Abstract
A prevalent and distinctive infectious interstitial pneumonia (IIP) of immunocompetent laboratory rats was suspected to be caused by a putative virus, termed rat respiratory virus, but this was never substantiated. To study this disease, 2 isolators were independently populated with rats from colonies with endemic disease, which was perpetuated by the regular addition of naive rats. After Pneumocystis was demonstrated by histopathology and polymerase chain reaction (PCR) in the lungs of rats from both isolators and an earlier bedding transmission study, the relationship between Pneumocystis and IIP was explored further by analyzing specimens from 3 contact transmission experiments, diagnostic submissions, and barrier room breeding colonies, including 1 with and 49 without IIP. Quantitative (q) PCR and immunofluorescence assay only detected Pneumocystis infection and serum antibodies in rats from experiments or colonies in which IIP was diagnosed by histopathology. In immunocompetent hosts, the Pneumocystis concentration in lungs corresponded to the severity and prevalence of IIP; seroconversion occurred when IIP developed and was followed by the concurrent clearance of Pneumocystis from lungs and resolution of disease. Experimentally infected immunodeficient RNU rats, by contrast, did not seroconvert to Pneumocystis or recover from infection. qPCR found Pneumocystis at significantly higher concentrations and much more often in lungs than in bronchial and nasal washes and failed to detect Pneumocystis in oral swabs. The sequences of a mitochondrial ribosomal large-subunit gene region for Pneumocystis from 11 distinct IIP sources were all identical to that of P. carinii. These data provide substantial evidence that P. carinii causes IIP in immunocompetent rats.
Publications first appearing in the mid-1990s describe the occurrence of distinctive lung lesions characterized by lymphohistiocytic interstitial pneumonia and perivascular lymphocytic cuffs in immunocompetent laboratory rats. 15,39 Gilbert et al 19 reported that these lesions interfered with interpretation of pulmonary histopathology in a liposome inhalation study. The lymphohistiocytic nature of the lesions and their eventual resolution, as separately described by Elwell et al 15 and Slaoui et al, 39 suggested an infectious etiology. However, standard diagnostic methodologies to detect known rodent pathogens, including serology for Mycoplasma, cilia-associated respiratory (CAR) bacillus and viruses, microbiologic culture, and special stains, were unrewarding. Early attempts to isolate and transmit an infectious agent led to the description of a novel enveloped virus, referred to as rat respiratory virus (RRV), that reportedly could be propagated in cell culture and was antigenically related to Hantaan virus. 9,35,36 In a limited evaluation of an experimental RRV antibody assay, however, we found that the assay results did not correspond to the known infectious interstitial pneumonia (IIP) status of colonies as established by pulmonary histopathology (C. B. Clifford, unpublished data). In the absence of polymerase chain reaction (PCR) and meaningful serologic tests, pulmonary histopathology was the only reliable diagnostic methodology available for surveillance of rat colonies.
The histopathologic criteria for diagnosing the pulmonary lesions of IIP were recently published by Albers et al. 1 These authors also found a consistent time course for lesion development in naive rats introduced into an endemically infected breeding colony, where animals were housed in open cages. At necropsy, small, irregular gray-white lesions were observed in the lung at 6 to 13 weeks postexposure. Histologically, lymphohistiocytic interstitial pneumonia and prominent perivascular lymphoid cuffing were observed from 5 to 13 weeks postexposure with peak severity from 7 to 8 weeks. These data also provided support for the transmissible nature of the interstitial pneumonia. Therefore, in presenting our studies, we will refer to this histopathologic diagnosis as IIP.
Just-published research has provided convincing evidence that Pneumocystis carinii, and not the putative RRV, causes IIP. 26 Consequently, the publication’s authors have proposed that this prevalent, once idiopathic disease be called Pneumocystis pneumonia. The genus Pneumocystis comprises obligate extracellular parasites that were once considered to be protozoa but are now classified as ascomycetes fungi. 11,42 Pneumocystis species are host specific and have yet to be described as infecting aberrant hosts. 14 The laboratory rat (Rattus norvegicus) has 2 formally described Pneumocystisspecies, P. carinii and P. wakefieldiae, 10,34 with P. cariniibeing much more common and the only species so far linked to IIP. Significant clinical disease (ie, pneumonia with eventual obstruction of alveoli with organisms and subsequent respiratory failure) due to Pneumocystisin any species, including rats, has typically been associated with acquired or congenital immune deficits,12,31 although several studies have found mild lesions in immunocompetent human and animal hosts, 40,41 including mice naturally or experimentally infected with P. murina. 6,8,17,18,20,44
This report provides the results of multiple investigations into the cause and pathogenesis of IIP that were performed with the ultimate objective of developing pathogen-specific PCR and serologic assays better suited than histopathology to routine, high-throughput colony surveillance. Furthermore, understanding the pathobiology of the agent responsible for IIP is fundamental to assessing the efficacy of biosecurity measures aimed at eliminating and excluding this infectious disease from production and research colonies.
Our data independently corroborate the cause-effect relationship between P. carinii infection and IIP just reported by Livingston et al. 26 To study IIP, 2 isolators were independently populated with rats from colonies with endemic disease, which was perpetuated by the regular addition of naive rats. After histopathology and PCR demonstrated Pneumocystis in the lungs of rats from both isolators and an earlier bedding transmission study, the relationship between Pneumocystisand IIP was explored further by analyzing specimens from 3 contact transmission experiments, diagnostic submissions, and barrier room breeding colonies, including 1 with and 49 without IIP. Immunocompetent WH and CD and the immunodeficient athymic RNU rat strains were exposed to IIP from 3 distinct sources. To model natural transmission and avoid nonspecific inflammation and other artifacts associated with intratracheal instillation, 6,8,18,37 rats in transmission experiments were exposed to infection by being housed on bedding from or in contact with contagious animals. The time course and severity of IIP were compared with the concentration of Pneumocystis in lung as measured by a real-time quantitative (q) PCR and the titer of Pneumocystis serum antibodies determined by an indirect immunofluorescence assay (IFA). In addition, the suitability of lung, bronchial, and nasal washes and oral swabs for Pneumocystis detection by PCR and the mitochondrial ribosomal large-subunit genotypes of Pneumocystis from 11 distinct sources were compared.
Materials and Methods
Animals
All rats discussed in this research report, except diagnostic submissions from external clients of the diagnostic laboratory were housed in Charles River's facilities and all facilities were accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. All experimental work on live animals was approved by the Charles River Institutional Animal Care and Use Committee. For most studies, the rats were housed in flexible film isolators. Each isolator was equipped with wire-topped solid-bottomed cages, bedded with irradiated kiln-dried aspen shavings (NEPCO, Warrensburg, New York). Animals had ad libitum access to sterile filtered water and irradiated feed (Lab Diet 5L79; Purina Mills, Richmond, Indiana) and were maintained on a light cycle of 12 hours light and 12 hours dark. In studies in which rats were removed from their isolator, they were then housed in filter-top microisolation caging, and all cage changes and animal manipulations were performed under a class II biosafety cabinet.
Infectious Disease Monitoring
Surveillance for adventitious infections was performed on the isolator and barrier colonies that were the source of the IIP-negative (naive) and IIP-positive rats used in transmission experiments. Surveillance included (1) serology for Sendai virus, pneumonia virus of mice, sialodacryoadenitis virus, Kilham’s rat virus, H-1 virus, rat minute virus and rat parvovirus-1, rat theilovirus (RTV), reovirus, Mycoplasma pulmonis, lymphocytic choriomeningitis virus, hantavirus, mouse adenovirus, CAR bacillus, Clostridium piliforme, and Encephalitozoon cuniculi; (2) culture for Bordetella bronchiseptica, Citrobacter rodentium, C. piliforme, Corynebacterium kutscheri, Pasteurella pneumotropica, Salmonella spp, and Streptococcus pneumonia; (3) PCR for parvoviruses, RTV, Helicobacterspp, M. pulmonis, Streptobacillus moniliformis, and Pneumocystis; and (4) necropsy with gross pathology, pulmonary histopathology for IIP, and microscopic examinations for enteric protozoa, endoparasites, and ectoparasites (Research Animal Diagnostic Services; Charles River, Wilmington, Massachusetts). Testing detected Pneumocystis in IIP-positive colonies but did not detect the other infectious agents.
Animal Sample Collection
Rats were euthanized by carbon dioxide inhalation. Immediately after euthanasia, blood was collected to prepare serum for serology and lung specimens for histopathology and Pneumocystis qPCR testing. In some studies, a nasopharyngeal wash, a bronchial wash, and an oral swab were also collected for testing by qPCR. Oral swabs were performed by opening the mouth of the animal and swabbing the tongue and oral cavity with a sterile cotton swab. To perform nasal and bronchial washes, the trachea was exposed and incised using an aseptic technique. A sterile pipette was then used to instill approximately 1 ml of trypticase soy broth (TSB) either rostrally into the nasopharynx (nasopharyngeal wash) or caudally into the lungs (bronchial wash). The TSB was washed several times through the nasopharynx or the bronchi, then aspirated and collected into sterile 1.7-ml tubes for further processing.
Histopathology
Lungs and other tissues were collected from rats after euthanasia. Lungs were gently inflated with 10% neutral-buffered formalin. All tissues were fixed by immersion in formalin. After at least a 24-hour fixation, representative samples were cut from the fixed tissues and processed into paraffin blocks. Sections approximately 4 microns thick were cut onto slides and stained with hematoxylin and eosin (HE) for light microscopic examination by a board-certified veterinary pathologist. For routine pulmonary histopathology, a single section that included portions of all lobes was examined. In certain situations, portions of lung were sampled for other purposes (PCR, bacterial culture, etc), and in those situations, less was available for histopathology. IIP was diagnosed in accordance with published criteria 1 by a board-certified pathologist (T. M. Albers), and all IIP diagnoses were confirmed by a second board-certified veterinary pathologist (M. A. Simon). For lungs from the transmission experiments, the pathologists ranked the severity of IIP on a scale of 0, for no lesions, to 4 indicating severe IIP. Additional sections were cut for PCR evaluation or for special histochemical stains, including a Grocott methenamine silver (GMS).
Pneumocystis qPCR
DNA was isolated by column purification (Qiagen, Inc., Valencia, California) from approximately 15 mg of lung tissue or paraffin-embedded lung or 100 μl of nasal wash, bronchial wash, or vortexed swab wash; the DNA was eluted with 200 μl of AE buffer (Qiagen) and tested by a qPCR targeting a region of the mitochondrial large subunit 18S rRNA gene conserved among Pneumocystis species. This target was selected using the blastx algorithm 5 to align GenBank accession AY532651 (http://www.ncbi.nlm.nih.gov/GenBank/index. html) and to identify regions specific for Pneumocystis. Primers were designed and analyzed with NetPrimer software (Premier Biosoft International, Palo Alto, California): PNEU18F (5′-TAACCTRCTAAATAGCCAGATTA-3′) and PNEU18R (5′-GGCTTCATGCCAAYAGTC-3′). The probe was analyzed using Primer Express software (Applied Biosystems, Foster City, Calfiornia): PNEU18P (5′-FAM-CCYGCGATCAGC-MGB-3′). All primers and probes in this study were synthesized by Sigma (St. Louis, Missouri) and Applied Biosystems, respectively. The positive control standard was prepared by amplifying a product from a Pneumocystis-positive RNU rat lung with HotStar Hifidelity polymerase (Qiagen) and ligating the product into a plasmid (pCR 4 Blunt TOPO; Invitrogen, Carlsbad, California), according to the manufacturer’s instructions. Plasmid DNA specimens from PCR-positive transformed bacteria were submitted to Tufts University Core Facility (Boston, Massachusetts) for DNA sequencing to verify product integrity and to confirm identification consistent with available GenBank accessions. The plasmid concentration was quantified via spectrophotometer (OD260/280) and diluted to create a positive control standard, which was included in every assay run. Reaction components and thermocycler parameters used for the qPCR are as described 33 and performed on an ABI 7300 real-time PCR instrument (Applied Biosystems). The instrument reported assay reactions as cycle threshold (CT) values. These were converted to Pneumocystis DNA (or genome) copies/reaction using a CT value of 3.3 to calculate 10-fold differences from a 100 plasmid-copy standard. 45 The Pneumocystis DNA copies/reaction were adjusted to copies per mg lung, per μl of bronchial or nasal wash, and per swab. The limit of detection for qPCR assay was determined to be 1 to 10 plasmid copies. A positive cutoff of 5 DNA copies was selected based on receiver operating characteristic (ROC) curve analysis. 30
Pneumocystis Mitochondrial DNA Sequencing
A portion of the large subunit ribosomal (rRNA) mitochondrial DNA was amplified with 2 overlapping primer sets, yielding a PCR product of 325 base pairs (primers subtracted). Primer set 1 was designed as described above by using GenBank accession AF337535 (http://www.ncbi.nlm.nih.gov/GenBank/index.html)—PCARMRF (5′-GTGATTGTGTACGTTGC-3′) and PCARMRR (5′-GGCTGTTTCCAAGCCCA-3′)—and primer set 2 was previously described (PAZ102-E and PAZ102-H). 27 Reaction components and thermocycler parameters for both primer sets were performed as described for primer set 2. 27 PCR products were submitted to Tufts University Core Facility for DNA sequencing. Sequences were aligned using Vector NTI software (Invitrogen).
IFA for Pneumocystis Serum Antibodies
To prepare IFA antigen, Pneumocystis-infected lungs, collected from RNU rats after 15 weeks of exposure in an IIP isolator, were homogenized with the Stomacher microbiologic blender (Bioscience International, Rockville, Maryland), and Pneumocystis were purified by differential filtration and centrifugation as described by Thomas et al. 43 Pneumocystis infection was verified by light microscopy of GMS-stained lung sections; qPCR measured the concentrations of Pneumocystisin lung homogenate and the purified suspension. Suspended Pneumocystis were dispensed and dried onto glass-slide wells, to which they were fixed by immersion in cold acetone. Slides were stored at ≤60°C. The IFA procedure was performed as described. 33 Briefly, serum incubation was followed by a wash step, incubation with FITC-labeled goat IgG anti-rat IgG (Molecular Probes, Eugene, Oregon), and a second wash. After being covered with mounting medium and a coverslip, slide wells were examined with an epi-illumination fluorescence microscope (Carl Zeiss AG, Oberkochen, Germany), and reactions were classified as positive, negative, or nonspecific according to the pattern and intensity of fluorescence. A positive control serum from naturally infected rats and a negative control serum were tested in every IFA run to verify assay sensitivity and specificity. Serum samples were screened at a 30-fold dilution. Serial 0.5 log10 dilutions of each positive serum were retested to determine antibody titers (ie, the reciprocal of the highest dilution that gave a positive reaction).
IIP Isolators for Transmission Experiments
Rats from infected sources were added to IIP isolators 1 and 2 in January 2007 and January 2009, respectively. The sources included Sprague-Dawley rats from a vendor in Europe and another in North America and genetically modified rats; they did not include the ZDF barrier-room colony to which naive WH rats were exposed in the previously published study. 1 To perpetuate IIP, we periodically added to each isolator several naive 3- to 4-week-old female Crl: CD(SD) (CD) rats or Crl: NIH-Foxn1rnu athymic nude (RNU) rats obtained from commercial colonies (Charles River), where quarterly pulmonary histopathology and qPCR had never detected either IIP or Pneumocystis, respectively. Naive CD rats were added each week and RNU rats were added monthly or bimonthly. Newly introduced rats were exposed to IIP indirectly by soiled bedding transfer or by direct contact with infective isolator rats. Perpetuation of IIP in each isolator was monitored by pulmonary histopathology performed on CD rats at times postexposure previously determined to be optimal for lesion detection. 1
Transmission Experiments
Pulmonary histopathology and Pneumocystis qPCR and IFA were performed on temporally collected respiratory and serum specimens from 4 separate transmission experiments involving 1 immunodeficient and 2 immunocompetent rat strains, 3 independent sources of infection, and exposure to infection by soiled bedding transfer, contact, and oronasal inoculation of infective lung homogenate. The transmission experiments, summarized in Table 1, include the following:
Summary of Infectious Interstitial Pneumonia (IIP) Transmission Experiments

aArchived lung and serum specimens from Albers et al. 1
The study published by Albers et al 1 in which naive WH (Crl: WI(Han)) rats were introduced to a barrier room containing an endemically infected breeding colony of ZDF rats (ZDF-Leprfa /Crl) housed in open cages. The study rats were housed on 50% soiled bedding from the cages of colony rats 8 weeks of age and older.
Contact exposure of naive CD rats by co-housing them for 2 weeks with infective rats in IIP isolator 1 or 2. The infective rats were exposed by direct contact 2 weeks earlier.
Contact exposure of naive athymic RNU rats in isolator1 or 2 as described for experiment II.
Contact exposure of naive CD rats to RNU index rats infected with Pneumocystis by oral and intranasal inoculation of 50 μL per site of lung homogenate shown by qPCR to contain approximately 10 5 Pneumocystis genome copies/100-μL dose. The infected homogenate was prepared from the lungs of an RNU rat exposed to infection in IIP isolator 1 for 20 weeks. Prior to animal inoculation, the Pneumocystis-infected lung homogenate was treated with penicillin-streptomycin (Sigma) and cultured for extraneous bacteria and fungi in thioglycollate broth and TSB incubated at 35°C and room temperature, respectively. After 7 days of incubation, no growth was observed in either broth. A control group of naive RNU rats was inoculated with uninfected RNU lung homogenate. Both the test and control groups were reinoculated as described at 3 and 7 days after the initial inoculations. Five days after the final inoculation, RNU sentinel rats were placed in contact with the inoculated rats. Pneumocystis was first detected by qPCR in the lungs of sentinels for the Pneumocystis-inoculated rats at approximately 8 weeks after the first inoculation. All lungs collected at the same time from control rat sentinels were Pneumocystis qPCR negative. Approximately 2 weeks later, naive CD rats were placed in contact with the inoculated RNU rats.
Statistical Analysis
All calculations were done in R. 7 The Pneumocystis qPCR positive cutoff in DNA copies/mg lung (or μl wash) was determined by ROC curve analysis of the results for known positive and negative lung specimens. 30 The chosen cutoff of 5 copies gave an estimated sensitivity of 93% and specificity of 100%. The Kruskal-Wallis rank sum test, a nonparametric counterpart to analysis of variance (ANOVA), was used to compare the Pneumocystis DNA copies/mg lung for experimentally infected CD rats (from transmission experiments II and IV) grouped by their IIP and serologic statuses. The Cochran test with pairwise comparisons and the McNemar test compared the positive rates by test method and by PCR specimen type. The Cochran and McNemar tests were used instead of Pearson’s χ 2 test because the former tests are more accurate when repeated measurements on the same animals are compared. Bonferroni’s approach controlled for compounding error. 16 Fisher exact test (the exact equivalent of Pearson’s χ 2 test) measured correspondence between finding IIP and Pneumocystis in rat lungs from diagnostic submissions.
Results
Experimental Transmission of IIP and Pneumocystis to Immunocompetent and Immunodeficient Rats
Pulmonary histopathology on an immunodeficient RNU rat from an IIP isolator demonstrated Pneumocystis in the same areas of the lung as IIP lesions (Figs. 1–4). Initially, this finding was considered incidental based on the putative viral etiology of IIP and the general understanding that Pneumocystispneumonia is principally a disease of immunodeficient hosts. The results of ongoing Pneumocystis qPCR testing and pulmonary histopathology of rats from the IIP isolator colonies, however, consistently showed in lungs from CD rats a correlation between the concentration of Pneumocystis DNA copies and the severity of IIP (data not shown). In addition, between January 2009 and July 2010, PCR failed to detect Pneumocystis in any of the more than 2500 bronchial washes from rats in 55 IIP-negative barrier-room colonies located in 10 Charles River facilities in North America, Europe, and Japan.
The role of Pneumocystis infection in the pathogenesis of IIP was investigated by analyzing respiratory and serum specimens collected at regular (usually weekly) intervals during the 4 transmission experiments summarized in Table 1. In these experiments, infection was transmitted to immunocompetent CD and WH rats and athymic RNU rats from 3 independent sources of IIP, including an enzootically infected ZDF breeding colony in a barrier room, and IIP isolators 1 and 2. To model natural transmission, rats were exposed to infection by bedding transfer (experiment I), direct contact with contagious animals in IIP isolators 1 and 2 (experiments II and III), or contact with RNU rats inoculated oronasally with infective lung homogenate prepared from an RNU rat in IIP isolator 1 (experiment IV).
In transmission studies with immunocompetent CD and WH rats, qPCR detected Pneumocystis in lungs several weeks before pulmonary histopathology demonstrated IIP (Figs. 5 and 6). For example, of the 12 CD rats tested at 1 week following contact exposure in experiment II, 5 (42%) were Pneumocystis qPCR positive, whereas none had IIP lesions (Fig. 6a, c). The percentage of lungs with Pneumocystis rose to 100% at 2 weeks postexposure. By comparison, IIP lesions were not seen until 3 weeks and then in just 1 of 12 rats (8.3%); the maximum IIP prevalence of 85.7% was reached 1 to 2 weeks later. Figures 1 and 2 are photomicrographs showing the close similarity in appearance between IIP in WH rats from our earlier study and CD rats.
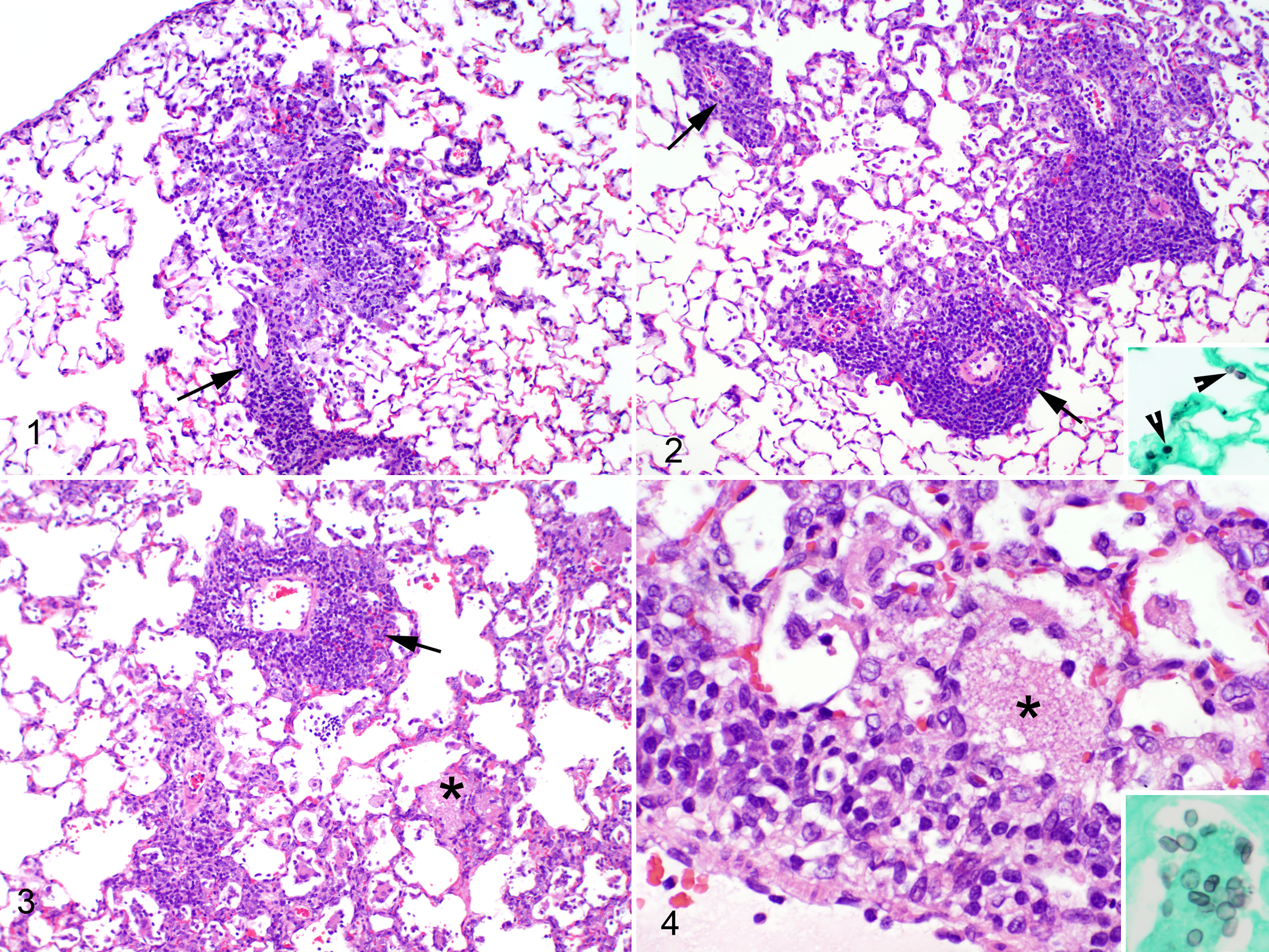
Lung, CD rat from the previously published study.
1
Depicted is an area of thickened alveolar septa surrounding alveoli infiltrated with lymphocytes and macrophages, that is, lymphohistiocytic interstitial pneumonia with prominent encirclement of blood vessels by thick bands of lymphocytes (cuffing, arrow). These lesions satisfy the histologic criteria established for infectious interstitial pneumonia (IIP). Hematoxylin and eosin (HE).
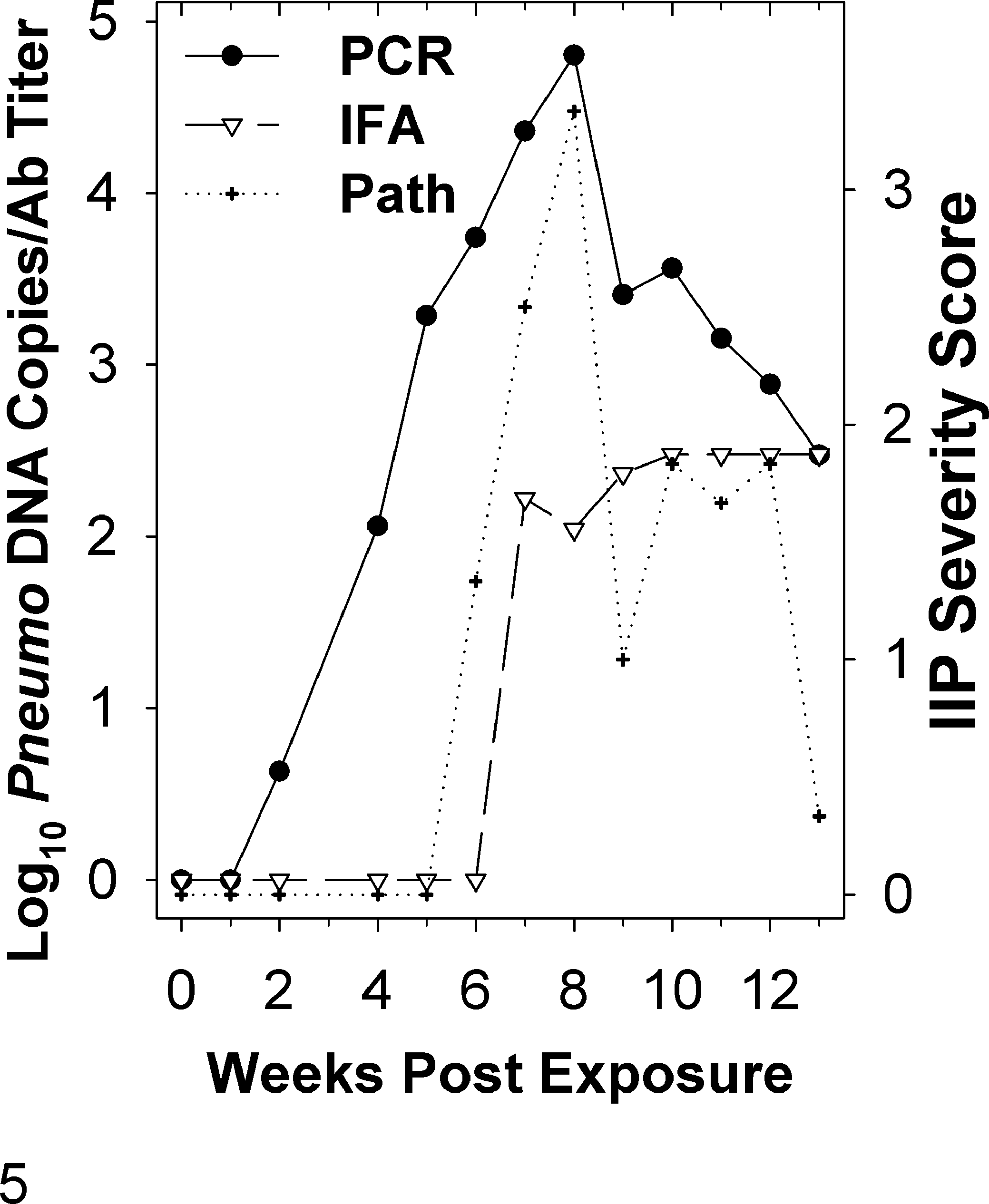
Infectious interstitial pneumonia (IIP) and Pneumocystis infection and humoral immunity in bedding-exposed WH rats from transmission experiment I (Table 1). Naive WH rats were introduced to a barrier room containing a ZDF rat colony with enzootic IIP; the naive rats were housed on 50% soiled bedding transferred from the cages of colony rats ≥8 weeks of age. Each graphed data point represents an average for 3 rats. The IIP severity scores, assigned by the pathologists who examined the lung sections, are descriptive. The Pneumocystis DNA copies/mg lung and serum antibody titers were measured by quantitative polymerase chain reaction (qPCR) and immunofluorescence assay (IFA), respectively. A value of 100 indicates ≤1 DNA copy/mg lung or an antibody titer of 30.
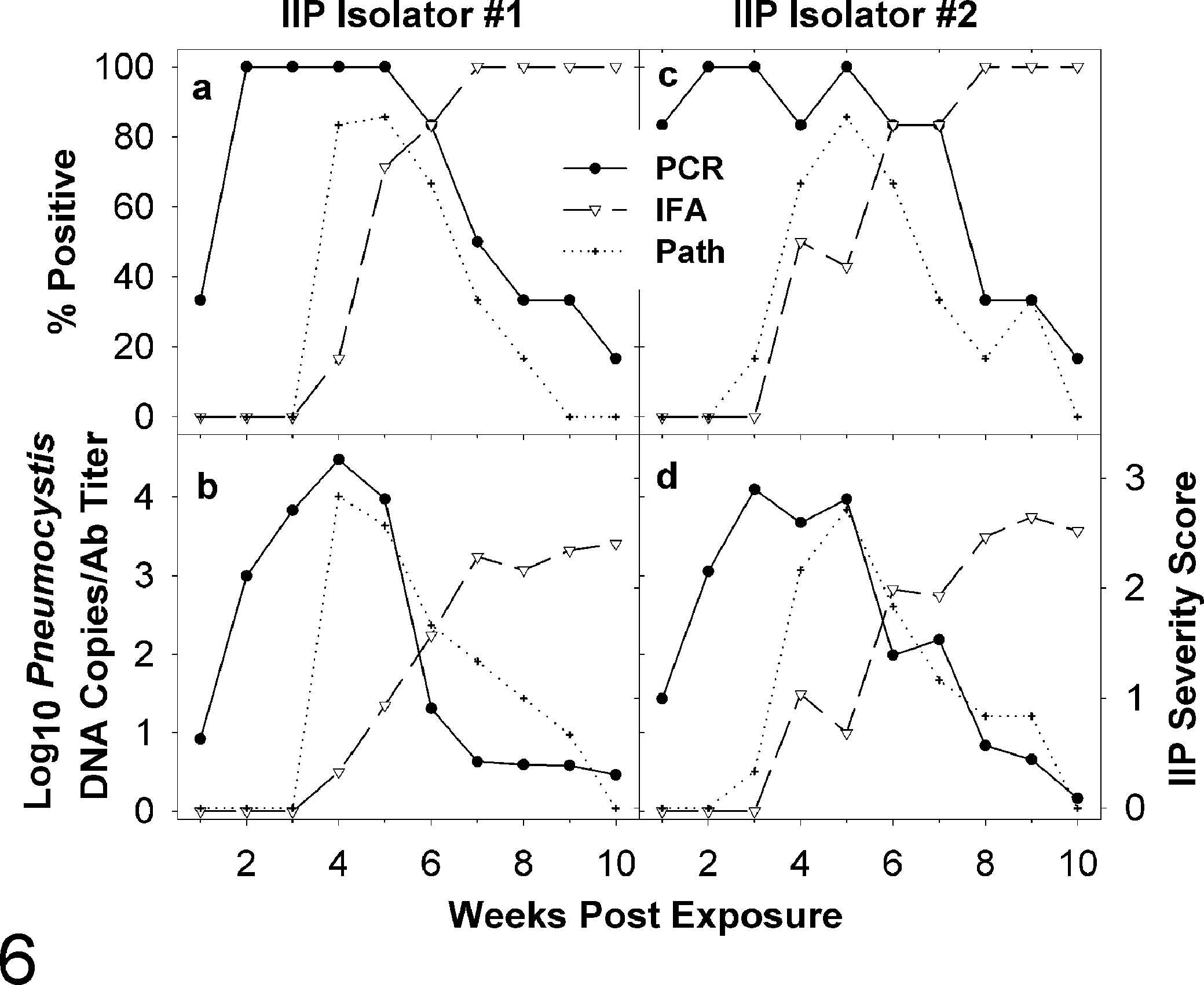
Infectious interstitial pneumonia (IIP) and Pneumocystisinfection and humoral immunity in contact-exposed CD rats from transmission experiment II (Table 1). Naive CD rats were added to both IIP isolators where they were co-housed with infective rats for 1 week; 6 rats were removed from each isolator per week, except on week 5, when 7 animals were removed. Graphs (a) and (c) show the percentages of contact-exposed CD rats from isolators 1 and 2 with IIP by pulmonary histopathology, ≥5 Pneumocystis DNA copies/mg lung by quantitative polymerase chain reaction (qPCR), and antibody titers ≥0.5 log10 by immunofluorescence assay (IFA). Graphs (b) and (d) show the mean IIP severity scores, Pneumocystis DNA copies/mg lung, and serum antibody titers. A zero value indicates a Pneumocystis concentration ≤1 or an antibody titer below 30. The IIP severity scores assigned by the pathologists who examined the lung sections are descriptive.
The severity of IIP and concentration of Pneumocystis in lungs peaked concurrently at 7 to 8 following bedding exposure in WH rats (Fig. 5) and at 3 to 5 weeks following contact exposure in CD rats (Fig. 6). The peaks in CD rats occurred at 3 weeks in rats co-housed with infected RNU rats (Table 2) and at 4 and 5 weeks for CD rats exposed by contact in IIP isolator 1 (Fig. 6b) and isolator 2 (Fig. 6d), respectively. In both the WH and CD rats, the concentration of Pneumocystis peaked at 10 4 to 10 5 DNA copies/mg lung. Evaluation of GMS-stained lung sections from contact-exposed CD rats at 3, 4, and 5 weeks postexposure found rare silver-positive cysts in alveoli, although the cyst forms were not seen in the areas of inflammation (Fig. 2).
Transmission of Pneumocystis to CD Rats Co-housed With RNU Rats Infected by Oronasal Inoculation of Infective Lung Homogenate (Transmission Experiment IV [Table 1])
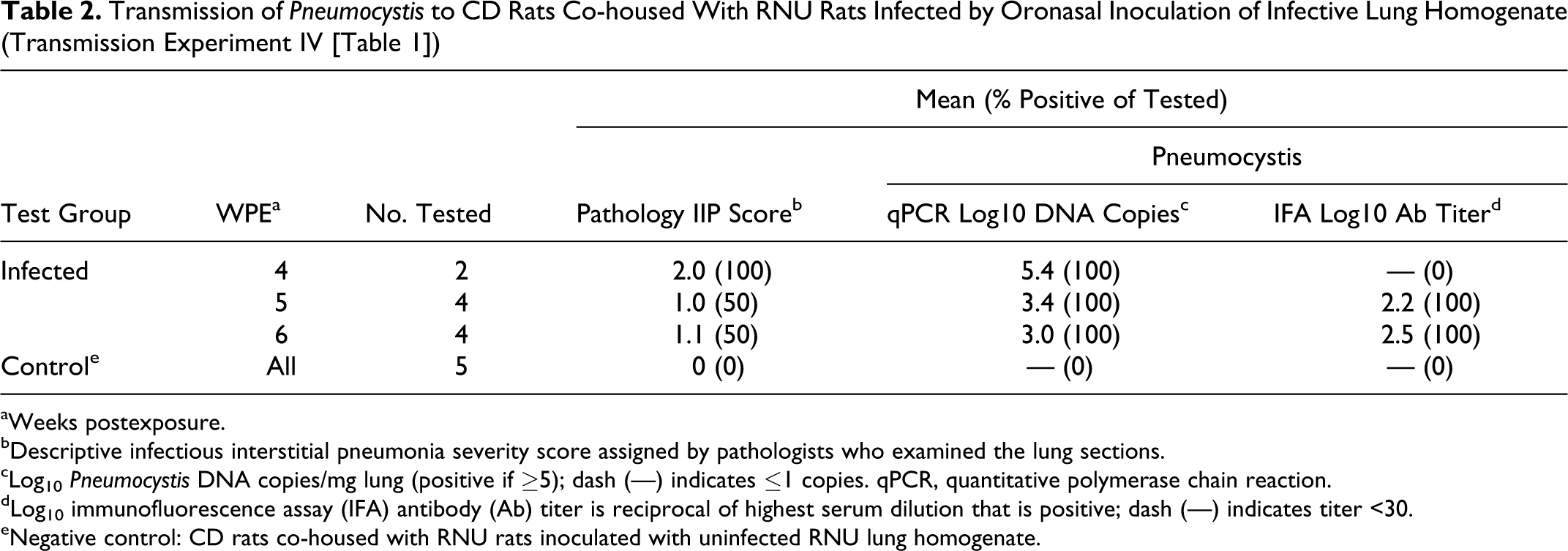
aWeeks postexposure.
bDescriptive infectious interstitial pneumonia severity score assigned by pathologists who examined the lung sections.
cLog10 Pneumocystis DNA copies/mg lung (positive if ≥5); dash (—) indicates ≤1 copies. qPCR, quantitative polymerase chain reaction.
dLog10 immunofluorescence assay (IFA) antibody (Ab) titer is reciprocal of highest serum dilution that is positive; dash (—) indicates titer <30.
eNegative control: CD rats co-housed with RNU rats inoculated with uninfected RNU lung homogenate.
Pneumocystis seroconversion by IFA coincided with the concurrent peaks in the prevalence and severity of IIP and the concentration of Pneumocystisin lungs. As the percentage of Pneumocystis-seropositive animals increased and IFA antibody titers rose, the prevalence and levels of disease and infection declined. All CD rats were seropositive by 5 weeks after being co-housed with oronasally inoculated RNU rats (Table 2) and by 7 and 8 weeks following contact with infective rats in IIP isolator 1 (Fig. 6a) and isolator 2 (Fig. 6c), respectively. At 10 weeks following contact exposure in the IIP isolators, Pneumocystis IFA serum antibody titers in CD rats ranged from 103 to 104, with a mean of approximately 4000; of the 12 animals sampled at this time, none had IIP, and qPCR measured low levels of Pneumocystis (ie, roughly 10 and 100 DNA copies/mg) in the lungs of just 2 rats.
In CD contact transmission experiment II, a total of 122 rats from the 2 IIP isolators were sampled from 1 to 10 weeks postexposure. Of the 53 rats that were Pneumocystis IFA seronegative, qPCR detected Pneumocystis in lungs from 46 (86.8%), which was significantly higher than the 14 (26.4%) diagnosed with IIP (P < .001, Cochran and McNemar tests). For these seronegative animals, the median concentration of organisms in lungs with and without IIP was 104.6 and 102.9 DNA copies/mg, respectively. This concentration difference was significant (P < .05, Kruskal-Wallis test) and consistent with the correlation between the level of Pneumocystis infection and prevalence of IIP shown in Fig. 6.
Of the 132 contact-exposed rats in transmission experiments II and IV, histopathology demonstrated IIP in 44 (33%), qPCR found Pneumocystis in the lungs of 84 (64%), and IFA detected serum antibodies in 77 (58%) (Table 4). The positive rates by Pneumocystis qPCR and IFA were not statistically different from one another (P > .05, Cochran and McNemar tests), but both were significantly higher than the percentage of rats in which histopathology found IIP (P < .001).
In the contact transmission experiment to immunodeficient RNU rats (experiment III), Pneumocystis was detected in all rats sampled from 2 weeks postexposure forward. The concentration of Pneumocystis continued to rise throughout the study. At 10 weeks postexposure, it reached 106 DNA copies/mg lung (Fig. 7), which was 10- to 100-fold higher than the peaks in immunocompetent hosts. IFA did not detect Pneumocystis antibodies in sera from any of the 40 RNU rats sampled between 1 and 10 weeks postexposure. In sharp contrast to the time course of IIP in immunocompetent animals, lesions consistent with IIP were not found in RNU rats until 10 weeks postexposure and then in just 2 of 4 rats (data not shown).
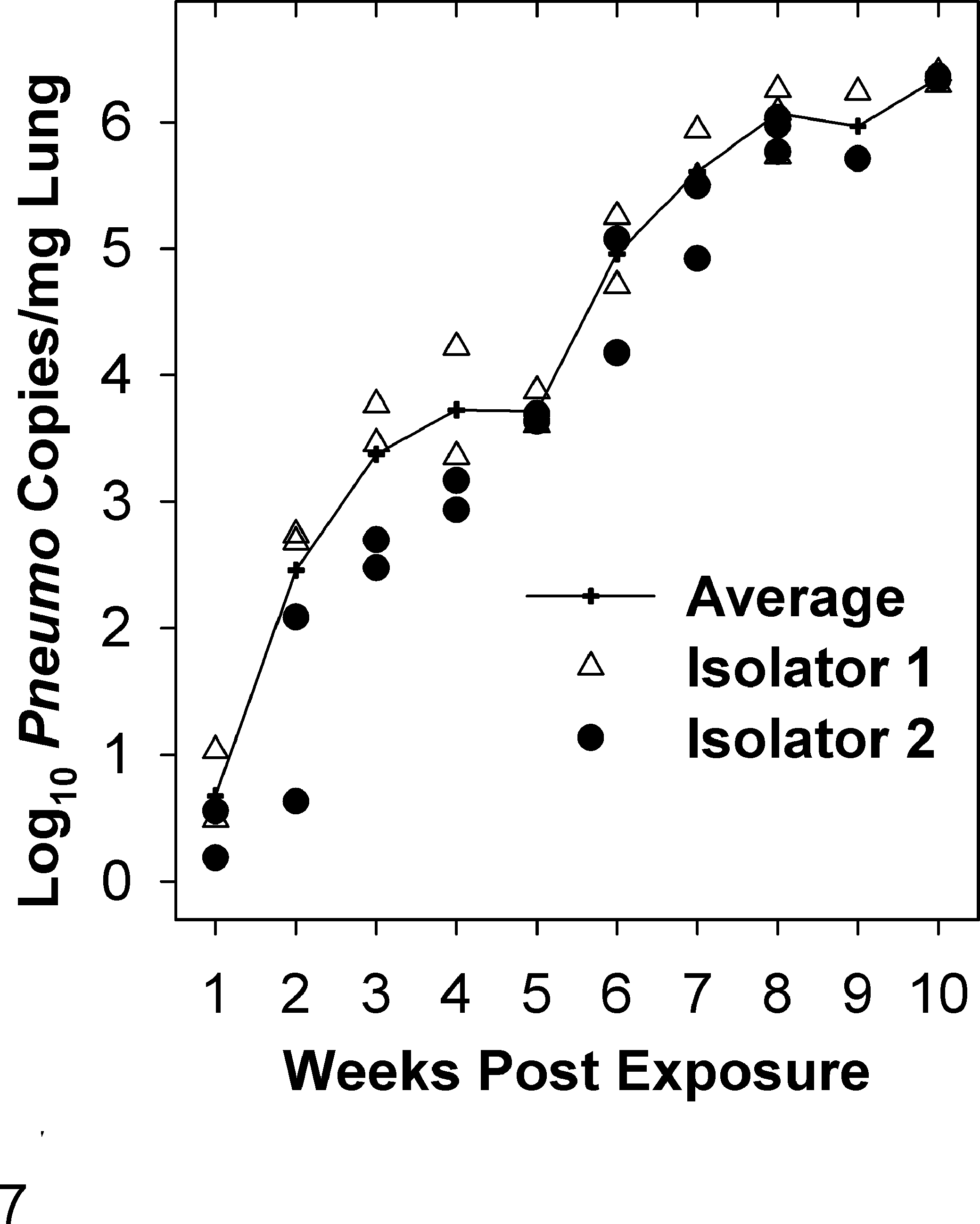
Pneumocystis infection in contact-exposed athymic RNU rats in transmission experiment III (Table 1). Naive RNU rats were added to both infectious interstitial pneumonia (IIP) isolators where they were co-housed with infective rats for 1 week. Two RNU rats were removed weekly from each isolator (ie, 4 rats per week) for testing. The triangles and circles represent the Pneumocystis quantitative polymerase chain reaction (qPCR) results for individual RNU rats from IIP isolators 1 and 2, respectively; 100 indicates ≤1 DNA copy/mg lung. IIP was only diagnosed at 10 weeks postexposure in 2 of the 4 rats evaluated at that time and was not found in any of the 36 experimental animals sampled prior to 10 weeks. All rats were Pneumocystis seronegative by immunofluorescence assay (data not shown).
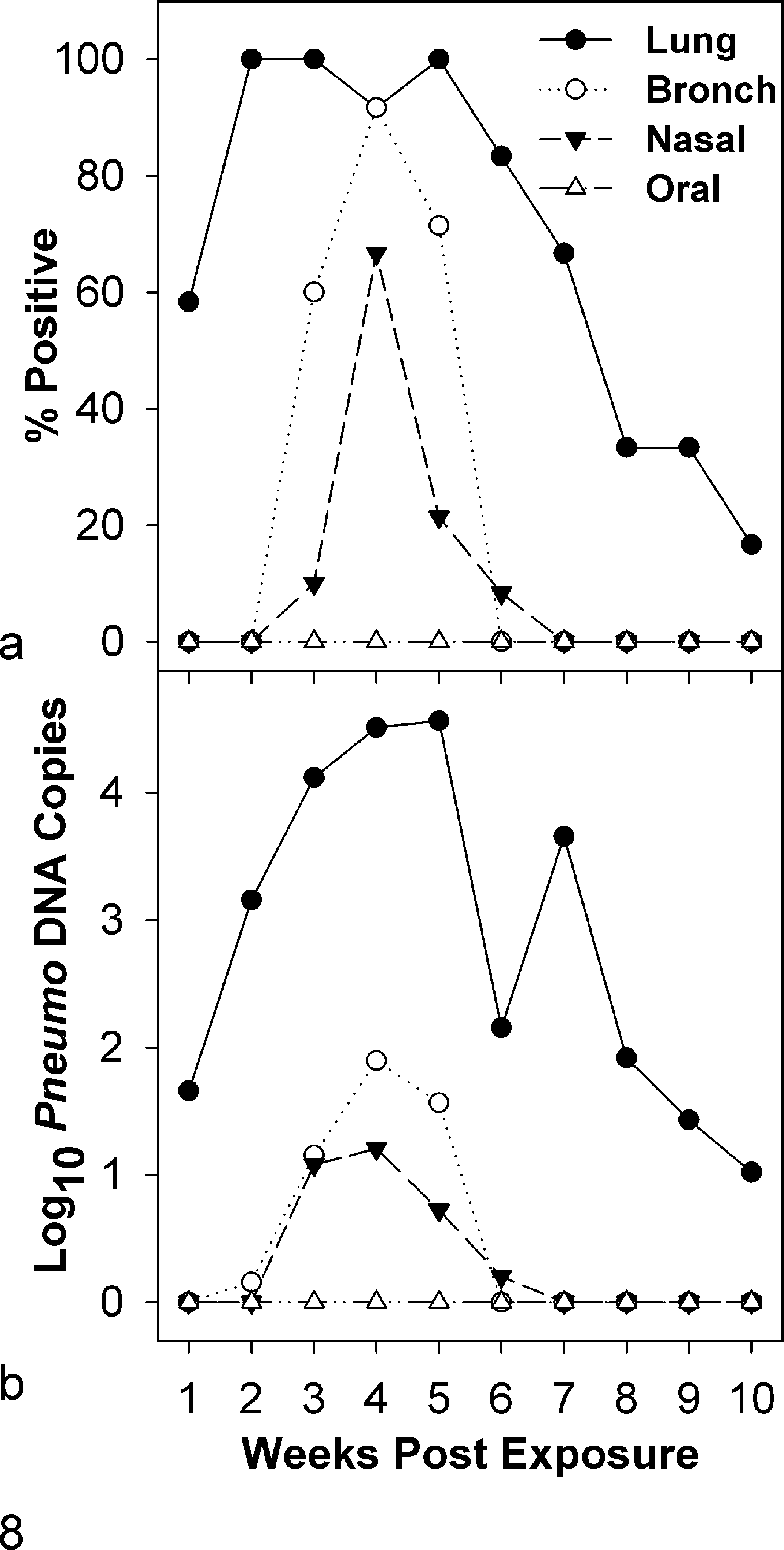
Detection of Pneumocystis in different respiratory specimen types by quantitative polymerase chain reaction (qPCR). Lung, bronchial wash, nasal wash, and oral swab specimens were collected at weekly intervals from contact-exposed CD rats during transmission experiment II (Table 1). Each graphed data point represents results for 12 rats (6 per infectious interstitial pneumonia [IIP] isolator). The bottom graph (b) shows the mean Pneumocystis genome copies/mg lung, μl wash or oral swab; 100 indicates ≤1 copy. The top graph (a) shows the percentage of samples with Pneumocystis concentrations ≥5 genome copies.
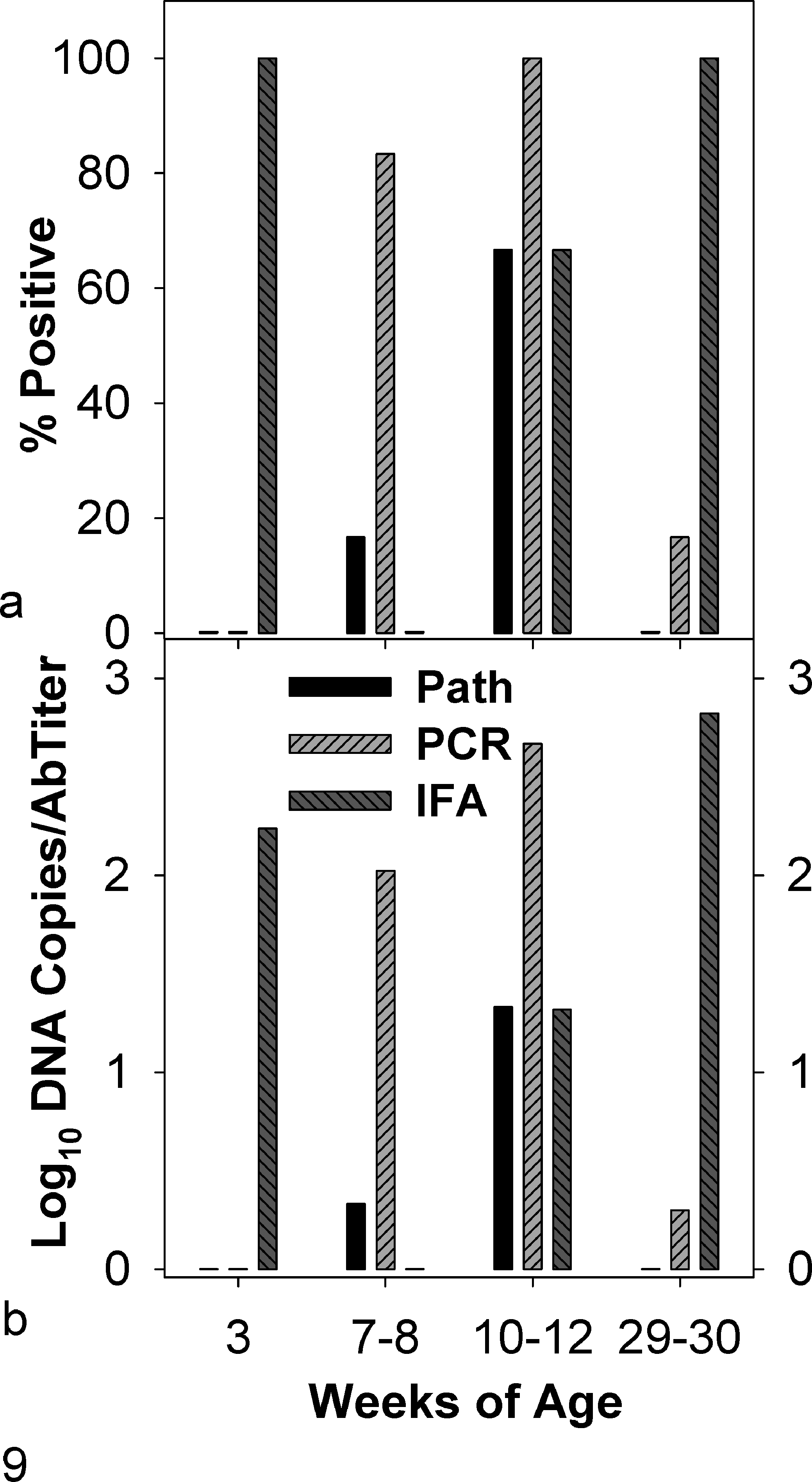
Infectious interstitial pneumonia (IIP) and Pneumocystis infection and humoral immunity in SD rats from an enzootically infected barrier-room breeding colony. The number of rats tested in the 29- to 30-week-old age group was 18; otherwise, 6 animals were tested per age group. Shown in the top (a) graph are the percentages of rats in each age group with IIP by pulmonary histopathology, ≥5 Pneumocystis DNA copies/mg lung by quantitative polymerase chain reaction (qPCR), or Pneumocystis antibody titers ≥30 by immunofluorescence assay (IFA). The mean Pneumocystis DNA copies/mg lung, antibody titers, and descriptive IIP severity scores, for each age group, are shown in the bottom (b) graph. A 100 value indicates a Pneumocystis concentration ≤1 copy/mg lung or an antibody titer below 30.
Comparison of Respiratory Specimen Types for Detection of Pneumocystis by qPCR
During transmission experiment II, bronchial wash, nasal wash, and oral swab specimens were collected in addition to lung from 120 (of 122) contact-exposed CD rats. qPCR first detected Pneumocystis in lungs at 1 week postexposure and in bronchial and nasal washes 2 weeks later but never demonstrated the organisms in oral swabs (Fig. 8, Table 3). From 2 to 5 weeks postexposure, the percentage of infected lungs averaged 98%; in association with the development of humoral immunity, the percentage decreased to 17% when the study concluded at 10 weeks. By comparison, Pneumocystis could only be demonstrated in bronchial and nasal washes between 3 and 6 weeks postexposure; during this period, 75% of the bronchial washes and 33% of the nasal washes were qPCR positive. Of the 120 rats from which upper and lower respiratory samples were collected during this 10-week study, Pneumocystis was detected in 68% of the lungs; this percentage was approximately 3 and 6 times higher than those for bronchial and nasal washes, respectively. In addition, the average Pneumocystis DNA copies/mg lung was 40- to 80-fold higher than the average copies/μl of bronchial or nasal wash (Table 3).
Quantitative Polymerase Chain Reaction (qPCR) Detection of Pneumocystis in Lung and Other Respiratory Specimens Collected From 120 CD Rats From 1 to 10 weeks After Contact Exposure During Transmission Experiment II (Table 1)
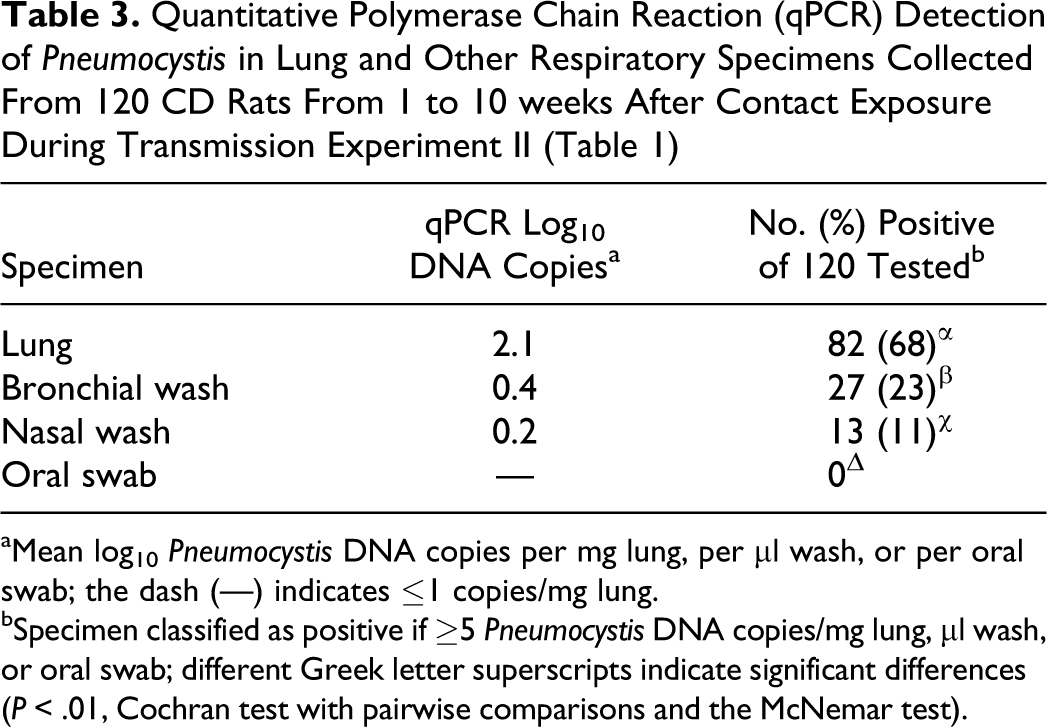
aMean log10 Pneumocystis DNA copies per mg lung, per μl wash, or per oral swab; the dash (—) indicates ≤1 copies/mg lung.
bSpecimen classified as positive if ≥5 Pneumocystis DNA copies/mg lung, μl wash, or oral swab; different Greek letter superscripts indicate significant differences (P < .01, Cochran test with pairwise comparisons and the McNemar test).
Detection of Pneumocystis in Immunocompetent Rats by Pulmonary Histopathology, Polymerase Chain Reaction (PCR), and Immunofluorescence Assay (IFA)
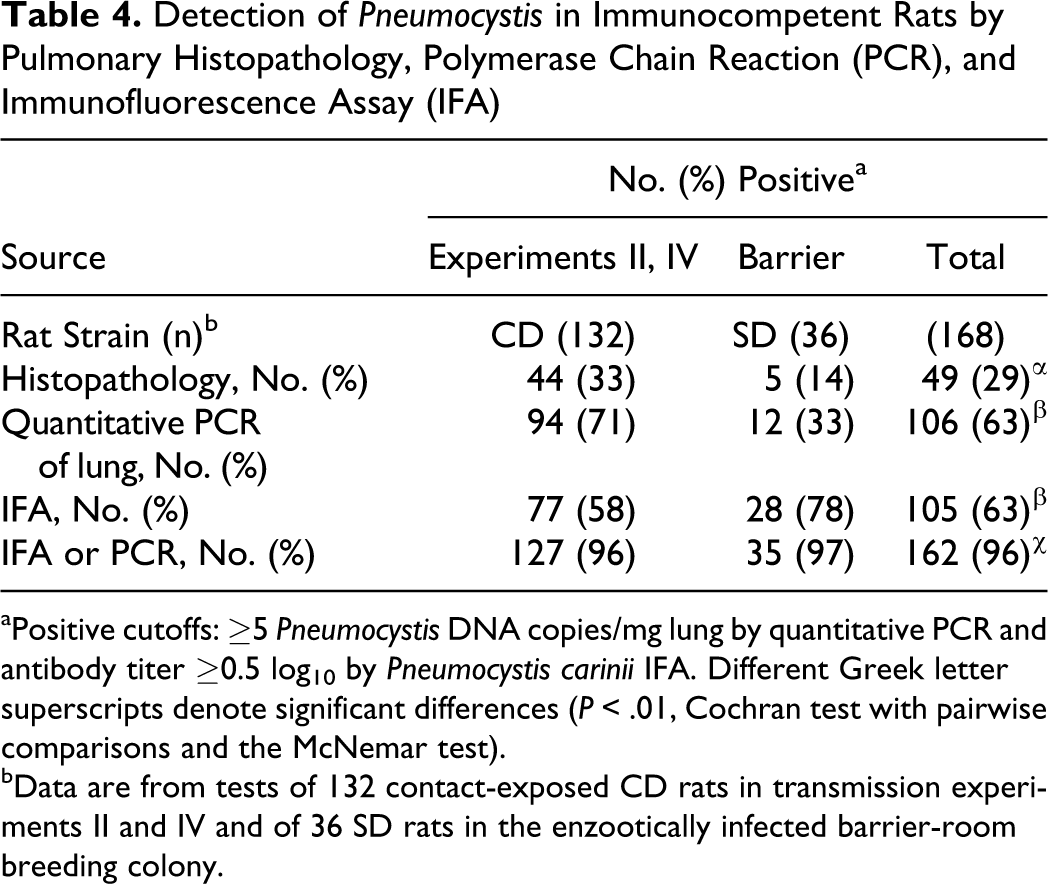
aPositive cutoffs: ≥5 Pneumocystis DNA copies/mg lung by quantitative PCR and antibody titer ≥0.5 log10by Pneumocystis cariniiIFA. Different Greek letter superscripts denote significant differences (P < .01, Cochran test with pairwise comparisons and the McNemar test).
bData are from tests of 132 contact-exposed CD rats in transmission experiments II and IV and of 36 SD rats in the enzootically infected barrier-room breeding colony.
Pneumocystis Survey of IIP-Positive Rat Populations
Lung sections from external diagnostic submissions where 1 or more rats were diagnosed with IIP were tested for Pneumocystis by qPCR. Of the 99 lungs sections evaluated, 38 were positive for IIP and Pneumocystis and 54 were negative for both. Of the 7 remaining sections, 2 had IIP without Pneumocystis and 5 were Pneumocystis positive without IIP. The 93% agreement between the histopathology and Pneumocystis PCR results was highly significant (P < .001, Fisher exact test).
SD rats from 4 age groups were acquired from a barrier-room breeding colony reported by the vendor to have enzootic IIP and Pneumocystis. This colony was unrelated to those used as sources of infection for the transmission experiments listed in Table 1. There were 18 rats 29 to 30 weeks of age and 6 rats in each of the younger age groups. As shown in Fig. 9, all of the 3-week-old rats had maternal antibody titers to Pneumocystis, and none was infected with Pneumocystis or had IIP lesions. 21 Pulmonary histopathology found IIP in one 7- to 8-week-old rat and four 10- to 12-week-old rats, whereas qPCR detected Pneumocystis in the lungs of 11 of the 12 rats in these age groups; the single Pneumocystis-negative rat did not have IIP. The average Pneumocystis concentration for the 5 IIP-positive rats was 103.7DNA copies/mg lung, which was 100-fold higher than the average concentration of 101.6in the remaining 6 qPCR-positive rats that were IIP negative. Pneumocystis antibodies were undetectable in sera from 7- to 8-week-old rats but were demonstrated in sera from four 10- to 12-week-old and all eighteen 29- to 30-week-old rats. None of the 29- to 30-week-old rats had IIP; 1 was Pneumocystis qPCR positive but with just 15 DNA copies/mg lung. Of the 24 colony rats evaluated, IIP was found in 5 (21%), and Pneumocystis was detected in lungs from 12 (50%); 28 (78%) were Pneumocystis seropositive.
A comparison of pulmonary histopathology, Pneumocystis qPCR, and P. carinii IFA results for 168 immunocompetent rats is shown in Table 4 and Figure 9. These included 132 contact-exposed CD rats from transmission experiments II and IV and 36 SD rats from the enzootically infected barrier-room breeding colony. Overall, the percentages of rats that were positive by qPCR and IFA were more than twice the percentage diagnosed with IIP by histopathology (P < .01, Cochran and McNemar test). Rats were positive for Pneumocystis by either qPCR or serology 96% of the time, which was significantly more frequent than the 63% by each method alone (P < .01). For the experimentally infected CD rats, the percentages that were positive by qPCR and IFA were 71% and 58%, respectively; 78% of the SD rats from the breeding colony were seropositive, whereas 33% were positive by qPCR.
Genotyping of Pneumocystis Linked to IIP
DNA was isolated from rat lungs representing 11 independent sources of Pneumocystis infection, including the 2 IIP isolators, 2 barrier-room colonies, and 7 paraffin-embedded lungs from external diagnostic submissions. A 325 base-pair segment of the mitochondrial rRNA gene from each of these Pneumocystis sources was sequenced. The sequenced gene segment corresponded to P. carinii nucleotide positions 21207 to 21531 in GenBank accession GU133622. The genotypes for all sources were the same and, according to Blastn analysis, identical to the sequence in GU133622; moreover, they matched the partial sequences available for P. carinii GenBank accessions U20169 and U20170.
Pneumocystis Survey of Immunocompetent Rats From IIP-Negative Charles River Barrier-Room Breeding Colonies
Charles River’s policy has been to screen rat colonies for IIP by performing quarterly histopathology on young adult rats and to depopulate and disinfect rooms containing colonies with IIP. Pulmonary histopathology detected IIP in 0.6% of the more than 13 000 rats evaluated since 2002 and in none of the 2500 animals screened in 2009 and 2010. We reported elsewhere 32 that the prevalence of IIP in 3900 rats from external sources between 2002 and 2008 was 6%.
As noted earlier, PCR did not detect Pneumocystis in any of the 2500 rat bronchial washes from IIP-negative colonies screened in 2009 and 2010. To corroborate that IIP-negative rat colonies are indeed free of Pneumocystis, we tested rats from IIP-negative colonies by Pneumocystis IFA serology and qPCR of lung specimens, instead of bronchial washes. The switch to lung was made based on data in Table 3 showing that Pneumocystis were found in lungs considerably more often and at much higher concentrations than in bronchial washes. For most colonies, lung and serum samples were collected from each of 4 retired breeder and 4 young adult rats; for some, however, sera were prepared from retired breeders only. As shown in Table 5, 440 lungs and 352 sera were collected from rats representing 49 barrier-room breeding colonies at 10 Charles River facilities on 3 continents. Pneumocystis were not detected by qPCR in any of the lung specimens, and all sera were Pneumocystis IFA negative.
Retired Breeder and 8- to 10-Week-Old Rats From Infectious Interstitial Pneumonia–Negative Charles River Barrier Colonies Tested Negative for Pneumocystis by qPCR of Lung and IFA
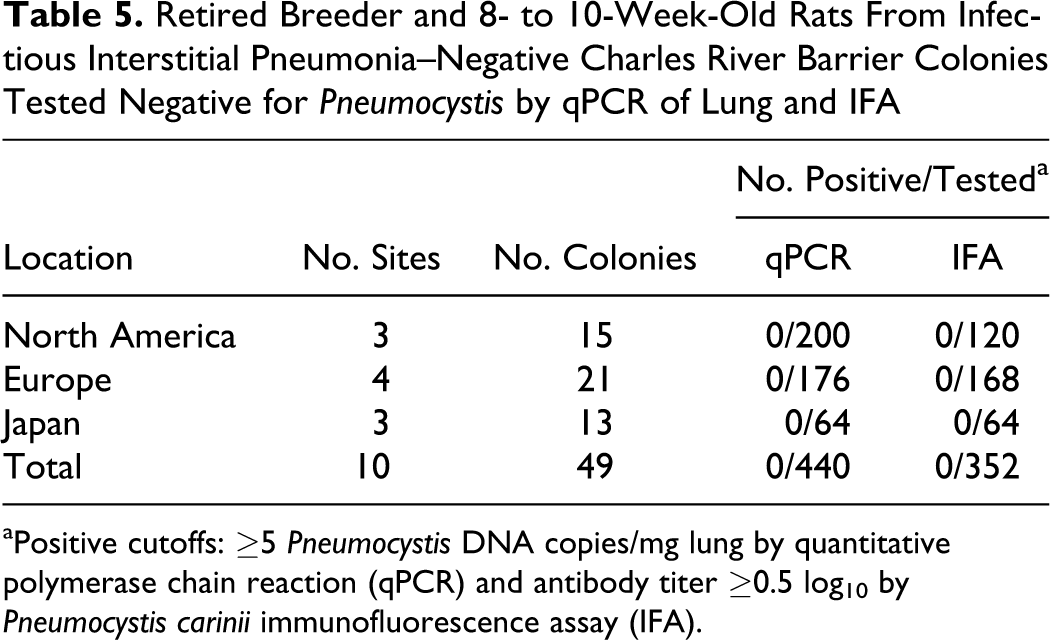
aPositive cutoffs: ≥5 Pneumocystis DNA copies/mg lung by quantitative polymerase chain reaction (qPCR) and antibody titer ≥0.5 log10by Pneumocystis cariniiimmunofluorescence assay (IFA).
Discussion
IIP, a prevalent and transient mild interstitial pneumonia of immunocompetent laboratory rats, had been attributed to a putative viral pathogen referred to as RRV, 9,36,38 but the nature of this agent was never proven. The data in this report provide persuasive evidence that IIP is caused by P. carinii, the most common Pneumocystis species of laboratory rats. Analysis of rat respiratory and serum specimens from 4 transmission experiments, a barrier-room breeding colony with enzootic IIP and Pneumocystis, external diagnostic submissions during 2009-2010, and more than 400 rats from 49 IIP-negative barrier-room colonies in North America, Europe, and Japan, showed that qPCR and IFA only detected Pneumocystis infection and serum antibodies in rats from colonies and experiments in which histopathology demonstrated IIP. In immunocompetent rats, the concentration of Pneumocystis in lungs correlated with the severity of IIP, and the resolution of pneumonia invariably followed the development of Pneumocystis humoral immunity and the clearance of Pneumocystis infection from lungs. The genotypes of IIP-associated Pneumocystis from 11 independent sources matched that of P. carinii. This cause-effect relationship between P. cariniiinfection and IIP that our data support is wholly consistent with findings just published by Livingston et al. 26 Our study was contemporaneous with theirs and was carried out without prior knowledge of their research. Thus, the 2 studies independently corroborate one another and, as far as we can ascertain, are the first reports of Pneumocystis pneumonia in immunocompetent rats.
As the lymphohistiocytic infiltrates of IIP are consistent with a viral etiology and one had been proposed, our preliminary investigations into the cause of IIP focused on finding novel viruses. To do this, lung and other tissues from transmission experiments were probed for viral antigens by Western immunoblotting and indirect IFA with IIP-convalescent rat serum. Nonspecific PCR techniques, such as single primer amplification (SISPA) 3 and random primer amplification, 4 were performed on nucleic acid extracts from nasal aspirate, bronchial wash, clarified fecal slurry, serum, urine, and skin specimens pretreated to deplete host nucleic acid and enrich viruses. These analyses failed to reveal viral antigens or genomic sequences despite being applied to a variety of specimens collected over a wide range of times postexposure (data not shown).
When pulmonary histopathology first demonstrated Pneumocystis in the lungs of an RNU rat from one of the IIP isolators used in transmission experiments and then PCR detected Pneumocystis in the lungs of immunocompetent CD rats from both IIP isolators, we were skeptical of a relationship between the infection and disease because Pneumocystisis primarily an opportunistic pathogen of immunodeficient hosts. Furthermore, we were unable to locate reports of lesions similar to IIP among the many studies of P. carinii in immunosuppressed rats as a model for human pneumocystosis. Finally, according to PCR test results reported by others, Pneumocystis were not detected in diseased rat lungs 36,38 and were very common in commercial rat barrier colonies. 22 –24,47
In immunocompetent rats from the transmission experiments and an endemically infected barrier-room breeding colony, seroconversion to P. carinii coincided with the concurrent peaks of IIP and Pneumocystis infection. Subsequently, in association with increasing antibody titers, P. carinii was cleared from lungs and the lesions of IIP resolved. Infected immunodeficient RNU rats, by contrast, did not seroconvert to or recover from infection with Pneumocystis during a 10-week transmission experiment. Interestingly, of the 40 RNU rats sampled over the course of this experiment, IIP lesions were diagnosed in just 2 at 10 weeks. These striking differences between the pathogenesis of Pneumocystis in immune-competent and immune-deficient hosts are consistent with the characteristic lymphohistiocytic interstitial pneumonia and perivascular cuffs of IIP being mediated by the adaptive immune response to infection. Once the infiltrating immunocytes have cleared the infection, the lesions they compose resolve. This link between IIP and the development of adaptive immunity that clears Pneumocystis infection provides a plausible explanation for cysts rarely being observed in areas of inflammation.
The time course of P. carinii infection in contact-exposed immunocompetent rats shown here is very similar to the time course reported for P. murina in immunocompetent mice exposed to infection by direct contact. 6,18,20,44 In experimentally infected animals of both species, the concentration of Pneumocystis in lungs peaked at approximately 1 month postexposure, and the infection was quickly cleared as the acquired immune response developed.
IIP peaked several weeks later and persisted longer in WH rats exposed to Pneumocystis in bedding than in CD rats exposed by direct contact. 1 This apparently was not due to the strain difference as the slower rate at which IIP progressed in the WH experiment was duplicated in CD rats exposed to infective bedding (data not shown). Thus, we believe that contact-exposed CD rats developed and recovered from IIP sooner because the P. carinii dose they received was much higher than the dose in soiled bedding. The pronounced effect of P. carinii dose on the development and progression of IIP is probably a function of the slow doubling time of Pneumocystis, reported to be 4.5 days in rats. 2
In the SD barrier-room breeding colony with endemic P. carinii, maternal antibodies apparently protected rats from infection until after weaning. Pneumocystis were detected in the lungs of nearly all rats (11/12) between 7 and 12 weeks of age. Lungs from 10- to 12-week-old rats had the highest prevalence of IIP (67%) and concentration of Pneumocystis (103 DNA copies/mg). IFA detected Pneumocystis serum antibodies in none of the 7- to 8-week-old rats and in 4 of 6 (67%) of the 10- to 12-week-old rats. By 29 to 30 weeks of age, all rats were P. carinii seropositive, with antibody titers that were on average 10-fold higher than those in the 10- to 12-week-old group. These results correspond with those from the transmission experiments in that the peaks of IIP and Pneumocystis infection were concurrent and coincided with seroconversion; in addition, the clearance of the Pneumocystis infection by the acquired immune response was followed by the resolution of IIP. This pattern of infection being delayed by passive immunity until after weaning and convalescence, following with the development of active immunity, is common for endemic infections in research animal breeding colonies. 29
Overall, comparative test results in this report for experimentally and naturally infected rats show that the positive rate for both Pneumocystis assays is over 60%, which is more than twice the rate at which pulmonary histopathology detected IIP. Combined, the Pneumocystis assays detected lung infection or serum antibodies in 96% of rats. Even though Pneumocystis assays proved to be significantly more sensitive than histopathology in this study, we have never found Pneumocystis in IIP-negative rat colonies or failed to detect Pneumocystis in rat populations with the disease. Therefore, in our hands, histopathologic monitoring of rat colonies for IIP has accurately predicted their Pneumocystis status. Moreover, our retrospective data for diagnostic submissions show a highly significant correlation between IIP and Pneumocystis in lung sections.
To determine the species of Pneumocystis connected with IIP in rats, we sequenced a region of the mitochondrial ribosomal large-subunit gene used by others to compare Pneumocystis species. 28,46 Pneumocystis from 11 distinct sources of IIP had identical sequences that matched the P. carinii sequences in GenBank. This outcome is not unexpected since P. carinii is much more common than the only other Pneumocystis species identified in laboratory rats, P. wakefieldiae. 10,34 Likewise, P. carinii was the only Pneumocystis species linked to IIP by Livingston et al. 26
As part of transmission experiment IV, RNU rats were inoculated oronasally with P. carinii–infected lung homogenate. Although we would have preferred to inoculate rats with a pure culture of P. carinii to strictly fulfill Koch’s postulates, this was not feasible because Pneumocystis have not been propagated continuously in culture. The P. carinii inoculum used by Livingston and coworkers was prepared from the lungs of an immunosuppressed Lewis rat. 26 By showing that rats given a 0.2-micron filtrate of the inoculum did not develop IIP, they conclusively demonstrated that rats must be infected with Pneumocystisto develop the disease. However, as the sources of P. carinii for their experiments and ours were infected rat lungs and not pure cultures, we cannot claim categorically that no other agent is involved the pathogenesis of IIP. For instance, a virus transmitted with P. carinii could contribute to the disease by facilitating Pneumocystis colonization of the lower respiratory tract in immunocompetent hosts. On the other hand, the participation of an infectious agent other than P. carinii in the pathogenesis of IIP is highly improbable for several reasons. First, routine surveillance of the IIP-positive colonies used in transmission experiments did not detect any known pathogen other than Pneumocystis. Furthermore, no evidence was found for a novel virus in specimens from diseased rats by the generic immunological and genetic methods discussed earlier. Finally, novel pathogens are infrequently found in long-used and intensively characterized laboratory animal species such as the rat.
On the basis of the strength of the evidence that the histopathologic diagnosis of IIP in immunocompetent rats is Pneumocystis pneumonia, we have replaced surveillance by pulmonary histopathology with Pneumocystis PCR and serologic assays better suited to routine, high-throughput screening. Since this switch, we have developed a Pneumocystis recombinant protein antigen multiplexed fluorometric immunoassay (MFIA) to replace IFA for screening (data not shown). Hence, we have achieved the key aim of this research to expand and improve the methodologies for diagnosing this prevalent disease of laboratory rats.
Footnotes
Acknowledgements
The authors would like to further acknowledge Brian Francis, Geomaris Maldonado, Bonnie Hayes, Rajeev Dhawan, Danielle Cleasby, and Michelle Wunderlich.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: All authors are or were employees of Charles River at the time the work was performed.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: All work was funded by Charles River.
