Abstract
The objective of the current study was to evaluate the feasibility of lymph collection from the bovine udder and to investigate if the lymphatic fluid might be of diagnostic value in cows infected with Mycobacterium avium subsp. paratuberculosis (MAP), the etiologic agent of paratuberculosis. Lymph fluid collection was attempted from 58 cows, and the reactions of the cows as well as the level of difficulty of the procedure were recorded in 56 animals. Lymph samples (51 in total) were tested for the presence of MAP by nested polymerase chain reaction. Collection of the lymphatic fluid caused no or mild signs of discomfort in 94.6% of the cows; in 51.8% of cows, lymphatic fluid was attained on the first attempt, while sample collection was unsuccessful in 12.1%. Mycobacterium avium subsp. paratuberculosis was detected in 43.1% of all lymph samples. The bacterium was present in 66.7% of cows with clinical Johne’s disease, in 42.8% of asymptomatic cows with a positive or suspicious enzyme-linked immunosorbent assay (ELISA) result in blood, and in 38.7% of cows with a negative ELISA result in blood. The present study shows that the procedure was well tolerated by most cows and can easily be performed on farm. The current report of the isolation of MAP from lymph fluid suggests that the present approach could be used for the early detection of Johne’s disease in cattle.
Introduction
The lymphatic system is an important part of the immune system in mammals and plays a major role in the defense against infections. 8 The bovine udder has many well organized afferent lymph vessels, which can be grouped into subcutaneous, parenchyma, and teat vessels. 8 The subcutaneous lymph vessels draining the mammary lymph nodes are large vessels that are visible through the skin and may be distinguished from the subcutaneous veins by their dorsocaudal course. 7 The large subcutaneous lymph vessels can best be seen at the lateral and caudal part of the hindquarters of the udder. Visibility varies according to the milk yield, stage of lactation, presence of udder edema, and individual variations in the development of the vessels. 12 A pulsatile flow of lymphatic fluid ranging from 13 to 45 ml/hr was recorded in afferent lymph vessels at the udder of healthy cows, 10 while a total lymph flow of up to 1 l per day was reported from the udder of lactating goats. 17
Although the composition of the lymphatic fluid reflects that of blood plasma, there are distinct differences between the 2 regarding cells, trace elements, enzymes, and metabolic parameters.8-10,21 Lymphatic fluid collected from the afferent lymphatic vessels of the udder is straw colored and is more or less opaque, depending on the cell content. 26 The quantity of white blood cells in bovine udder lymph varies from 10,000 to 2.5 million cells/ml. 27 In another study, 1–2.5 million white blood cells/ml of lymphatic fluid, with macrophages representing 2–4% of these cells, were reported. 21 Variations in cell counts are mainly related to the health, lactation status of the sampled animals, and sampling time.21,26
Bovine udder lymph has been collected either by surgical procedures to study lymph flow and composition10,26 or by puncture of superficial lymphatic vessels with11,20 or without (Dübeler I: 2006, Antioxidativer Status in Euterlymphe und Blut von gesunden und kranken Kühen [Antioxidant status in udder lymph and blood of healthy and sick cows]. Thesis, University of Veterinary Medicine Leipzig, Germany. In German)8,9,12 local anesthesia before puncture. Puncture of lymph vessels has been performed in the study of lymph composition in relation to the health status of cows and has been described as safe and easy to perform. No further detail is given concerning the feasibility or use of this procedure (Dübeler I: 2006, Antioxidativer Status).8,9
Paratuberculosis, or Johne’s disease (JD), is caused by Mycobacterium avium subsp. paratuberculosis (MAP) and can be found worldwide.2,14 The disease mainly affects domestic and wild ruminants, with an estimated prevalence of 68.1% among U.S. dairy operations in 2007 (APHIS Info Sheet April 2008, http://www.aphis.usda.gov), and up to 68.0% among cattle herds in parts of Europe. 25 Paratuberculosis is listed by the World Organization of Animal Health 37 and can cause considerable economic losses in affected farms. An estimated loss of $200 million ± $160 million to the U.S. economy was reported in 1996 due to reduced milk yield in JD-positive farms. 18
Fecal–oral transmission is the most common route of disease transmission, with infected animals shedding high amounts of MAP in the feces. 20 Additional sources of infection come from MAP found in the colostrum and milk of cows with advanced paratuberculosis 30 as well as prenatal infection in utero. 36 After ingestion, MAP is taken up by M cells in the Peyer patches, which are a part of the gut-associated lymphoid tissue, and is passed to macrophages within the Peyer patches. 22 Within the macrophages, MAP can migrate to mesenteric lymph nodes and via the ductus thoracicus to the bloodstream, leading to hematogenous spread of infected macrophages.6,19
A mean incubation period for JD of 5 years has been reported in dairy cows. 15 Because of intermittent and low levels of MAP shedding, 34 as well as a delayed and highly variable immune response to the organism, 23 diagnosis of paratuberculosis infection is difficult in the early stages of the disease.
Cultivation of MAP in fecal samples has a high specificity and a sensitivity that ranges between 39.0% and 92.0%, depending on the culture method used and infection stage of tested animals. 34 Fecal culture is considered the gold standard in the diagnosis of paratuberculosis. 37 Polymerase chain reaction (PCR)-based tests are the second widely used method for direct MAP detection in feces, milk, blood, and tissue samples of cattle. The overall specificity of the PCR technique was estimated to be 97.0%, with a sensitivity of 60.0%, 1 depending on different primers, techniques, protocols, and study designs. 3 Enzyme-linked immunosorbent assays (ELISAs) to detect specific antibodies against MAP are the most broadly used indirect test for JD. Sensitivity and specificity of ELISAs vary widely, depending on the test kit used. The sensitivity of the ELISAs range between 7.0% and 94.0%, and the specificity between 41.0% and 100%. 24
The first objective of the current study was to evaluate the feasibility of the collection of lymphatic fluid under field conditions by puncture of lymph vessels (lymph puncture) superficial to the bovine udder and to assess the cow’s reaction to the procedure. The second objective was to determine if the lymphatic samples taken at the udder could be used for detection of MAP by PCR in cows infected with JD. The overall aim of the study was to challenge the hypothesis that lymphatic fluid from the udder of cows infected with JD is of earlier diagnostic value than blood or milk of these cows because MAP may occur in macrophages within lymphatic fluid in early stages of the disease.
Material and methods
Study population
Cows selected for the study originated from 3 dairy herds known to be infected with MAP, each located in north central Florida. Animals were selected with the aim to sample as many cows with the suspicion of a MAP infection as possible. Overall, 58 cows were included in the study, with 48 cows selected from the University of Florida Dairy Unit facility (Gainesville, FL), and a further 10 cows from 2 different large commercial dairies. The university farm consisted of approximately 450 lactating cows; the 2 commercial dairy farms housed approximately 3,500 and 1,500 lactating cows, respectively. Animals were kept in free stall barns and were milked in a double herringbone milking parlor in all 3 farms. The predominant breed was Holstein, with a study population of 94.8% (55/58) Holstein cows, 3.5% (2/58) red Holstein, and 1.7% (1/58) Holstein crossbred cows. The mean age of study cows was 5.0 years with a maximum of 8.8 and a minimum of 2.0 years. All cows were lactating at the sampling date. Days in milk and pregnancy status were not recorded.
Of the 58 selected cows, 7 showed clinical signs of JD (i.e., diarrhea and weight loss), while another 7 were chosen because they were among the older group of cows in the herd and had a positive or suspicious MAP serum ELISA result. A low body condition score was assigned to 41 animals. Additionally, 3 cows sampled at random occasions were included in the study population.
Collection of lymph samples
Lymph samples were collected after milking with cows restrained in an examination parlor, which is an area common to most dairies where routine rectal palpation and medical examinations are performed. The cows were not sedated, and beyond the occasional lifting of the tail, no additional restraining methods were applied.
Superficial lymphatic vessels of the udder were identified as slightly contoured, up to 2-cm-wide vessels which, in contrast to blood vessels, did not move when the skin of the udder is moved (Dübeler I: 2006, Antioxidativer Status). Furthermore, lymphatic vessels were distinguished from superficial blood vessels by their diagonal orientation, crossing under the superficial blood vessels as seen in Figure 1. After cleaning the puncture site with an iodophor scrub and 70% alcohol, a 18-gauge (1.27-mm) needle was placed at an angle of approximately 45° through the skin and inserted a few millimeters into the lymph vessel as described. 7 Then, by either dripping or slow flow, the lymphatic fluid was collected in a sterile tube containing 0.75 ml of ethylenediamine tetra- acetic acid (EDTA a ) to prevent clotting. The collected samples were cooled for transport and stored at −20°C until further processing in the laboratory.
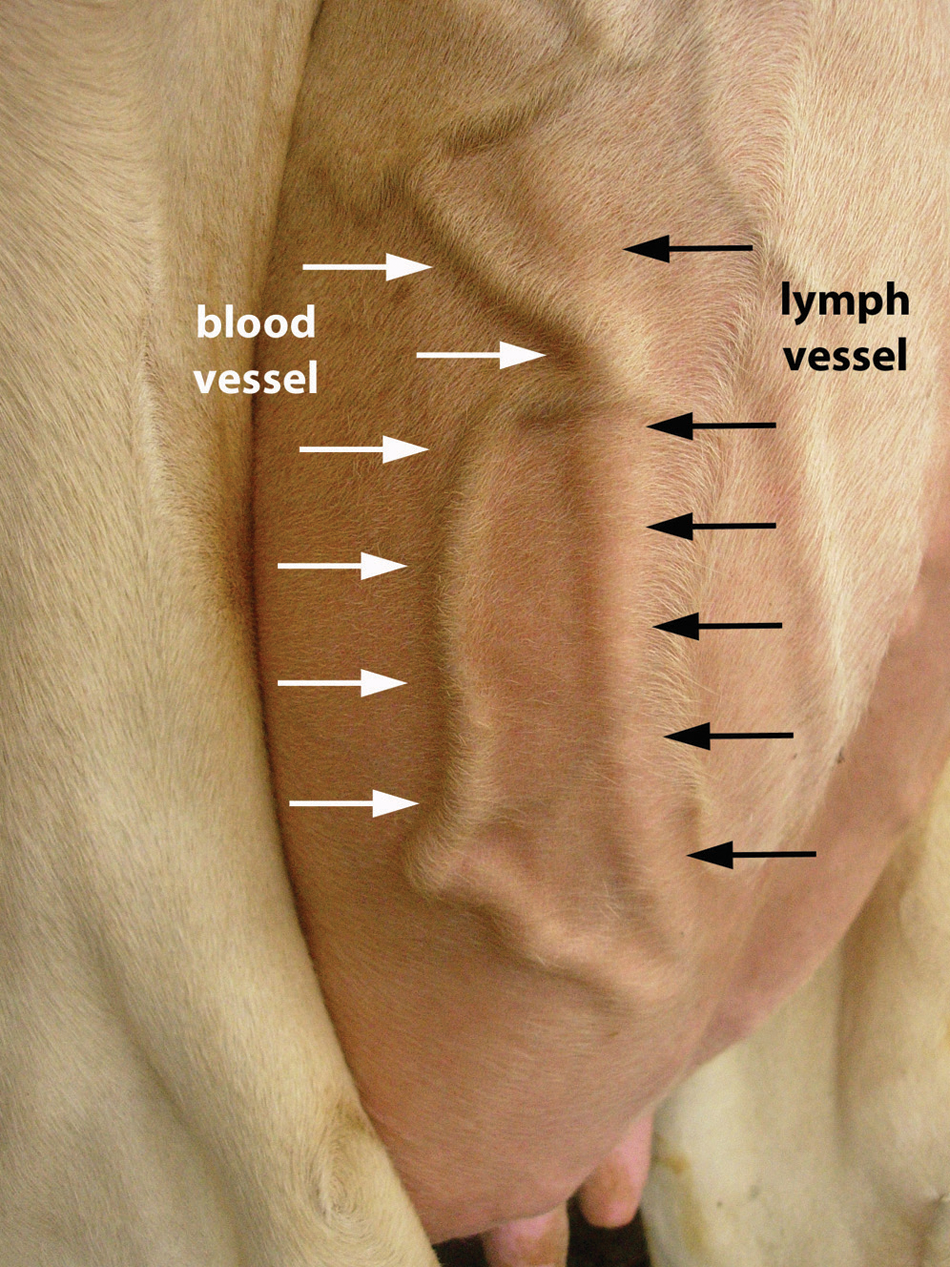
Subcutaneous lymph and blood vessel at the lateral part of the left hindquarter of a bovine udder.
Rating of the lymph collecting procedure
To assess the feasibility of the collection of bovine udder lymph under field conditions, the reaction of the cows to the lymph puncture as well as the level of difficulty of the procedure were recorded. The reaction of the cows to the lymph collection was rated as follows: 1 = no signs of discomfort, 2 = mild signs of discomfort, 3 = moderate signs of discomfort, and 4 = serious signs of discomfort with kicking in response to the puncture of the lymphatic vessel. The level of difficulty was rated as follows: 1 = easy, lymph gained at first attempt; 2 = easy but more than 1 attempt necessary; 3 = moderate difficulties but collection possible; 4 = very difficult but collection of lymph fluid possible; and 5 = collection not obtained.
Collection of blood samples
Blood samples were taken simultaneously to the lymphatic samples by venipuncture of the tail vein, using a commercial system a and sterile tubes without additive. a Blood samples were cooled for transport. After arrival at the laboratory, serum was harvested by centrifugation and stored at −20°C until the ELISA was performed.
DNA extraction
A modified protocol published for MAP DNA extraction in blood was used for DNA extraction. 28 Lymphatic fluid was centrifuged at 1,000 × g for 10 min. The supernatant was removed, and the pellet was washed twice (1,000 × g/10 min.) in 1 ml of phosphate buffered saline. After the second wash, the pellet was resuspended in 50 µl of 0.2 N NaOH and heated at 110°C for 20 min. The remaining sample was again centrifuged at 1,000 × g for 15 sec. The supernatant was transferred in a sterile tube and stored at −20°C until further processing.
IS900 nested polymerase chain reaction
MAP DNA in lymphatic samples was detected by a modified nested PCR described for use in blood samples. 28 The PCR was performed using a commercial reaction mix b according to the manufacturer’s specification and a thermocycler. c To avoid cross-contamination of PCR reactions, precautions that included preparation of samples in an ultraviolet-treated laminar flow hood were applied. For detection of MAP, 1 µl of the product of the DNA extraction was used in the first reaction, with primers P90 and P91, targeting a 413-bp sequence of the IS900 in MAP.4,33 The protocol for the first reaction of the nested PCR was 35 cycles at 94°C for 30 sec, 58°C for 15 sec, and 72°C for 60 sec. Primers J1 and J2, overlapping a span of 333-bp region within the insertion sequence,4,33 were used for the second reaction, with 1 µl of the product of the first reaction. The protocol for the second stage consisted of 36 cycles at 94°C for 30 sec, 63°C for 15 sec, and 72°C for 60 sec.
MAP DNA isolated from a cow with clinical JD confirmed by necropsy was used as positive control, and sterile water was included as negative control in each of the PCR reactions. After the second PCR reaction, 10 µl of the reaction product was run on 1.5% agarose gel by electrophoresis in Tris–acetic acid–EDTA running buffer. d Gel inspection was performed after a running time of 45 min at 100 V (volt) using ultraviolet light e and commercial software. f
Enzyme-linked immunosorbent assay
Two commercial kits were used for detection of MAP-specific antigens in blood serum according to the manufacturer’s specifications. In the first kit, g samples with a S/P value (sample-to-positive ratio) under 0.1 were rated as negative. Samples with an S/P value between 0.10 and 0.25 were rated as suspicious, between 0.26 and 0.40 as low positive, between 0.41 and 1.0 as positive, and above 1.0 as strong positive. For the second kit, h cutoff values were a S/P value under 0.60 for negative, 0.60–0.70 for suspicious, 0.71–1.25 for low positive, 1.26–2.50 for positive, and greater than 2.50 for strong positive samples.
Results
Feasibility of lymph collection
Data concerning the feasibility of lymph collection were not recorded in 2 cows. The reaction of the remaining 56 cows to the lymph puncture at the udder and the degree of difficulty to perform the procedure in cows are presented in Table 1. Collection of the lymphatic fluid caused no or mild signs of discomfort in 94.6% (53/56) of the cows. Only 1 (1.8%) cow showed serious signs of discomfort with kicking as a reaction to the procedure. In 51.8% (29/56) of the cows, lymphatic fluid was obtained on the first attempt. Lymph collection was not successful in 12.1% (7/58) of cows.
Relation between cow reaction to lymph collection and level of difficulty of lymph collection from the bovine udder (n = 56).*
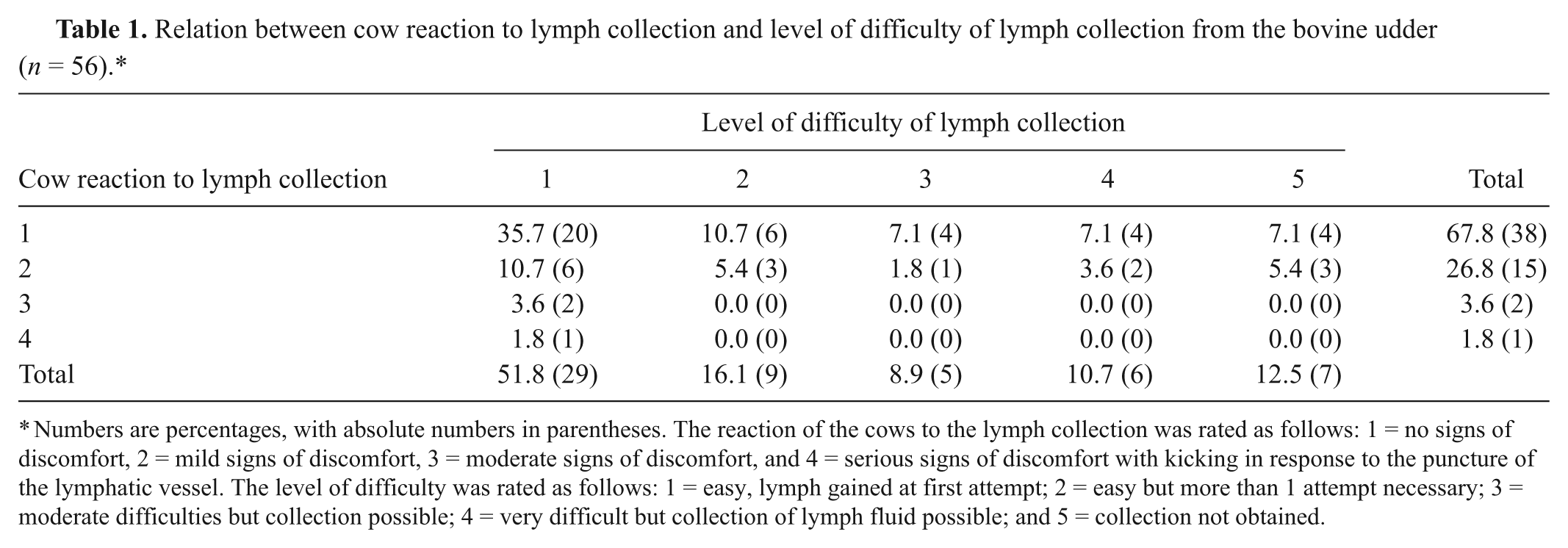
Numbers are percentages, with absolute numbers in parentheses. The reaction of the cows to the lymph collection was rated as follows: 1 = no signs of discomfort, 2 = mild signs of discomfort, 3 = moderate signs of discomfort, and 4 = serious signs of discomfort with kicking in response to the puncture of the lymphatic vessel. The level of difficulty was rated as follows: 1 = easy, lymph gained at first attempt; 2 = easy but more than 1 attempt necessary; 3 = moderate difficulties but collection possible; 4 = very difficult but collection of lymph fluid possible; and 5 = collection not obtained.
Table 1 also shows the number of cows for each combination of cow reaction and rating of the lymph sampling. In 35.7% (20/56) of the cows, collection of the lymphatic fluid was possible at the first attempt, and the animals showed no signs of discomfort in response to the procedure. Even the cows from which the collection was difficult or not possible showed no or very little sign of discomfort. Conversely, the procedure was easy to perform in the 2 cows showing moderate signs of discomfort and in the 1 cow with serious signs of discomfort (Table 1).
A mean value of 1.9 ml of lymph fluid with a maximum of 4.0 ml and a minimum of 0.2 ml was collected from the sampled cows. No excessive or prolonged bleeding, dermatitis, reduced milk yield, or mastitis related to the lymph collection was evident in cows of the current study.
Appearance of lymphatic fluid
The majority of the lymph samples were colorless (56.9%, 29/51), with the remaining specimens showing a yellowish coloration in 19.6% (10/51) and a reddish coloration in 23.5% (12/51) as seen in Figure 2. Approximately half of the lymph samples showed a certain degree of opacity, which was rated as turbid in 15.7% (8/51) and cloudy in 39.2% (20/51). The remaining 45.1% (23/51) of collected samples were clear (Fig. 2).

Appearance of lymphatic fluid collected from the bovine udder (n = 51).
Detection of MAP in lymph samples
Mycobacterium avium subsp. paratuberculosis was detected in 43.1% (22/51) of the lymph samples by nested PCR for IS900. Lymph samples from 66.7% (4/6) of the cows showing clinical signs of JD were positive for the bacterium by PCR (Table 2). From the 14 cows without clinical JD but positive or suspicious blood ELISA results, 42.8% (6/14) had positive lymph PCR results (Table 2). Finally, PCR for MAP identified 38.7% (12/31) positive lymph samples collected from cows without signs of clinical JD and a negative blood ELISA result (Table 2).
Detection of Mycobacterium avium subsp. paratuberculosis by nested polymerase chain reaction in lymphatic fluid from cows with and without clinical Johne’s disease (n = 51).*
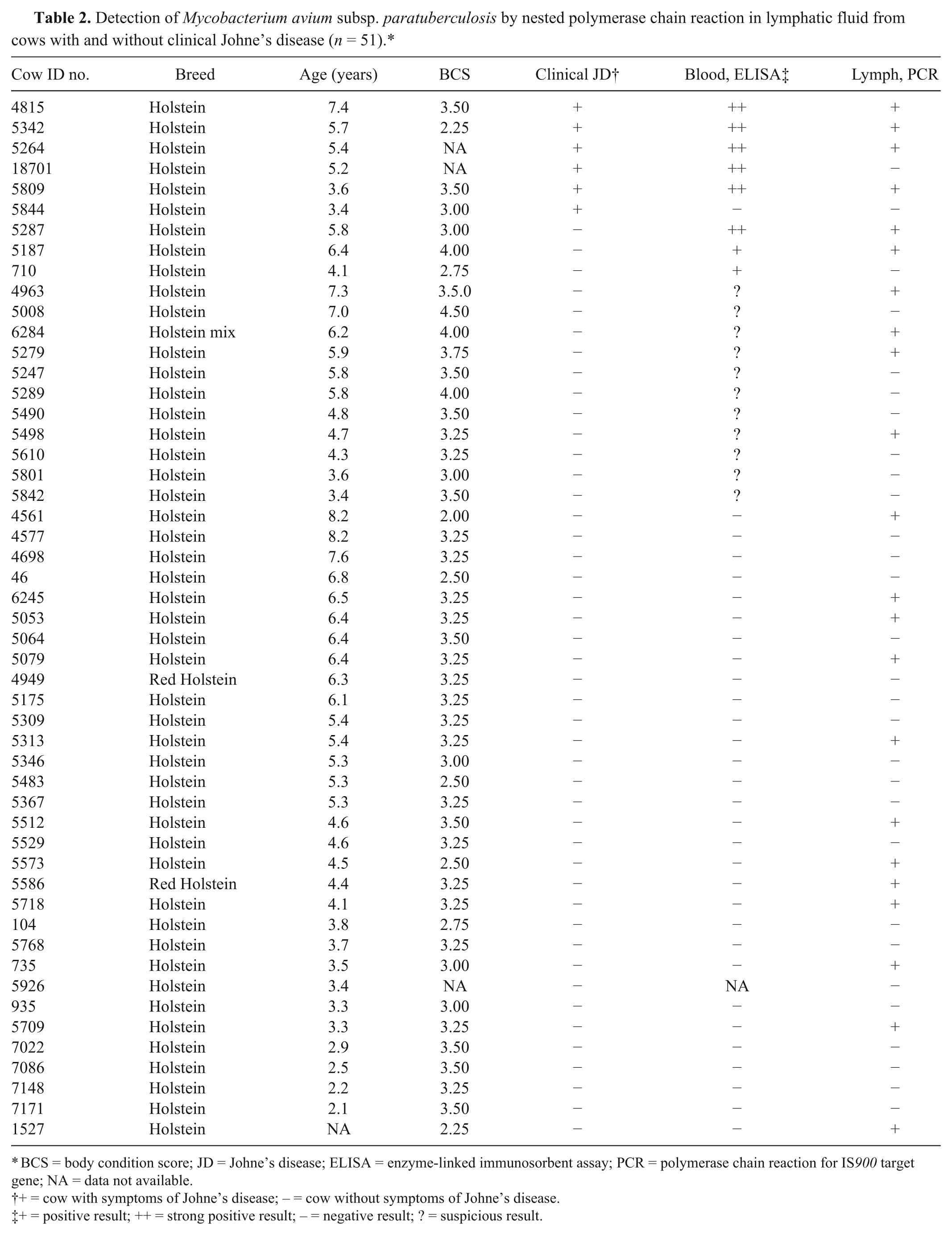
BCS = body condition score; JD = Johne’s disease; ELISA = enzyme-linked immunosorbent assay; PCR = polymerase chain reaction for IS900 target gene; NA = data not available.
+ = cow with symptoms of Johne’s disease; − = cow without symptoms of Johne’s disease.
+ = positive result; ++ = strong positive result; − = negative result; ? = suspicious result.
Discussion
The results of the present study show that the procedure of lymph collection was well tolerated by the majority of sampled cows. Even those cows in which the sampling was rated as difficult or impossible showed no or only slight signs of discomfort. Tail restraint was necessary in some cows. Lymphatic fluid was successfully collected in 87.9% (51/58) of the cows, and lymphatic fluid was obtained on the first attempt in most cows. Lymph vessels were most visible at the caudal and lateral part of the hindquarters of the udder; it was there that lymph puncture was performed in all cows in the present study.
In those cases where blood vessels were accidentally punctured, bleeding stopped after a few seconds of manual compression. Small hematomas occurred in some of these cases. Herd records showed no reduction in milk yield, and no occurrence of mastitis or any other alterations in cows included in the study, even in cases were blood vessels had been punctured or lymph collection was difficult or impossible. Sample collection might be even simpler and show an even higher success rate than in the present study if performed before milking, due to the improved visibility of the lymph vessels in udders filled with milk. Unfortunately, separation and restraining of cows before entering the milking parlor is hampered by milking and treatment routines in most commercial dairy farms.
From the results of the present study, it can be stated that the collection of lymphatic fluid from superficial lymph vessels of the bovine udder is a safe and relatively easy procedure to perform under field conditions. The sampling is well tolerated by most cows and can be performed in treatment parlors available in most dairy farms without interfering with the routine management activities of the farm. Beside the application for detection of MAP as discussed in the current work, lymphatic fluid might also be useful in the study of other aspects of bovine medicine such as mastitis, leucosis, or lymph-associated Bovine immunodeficiency virus.
In contrast to the findings of another study, which described lymphatic fluid from the bovine udder as straw colored, 26 the majority of the samples collected in the present study were colorless. Only 19.6% showed a yellowish color, and 23.5% of the collected samples were reddish, indicating contamination with blood. Venipuncture of a blood vessel prior to a lymph vessel was recorded in only 5 of the 12 cows showing a red-colored lymphatic fluid. This observation might be explained by the unnoticed accidental puncture of small blood vessels in the remaining 7 cows. Discoloration of the lymph due to red blood cells and heme products entering the lymph for up to 3 days after a surgical intervention at the udder was recorded in a previous study. 26 Therefore, trauma of the teat or the udder within a few days before lymph collection could also be a possible cause for a red color of the lymph samples, although no signs or history of teat or udder injuries were present in these cows during the present study.
While the lymph fluid was cloudy or turbid in most specimens of the present study, as previously reported, 26 45.1% of the samples were clear. The differences in the opacity of lymph samples is most likely caused by different amounts of white blood cells within the lymph, related to health and lactation status of the cow, as well as sampling time.21,26
The current explorative study reports the use of lymph for the detection of MAP. In addition to feces and intestinal tissue samples, which are routinely used for MAP detection, the organism has also been regularly detected in milk, various extraintestinal tissues, blood, and semen samples.3,35 In a previous study, MAP was isolated by bacteriologic culture from peripheral tissue fluid from a collection chamber implanted in the right paralumbar fossa of 2, and in blood of 1 out of 7, cows with advanced JD. 16 Culture of supramammary lymph nodes from asymptomatic cows infected with MAP was positive in 46.0% of cows heavily shedding MAP into feces, in 22.0% of intermediate, and 6.0% of light shedders. 31 Comparison of the results of the present study with the 2 previous studies is difficult due to the different sample material and methods used. Nevertheless, the current results indicate that lymph fluid might be a better sampling material for the detection of paratuberculosis in cattle than peripheral tissue fluid and supramammary lymph nodes. Only culture of supramammary lymph nodes from heavy MAP-shedding asymptomatic cows revealed a higher percentage of positive samples than in lymph fluid 31 from asymptomatic cows with an unknown amount of MAP shedding. Furthermore, as the collection of peripheral tissue fluid and the biopsy of the supramammary lymph node require special procedures and equipment, the procedures are not easily applicable to a large number of animals due to animal welfare, time, and cost.
The same primers as in the present study were used for detection of MAP by nested PCR in blood in 2 other studies.5,28 In the first study, 80.0% of the cows with clinical JD, 38.2% of cows with subclinical JD and positive or suspicious blood ELISA results, and 16.7% of subclinical cows with a negative serum ELISA result were MAP positive in blood. 5 In the second study, only 2.1% of cows without clinical JD originating from herds with a history of JD tested positive for MAP by nested PCR in blood samples. 28 Comparing the results of the first study 5 to the current study, MAP was detected in cows with clinical JD to a greater extent in blood than in lymph. In the present study, asymptomatic cows with negative or suspicious blood ELISA result showed more positive lymph than in blood samples. When comparing ELISA results to the detection of MAP in lymphatic fluid, it has to be considered that the blood ELISA detects specific antibodies against the organism, while PCR is detecting the organism itself. Therefore comparison of the results of these 2 methods is limited as they may detect a different subset of animals in different stages of MAP infections. 23 Comparing the results of the present study to those of the second study mentioned above, 28 MAP was detected in a greater percentage of lymph samples in the present study. A possible explanation for the greater number of MAP-positive lymph samples compared to blood samples might, at least in part, be the lack of PCR inhibitors in lymph fluid such as heme, hemoglobin, lactoferrin, and immunoglobulin G, which are present in blood samples. 29 The PCR protocol used in lymph in the present study was standardized for the detection of MAP in blood 5 and showed a complementary sensitivity of 64% when combined with fecal culture, and of 4% when combined with fecal PCR. 28 Unfortunately, the analytical specificity and sensitivity of the PCR essay are not known and need further investigation.
Although some studies have shown that lymphocytes do not migrate efficiently between different organ systems,6,13 dissemination of macrophages containing MAP through the blood and lymph system is suggested to be responsible for dissemination of the bacterium throughout the body.19,31,32 The bovine mammary gland appears to be regularly infiltrated by MAP and is seen as a possible reservoir for the bacterium,5,28 possibly caused by the high concentration of iron and iron-rich enzymes. The results of the present study indicate that the MAP-containing circulating macrophages can be detected in lymph fluid by PCR and might be used for the diagnosis of JD in cows.
As the current study was of an explorative nature, the precise infection status of the included cows was unknown and no known negative animals were tested, so no statements concerning sensitivity or specificity of this method can be made, which was not an aim of the study. The results of the present study indicate that the detection of MAP circulating within macrophages in the lymphatic system might be of use for the early detection of JD in cattle. Further studies to evaluate this new method for the diagnosis of JD in cattle are being investigated. The comparison of MAP detection in lymphatic fluid to the detection of the organism in feces, blood, and milk in different stages of the infection would be beneficial to determine its use as potential diagnostic test for JD in the future.
Footnotes
Acknowledgements
The authors thank the farm managers and workers, most notably Mr. Eric Diepersloot and his team, for their willingness to support this study, as well as Dr. Maureen Long and her team, especially Dr. Claudio Verdugo and Mr. Elliot Williams for their patient and competent support and cooperation.
a.
Vacutainer® Becton, Dickinson and Co., Franklin Lakes, NJ.
b.
Hot Master Mix®, Eppendorf North America, Westbury, NY.
c.
Gene Amp PCR System 9600, Perkin Elmer, Waltham, MA.
d.
Continental Lab Products, San Diego, CA.
e.
Gel Doc 2000, Bio-Rad Laboratories, Hercules, CA.
f.
Quantity One Version 4.5.2, 1-D Analysis-Software; Bio-Rad Laboratories, Hercules, CA.
g.
IDEXX HerdChek® M. pt. ELISA, IDEXX Laboratories, Westbrook, MA.
h.
IDEXX MAP Ab Test, IDEXX Laboratories, Westbrook, MA.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This study was financially supported by the University of Veterinary Medicine Vienna, Austria, PL4/2, and materially supported by the University of Florida Johne’s Research Laboratory.
