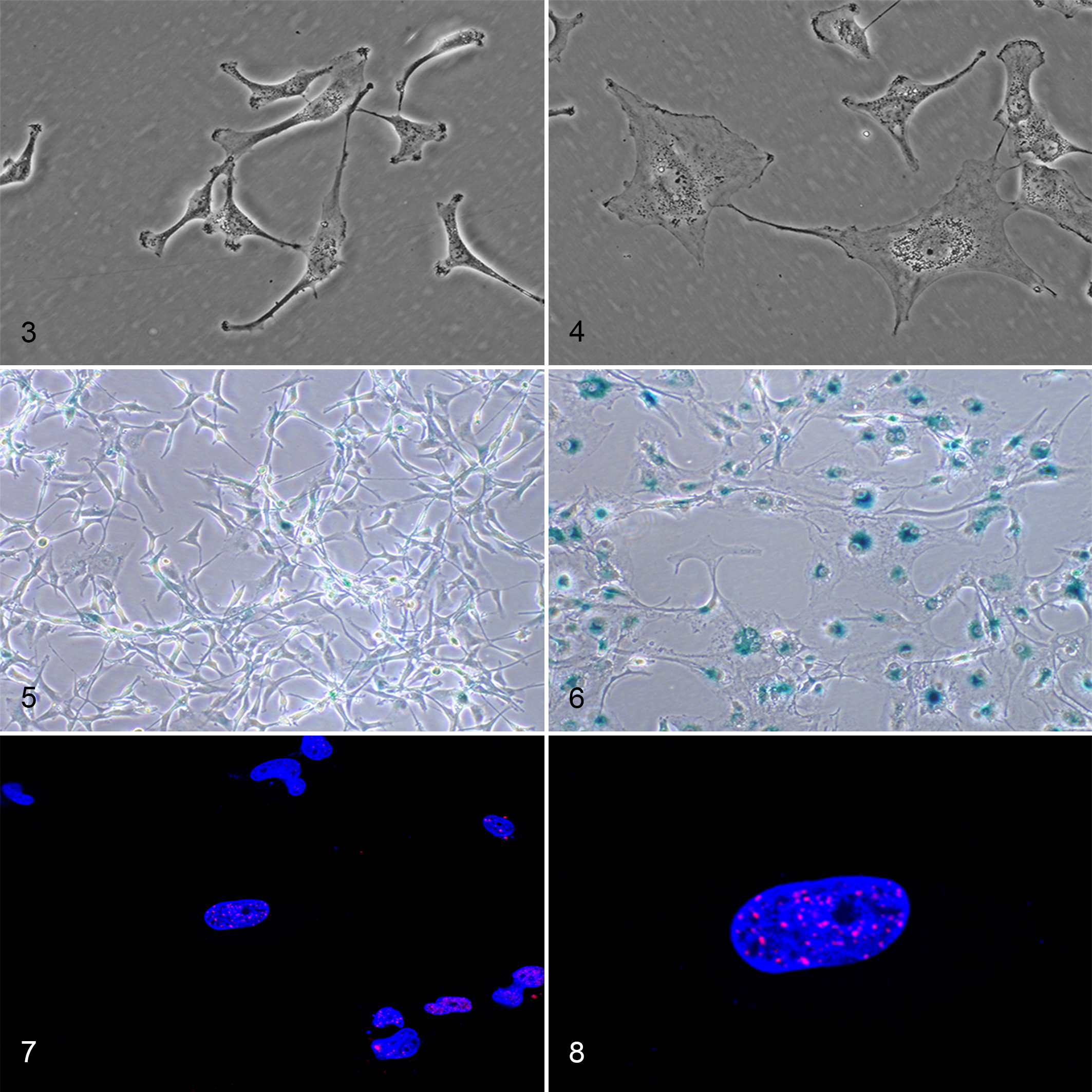
Researchers use several methods to identify senescent cells, including changes in cellular morphology and senescence-associated alterations in protein expression. 15,25,37,104 In culture, senescent cells are typically enlarged, flattened, multinucleated, and vacuolated (Figs. 3, 4). 59,75 Similar morphologic changes are also reported in tissues but may be restricted by the surrounding tissue architecture and thus more difficult to appreciate. 15,59 In these cases, it may be easier to identify histologic features of chronic inflammation associated with cellular senescence such as fibrosis and mononuclear cell inflammation. 31,49,101,125 Senescent cells also accumulate lipofuscin, a yellow-brown lipid-containing residue formed through lysosomal digestion, which can be visualized histologically or highlighted using histochemical stains such as Sudan Black B. 124,139 In addition, the senescence-associated β-galactosidase (SA-β-gal) assay is probably the best described and most commonly used staining technique. 37 It can be used in vitro or in vivo and highlights senescent cells with blue dye by taking advantage of increased β-galactosidase activity within the lysosomes of aged cells (Figs. 5, 6). 78 Finally, altered protein expression in senescent cells can be detected using a variety of techniques including western blotting, quantitative real-time polymerase chain reaction, or immunohistochemistry. 104,146 Double-strand DNA breaks can be identified by γH2AX or 53BP1 labelling in both replicative and stress-induced cellular senescence (Figs. 7, 8). 34,104,113 Following activation of the DNA damage response pathway, early senescent cells have increased nuclear expression of p53 and p21 while late senescence is best characterized by elevated p16. 59,132,138 Activation of these pathways induces cell cycle arrest and formation of senescence-associated heterochromatin foci (SAHFs) identified by DAPI staining, trimethylated H3K9, or heterochromatin protein 1. 75,102,122 These SAHFs contribute to the stability of cellular senescence by repressing genes, such as those associated with cellular proliferation. 102 Functionally, senescent cells adopt SASP characterized by increased secretion of chemokines, growth factors, matrix metalloproteases, and inflammatory cytokines. 32,109,151 Over 300 SASP proteins have been characterized and can vary by cell type, stressor, and time course but frequently include cytokines, such as interleukin (IL)-1, IL-6, and IL-8. These SASP proteins can be identified by increased mRNA production within cell or tissue samples or through the detection of secreted proteins in media or serum. 12,104,106 Finally, senescent cells upregulate the expression of anti-apoptotic BCL-2 family proteins such as BCL-XL resulting in resistance to apoptosis and subsequent tissue persistence. 121,147,157
Models of Cellular Senescence: Cell Culture
Primary cells proliferate in culture for a finite period of time. 56,57 Serial passaging of human cells has been used as an experimental method to study cellular senescence and aging in vitro. 56,115,143 Cells in culture may also accumulate DNA damage over time or following application of an acute stressor such as radiation, 109,144 chemotherapy, 51,109 or oxidative stress (hydrogen peroxide) 44,109,149 resulting in stress-induced senescence. These studies can be used to isolate the effect of specific stimuli on individual cells and to investigate the potential roles of cellular senescence in disease processes. 32,109,151 These types of experiments are also helpful to define the secretory proteins produced by senescent cell types. In addition, experiments involving transwells, co-culture, or conditioned media exposure can be used to define the effects of senescent cells and their associated secretory factors on neighboring cells. 32,114,143
Mouse Models: Senescence-Accelerated Mouse—Prone (SAMP)
While maintaining an inbred colony of AKR/J mice at Kyoto University, researchers identified an aging phenotype in a subset of litters. 136,137 Features of these mice included a poor hair coat, reduced activity, and decreased lifespan. 136 Aging features are thought to develop due to elevated oxidative stress, are inherited by subsequent generations, and have been further separated into several distinct SAMP phenotypes. 27,107,111 Litters of inbred AKR/J mice that did not experience an accelerated aging process are referred to as senescence-accelerated mouse—resistant (SAMR) and are typically used as controls for SAMP experimental studies. 136,137
Organismal senescence, such as that observed in SAMP mice, is the process of biological aging accompanied by the gradual deterioration of functional characteristics. 20,66,135 Critically, organismal senescence is a term applied to the entire organism and is not equivalent to cellular senescence, which is a mechanism of cell cycle arrest within individual cells. 20,66 Although organismal senescence is associated with increased numbers of senescent cells, not all cell types within an aged individual undergo cellular senescence. 21,66 As such, cellular and organismal senescence are not mutually inclusive but do appear to maintain a critical interplay. In the case of aged SAMP mice, organismal senescence is associated with increased cellular senescence in a variety of cell types including astrocytes, 47,48 endothelial cells, 107,152 progenitor cells, 133 retinal epithelial cells, 43 and fibroblasts. 77
The role of cellular senescence in promoting neurodegeneration is a key area of aging research. 10 SAMP8 mice experience age-dependent deficits in learning and memory and develop a variety of age-associated neuropathologic changes similar to those described in aging humans. 98 By as early as 5 months of age, neuropathologic changes include astrogliosis, microgliosis, and neurodegeneration. 68,134 SAMP8 mice also accumulate amyloid, 4,86 have increased nitric oxide synthase activity, 4,63 and demonstrate age-associated hyperphosphorylation of tau further underscoring their utility as a model of brain aging and Alzheimer’s disease. 23,98 Senescent SAMP8-derived astrocytes have been identified as a key contributor to neurodegeneration and demonstrate critical impairment of their normal neuroprotective functions. 47,48 A similar mechanism for astrocyte-mediated neurotoxicity in humans has also been shown to induce neuronal toxicity in vitro and may contribute to neurodegeneration in patients with Alzheimer’s disease, amyotrophic lateral sclerosis, and radiotherapy-induced brain injury. 10,83,142,143
In humans, cellular senescence has been suggested to promote a variety of musculoskeletal diseases including sarcopenia, 8,131 muscular dystrophy, 8,74 and osteoporosis. 8,42,133 Adult SAMP6 mice have a significantly higher incidence of spontaneous leg fractures attributed to osteoporosis. 90,130,133 Induction of cellular senescence in bone marrow progenitor cells disrupts their differentiation favoring adipogenesis over osteoblastogenesis. 3,62,133 This mechanism has been suggested to result in inefficient osteoblastic activity and osteoporosis in SAMP6 mice and in aged humans. 8,72,133
Critically, although SAMP mice are separated into distinct phenotypes, organismal senescence may be associated with age-related dysfunction in multiple organs within the same mouse. For example, although most frequently used for neurodegeneration research, SAMP8 mice also develop age-associated vascular disease and have been used to study cellular senescence in endothelial cells. 69,107,158 In humans, increased numbers of senescent cells have been suggested to contribute to atherosclerosis, 29,49,94 impaired angiogenesis, 144,148 and cardiac fibrosis. 5 While the development of accelerated aging in multiple organs can complicate the interpretation at a cellular level, these models may be more representative of the physiologic effects of aging on the entire organism.
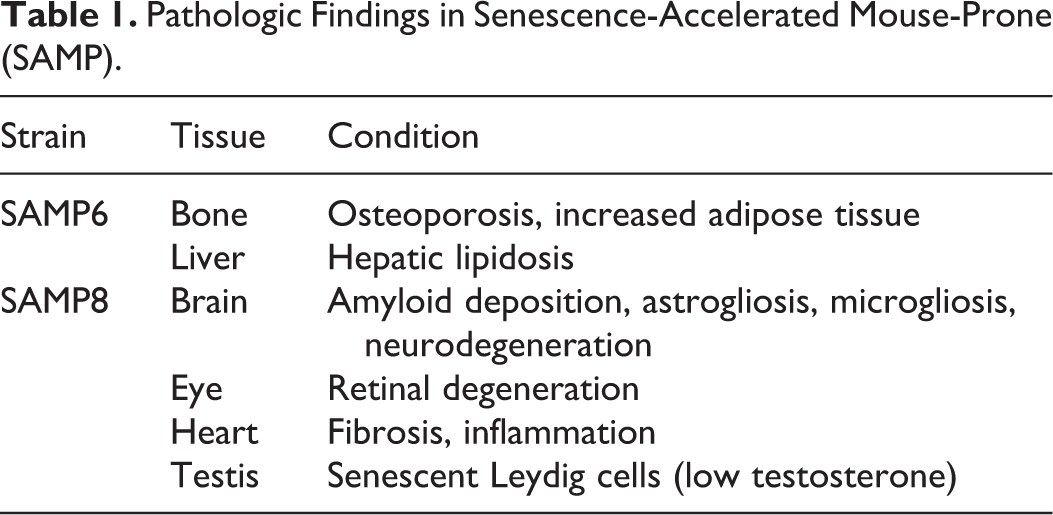
In addition to these neurologic and musculoskeletal alterations, SAMP6 and SAMP8 mice also develop a variety of other age-associated conditions including retinal degeneration, 43 testosterone deficiency, 107 myocardial fibrosis, 69 and hepatic lipidosis 103 (Table 1). Finally, there are more than 10 SAMP mice strains, each of which can develop a variety of age-related conditions such as renal fibrosis (contracted kidneys), immune dysfunction, and degenerative joint disease. 61,129,135 Although SAMP studies do not always examine cellular senescence in the development of age-related disease, each of these conditions has been associated with induction of cellular senescence in other experimental models suggesting that cellular senescence may also contribute to the development of these conditions in SAMP mice. 40,65,85,88,97,110
Pathologic Findings in Senescence-Accelerated Mouse-Prone (SAMP).
Mouse Models: Targeting Senescence Pathways (p53/p21/p16)
Inhibiting senescence pathways is another way to investigate the role of cellular senescence in diseases, including targeting the induction (p53/p21) or persistence (p16) of senescence. 59 These knockout mice may also be crossed to disease models to study the roles of these pathways in the progression of age-associated disease.
Cellular senescence studies using p21- and p53-null mice have identified critical roles for cellular senescence in embryonic development and in the promotion of premature aging syndromes such as Hutchinson-Gilford progeria syndrome and ataxia telangiectasia. 14,100,145,154 However, studying these processes in p53- and p21-null mice is complicated by the loss of beneficial p53 pathway functions. 46,59,76 Critically, p53-mediated induction of cellular senescence is an important mechanism of tumor suppression. 22,26,138 Thus, mouse models that inhibit cellular senescence have an increased incidence of several neoplasms which can affect remarkably young mice (Table 2). 41,89,126,127 The p53-null mice develop tumors by 10 weeks of age with almost 75% of mice affected by 6 months of age. 41 As a result, mice are euthanized early making them difficult to study over time. 41 The most common tumors in these mice include lymphoma (77%) and hemangiosarcoma (27%). 41 Because the majority of mice succumb to cancer, there are only rare cases of infectious or inflammatory diseases reported (eg, abscesses, gastroenteritis, myocarditis). 41 An alternative method to target the p53 pathway is through deletion of its downstream effector, p21. 54,59 As expected, p21-null mice also develop tumors earlier than wild-type counterparts (16 months vs >2 years). 89 Tumor types are similar to those reported in p53-null mice but with a longer latency period and include histiocytic sarcoma (52%), hemangioma/hemangiosarcoma (22%) and lymphoma (14%). 41,89 Although 55% of male p21-null mice succumb to their tumors, the most common cause of death in female p21-null mice is severe autoimmune glomerulonephritis which affects approximately 60% and is thought to be due to an abnormal proliferation of T cells. 89
Mouse Models Targeting Senescence Pathways.

a Contributing cause of death in p21-null mice varies by sex. Glomerulonephritis (GN) is the primary cause in 60% of females (males, 26%) while cancer is the cause in 55% of males (females, 26%).
Finally, p16 has been characterized as the best in vivo marker for senescent cells and its knockout has produced another commonly used model to study cellular senescence. 84,126 Studies in p16-null mice have suggested that inhibiting cellular senescence in the kidney leads to enhanced recovery of renal function following ischemia-reperfusion injury and reduces interstitial fibrosis and tubular atrophy in renal transplant models. 17,40,80 Although these mice develop tumors later than p53 knockout mice, approximately 25% of p16-null mice develop tumors by 10 months of age. 41,89,126 Spontaneous tumors reported in these mice include malignant spindle cell neoplasms (29%), angiosarcoma (23%), osteosarcoma (18%), histiocytic lymphoma (18%), and melanoma (12%). 127 With loss of p16 function, these mice are also highly susceptible to carcinogen-induced tumors. 126,127 Other reported changes in p16-null mice include elevated T cell proliferation, absolute T cell counts, and thymic hyperplasia. 126
Classically, the induction of cellular senescence in damaged cells is described as an important barrier to carcinogenesis. This theory is supported by the increased tumor burden observed in these knockout mice; however, the role of cellular senescence in cancer progression may be more complicated (Fig. 9). 32,38 For example, while induction of cellular senescence is initially associated with inhibition of tumor initiation or regression, persistence of senescent tumor and nontumor cells has been shown to promote tumor recurrence through SASP. 32,38,64,81 In addition, some reports suggest that tumor cells can either escape or inhibit the induction of cellular senescence allowing them to reenter the cell cycle. 13,46,93 The contrasting roles of cellular senescence in cancer are being further investigated in senolysis mice, which are used to study the roles of cellular senescence in many age-associated diseases including cancer and cancer recurrence. 38
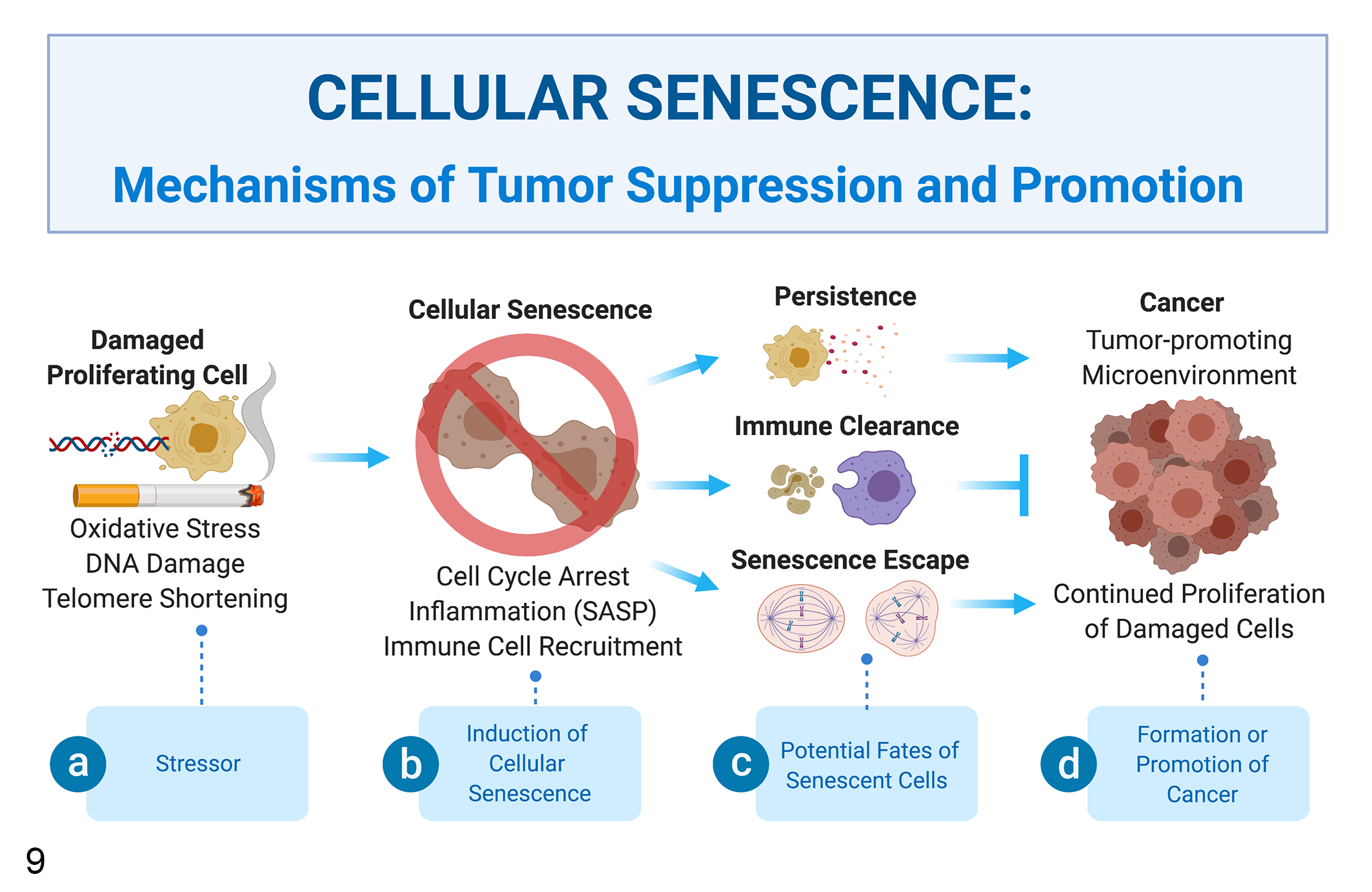
Mechanisms of tumor suppression and promotion. (a) Induction of cellular senescence is an important mechanism of tumor suppression which prevents tumor initiation by inhibiting proliferation of damaged cells. (b) Senescent cells undergo a permanent cell cycle arrest and adopt the senescence-associated secretory phenotype (SASP), which recruits leukocytes to remove senescent cells. (c, d) There are several potential fates for cells undergoing senescence. Normal tissue function can be restored if macrophages remove senescent cells; however, this process is not always efficient, leading to the persistence of senescent cells that can promote tumor recurrence and the side effects of cancer therapy through SASP. Some reports also suggest that tumor cells can either escape or inhibit the induction of cellular senescence allowing them to reenter the cell cycle leading to tumor initiation further underscoring the contrasting functions of cellular senescence in tumor suppression and promotion.
Mouse Models: Senolysis
Recently, several transgenic mouse models have been developed that allow for direct visualization and elimination of senescent, p16-positive cells including the p16-3MR (tri-modal reporter) and the INK-ATTAC (apoptosis through targeted activation of caspase 8) models. 11,39 These models identify p16-positive senescent cells through expression of red or green fluorescent protein (RFP/GFP). 11,39 This allows researchers to image and quantify fluorescent protein expression in live mice as they age or to study the accumulation of senescent cells following introduction of an acute stressor, such as chemotherapy. 9,38 Finally, these mouse models were designed to allow for specific targeting and removal of senescent cells. 11,39 This allows researchers to investigate the therapeutic value of senolysis in specific disease contexts. 11,39 Through the use of these models, the contributory roles of cellular senescence and the beneficial effects of senolysis have been demonstrated in several age-associated diseases including tau-related neuropathology and cognitive decline, 19 chemotherapy-induced fatigue, 38 cancer recurrence, 38 osteoporosis, 42 myocardial hypertrophy, 5 and cardiac fibrosis. 5
Although fluorescent reporter genes offer a great opportunity to identify and study the spread of senescence, they are not without potential side effects. 6 For example, GFP has been reported to cause injury through production of reactive oxygen species, promotion of apoptosis, and induction of immunogenicity. 6,16 This is reported to be more severe in BALB/c than C57BL/6 mice suggesting that the background strain is important when evaluating mouse models for GFP immunogenicity. 16 In addition, transgenic mice using the INK-ATTAC model are injected with AP20187 while the p16-3MR model is controlled by ganciclovir administration. 11,39 Although side effects of either treatment are not well described in these models, any intraperitoneal injection has the potential to cause ileus and peritonitis. 141 Finally, inhibiting cellular senescence has been shown to impede wound healing which should be monitored in these mice. 9,39
Evidence for Cellular Senescence in Domestic Animals
Although cellular senescence is not a newly discovered mechanism, researchers are still characterizing its potential role in human diseases, 101 including neurodegeneration, 10,19 pulmonary fibrosis, 31 wound healing, 39 and osteoarthritis. 65 As researchers aim to identify ways to modulate cellular senescence in disease, it will become important to understand the comparative role of cellular senescence in veterinary diseases. In addition to mouse model studies, there are reports of cellular senescence contributing to disease or cellular dysfunction in domestic animals. For example, dogs have increased numbers of senescent cells in the aged testis, 92 in chronic hepatitis, 73 and within the nontumor tissue surrounding intraocular tumors. 91 In cats, cellular senescence in increased in feline chronic kidney disease and contributes to diminished self-renewal and reduced multipotency of mesenchymal stem cells in vitro. 79,116 Although classic markers of cellular senescence are not always examined, there is also good evidence that aging alters cellular functions in animals consistent with the induction of cellular senescence. Examples include reduced proliferation, cellular dysfunction, and features of immunosenescence in aged horses, 18 cats, 36 and dogs, 36,117,150 in horses with asthma, 18 and in dogs with cancer. 150 In addition, senescence-associated histologic changes are reported in aged animals including fibrosis and mononuclear cell inflammation. 30,105,116 There is also evidence that SASP-associated cytokines such as IL-1, IL-6, and IL-8 are produced in animals with chronic inflammatory diseases that are attributed to cellular senescence in humans including pulmonary fibrosis, chronic kidney disease, and arthritis. 24,119,156 In the future, further examination of these and other age-related diseases may better identify the potential roles for cellular senescence in promoting pathology in domestic species.
Conclusions
Cellular senescence is a normal process by which aged and damaged cells are inhibited from replicating. 22 It is important in normal embryonic development and serves as a critical tumor suppressor mechanism. 22,100 Secretion of SASP proteins is thought to induce removal of senescent cells through inflammatory cell recruitment, thereby promoting healing and restoring tissue homeostasis. 49,100,101 However, surviving senescent cells that are not cleared by the immune system continue to secrete SASP proteins and contribute to organ dysfunction. 101 Like other biologic processes, senescence programming is critical in specific contexts, but its persistence can lead to an exuberant and long-lasting inflammatory response that can contribute to further tissue injury. 39,101
In this review, we discussed several mouse models used to investigate the roles of cellular senescence in aging and cancer; however, this list is not exhaustive. Importantly, cellular senescence may contribute to aging phenotypes in a variety of mouse models. For example, progeroid syndrome models, which have been reviewed previously, have increased numbers of senescent cells that promote premature aging. 11,55 Sirtuin-deficient mouse models, such as SIRT6 null mice, also experience premature aging and have demonstrated a role for NF-κB signaling in regulating cellular senescence and SASP. 28,70,123 Finally, mouse models with deficient DNA damage repair mechanisms or models of traumatic injury, such as traumatic brain injury or posttraumatic osteoarthritis, may be used to investigate the roles of premature or stress-induced senescence in injured tissues. 50,53,65,118,140 These experimental models demonstrate the sudden increase in senescent cells following injury and, when incorporated into a senolysis mouse model, can be used to characterize the therapeutic benefit of senolysis on disease progression. 65
Currently, there is intense interest in defining the roles of cellular senescence in human diseases. 101 This interest goes beyond simply characterizing senescent cells in vitro but rather identifying ways to restore or remove senescent cells to benefit patients. 108 One of the first methods by which this was pursued was through the use of therapeutics which target the production or secretion of SASP proteins such as cytokine-directed antibodies, 106,120 NF-κB inhibitors, 96 or sirtuin activators. 71,95,108 More recently, anti-SASP therapies have been augmented by senolytic drugs, which reduce the organismal burden of senescent cells. 7,8,42,108 These senolytics have shown promise in preliminary clinical trials in humans. 60,67 As therapeutic interventions evolve, defining the roles of cellular senescence in veterinary diseases will be important not only for understanding chronic inflammation in the context of aging but also for facilitating the translation of anti-senescence therapies between humans and animals.
Footnotes
Acknowledgements
Jessica Beck is supported through a Research Fellowship in the Laboratory of Human Carcinogenesis and by a Cancer Research Training Award in the Comparative Biomedical Scientist Training Program at the National Cancer Institute in partnership Purdue University. The Laboratory of Human Carcinogenesis is supported by the Intramural Research Program of the NIH, NCI. Figures 1, ![]() , and 9 were created using Biorender Software.
, and 9 were created using Biorender Software.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
