Abstract
Caseous lymphadenitis is an infectious and contagious disease caused by Corynebacterium pseudotuberculosis, with a worldwide distribution and high prevalence in small ruminant populations. This disease causes significant economic loss in small ruminants through reduced meat, wool, and milk production. C. pseudotuberculosis can also affect horses, domestic and wild large ruminants, swine, and man. It is considered an occupational zoonosis for humans. As part of in vitro investigations of the pathogenesis of C. pseudotuberculosis, this study analyzed its capacity to adhere to and invade the FLK-BLV-044 cell line, derived from ovine embryonic kidney cells. C. pseudotuberculosis showed a measurable capacity to adhere to and invade this cell line with no significant differences between the four strains assessed. The incubation of the cell line at 4ºC, pre-incubation with sugars, complete and heat inactivated antiserum, and heat-killed and ultraviolet-killed bacteria produced a significant (P < 0.05) decrease in the invasion efficiency or inability to invade the cell line. Plate counting and fluorescence studies showed intracellular bacteria for up to 6 days. Non-phagocytic cells may therefore act as a suitable environment for C. pseudotuberculosis survival and play a role in the spread of infection and/or maintenance of a carrier state.
Caseous lymphadenitis, also called pseudotuberculosis, is a prevalent disease with a worldwide distribution, causing lesions in sheep and goats. 6 This disease does not produce high mortality but causes important economic losses by reducing production of meat, wool, and milk.1,7 This pathogen has also been isolated from abscessed tissues in other species and is associated with conditions such as ulcerative lymphangitis and pigeon fever in horses, cattle, camels, buffaloes, and humans. 7 This disease shows three clinical forms—cutaneous, visceral, and a mixed form—but the cutaneous and visceral forms are the most frequent. The cutaneous form is characterized by abscess formation in superficial lymph nodes and subcutaneous tissues, while the visceral form is characterized by abscesses that can develop internally in spleen, kidneys, lungs, and liver.7,15
Little data exist on the pathogenesis of C. pseudotuberculosis in animal tissues, and the potential for this pathogen to adhere to and invade ovine cell lines has not been previously studied. The precise nature of the mechanism responsible for adherence and the interaction with animal cell receptors to facilitate bacterial entry into host cells may be of importance in understanding the pathogenicity of C. pseudotuberculosis in small ruminants. Recently an invasion-associated protein has been described in corynebacterias.19,27 The invasion and persistence in non-phagocytic cells could be used by C. pseudotuberculosis as a tactic to avoid hostile environments in the tissues and to escape phagocytosis. This would aid progression of infection, with a level of protection against host defenses, providing a suitable environment for its proliferation.3,9
We hypothesised that C. pseudotuberculosis may exploit non-phagocytic cells to avoid the host immune defenses and establish a favorable ecological environment for subsequent dispersion of infection to other tissues. Investigations of intracellular behavior using an in vitro model may be useful for investigation of interactions between caprine and ovine pathogens and their host and assist development of new therapeutics.
Materials and Methods
Bacterial Strains and Culture Conditions
Four C. pseudotuberculosis strains were used: IUSA-1, IUSA-2 and TARA-1 (clinical isolates from goats and sheep in the Canary Islands), and CECT-808-T (a reference strain from the Spanish Type Culture Collection). Strains were cultured on brain heart infusion broth (BHIB) (Pronadisa) or on blood agar base (Pronadisa) supplemented with 5% (v/v) sheep’s blood and incubated at 37ºC for 48 to 72 hours. In both media, 0.5% (v/v) Tween 80 was added to reduce cell aggregation. All strains were stored at –80°C in BHIB with 15% glycerol.
The strains were confirmed as C. pseudotuberculosis by multiplex polymerase chain reaction (PCR) based on primers targeting the 16SrRNA and pld genes.4,14,20 The PCR was performed at a final volume of 25 µL, containing 1.5 U Taq DNA polymerase (Invitrogen), 1X PCR buffer, 15 mM MgCl2, 2 mM dNTPs, 1 µM of each of the primers (16S-F/16S-R and PLD-F/PLDR2), and 10 ng DNA extracted from cultured C. pseudotuberculosis.
Ovine Cell Line
The fibroblast-like cell line FLK-BLV-044 (DSMZ catalog code: ACC 153) derived from ovine embryonic kidney cells was cultured using Dulbecco’s Modified Eagle’s Medium (DMEM) (Sigma) supplemented with 10% fetal bovine serum. The cells were incubated at 37°C in 5% CO2, and before reaching total confluence were subcultured after detaching them with trypsin-EDTA (Sigma) solution. The cells were plated at a density of 105 cells per well in 24-well tissue culture plates (Corning).
Invasion and Adherence Assays
The adherence and invasion assays were based on a gentamicin protection assay described previously.12,23 Strains were incubated for 48 hours in BHIB at 37ºC, and viable bacteria were determined by serial dilutions in sterile phosphate buffered saline (PBS) followed by plate counting on sheep blood agar (SBA). To determine the effect of bacterial concentration on invasion efficiency (percentage of initial inoculum internalized), the bacteria were added to each tissue-culture well to give a multiplicity of infection (MOI) of 5, 25, 50, 100, and 150. The mixture was centrifuged for 5 minutes at 33g to promote adherence of the bacteria to the host cells. Four hours post infection, the ovine cells were incubated for 2 hours with DMEM containing gentamicin (200 µg/mL), an antibiotic that cannot penetrate intact eukaryotic cells, so it will kill susceptible extracellular but not intracellular bacteria. Each well with the ovine cells was washed three times with PBS and lysed with 100 µL of 0.2% Triton X-100 (Sigma). An additional 400 µL of PBS was added per well, mixed and serially diluted in PBS, and internal bacteria determined by plate counting on SBA. The noninvasive E. coli DH5-α was used as a negative control.
In order to analyze C. pseudotuberculosis invasion in the FLK-BLV-044 cell line, the number of bacteria recovered from the cells over time was assessed at a MOI of 100. Extracellular bacteria were removed after 30, 60, 90, 120, 150, 180, 210, and 240 minutes post infection by three washes with PBS, and the ovine cells incubated for 2 hours with DMEM containing gentamicin (200 µg/mL) and then processed as described for the invasion assay.
To assess intracellular persistence of C. pseudotuberculosis in the FLK-BLV-044 cell line at MOI 100, at 4 hours post infection the bacteria were killed by gentamicin (2 hours, 200 µg/mL), and the culture medium was replaced by a medium containing 20 µg/mL of gentamicin, and incubated for a further 24, 48, 72, 96, 120, and 144 hours to give sufficient time for potential intracellular survival or replication.
In order to study the influence of antibodies on bacterial invasion, strains were pre-incubated at 1:1 (v/v) for 30 minutes at 37ºC with a complete and heat inactivated polyclonal antiserum against C. pseudotuberculosis (IUSA-1) produced by using previously described protocol. 21 After pre incubation, the mixture at MOI 100 was used to infect the ovine cells as described previously for the invasion assay.
The effect of the cell metabolism on bacterial invasion was examined by performing C. pseudotuberculosis infections at 4ºC. 13 In order to investigate bacterial ligand blocking, the strains were grown for 48 hours in BHIB with mannose and glucose (1%). After incubation, bacterial inoculum was used to infect the cells as described previously at MOI 100, which were then processed in the standard assay. In all experiments, the invasion efficiency was calculated as the average of the total number of colony forming units recovered compared to the initial inoculum.
In the adherence assay, bacteria were used to infect the ovine cells at a MOI of 100, and at 4 hours post infection, external nonadhered bacteria were removed by washing the wells three times with PBS and ovine cells lysed by adding Triton X-100. Then serial dilutions of the disrupted mixture were plated on SBA and incubated for 48 hours at 37ºC to obtain the total bacteria (adherent and invasive bacteria). Bacterial adhesion efficiency was calculated as the difference between the total and invasive bacteria determined by plate counting.
Fluorescence Microscopy of C. pseudotuberculosis Interactions With the FLK-BLV-044 Cell Line
For immunofluorescence studies, FLK-BLV-044 ovine cells were seeded on 12 mm diameter coverslips in 24 well plates at a density of 105 cells per well and then infected with C. pseudotuberculosis (IUSA-1) at MOI 100 for 4 hours, after which the ovine cells were washed three times with PBS and fixed in cold paraformaldehyde (4% in PBS) for 20 minutes at room temperature. Bacteria were detected using polyclonal rabbit antibodies against C. pseudotuberculosis strain IUSA-1 and developed with two commercial conjugates containing fluorescent dyes. The antibody reagents were all diluted 1:500 in PBS containing 1% bovine serum albumin (Sigma) (PBS-BSA) and applied for 20 minutes at room temperature. In the first stage, after the coverslips were carefully washed twice in PBS, extracellular bacteria were identified by incubating with anti–C. pseudotuberculosis antibody. This was followed by careful washing of the coverslips in PBS and development of bound antibodies by incubation with anti-rabbit IgG antibody conjugated with Alexa Fluor 594 dye (Invitrogen). Following this initial fluorescent dye staining, the ovine cells were permeabilized with detergent, to enable staining of both intracellular and extracellular bacteria with the second fluorescent dye. The ovine cells were thus subjected to PBS containing 0.1% Triton X-100 for 4 minutes at room temperature and carefully washed four times with PBS. The coverslips were then incubated with polyclonal anti–C. pseudotuberculosis antiserum as described previously, and bound antibodies developed with anti-rabbit IgG antibody conjugated with Alexa Fluor 488 dye (Invitrogen). After washing, the coverslips were mounted on glass slides with Prolong Gold antifade reagent with DAPI (Invitrogen). All preparations were examined by epifluorescence microscopy with a ×40 objective. Digital images were acquired using a Zeiss AxioCam HRc camera and merged using Photoshop CS3 (Adobe) software.
In order to investigate the effect of bacterial inactivation by high temperature or ultraviolet light on the ability of C. pseudotuberculosis to invade ovine tissue culture cells, a 400 µL volume of a BHIB culture of strain IUSA-1 was heated at 80ºC for 20 minutes in a thermoblock or exposed to UV light for 18 hours. After both treatments, subculture to SBA confirmed the nonviability of the bacteria in each assay. Then heat-killed and UV light-killed C. pseudotuberculosis at MOI 100 were used to infect the ovine cells and processed for immunofluorescence as described before.
Statistical Analysis
Statistical analyses were carried out using SPSS version 17.0. Data were analyzed by one-way ANOVA and Student’s t-test, considering P < 0.05 as significant. The experiments were performed in triplicate, and numerical data and bars are shown as mean values with standard deviations.
Results
The number of intracellular bacteria detected increased with increasing initial inoculum to MOI 100, with significantly (P < 0.05) higher invasion efficiency above an MOI of 25 (Fig. 1). The degree of invasion and adherence in the FLK-BLV-044 cell line are shown on Fig. 2. Maximal and minimal adherence efficiencies were observed for strains IUSA-1 (2.01%) and TARA-1 (1.5%) (Fig. 2a), but no statistically significant differences were found between strains. Maximum invasion efficiency was observed for strain IUSA-1 (0.17%), whereas strain IUSA-2 showed the minimal invasion efficiency of 0.10% (Fig. 2b), but no statistically significant differences were observed between strains. The noninvasive control E. coli DH5-α was tested over a similar range of MOI and the invasion efficiency was 0.005%. In addition, we were unable to obtain intracellular E. coli from experiments carried out at 4ºC (data not shown). The invasion process was time dependent, increasing over 240 minutes (Fig. 3) with intracellular bacteria detected after only 30 minutes. In intracellular survival experiments, all strains were able to grow up to 24 hours post infection and then survive up to 6 days (Fig. 4). After this period, the tissue culture cells began to show signs of degradation, and the cellular monolayer detached.
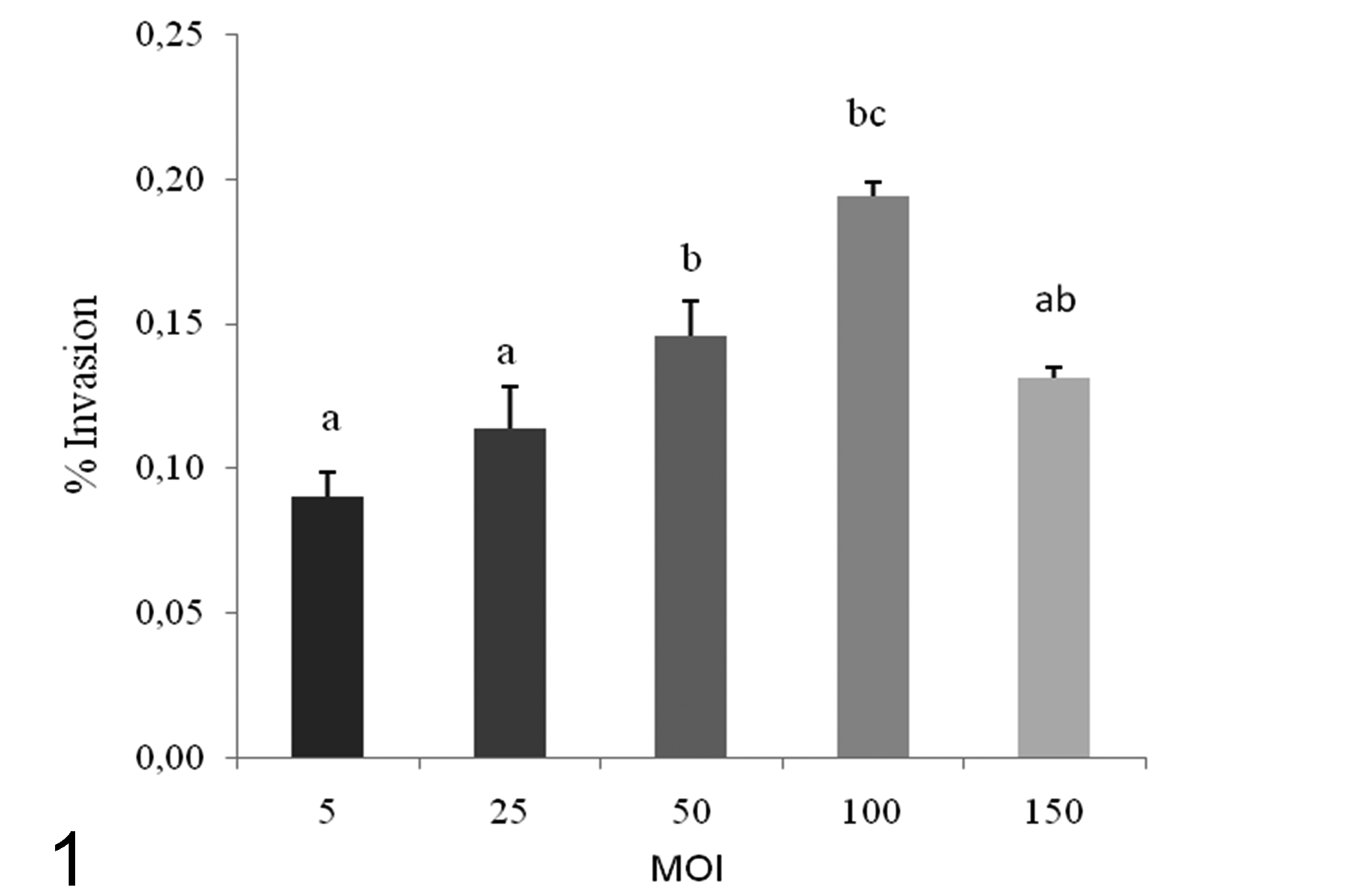
Effect of bacterial concentration using different multiplicity of infection (MOI) on invasion of C. pseudotuberculosis (IUSA-1) for cell line FLK-BLV-044. Results from three independent experiments are given as mean ± SD (standard deviation) intracellular bacteria expressed as a percentage of the original inoculum. In bars, different letters (a, b, c) indicate statistically significant differences (P < 0.05) between treatments.
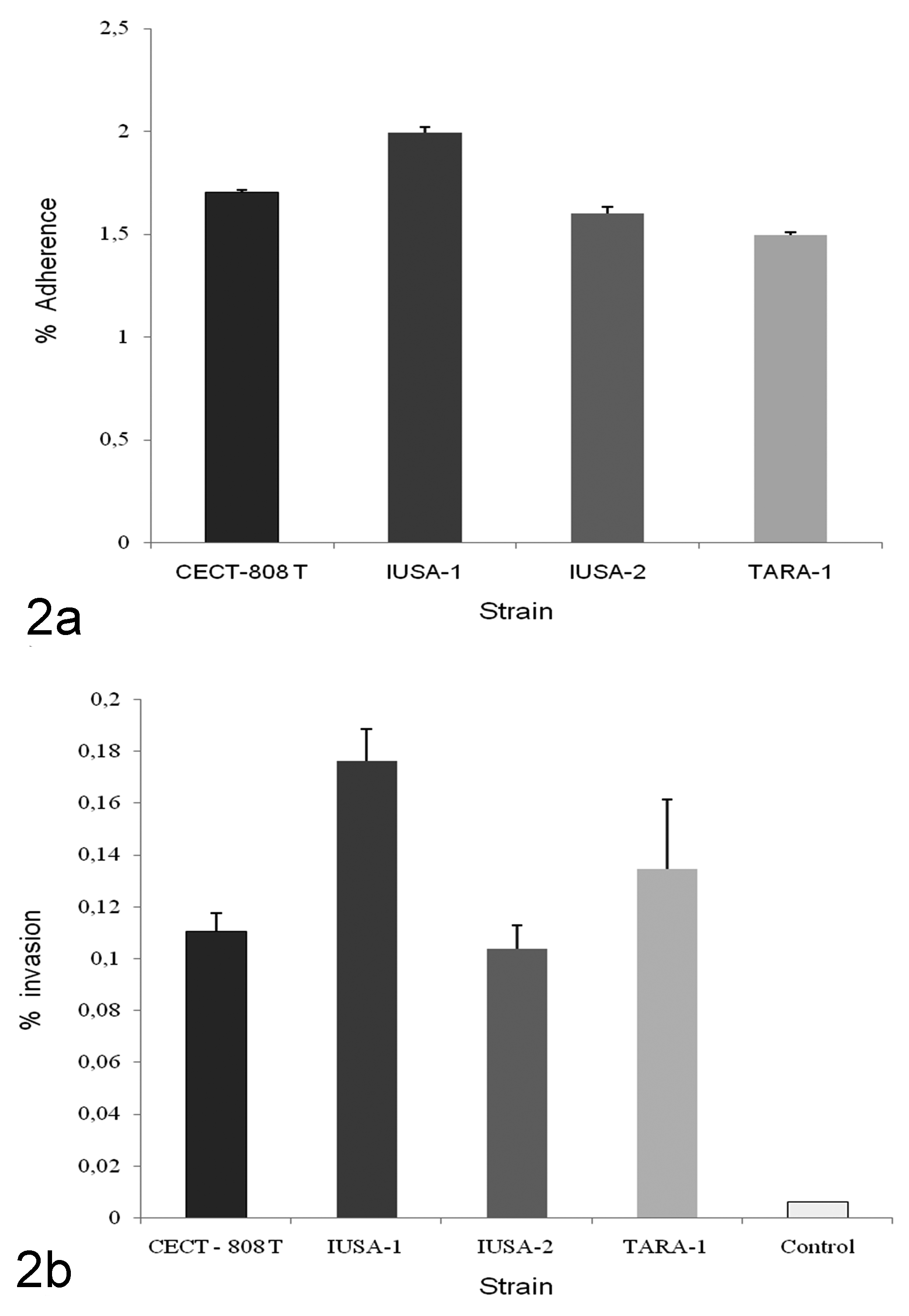
Adherence (a) and invasion (b) of C. pseudotuberculosis IUSA-1, IUSA-2, TARA-1, and CECT-808 T for cell line FLK-BLV-044 at a multiplicity of infection of 100 in a gentamycin protection assay for 4 hours. E. coli DH5-α was used as negative invasion control. All assays were performed on three separate occasions in triplicate wells and results are given as mean ± SD expressed as a percentage of the original inoculum.
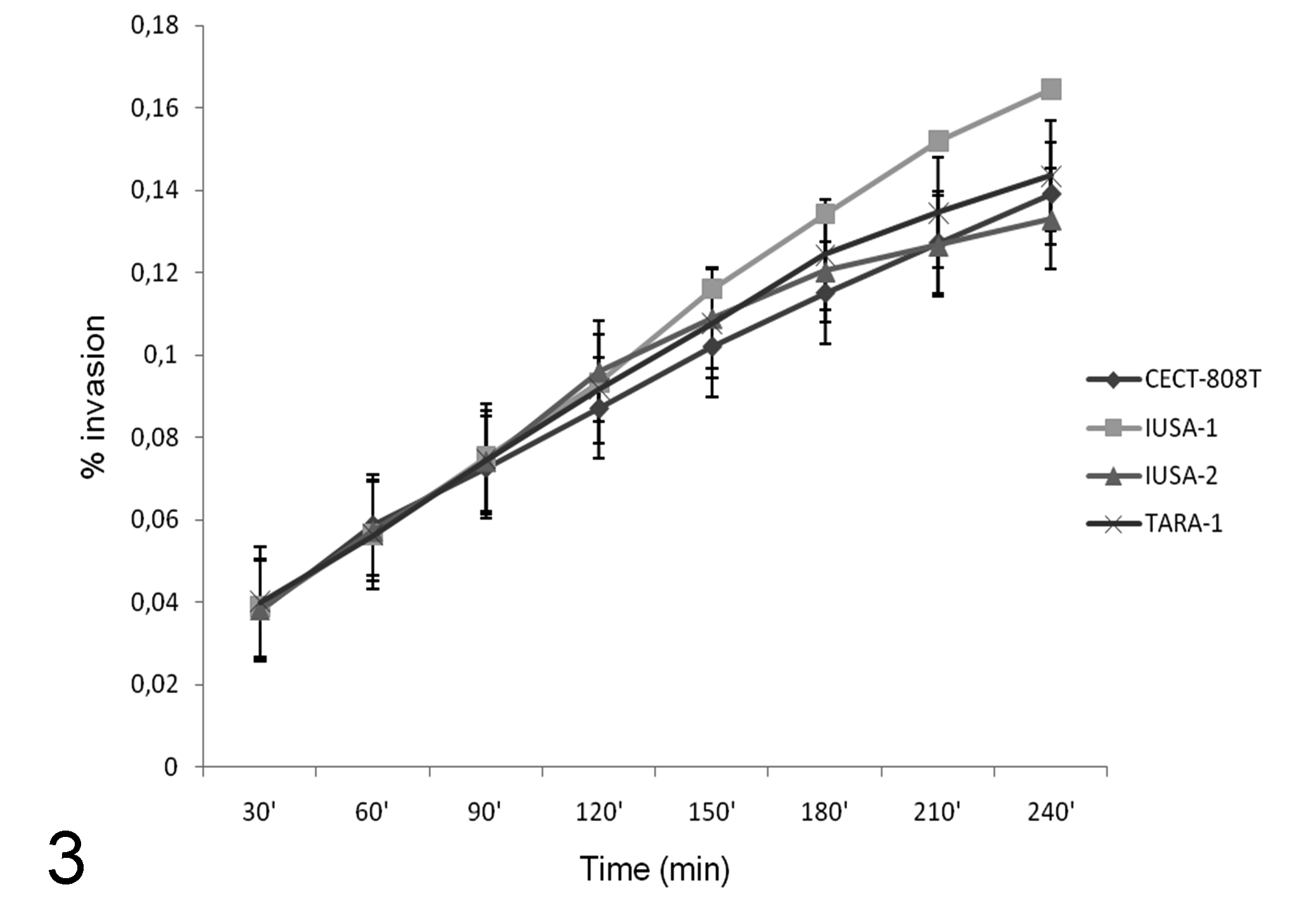
Invasion of C. pseudotuberculosis strains in the cell line FLK-BLV-044 at a multiplicity of infection of 100. Results of gentamycin protection assays in three independent experiments per strain using different periods of infection are given as mean ± SD intracellular bacteria expressed as a percentage of the original inoculum.
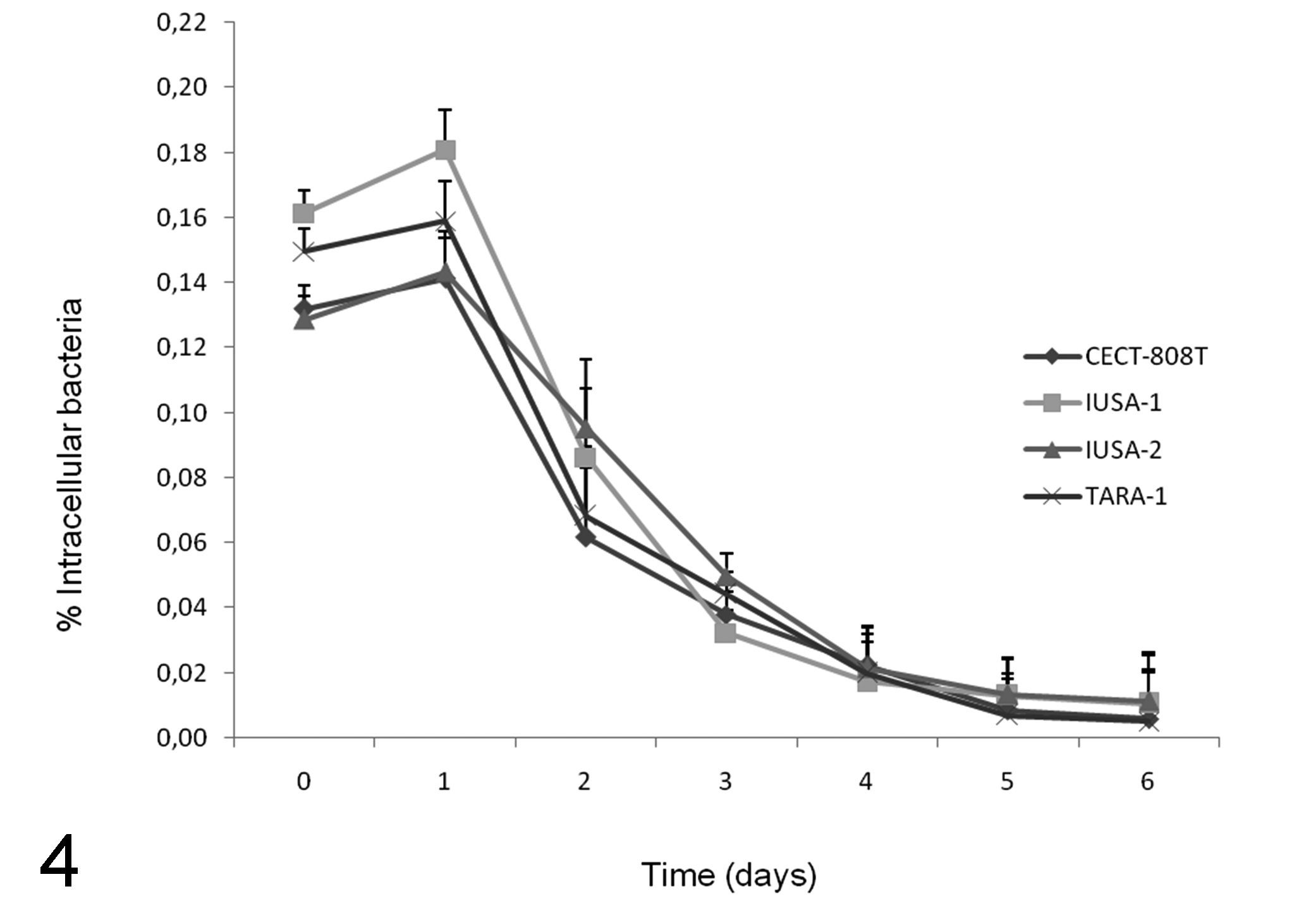
Intracellular survival of C. pseudotuberculosis strains in the cell line FLK-BLV-044 over time at a multiplicity of infection of 100. Cells were infected for 4 hours, treated with gentamicin and incubated for up to 6 days. Each invasion assay was performed on three separate occasions, and results expressed as mean ± SD intracellular bacteria expressed as a percentage of the original inoculum.
Incubation of the cell line at 4ºC, pre-incubation in the presence of glucose-mannose (1%), and pre-incubation with a complete and heat inactivated antiserum produced a significant (P < 0.05) decrease in the invasion efficiency by C. pseudotuberculosis compared to the control assay (Fig. 5).
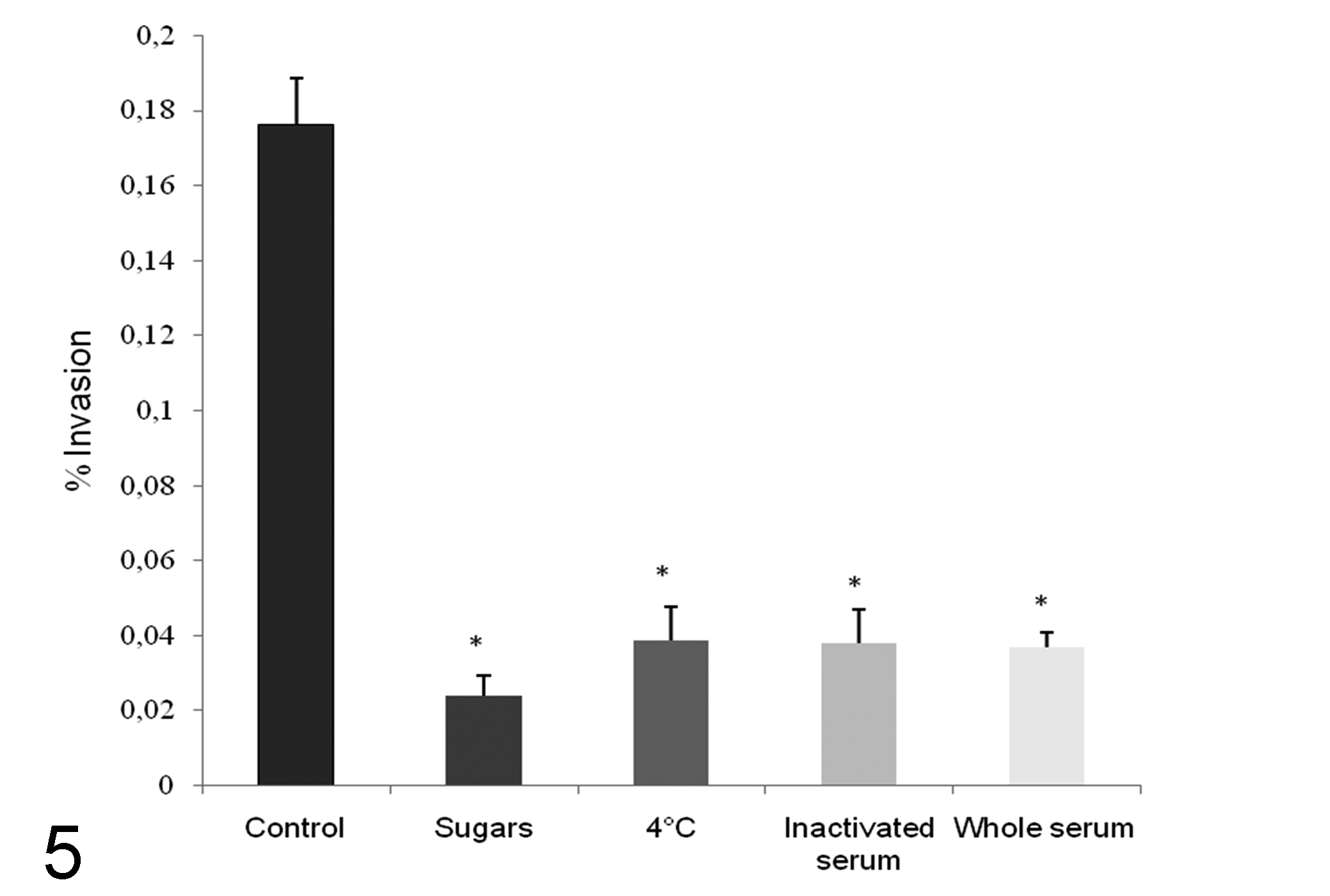
Effect of pre-incubation with sugars, reduced cell metabolism, and anti–C. pseudotuberculosis antisera on the invasion of FLK-BLV-044 cells by C. pseudotuberculosis strain IUSA-1 at a multiplicity of infection of 100. Bacteria in brain heart infusion broth were incubated in the presence of 1% mannose and glucose, heat inactivated or whole antiserum for 30 minutes at 37ºC before infection, or added to the monolayers previously incubated at 4ºC. Results of the mean ± SD intracellular bacteria expressed as a percentage of the original inoculum from three independent experiments per treatment are shown. Asterisks denote significant differences (P < 0.05) between treated and nontreated control organisms.
Immunofluorescence microscopy confirmed the intracellular location of C. pseudotuberculosis (Fig. 6). The immunofluorescence also demonstrated that heat-killed and ultraviolet-killed bacteria were unable to invade the cell line (data not shown).
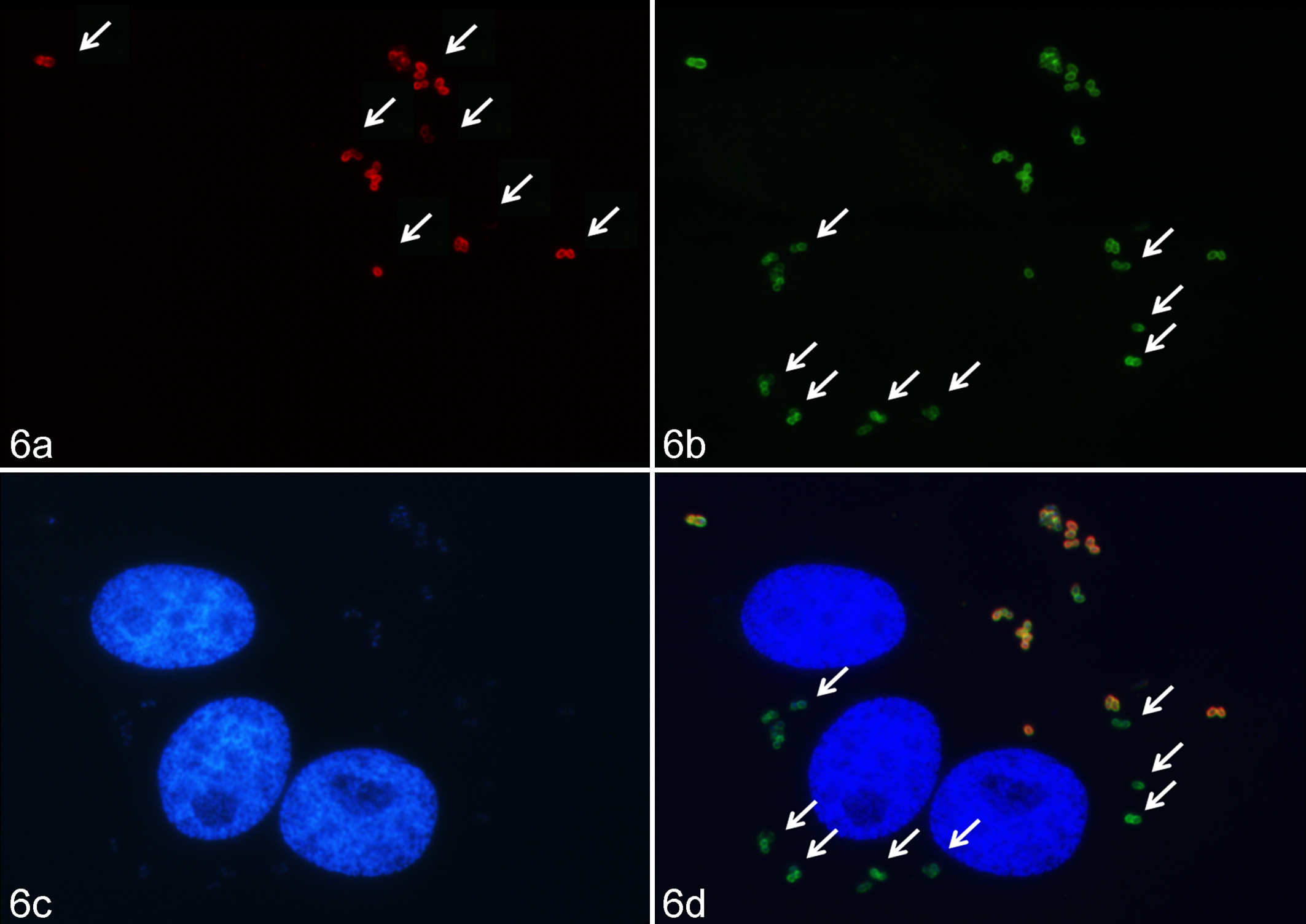
Adherence and invasion of C. pseudotuberculosis IUSA-1 at a multiplicity of infection of 100 for 4 hours in the cell line FLK-BLV-044 under fluorescence microscopy. (a) Extracellular bacteria were detected with anti–C. pseudotuberculosis sera and Alexa Fluor 594 conjugate on non-permeabilised cells (arrows). (b) Total bacteria were detected on permeabilized cells after bound anti–C. pseudotuberculosis sera was developed with Alexa Fluor 488 conjugate (arrows). (c) The DNA of tissue culture cell nuclei was stained in blue with DAPI. (d) In the merged images, extracellular bacteria are shown in orange or yellow (mixture of red and green), intracellular bacteria in green, and DNA of tissue culture in blue. Arrows indicate some intracellular bacteria.
Discussion
Studies of adherence and cell invasion are currently assisting the understanding of different aspects of the pathogenesis of infectious diseases. This study is the first to investigate the kinetics of adherence, invasion, and intracellular survival of C. pseudotuberculosis in non-phagocytic cells. The capacity of some bacteria to invade non-phagocytic cells is considered an important virulence factor of several animal and human bacterial pathogens, including Yersinia, 11 Salmonella, 5 and Shigella.22,25
Our results from fluorescence microscopy and gentamicin protection assay demonstrated C. pseudotuberculosis adherence to and invasion of FLK-BLV-044 cells, with similar efficiencies among the strains tested and a possible correlation between the efficiency of adherence and invasion. The maximum invasion efficiency observed in the present study is lower than showed by Brucella 24 and Salmonella 18 but similar or higher than those reported in Campylobacter,2,10 Burkholderia,17,26 Hafnia alvei, 21 and Prevotella. 8 In the time-course analysis, at 30 minutes post infection a few internal bacteria were detected in FLK-BLV-044 cells, but the internalization was significantly (P < 0.05) higher after 240 minutes. The invasion process was also dependent on bacterial quantity and also suggested saturation of cellular receptors above MOI 100. 16 In addition, after invasion, C. pseudotuberculosis remained viable for up to 144 hours, raising the possibility that C. pseudotuberculosis may persist in non-phagocytic cells in different tissues of sheep and goats, facilitating progression of the infection in the animals.
The invasion process was dependent on temperature, suggesting that cell metabolism plays an active role in the invasion process. Finally, the incubation of bacteria prior to invasion with sugars, inactivated and whole antiserum, appeared to block specific bacterial receptors and/or cellular ligands.
Immunofluorescence demonstrated that heat- or UV-killed bacteria were unable to invade the FLK-BLV-044 cell line, suggesting that invasion requires either viable C. pseudotuberculosis or intact bacterial surface components to interact with the ovine cell surface. Further studies of the invasion and persistence of C. pseudotuberculosis should aid the development of therapeutics and vaccines and may assist in new aspects relevant to responses against C. pseudotuberculosis or other facultative intracellular pathogens.
In conclusion, these in vitro findings raise the possibility that C. pseudotuberculosis can persist and disseminate in vivo through non-phagocytic cells, facilitating diffusion of the infection and providing a certain level of protection against animal defenses and a suitable environment for its proliferation, facilitating the establishment of a carrier state.
Footnotes
Acknowledgements
We would like to thank Spanish Agency for International Development (AECID) for their program of PhD scholarships for foreign students for J. Valdivia.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
