Abstract
Peritonitis due to infections with green algae was diagnosed at slaughter (in Texas and South Dakota) in 2 cows. One cow also had a generalized lymphadenitis. The intralesional green algae were histologically similar to those previously associated with bovine lymphadenitis. Amplified and sequenced algal ITS2 genes had higher homology with the genus Scenedesmus than with Chlorella.
Clinical disease associated with disseminated green algal infections has been reported mainly in sheep,2,6,10 with 1 report in a dog. 6 Green algal infections of the skin of a gazelle and intestine of a dromedary have also resulted in clinical disease. 6 Infections with green algae in cattle, however, typically result in a granulomatous lymphadenitis that is only detected at slaughter. 7 In this report, peritonitis resulting from green algal infection was detected in 2 cows at slaughter.
Case Histories
An aged cow (cow No. 1), consigned to a Texas slaughter establishment in late April, was clinically normal prior to slaughter, but multiple green masses that thickened the parietal peritoneum were detected at postmortem examination. A 6-year-old beef cow (cow No. 2) from South Dakota, presented for slaughter in early June, was also clinically normal antemortem, but multiple firm green plaques and nodules that thickened the parietal peritoneum (Fig. 1) were detected postmortem. In addition, a firm green nodule was in the liver, and lymph nodes throughout the carcass were firm, enlarged, and bright green.
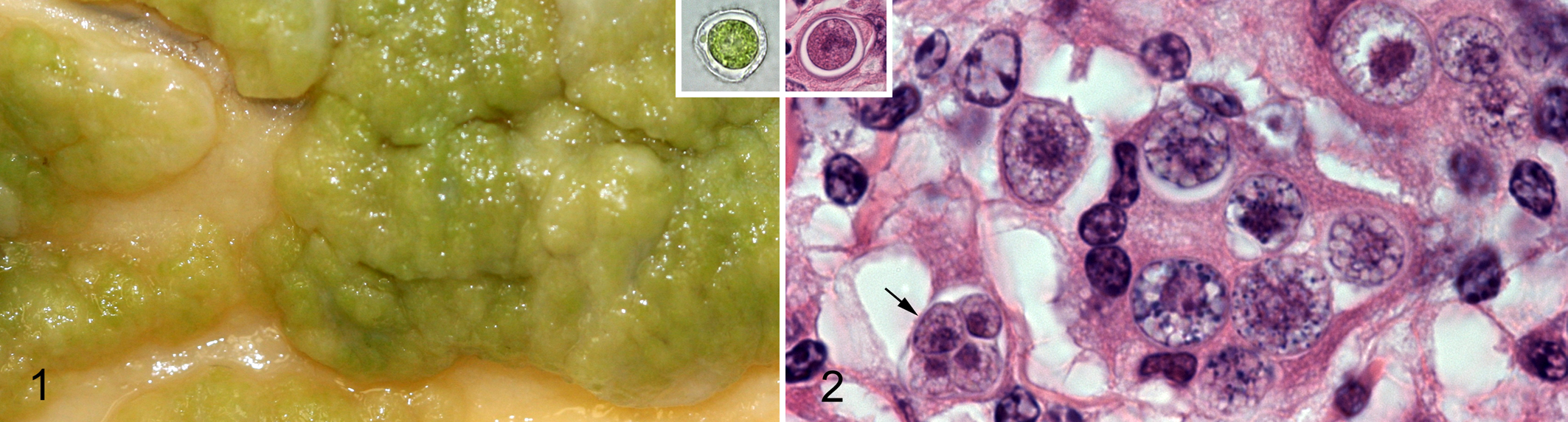
Peritoneum; cow No. 2. The peritoneum is thickened by coalescing green nodules. Inset: Unstained impression smear. The algal cell is 10 μm in diameter with green cytoplasmic pigment and a thick refractile cell wall.
Cytologic and Histologic Findings
In unstained impression smears of formalin-fixed tissue from both cows, numerous 10- to 15-μm, irregularly round to ovoid organisms with green cytoplasmic pigment and thick refractile cell walls were observed within macrophages and extracellularly (Fig. 1, inset). Representative formalin-fixed tissue samples were routinely processed, embedded in paraffin, sectioned at 4 μm, and stained with hematoxylin and eosin (HE) or periodic acid–Schiff (PAS).
Histologically, in both cows, the peritoneal mesothelial layer was multifocally elevated by accumulations of macrophages and multinucleated giant cells; this inflammation did not extend through the underlying connective tissue into adipose tissue or skeletal muscle. The lymph nodes of cow No. 2 were infiltrated by a similar population of leukocytes, as was a nodule in the liver. Occasional aggregates of plasma cells and scattered eosinophils accompanied the macrophages and multinucleated giant cells. Numerous irregularly round to ovoid organisms (sporangia), 4- to 15-μm in diameter (Fig. 2), were in the cytoplasm of macrophages and multinucleated giant cells. Extracellular sporangia were also observed. The organisms had a lightly eosinophilic cytoplasm that was often retracted from the cell wall. In most organisms, cell walls were uniformly thin without ornamentation, but the outer cell wall of some organisms had bulges (Fig. 2, inset). The cytoplasm often appeared refractile or finely vacuolated in HE-stained sections; under polarized light, each algal cell contained brightly birefringent crystals, each with a centrally located cruciate shadow (starch granules in chloroplasts). The algal cells had numerous PAS-positive cytoplasmic granules. Many sporangia had undergone endosporulation with the formation of 2 to 8 wedge-shaped sporangiospores. Some organisms were necrotic; adjacent macrophages contained crescentic (collapsed) sporangia with fragmented cell walls.
Transmission Electron Microscopy
One-millimeter cubes were removed from a paraffin block (cow No. 2) and de-paraffinized in xylene, rehydrated in a graded ethanol series, and then fixed by immersion in a solution of 2% glutaraldehyde, 2% paraformaldehyde, and 0.2% picric acid in a cacodylate buffer. These tissues were routinely processed and embedded in Epon; ultrathin sections were placed on coated grids, contrasted with lead citrate and uranyl acetate, and examined in a transmission electron microscope. Ultrastructurally, irregularly round individual algae with a single nucleus averaged 8 to 12 μm in diameter; some sporangia had asymmetric bulges in the outer cell wall (Fig. 3). Cell walls were composed of outer trilaminar and inner microfibrillar components (Fig. 3, inset). The cytoplasm contained osmiophilic globules, electron-lucent vacuoles, and numerous round to ovoid, electron-lucent starch granules within thylakoid stacks (Fig. 4). Pyrenoids were not identified.
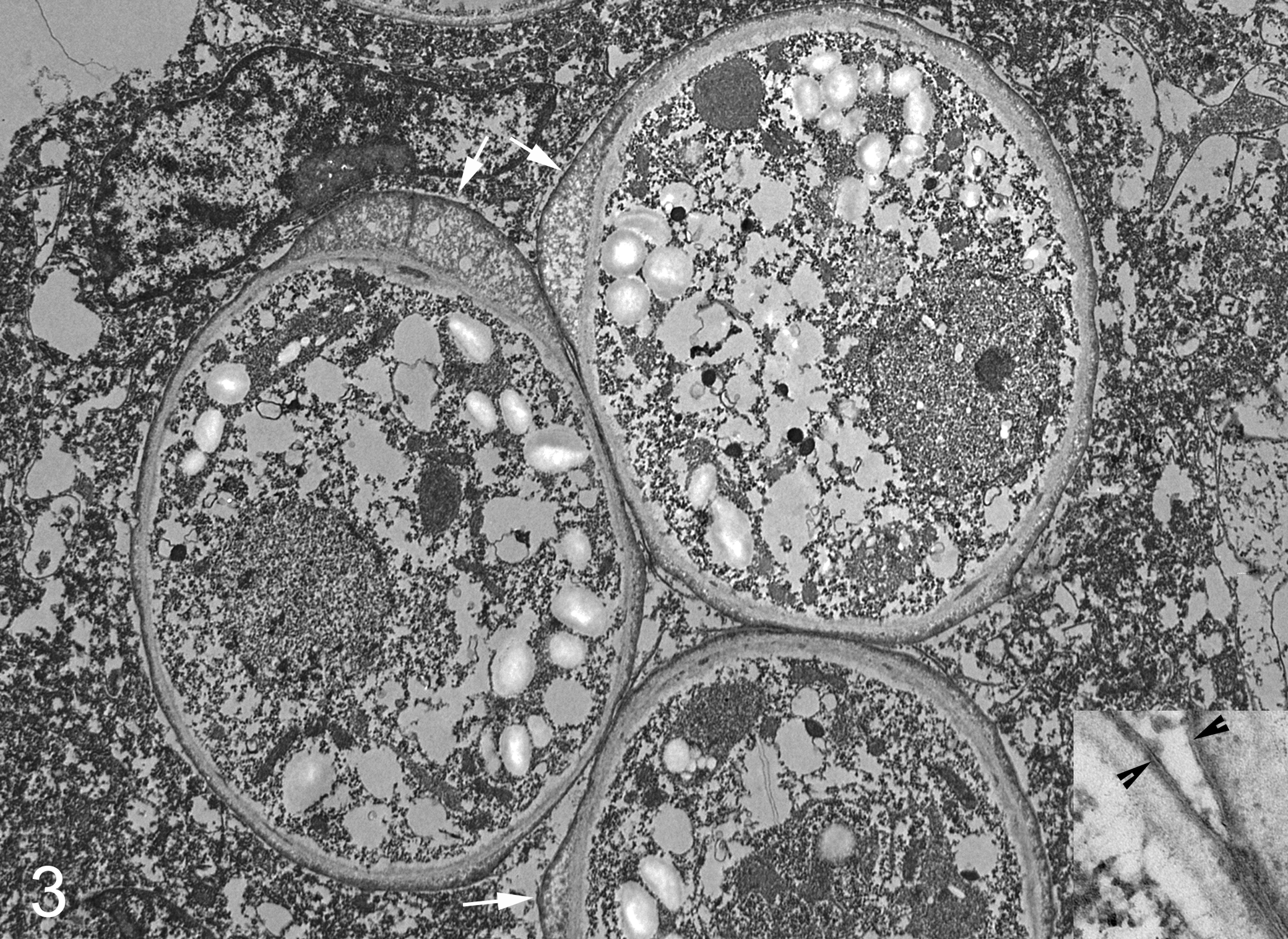
Lymph node; cow No. 2. The 8- to 12-μm-diameter sporangia have cell walls that are focally thickened (arrows). The cytoplasm of the sporangia contain numerous round to ovoid starch granules. Electron microscopy. Inset: Apposed cell walls of 2 sporangia. Note outer trilaminar layers (arrowheads) and inner microfibrillar layers. Electron microscopy.
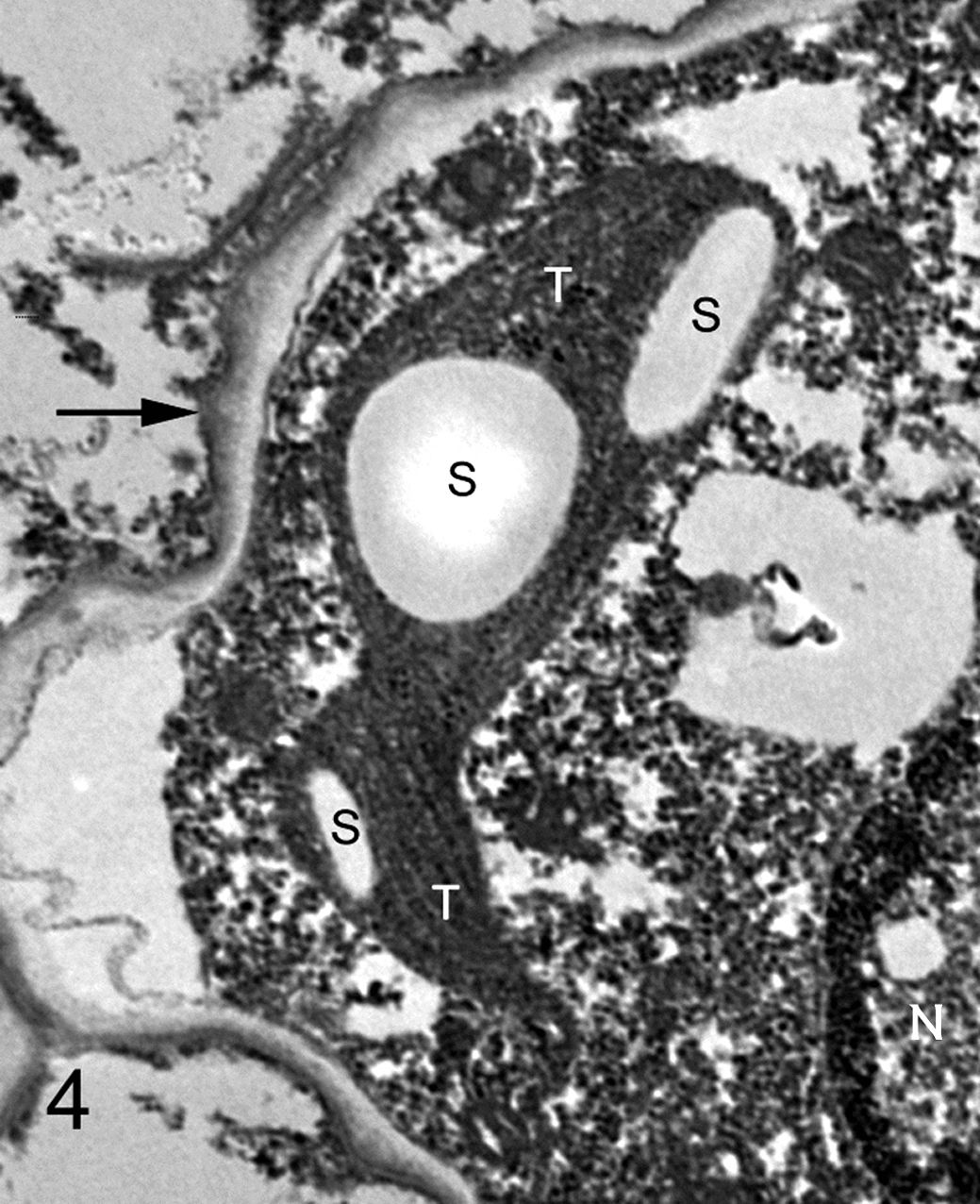
Lymph node; cow No. 2. Higher magnification of an algal cell containing a nucleus (N) and starch granules (S) embedded within thylakoid stacks (T). The cell wall is indicated by an arrow. Electron microscopy.
Molecular Diagnostics
DNA was extracted from paraffin-embedded tissues from each cow and from the unfixed and formalin-fixed, paraffin-embedded (FFPE) control algal culture. Extracted DNA samples were quantified and used in a polymerase chain reaction using primers ITS3 and ITS4 to amplify the entire internal transcribed spacer 2 region (ITS2) and segments of the bracketing 5.8 S rRNA gene and large subunit (LSU) rRNA gene. 8 The PCR conditions were incubation at 94°C for 10 minutes, 30 cycles of 94°C for 30 seconds, 50°C for 30 seconds, and 72°C for 45 seconds, with a final extension at 72°C for 10 minutes. The PCR assay contained 2 positive controls—unfixed and FFPE green algal culture (Carolina Biologic Supply, Burlington, North Carolina)—and 1 negative control. Amplified products were electophoresed in a 2% agarose gel containing ethidium bromide. Amplification produced approximately 430 base pair fragments (Fig. 5). The PCR-amplified products were sequenced and compared with sequences in the ITS2 database at http://its2.bioapps.biozentrum.uni-wuerzburg.de/cgi-bin/index.pl. 3 The ITS2 sequences generated from both unfixed and paraffin-embedded control algae matched (100%) the ITS2 sequence reported for this algal strain (AY948419). The ITS2 sequences and partial LSU sequences obtained from algae infecting cow No. 1 (GenBank accession number JQ615587) and cow No. 2 (GenBank accession number JQ615588) were identical. However, there were differences in a few nucleotides in the adjacent partial 5.8 S sequences. Comparison of the 248 nucleotide ITS2 segments (obtained by hidden Markov modeling) 3 to the ITS2 database did not identify ITS2 sequences that were 100% homologous. The green algae in the database with ITS2 sequences that most closely matched and were predicted to generate high-quality models of the subject ITS2 genes included Scenedesmus Pic6/16T-1 W (87%) and Scenedesmus pectinatus (88%).
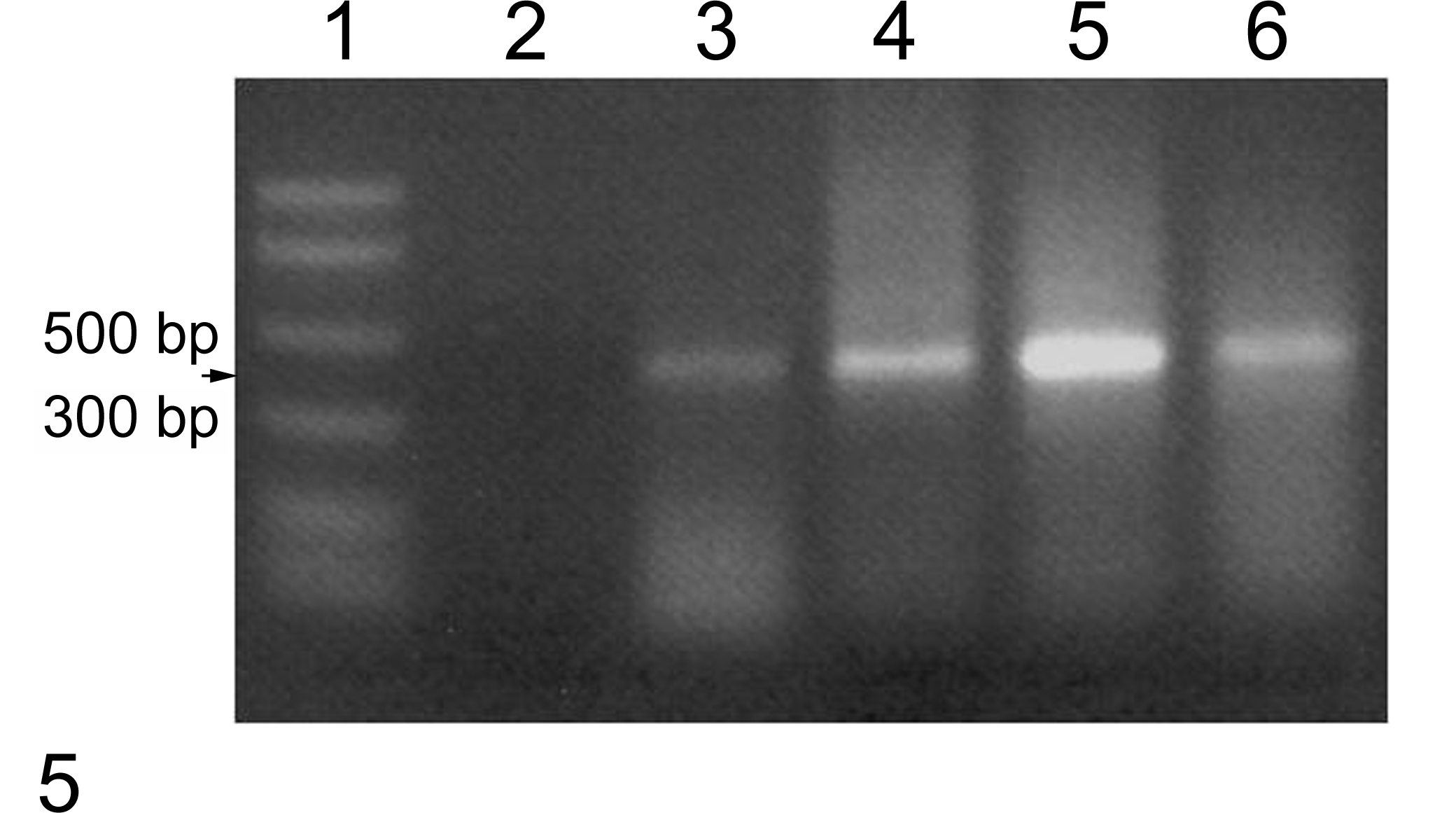
Agarose gel electrophoresis of polymerase chain reaction–amplified algal rDNA using primers ITS3 and ITS4. Lane 1, DNA ladder; lane 2, negative control; lane 3, positive control (unfixed algal culture); lane 4, positive control, (formalin-fixed, paraffin-embedded algal culture); lane 5, sample from cow No. 1; lane 6, sample from cow No. 2.
Discussion
The first reported algal infections were with the achlorophyllous algae Prototheca, ubiquitous saprophytes that can cause disease in humans and animals. 6 Cordy first recognized green algal infections of animals with his description of an affected lamb liver in 1973. 7 Some earlier and subsequent reports of protothecal infections in cattle were then retrospectively recognized as infections with green algae. 7 In cattle, green algal infections have mainly affected the retropharyngeal lymph nodes, 7 but there is 1 report of peritonitis (originally reported as due to Prototheca). 2
The green algae causing animal disease generally have been assumed to be unknown species of Chlorella, unicellular green algae that, like Prototheca, are now distributed mainly within the phylum Chlorophyta, class Trebouxiophyceae, order Chlorellales, and family Chlorellaceae. 4 “True” named Chlorella species are now limited to C. vulgaris, C. lobophora, and C. sorokiniana; these are spherical unicellular microalgae without cell wall ornamentation or mucilage. 4 Ultrastructurally, the cell walls of these algae lack the outer trilaminar layer that is associated with the production of sporopollenin. 1 Few reports7,10 indicate the successful culture of the green algae infecting warm-blooded animals; the green algae that infected a human wound 2 were not successfully cultured. The strains of green algae cultured from bovine lymph nodes were spherical to ellipsoidal organisms with some similarity to the then-classified Chlorella, now Scenedesmus fusca, but the growth characteristics were more consistent with those of unknown unicellular strains of Scenedesmus (Chlorophyta, Chlorophyceae, Sphaeropleales, Scenedesmaceae). 7 Scenedesmus are abundant in fresh water but also can survive in the soil of desert environments. These algae are often elongated and coenobial (colony forming); however, there are unicellular species with more coccoid shapes. 5 Some Scenedesmus species have been reported to cause granulomas in the skin, skeletal muscle, spleen, and kidneys of cichlids. 9
The presence of a cell wall with a sporopollenin layer in the green algae causing peritonitis in these cows is a characteristic of organisms classified in the genus Scenedesmus but not of those remaining in the genus Chlorella. 1 The identical ITS2 segment derived from algae infecting cattle slaughtered in widely separated states seems to suggest an enhanced susceptibility to this organism rather than a common source. In summary, 2 cases of peritonitis in cattle were caused by infections with green algae that appear to be more closely related to Scenedesmus than to Chlorella. Thus, infections with green algae, formerly known as chlorellosis, would more accurately be designated “chlorophytosis.”
Footnotes
Acknowledgements
We thank Drs. Milton Scroggs and Patrick McCain for submission of these samples.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the authorship and/or the publication of this article.
Funding
The author(s) received no financial support for the research and/or authorship of this article.
