Abstract
Several outbreaks of necrotic enteritis-like disease in lorikeets, from which Clostridium perfringens was consistently isolated, are described. All lorikeets had acute, segmental, or multifocal fibrinonecrotizing inflammatory lesions in the small and/or the large intestine, with intralesional gram-positive rods. The gene encoding C. perfringens alpha toxin was detected by PCR (polymerase chain reaction) on formalin-fixed, paraffin-embedded (FFPE) tissues in 20 out of 24 affected lorikeets (83%), but it was not amplified from samples of any of 10 control lorikeets (P < .0001). The second most prevalent C. perfringens toxin gene detected was the beta toxin gene, which was found in FFPE from 7 out of 24 affected lorikeets (29%). The other toxin genes were detected inconsistently and in a relatively low number of samples. These cases seem to be associated with C. perfringens, although the specific type involved could not be determined.
Clostridium perfringens is an important cause of enteric diseases in animals. The C. perfringens species is currently divided into 7 types (A to G) on the basis of the presence of 6 major toxin genes, that is, alpha (cpa), beta (cpb), epsilon (etx), iota (itx), enterotoxin (cpe), and necrotic enteritis B-like toxin (netb). 14 Type G strains, encoding alpha- and NetB toxins, commonly cause necrotic enteritis (NE) in chickens and other poultry species. 7 Experimental and epidemiological evidence indicates that NetB toxin is essential for development of NE. 7,14 In addition, some authors suggest that C. perfringens type A (that encodes alpha toxin [CPA]) and C (that encodes CPA and beta [CPB] toxins) strains may also produce NE in poultry. 11 NE is a multifactorial disease, and coccidiosis, stress, energy, and protein-rich diets are known predisposing factors in chickens. 16 Our knowledge about C. perfringens–associated enteritis in exotic birds is limited. 2,3,5,6,10,12 It has previously been suggested that this microorganism may cause a NE-like disease in both free-ranging and captive lorikeets. 6,10
Between 2000 and 2018, 24 of the 67 (36%) lorikeets necropsied at the Institute of Animal Pathology of the University of Bern were diagnosed with NE-like disease, which represented the most frequent diagnosis. A consistent finding was the isolation of C. perfringens from the intestine of affected birds. We here describe the pathologic features of the disease and the results of PCR (polymerase chain reaction) analyses for the detection of C. perfringens major toxin genes on DNA extracted from formalin-fixed, paraffin-embedded (FFPE) tissue of lorikeets.
The 24 coconut lorikeets (Trichoglossus haematodus) diagnosed with NE-like disease belonged to a zoological institution (A, n = 21) or a private aviary (B, n = 3), and had died spontaneously as part of 5 different outbreaks. Overall mortality in these outbreaks ranged from 29% to 60%. None of the outbreaks were associated with introduction of new birds, or other identifiable causes of stress. The clinical histories included either sudden death or nonspecific clinical signs such as apathy and separation from the flock shortly before death. Diseased lorikeets included juveniles (<2 year old, n = 5), adults (≥2 year old, n = 13), and birds for which the age was not recorded (n = 6); the age range was 7 months to 9 years. There were equal numbers of males and females. A full postmortem examination was performed, and samples of small and large intestine, lung, heart, and kidney in all cases, and of brain, pancreas, liver, spleen, and skeletal muscle in most cases, were fixed by immersion in 10% buffered formalin, pH 7.2, for 24 to 72 hours. Tissues were routinely processed for histology and stained with hematoxylin and eosin (H&E); additional intestinal sections were also stained with Gram. The necropsy reports were reviewed.
Gross lesions were observed in 14 of the 24 lorikeets (58%) and were limited to the intestine. They consisted of segmental or multifocal, fairly well-demarcated transmural hyperemia and/or hemorrhage, focally or multifocally ulcerated mucosa, and multifocal to diffuse fibrinonecrotic membranes (Fig. 1). Of the lorikeets that had gross abnormalities, 6 lorikeets had lesions exclusively in the small intestine (43%), 3 lorikeets (21%) had lesions only in the large intestine, and 5 lorikeets (36%) had lesions in both the small and the large intestine. This is in contrast with previous reports in lorikeets and other exotic birds with NE-like disease, in which lesions were restricted to the small intestine. 6,10,15
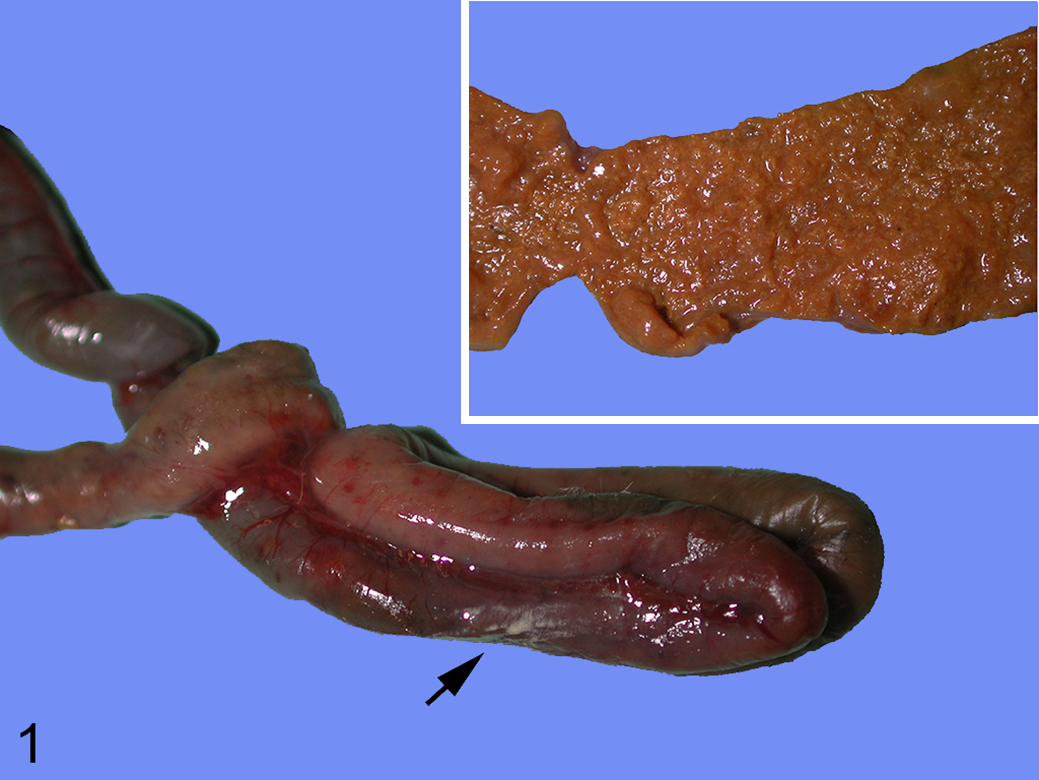
Necrotizing enteritis, small intestine, lorikeet. There is multifocal reddening of the intestinal wall, which corresponds to necrotizing enteritis visible from the serosal surface. The intestinal serosa has multifocal white areas, which correspond to areas of transmural inflammation and necrosis (peritonitis, arrow). Inset: The mucosa is diffusely necrotic.
Despite gross lesions being reported in only 14 cases, all 24 lorikeets had histologically moderate or severe fibrinonecrotizing lesions in the intestine (Fig. 2). The lesions were transmural in 13 (54%) cases, causing secondary peritonitis. The superficial epithelium and the lamina propria had extensive areas of necrosis. A fibrinonecrotic membrane composed of fibrin, viable and degenerate leukocytes, blood, and cellular debris covered the necrotic mucosa. Leukocyte infiltration of the mucosa and submucosa, mostly heterophilic with fewer lymphocytes, plasma cells, and macrophages, was a prominent feature in 13 (54%) and mild in 11 (46%) birds. The inflammatory cells formed a band between the viable and the non-viable tissue. Myriad non-sporulated, gram-positive rods with morphology compatible with Clostridium spp. were observed within the fibrinonecrotic membrane, lamina propria, and submucosa in all lorikeets (Fig. 3). Fibrin thrombi were present in small arterioles and venules of the mucosa and/or submucosa in 88% of the cases. In addition, a few discrete foci of hepatocellular necrosis were randomly scattered throughout the hepatic parenchyma in 2 lorikeets (8%). No other significant microscopic lesions were observed in any bird. Overall, these lesions resembled acute C. perfringens–induced NE in poultry.
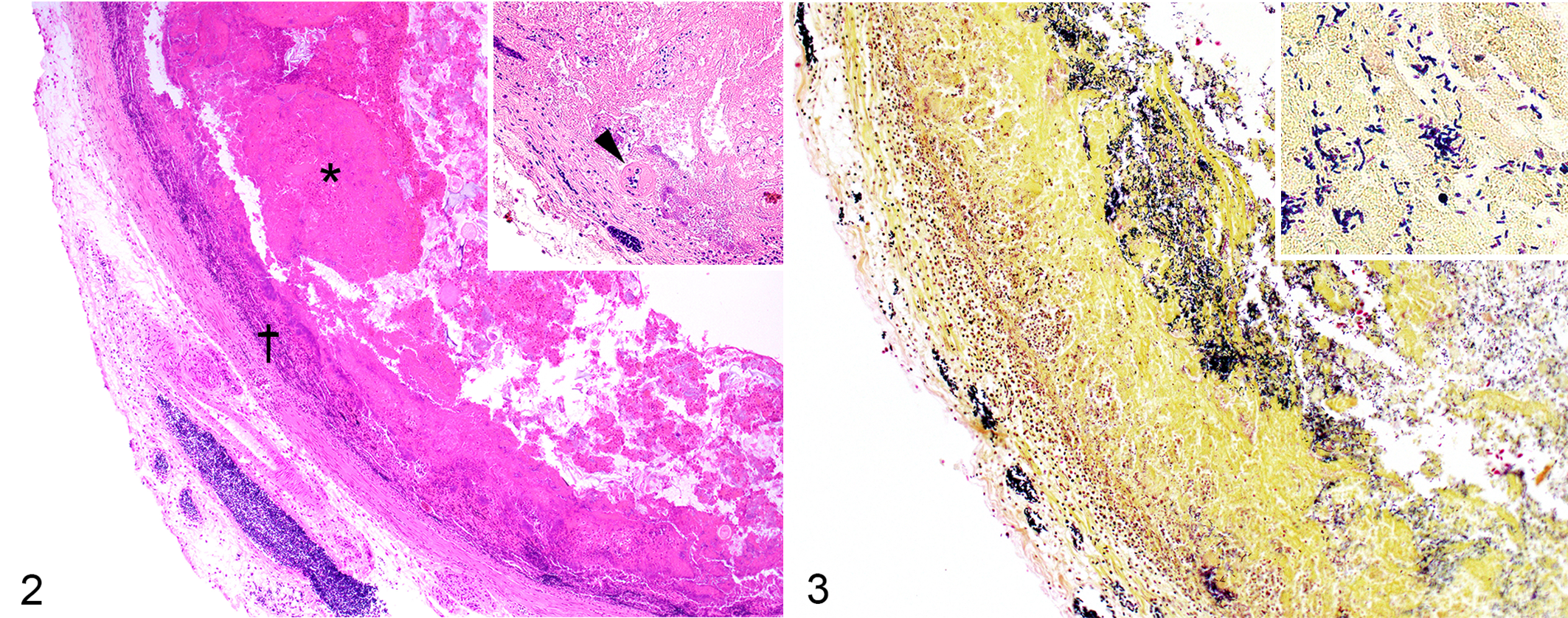
Transmural fibrinonecrotizing enteritis, small intestine, lorikeet. The mucosa is diffusely necrotic and covered by a fibrinonecrotic membrane (asterisk). There is loss of the mucosal/submucosal boundary and transmural infiltration with inflammatory cells (dagger). Inset: The mucosa contains a fibrin thrombus within a small vessel (arrowhead).
Bacteriological investigations were initiated soon after the necropsies for 16 of the 24 lorikeets (Table 1). Intestinal contents were incubated anaerobically for 24 hours at 37 °C on membrane C. perfringens agar plates (mCP; Oxoid). Yellow, circular, opaque colonies typical for C. perfringens were obtained in 12 cases (75%). In 7 of these cases, several of these colonies were pooled for DNA extraction and PCR detection of C. perfringens toxin genes as previously described. 1 These genes included cpa, cpb, etx, iap, cpe, and cpb2 (beta2 toxin). All samples were positive for cpa. cpb2 was detected in samples of 2 lorikeets (Table 1). PCR was negative for the other toxin genes in all samples tested.
Results of Clostridium perfringens Culture and PCR Typing in 24 Lorikeets With Necrotic Enteritis-Like Disease.
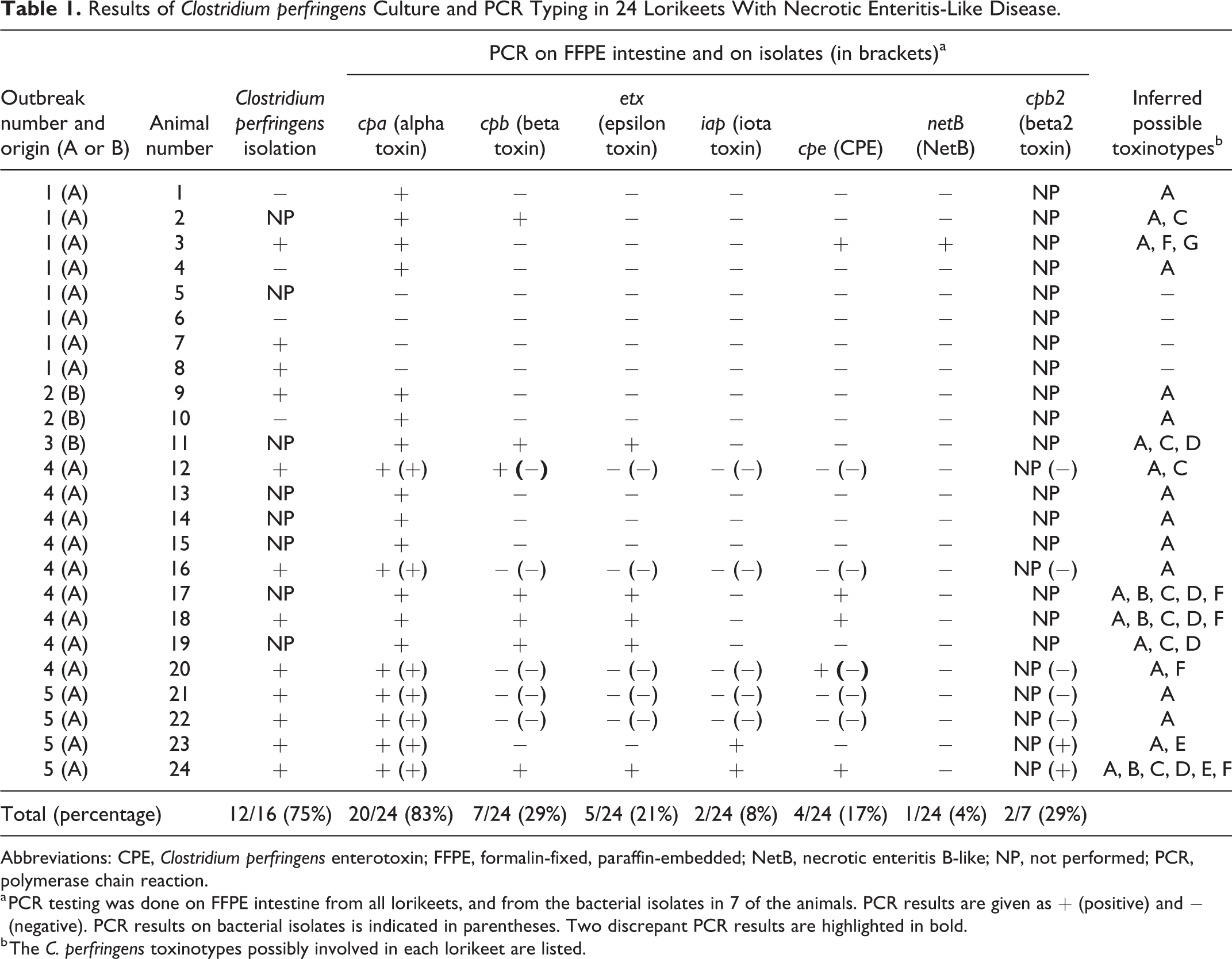
Abbreviations: CPE, Clostridium perfringens enterotoxin; FFPE, formalin-fixed, paraffin-embedded; NetB, necrotic enteritis B-like; NP, not performed; PCR, polymerase chain reaction.
a PCR testing was done on FFPE intestine from all lorikeets, and from the bacterial isolates in 7 of the animals. PCR results are given as + (positive) and − (negative). PCR results on bacterial isolates is indicated in parentheses. Two discrepant PCR results are highlighted in bold.
b The C. perfringens toxinotypes possibly involved in each lorikeet are listed.
Causes of necrotizing enteritis in lorikeets include bacteria such as Salmonella spp. 20 and C. colinum, 13 and parasites such as coccidia. 17 Intestinal content from 16 animals was inoculated into enrichment in Muller-Kauffmann Tetrathionate-Novobiocin Broth (Oxoid, Ref: BO1224K) followed by subculture on Brilliance Salmonella (Oxoid, Ref: PO5098A) and Brilliant Green Agar (Modified) (Oxoid, Ref: PO5033A). No Salmonella spp. were isolated in any of these 16 cases. No parasites were detected by using a combined sedimentation-flotation method with ZnCl2 on intestinal contents in 3 lorikeets. No coccidia were detected on histological sections of any of the birds. Because no specific medium for C. colinum was used, a co-infection by this microorganism cannot be ruled out. 13
Because the above-mentioned C. perfringens toxinotyping PCR protocol was only performed on isolates from a subset of the lorikeets and did not include netb, we retrospectively evaluated the presence of this toxin gene and the other typing toxin genes 14 on DNA extracted from FFPE intestinal samples of all lorikeets affected by NE-like disease and from 10 control lorikeets without necrotizing intestinal lesions. Total DNA was extracted from three 10-μm-thick paraffin sections using a QIAmp DNA FFPE Tissue Kit (Qiagen). Primers that were specific for short fragments of the main C. perfringens toxin genes were designed (Supplemental Table S1). DNA extracted from FFPE intestinal sections, from which the corresponding C. perfringens type had been isolated, was used as positive control. PCR amplicons were visualized in ethidium bromide–stained 1% agarose gels (Agarose SFP; Amresco). Because all strains of C. perfringens produce CPA, the amplification of cpa was considered indicative of the presence of C. perfringens DNA in the sample. Cpa was detected in 20 of the 24 lorikeets affected by necrotizing intestinal lesions (83%; Table 1), but in none of the 10 control lorikeets (P = .0001, χ2 with Yates’ correction; GraphPad Prism). This is in agreement with previous data indicating that C. perfringens is uncommonly found in healthy lorikeets or other psittacines. 15 C. perfringens was, however, isolated from the intestine of 2 lorikeets negative for cpa in the FFPE samples. Thus, altogether, C. perfringens was detected in the intestine of 22/24 lorikeets (92%) affected by necrotizing intestinal lesions. In 10 (42%) of the lorikeets with NE-like disease, cpa was the only toxin gene detected. The second most prevalent C. perfringens toxin gene detected was cpb (29%), while the other toxin genes were detected inconsistently and in a relatively low number of samples (Table 1).
The role of CPA in intestinal diseases of animals has been suggested but never definitively proven, 4,19 and there is no evidence to conclude from our results that CPA was responsible for the lesions observed in our birds. We cannot, however, completely rule out a role for this toxin in the pathogenesis of the NE-like disease. CPB is responsible for necrotizing enteritis in several animal species including birds, 11,16 and the lesions described in these lorikeets were very similar to those described in mammals and birds affected by this toxin. 11,18 It is therefore possible that CPB was responsible for the necrotizing intestinal lesions observed in at least some of these lorikeets. This is in agreement with a previous report that identified CPB in the intestine of lorikeets with necrotizing enteritis. 10 While NetB-producing type G strains have been shown to play a major role in NE in chickens and other poultry species, 7 netB was detected in only one lorikeet in this study.
In summary, our results suggest that, in lorikeets, a disease similar to the NE from chickens is associated with the presence of C. perfringens in the intestine of the lorikeets. C. perfringens type A was identified in 83% of lorikeets with NE-like disease. We could not, however, conclusively demonstrate a role of a C. perfringens toxin in the pathogenesis of this disease. It is possible that other yet unknown toxins contributed to the necrotizing intestinal lesions in these lorikeets. 18 Similarly, CPA was considered the key virulence factor for NE in broiler chickens for many years until recent evidence demonstrated that NetB, and not CPA, is the main virulence factor of NE-producing type G strains. 7 NetF-positive type A strains may be involved in canine hemorrhagic gastroenteritis and equine necrotizing enteritis, although definitive evidence of the role of NetF in these diseases is lacking. 9 Moreover, several previously unknown toxin genes were identified in isolates from turkeys, 8 indicating a much more diverse picture of pathogenic C. perfringens type A isolates. However, most studies (including ours) lack consistent isolation and full characterization of C. perfringens isolates from the intestine of diseased animals. Fulfillment of Koch’s postulates is also lacking. Therefore, conclusions on a causal relationship of particular pathogenic strains of C. perfringens type A with NE-like disease in lorikeets or other exotic birds cannot be drawn. To investigate the causal relationships of different C. perfringens with NE-like disease in animals, whole-genome sequencing of C. perfringens isolates should be considered, along with experimental work to fulfill Koch’s postulates.
Supplemental Material
Supplemental Material, Supplemental_materials-Grau-Roma_et_al - Clostridium perfringens–Associated Necrotic Enteritis-Like Disease in Coconut Lorikeets (Trichoglossus haematodus)
Supplemental Material, Supplemental_materials-Grau-Roma_et_al for Clostridium perfringens–Associated Necrotic Enteritis-Like Disease in Coconut Lorikeets (Trichoglossus haematodus) by Llorenç Grau-Roma, Mauricio Navarro, Sohvi Blatter, Christian Wenker, Sonja Kittl, Francisco A. Uzal and Horst Posthaus in Veterinary Pathology
Footnotes
Acknowledgements
We kindly thank Ms S. J. Uzal for critical review of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
