Abstract
Weaned pigs from a line bred for increased feed efficiency were enrolled in a study of the role of host genes in the response to infection with Porcine Reproductive and Respiratory Syndrome Virus (PRRSV). Four of the pigs were euthanatized early in the study due to weight loss with illness and poor body condition; 2 pigs before PRRSV infection and the other 2 pigs approximately 2 weeks after virus inoculation. The 2 inoculated pigs failed to produce PRRSV-specific antibodies. Gross findings included pneumonia, absence of a detectable thymus, and small secondary lymphoid tissues. Histologically, lymph nodes, spleen, tonsils, and Peyer’s patches were sparsely cellular with decreased to absent T and B lymphocytes.
One hundred pigs from each of 2 Yorkshire lines divergently selected for feed efficiency 1 were enrolled, at 2–4 weeks of age, in a study of the genetic influence on response to Porcine Reproductive and Respiratory Syndrome virus (PRRSV) infection. Pigs were arbitrarily distributed into pens of 16–18 pigs, with 1 week’s acclimation before inoculation with PRRSV isolate NVSL97-7985. 5
Four pigs from the increased feed efficiency line developed poor body condition and illness and were euthanatized prematurely. Pig No. 4, a gilt, weaned at 16 days of age, was infected with PRRSV at 38 days and euthanatized at 54 days. Pig No. 6, a barrow, weaned at 27 days of age, was infected with PRRSV at 36 days and euthanatized at 47 days. Pig Nos. 109 and 124 were barrows that had been weaned at 21 and 28 days of age, respectively, and were euthanatized at 34 and 41 days of age, respectively, before they were inoculated with PRRSV. The pigs were from 4 different litters, but pig Nos. 4, 6, and 124 had the same sire.
At necropsy, all of the 4 pigs had rough hair coat and were smaller than other pigs in the study. Thymus was not visible, lymph nodes were small, and ileal Peyerś patches were inconspicuous. Pig No. 4 had fibrinosuppurative bronchopneumonia, pig No. 6 had fibrinous synovitis in both tarsal joints, and pig No. 124 had dermatitis.
Lung, heart, kidneys, stomach, liver, lymph nodes, spleen, tonsils, nasal turbinates, and intestine were submitted to the Kansas State Veterinary Diagnostic Laboratory for histopathology. The lymph nodes (Fig. 1) and spleen (Fig. 2) from the 4 pigs had abnormal architecture with decreased numbers of lymphocytes and absence of lymphoid follicles. The tonsils and Peyer’s patches also had markedly reduced numbers of lymphocytes and absence of follicles. Pig Nos. 6 and 109 had interstitial pneumonia, and pig No. 109 had cytomegaloviral inclusions in epithelial cells of nasal submucosal glands and renal tubules.
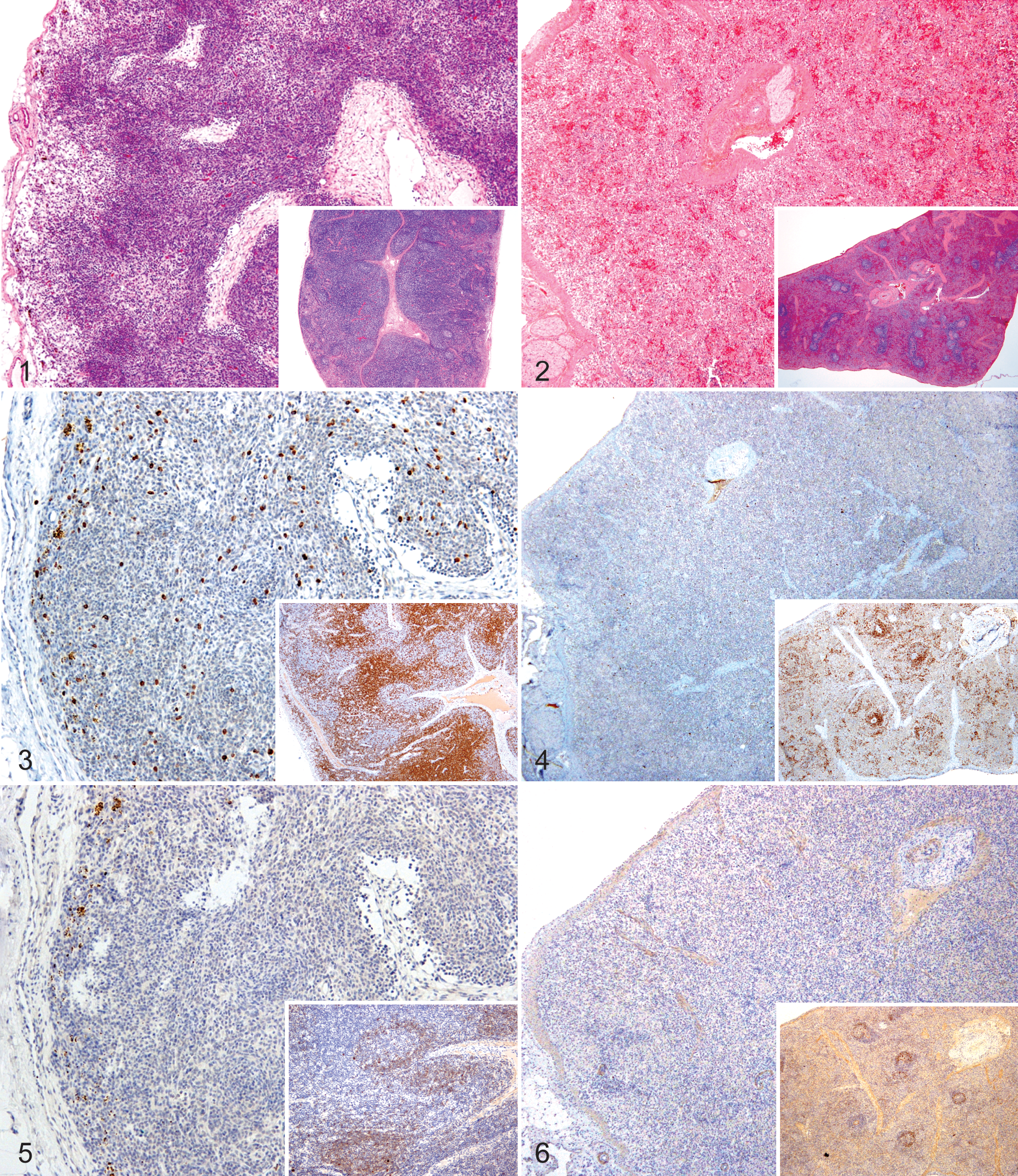
T and B lymphocytes were identified by immunohistochemistry (IHC) using rabbit anti-human CD3 polyclonal antibody or mouse anti-human CD79α monoclonal antibody, respectively, followed by biotinylated goat anti-rabbit or anti-mouse immunoglobulin with avidin-HRPO and DAB chromagen (Ventana Medical), and counterstained with hematoxylin. CD3+-positive lymphocytes were markedly decreased in number in lymph nodes (Fig. 3) and were nearly absent from the spleen (Fig. 4). CD79α+-positive lymphocytes were few in the lymph nodes (Fig. 5) and absent from the spleen (Fig. 6). Histiocytic cells comprised the predominant cell population in lymphoid tissues. Hemosiderin-laden macrophages were identified by positive reaction with Prussian blue staining in lymph node sections.
Viremia was quantified by standardized RT-PCR assays (Applied Biosystems, Foster City, California, or Tetracore, Rockville, Maryland). Results were reported as copies of RNA per PCR reaction volume. On day 0, all pigs were PRRSV-negative by RT-PCR. By day 4 post inoculation, all pigs were PRRSV-positive by RT-PCR, and they remained positive through day 11 post inoculation (Table 1). PRRSV-specific serum antibodies were detected using a standardized ELISA test (PRRS X3, IDEXX Laboratories, Westbrook, Maine). Results were reported as a sample/positive (S/P) ratio. On day 4 after inoculation, all pigs were seronegative by ELISA. On day 11, pig Nos. 4 and 6 remained negative, whereas all other infected pigs had an antibody response to PRRSV. These data suggest that the affected piglets were incapable of initiating an antibody response after PRRSV inoculation.
ELISA and RT-PCR Results After Infection With Porcine Reproductive Respiratory Syndrome Virus.
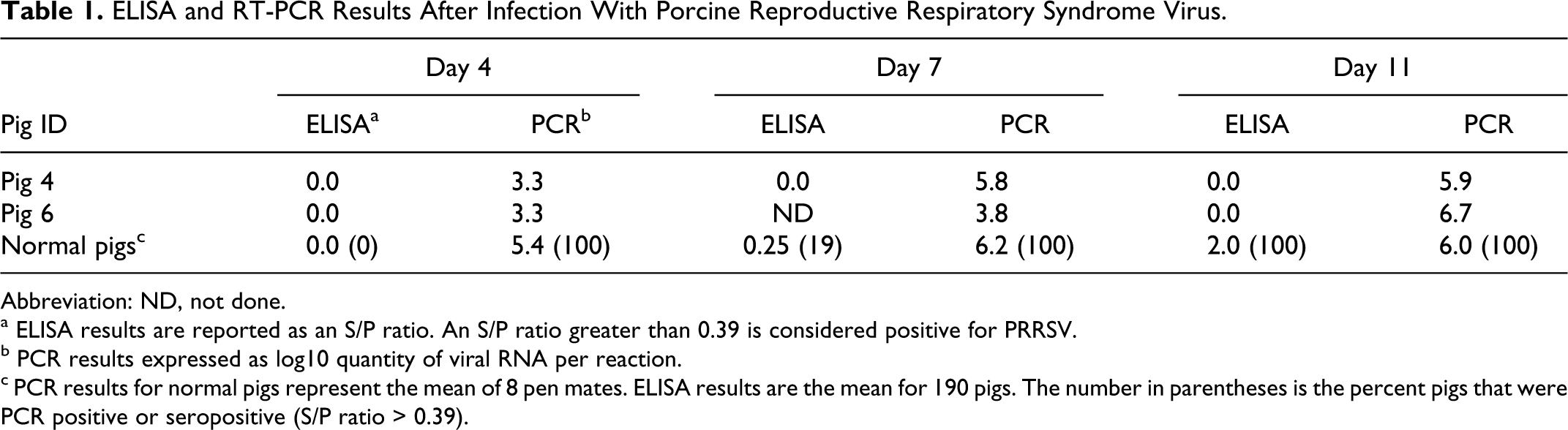
Abbreviation: ND, not done.
a ELISA results are reported as an S/P ratio. An S/P ratio greater than 0.39 is considered positive for PRRSV.
b PCR results expressed as log10 quantity of viral RNA per reaction.
c PCR results for normal pigs represent the mean of 8 pen mates. ELISA results are the mean for 190 pigs. The number in parentheses is the percent pigs that were PCR positive or seropositive (S/P ratio > 0.39).
Infection by porcine circovirus type 2 (PCV2) can result in lymphocyte depletion in lymphoid tissues. Co-infection with PCV2 and PRRSV can result in postweaning multisystemic wasting syndrome.3,4 Although all inoculated pigs were positive for PRRSV by RT-PCR, PCV2 antigen was not detected by IHC in lymph node or lung sections from the 4 affected piglets, indicating that they did not have PCV-associated disease. There were no clinical signs or morphological lesions to suggest infection by any other pathogens known to cause lymphoid depletion in pigs, indicating that lymphoid hypoplasia was not a result of infection. Subsequent matings of the parents of these pigs have produced approximately 22% of piglets per litter with similar histologic lesions, suggesting that the affected pigs had a primary immunodeficiency. 2
Although the PRRSV isolate used in this study normally results in seroconversion within 11 days post inoculation, the 2 inoculated pigs failed to mount a measurable antibody response. All 4 pigs had a near absence of T and B lymphocytes in undersized secondary lymphoid tissues. No pathogens known to cause secondary immunodeficiencies were detected, and subsequent matings of the parents of the affected pigs produced offspring with similar lesions. Collectively, these findings are consistent with an inherited primary immunodeficiency disorder.
Footnotes
Acknowledgement
The authors thank Dr. Amanda Beck (KSU) for in-house review of the manuscript for accuracy of the pathology descriptions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The feed efficiency lines of pigs were developed with funds from the Iowa Agriculture and Home Economics Experiment Station, Ames, Iowa (project no. 3600) and from the ISU Center for Integrated Animal Genomics, and supported by Hatch Act. The PRRSV infection trial was part of Agriculture and Food Research Initiative Competitive Grant no. 2011-68004-30336 from the USDA National Institute of Food and Agriculture.
