Abstract
Recently, confirmed occurrences of persistent bovine viral diarrhea virus (BVDV) infection in North American alpacas have raised concerns about the role of persistently infected (PI) alpacas in transmission of virus among herds, yet only limited pathological descriptions of persistent infections in alpacas have been reported. The objective of this study was to characterize BVDV antigen distribution in 10 PI alpacas of varying age and to compare viral antigen distribution and localization in tissues of PI alpacas with 5 PI calves of varying age. Ocular dysplasia was evident in 1 PI alpaca, constituting the first reported congenital ocular lesion in PI alpacas. Viral antigen was widely distributed in alpaca tissues and was prominent in neurons, endothelial cells, and vascular tunica media myocytes but had limited distribution in lymphoid tissues and moderate distribution in epithelium of several organ systems of alpacas. Macrophages in the alpaca gastrointestinal system submucosa and lymph node medullary sinuses often had prominent labeling. In addition, only 1 alpaca had antigen labeling in the bone marrow in contrast to PI cattle. Labeled cells in calf tissues were more widely distributed, occurring prominently in lymphoid and epithelial tissues. Common features of the 2 host species were widespread antigen labeling and absence of lymphoid depletion.
Keywords
Bovine viral diarrhea virus (BVDV) is a member of the genus Pestivirus in the family Flaviviridae, as are classical swine fever virus and border disease virus. BVDV isolates are grouped into 2 biotypes, noncytopathic (ncp) and cytopathic (cp), based on the virus’s effects in cell culture 28 , and into 2 genotypes, I and II. 31 BVDV infects cattle worldwide and causes substantial production losses. In addition to affecting sheep and swine, BVDV infections have been described in other species, including goats, exotic ruminants, Old World camelids, and New World camelids.4,11,16,18,23 Recently, confirmed reports of clinical cases of BVDV infections in North American alpacas leading to reproductive failure and persistently infected (PI) crias have raised the importance regarding occurrence of PI carrier animals.3,33 A recent experimental study showed transmission of BVDV from PI alpacas to naïve alpacas. 10
BVDV infection in cattle has several outcomes, which include subclinical infections, clinical BVDV with enteric or respiratory disease, hemorrhagic syndrome, venereal infections, reproductive failure (embryonic death, abortions, stillbirths), congenital defects, persistent infection, and mucosal disease.1,2,8,9 Lesions and antigen localization reflect viral tropism for lymphoid, neural, and epithelial tissues.6,7,17,21 Bovine fetuses infected in utero between 30 and 120 days of gestation with ncp BVDV are usually born PI and immunotolerant.13,20,27 PI cattle continuously shed large amounts of virus and are the major source of BVDV. 22
Although numerous pathological descriptions of persistent BVDV infections in cattle have been published,5,9 only a few corresponding descriptions of persistent infections in alpacas exist.11,12,19 The objectives of this study were to characterize viral antigen distribution in 10 PI alpacas and compare it with that of 5 PI calves.
Materials and Methods
Animals
Alpacas for the present study were donated by alpaca breeders who were members of the Alpaca Owners and Breeders Association; alpacas were registered with the Alpaca Registry, Inc, and were confirmed PI for BVDV type 1b.3,33 All were naturally occurring infections identified through diagnostic testing and then enrolled prospectively in this targeted study. Signalment and diagnostic testing are summarized in Table 1. Calves were identified by routine diagnostic testing for detection and removal of PI animals from cattle herds in Nebraska and Colorado. Ten alpacas (A1-A10) and 5 calves (C1-C5) were euthanatized after confirmation of PI status by polymerase chain reaction (PCR) or immunohistochemistry (IHC) between 2006 and 2007. One alpaca, A0, was negative for BVDV on PCR and IHC and served as a negative control. Alpacas A1–A10 and calves C1–C5 were confirmed positive for BVDV by virus isolation or repeated positive antemortem PCR and by IHC as previously reported. Specifically, PI alpacas were confirmed by a minimum of 2 consecutive positive whole-blood PCR results at greater than 3-week intervals and subsequent IHC at necropsy.3,33
Summary of Immunohistochemical Scores and Means for Alpaca and Bovine Tissues.
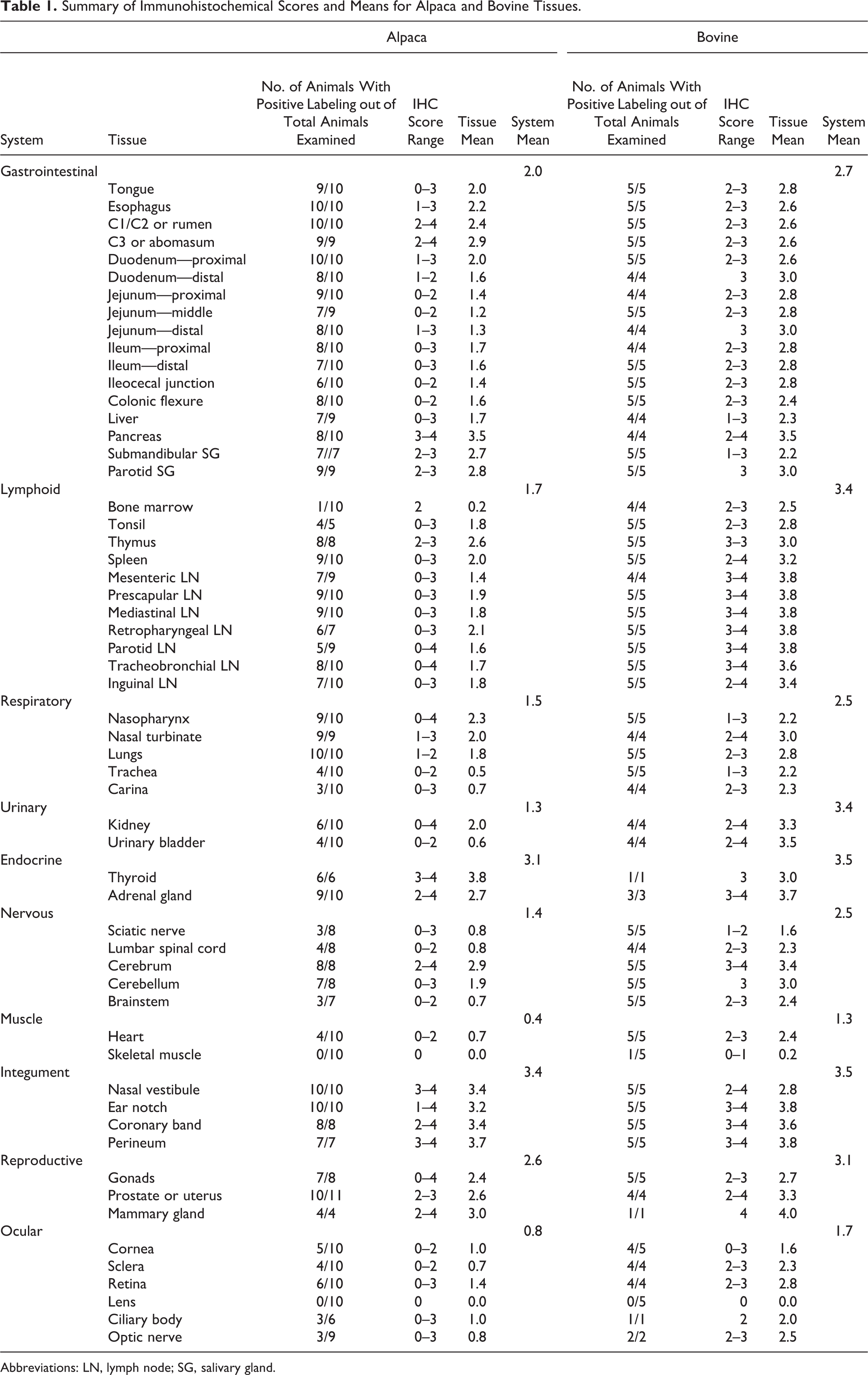
Abbreviations: LN, lymph node; SG, salivary gland.
Necropsy and Tissue Collection
Systematic necropsy was performed to collect tissues, and all gross abnormalities were recorded at necropsy by the pathologist (JNH). One pathologist (JNH) was responsible for all necropsies at the primary site, and 2 alpacas and 3 calves were examined by clinicians at different sites. A standard set of tissues was collected in all animals, and ancillary testing was not pursued on secondary lesions. Gross thymic atrophy was subjectively evaluated based on visual appraisal of the organ size. Systems sampled for BVDV histological analysis and IHC included the digestive system (tongue, salivary gland, esophagus, C1–C2/rumen, C3/abomasum, duodenum, jejunum, ileum, colon, ileocecal junction, exocrine pancreas and liver); hemolymphatic system (tonsils, thymus, spleen, bone marrow, and mesenteric, prescapular, mediastinal, retropharyngeal, parotid, tracheobronchial, and inguinal lymph nodes); respiratory system (nasal turbinates, nasopharynx, trachea, lungs, tracheobronchial junction); urinary system (kidneys, urinary bladder); endocrine system (adrenal gland, thyroid gland); muscle (cardiac and skeletal muscle); integument (coronary band, pinna, nasal vestibule, perineum); reproductive system (testis, prostate, ovary, uterus, mammary gland); eye; and nervous system (sciatic nerve, brain, lumbar spinal cord). Tissues collected were fixed in neutral-buffered 10% formalin.
Histopathological Analysis
Tissues collected at necropsy were processed and embedded in paraffin after 48–72 hours of fixation in neutral-buffered 10% formalin. Tissue was sectioned at 5 μm, stained with hematoxylin and eosin, and examined for lesions by light microscopy. Histological lesions were recorded and given a morphological diagnosis by a single pathologist (JNH), who was aware of the positive PI BVDV status of the animals. Thymus atrophy (involution) was assessed subjectively by evaluating cell density, size of lobules, and the presence of adipose tissue.
Immunohistochemical Labeling
Paraffin-embedded tissues were sectioned at 5 μm and labeled for detection of BVDV antigen by use of an avidin–biotin–alkaline phosphatase method. Sections were deparaffinized in xylene, rehydrated in a series of graded alcohol solutions, and treated with protease XIV (Sigma Chemical, St. Louis, MO) in 0.5 M Tris-buffered saline solution (TBSS; pH, 7.6) for 15 minutes at 37°C. Sections were blocked for 30 minutes at 21°C in TBSS with 4% equine serum. After blocking, primary antibody, anti-BVDV monoclonal antibody 15C5 15 directed against Erns/gp 48 (diluted 1:1000 in TBSS) was added, and sections were incubated for 1 hour at 21°C. Sections were washed twice in TBSS with 1 mM EDTA and 0.05% Tween 20 (4 minutes per wash). Biotinylated horse anti-mouse immunoglobulin (Vector Laboratories, Burlingame, CA) diluted 1:200 in TBSS with 2% normal bovine serum and 4% horse serum was applied, and slides were incubated for 30 minutes at 18°C to 24°C. Slides were washed as described, and a conjugated avidin–alkaline phosphatase complex (Vector Laboratories, Burlingame, CA) conjugated to streptavidin was added and allowed to incubate for 15 minutes. After washing, substrate (Biored Fast Red, Vector Laboratories, Burlingame, CA) was applied to the tissue sections, and slides were allowed to incubate for 15 minutes at 20°C to 22°C in darkness. Slides were washed in ddH2O for 2 minutes, counterstained in Mayer’s hematoxylin counterstain, and dehydrated. A coverslip was applied, and each slide was examined for labeling. Slides for immunohistochemical evaluation received an antigen labeling score assigned by a single pathologist (JNH) to subjectively record distribution of BVDV antigen in alpacas. Scoring of labeling distribution was performed on a 5-point scale: 0, negative; 1, single cell or single small group of cells with labeling; 2, clearly positive, mild scattered, or regional labeling of cells; 3, moderate scattered or regional labeling of cells; and 4, widespread coalescing to diffuse labeling of cells. Staining was performed in a single laboratory using identical protocols and equipment, and standardized control panels of positive skin tissues with varying levels of antigen staining intensity were included to ensure consistent staining across the platform. Bovine skin from a positive PI BVDV calf was used for the positive control and a BVDV negative ear notch for the negative control. Positive controls were placed at multiple locations throughout the run to ensure staining consistency. Irrelevant slides were performed using a negative mouse antibody (Ventana Laboratories, Tucson, AZ). Alpaca A0 was negative for BVDV, and the same IHC procedure using primary antibody, anti-BVDV monoclonal antibody 15C5 15 directed against Erns/gp 48 was performed in all tissues similar to the positive PI alpacas in this study. Animals were processed as they were presented to the study in the years 2006–2007. Mean system antigen scores were calculated across all tissues from the indicated system collected from each BVDV PI animal, and then the tissue means were averaged to provide a mean antigen labeling score for each system. The system average reported here was then calculated from an average of the 10 alpacas or 5 calves for each system. Cellular localization of BVDV antigen in calves was recorded but being well documented elsewhere6,17,29,32 will only be described when distinct from findings in the alpacas.
Preparation of Nucleic Acid From Tissue
Samples from 2 calves and 8 alpacas were obtained from frozen thymus using a 5-mm punch biopsy tool. The tissue from each animal was weighed, and 110 mg was collected into a vial containing ceramic beads (Roche Diagnostics, Indianapolis, IN) and 1 ml of sterile phosphate-buffered saline. Tissue suspensions were homogenized (Bertin Technologies, Montigny-le-Bretonneux, France) for 30 seconds at 6500 rpm. DNA and RNA were co-eluted from 50 μl of the homogenate using the MagMax 96 Viral RNA Isolation Kit (Ambion, Grand Island, NY), per manufacturer’s protocol. Duplicate extractions were performed, and the RNA was measured on a fluorometer. A housekeeping gene, 18 s, was monitored to ensure that equivalent RNA was tested in each well for each alpaca or calf tissue. ATCC-524 New York-1 BVDV (lot number 142 0301) with a viral concentration of 106.75 TCID50 per 1.0 ml was extracted as described for the samples. Two independent 10-fold serial dilution series were tested in duplicate and analyzed concurrently with the samples. The 10-fold dilutions were averaged to form 1 standard curve, and regression analysis was used to determine a quantitative PCR–derived titer for each alpaca thymus.
Quantitative Real-Time Polymerase Chain Reaction
Previously reported primers and probes 24 directed to the 5′ untranslated region of BVDV were used at concentrations of 0.16 μM (primers) and 0.1 μM (probe). As an endogenous control, 18 S ribosomal RNA was coamplified using published primers and probe 24 at concentrations of 0.16 μM and 0.1 μM (probe). Both sets of primers were combined in a 25-μl PCR reaction mix containing 1X Multiplex master mix and 1X Multiplex RT-PCR enzyme (Ambion, Grand Island, NY) and 5 μl of sample or control. An Applied Biosystems 7500 Fast Real-Time PCR instrument was used in standard mode with the following parameters: reverse transcription at 50°C for 10 minutes; activation of polymerase at 95°C for 10 minutes; amplification for 40 cycles of 95°C for 15 seconds and 60°C for 1 minute.
Statistical Analyses
For each organ system, mean antigen labeling scores were compared between alpacas and the calves using t-tests and statistical analysis software (SAS, Cary, NC).
Results
Macroscopic Examination
Lesions observed in individual alpacas are summarized in the supplemental data (to view online supplement, please go to http://vet.sagepub.com/supplemental). Notable gross lesions included an enlarged liver with multifocal necrosis (A1); dark, firm, and edematous lungs with white exudates present in bronchioles (A2); enlarged tonsils with abundant white to green exudates (A5); and growth retardation with serous atrophy of fat (A7). Seven of the 10 PI alpacas had thymic atrophy as determined by decreased volume. Gross lesions were not observed in the PI calves examined.
Microscopic Examination
Alpaca A7 had unique microscopic ocular lesions that included a subcapsular and hypermature cataract with multifocal mineralization and liquefied lens protein (Fig. 1). The iris was focally thin, which is consistent with an iris coloboma and anterior synechiae (Fig. 1). There was fibrovascular tissue consistent with a cyclitic membrane along the posterior surface of the lens, to which the detached, disorganized, and gliotic retina was attached (Figs. 2, 3). Immunolabeling for BVDV antigen was detected in the disorganized retina (Fig. 4).
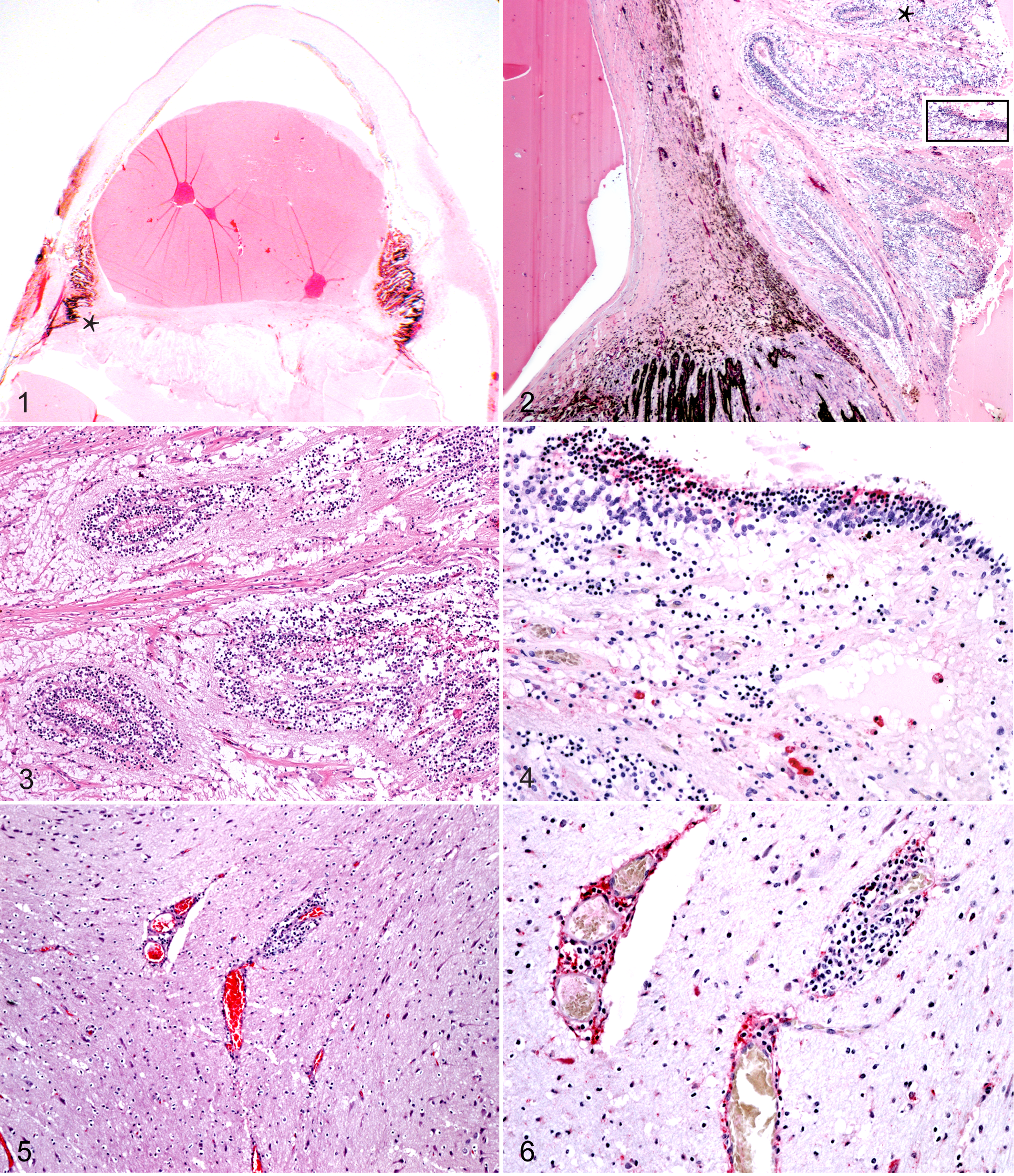
Eye; alpaca A7. Sub gross of eye. The iris is compressed against Descemet’s membrane
by a liquefied lens with a subcapsular and hypermature cataract. A band of fibrous
tissue separates the lens from the detached, disorganized, and thickened retina at the
level of the ciliary body.
In alpaca A1, multifocal scattered areas of hepatocellular coagulative necrosis with multifocal basophilic dystrophic mineralization and severe hepatic fibrosis were evident. A marked diffuse pleocellular inflammatory infiltrate composed of neutrophils, lymphocytes, and macrophages obscured most of the normal hepatic architecture. Alpaca A9 had multifocal random suppurative hepatitis.
The chronic bronchointerstitial pneumonia in A3 consisted of a diffuse infiltrate of macrophages that thickened alveolar septa and often filled alveolar lumina. Edema and fibrin were present in alveolar septa and lumina. Prominent lymphocytic cuffs with mild lymphocytolysis surrounded hyperplastic bronchi and bronchioles. Moderate numbers of neutrophils and cellular debris were evident in airways, and leukocyte exocytosis was prominent. Tonsilar tissue in A5 was abscessed, which precluded complete histological evaluation. Multifocal perivascular nonsuppurative meningoencephalitis (Fig. 5) was present in A3. In the alpaca (A3) with meningoencephalitis, immunolabeling for BVDV antigen was detected in scattered lymphocytes and occasional endothelial cells within the perivascular cuff (Fig. 6). Other histological lesions seen in individual PI alpacas included diffuse fungal glossitis and esophagitis (A4), lymphoplasmacytic enteritis and multifocal erosive rhinitis (A6), and erosive fibrinosuppurative glossitis (A7).
The small intestine of each of the calves had a mild diffuse lymphocytic and histiocytic infiltrate. Coccidia were noted in epithelia of calves C1 and C2. Calves C3 and C5 had thymic atrophy (involution).
Immunohistochemical Analyses
Means and ranges of distribution of BVDV antigen labeling in tissues from alpacas and calves are presented in Table 1. Alpaca A0 was a negative control and had no detectable BVDV antigen labeling in any tissues, and so it was excluded from Table 1 and further analysis. Tissues from the PI alpacas were characterized by widely distributed BVDV antigen labeling. Skin had the highest tissue antigen labeling in alpacas and calves, and 100% of PI animals were positive. Other tissues with consistently high labeling scores in alpacas were C3 compartment of stomach, cerebrum, parotid and submandibular salivary glands, thyroid, thymus, esophagus, nasal turbinate, and lung tissue. Bone marrow, trachea, kidney, and retina were inconsistently stained and had low scores (≤2.0) in the alpacas.
BVDV antigen labeling scores were consistently lower in alpacas compared with calves for comparable tissues. Lower mean BVDV antigen scores in alpacas compared with calves (Table 2, Fig. 7) were found in gastrointestinal tract (P < .0001); lymphoid tissue (P < .0001), including mesenteric lymph node; bone marrow (P < .0001), which was infrequently and minimally labeled in alpacas; urinary system (P < .0014); respiratory system (P < .0002), including lung; and nervous tissue (P ≤ .05). The exceptions were pancreas and skin, which had equivalent scores in both species, and alpaca stomach compartment C3, which scored higher than calf abomasum.
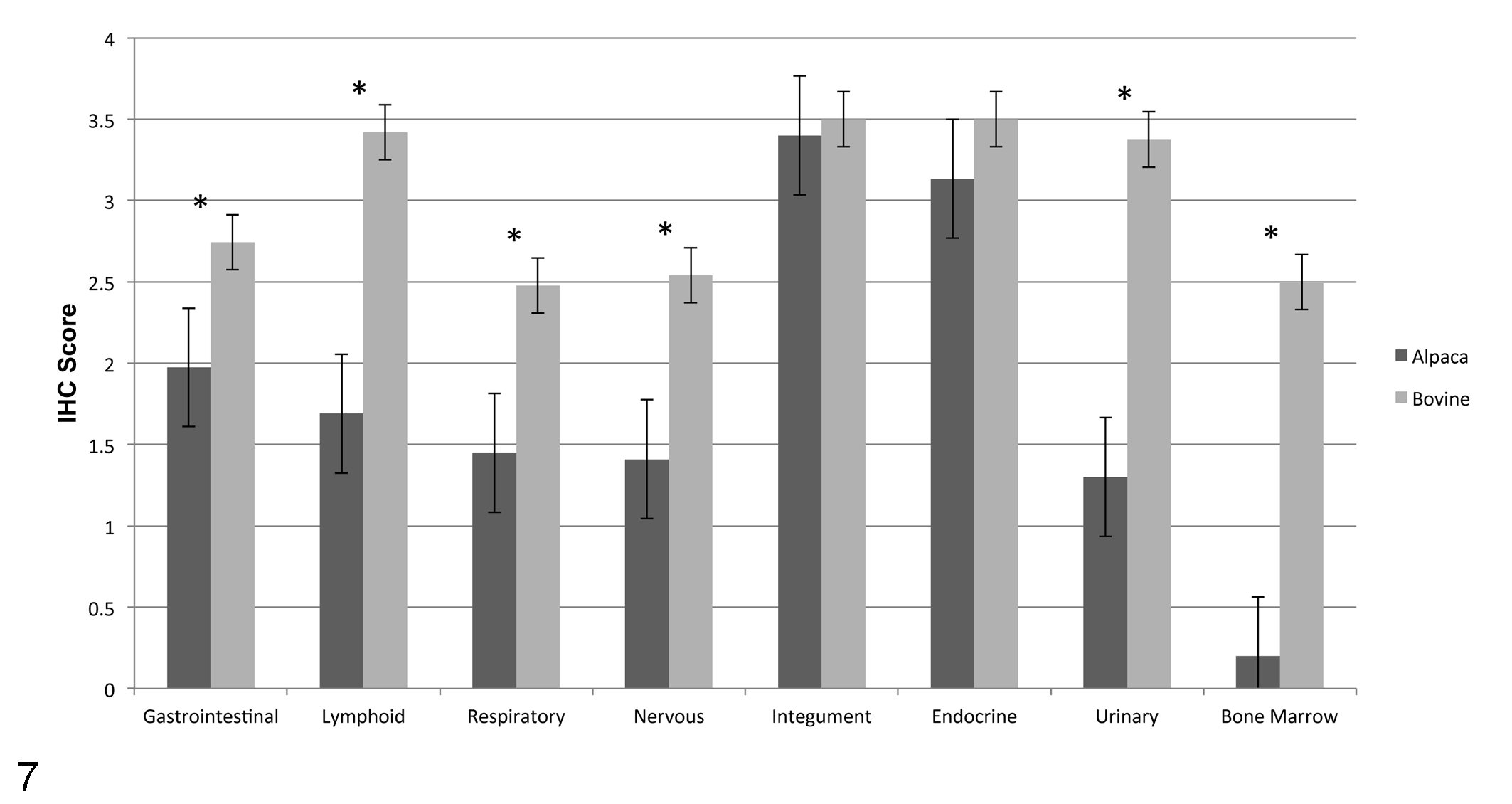
Bar graph representing the mean ± SEM comparing differences in BVDV systemic antigen labeling between persistently infected alpacas and persistently infected calves. Differences in distribution of BVDV antigen labeling were statistically significant in the gastrointestinal, lymphoid, respiratory, nervous, and urinary systems and in bone marrow (P ≤ .05; asterisks).
BVDV antigen was prominently localized in nervous tissue, vascular tunica media, macrophages and epithelium of endocrine organs, salivary glands, and pancreas. Scattered small vessels had prominent antigen labeling in the smooth muscle of the tunica media in most alpaca tissues. Less consistent labeling of vascular adventitial spindle cells and occasionally endothelial cells was evident. In alpaca salivary glands, labeling was most often in duct epithelium. Alpaca pancreatic BVDV antigen labeling was present in islets, stroma, and acinar cells. In the adrenal gland, antigen was most common in the zona fasciculata, although all cell types of the alpaca adrenal gland contained antigen. Thyroid follicular epithelial cell labeling of alpacas was diffuse and cytoplasmic.
BVDV antigen was distributed in alpaca esophagus and compartmental stomach as follows: scattered squamous epithelial cells of the esophagus, multifocal prominent labeling of chief cells, cardiac gland epithelium, and gastric pit epithelium of the forestomach. Staining was also prominent in scattered superficial submucosal macrophages and fibroblasts throughout the entire gastrointestinal tract. Prominent multifocal labeling of myenteric plexus neurons was evident. Limited antigen labeling was detected in parietal cells, epithelium of duodenal Brunner’s glands, crypt epithelium, enterocytes of small and large intestine, and macrophages of the gut associated lymphoid tissue. Alpacas lacked antigen labeling in sites that were prominent in calves, including lymphocytes, macrophages, and dendritic cells of the Peyer’s patch and in gastrointestinal associated lymphoid tissue. Scattered antigen labeling was observed in macrophages of the intestinal mucosa, in myocytes of the muscularis mucosa, and occasionally within crypt epithelial cells.
Alpaca lymphoid system labeling of lymphocytes was inconsistent. Nine of 10 alpacas had no labeling in bone marrow compared with calves (Figs. 8, 9). BVDV antigen labeling in the thymus occurred in scattered lymphocytes and epithelial cells. Lymph node antigen labeling was prominent in medullary macrophages of alpacas and calves (Figs. 10, 11). Occasional lymphocytes, macrophages, and dendritic cells of germinal centers contained antigen, which were identified by morphological features and location. Endothelial cells forming lymphatic sinuses were also labeled. BVDV antigen presence in the spleen occurred prominently in macrophages, although faint labeling was present in lymphocytes and endothelial cells. Tonsilar surface and crypt squamous epithelium stained prominently, and rare lymphocytes also stained.
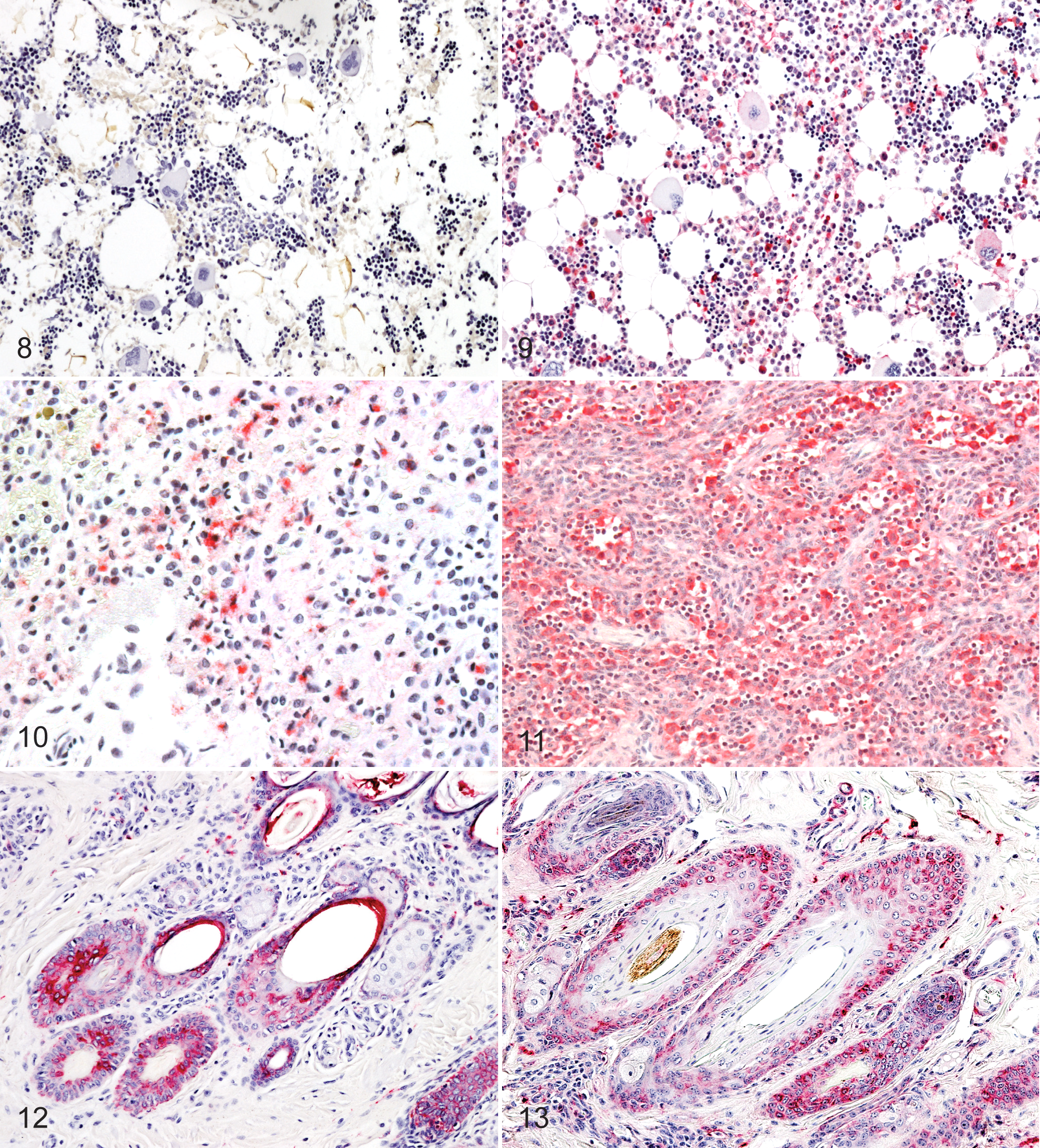
Bone marrow; alpaca A2. BVDV antigen labeling was not present in any of the
hematopoietic cell lines. Avidin–alkaline phosphatase IHC, hematoxylin counterstain.
In the skin, numerous foci of keratinocyte labeling were seen in all layers of the epidermis, except stratum corneum. Multifocal antigen labeling was present in keratinocytes of most hair follicles in alpacas and calves (Figs. 12, 13). Distribution of BVDV antigen was present in the suprabulb region, isthmus, and infundibulum of the hair follicles. Scattered histiocytes, dendritic cells, and fibroblasts in the dermis were also labeled.
In the nervous tissue, antigen labeling was most prominent in the cerebrum and cerebellum, whereas antigen was inconsistently found in the brainstem. Antigen was most localized in neurons, endothelial, and microglial cells. Labeling of the alpaca lumbar spinal cord was variable, faint, and scattered in both white and gray matter. Labeling was most prominent in neurons and less so in scattered microglial cells. Multifocal labeling of choroid plexus epithelium and meningeal endothelial cells and fibroblasts was noted. Three alpacas had antigen localized in the Schwann cells of the sciatic nerve, and 3 alpacas had scattered Schwann cell labeling in optic nerve.
In the urinary tract, marked antigen labeling was present in renal proximal and medullary tubular epithelial cells. Four alpacas had single or small group of cells staining in the urinary bladder.
In the lung, labeling was observed in pneumocytes, alveolar macrophages, and bronchiolar epithelium. Occasional labeling of bronchial and tracheal chondrocytes was detected. Labeling was present in rods, cones, and ganglion cell layers of the retina. Focal moderate labeling of ciliary body epithelium was present. Ovarian germinal epithelium often labeled, as did granulosa cells, thecal cells, and endometrial glands. Moderate labeling was evident in scattered Sertoli cells and prostatic epithelium. Four alpacas had mild BVDV antigen labeling in cardiac but not skeletal muscle.
Quantitative Real-Time Polymerase Chain Reaction
Viral loads calculated using quantitative real-time PCR were comparable among the 8 alpacas and 2 calves examined. The 8 alpacas had BVDV viral load in thymus ranging from 1 × 104.8 to 1 × 106.3 TCID50/ml, whereas the 2 calves had thymic BVDV viral load of 1 × 105.1 TCID50/ml and 1 × 106 TCID50/ml.
Discussion
BVDV antigen was widely distributed in tissues of alpacas PI with BVDV type 1. There are distinct differences, however: Unlike findings in cattle, lymphoid tissues and bone marrow exhibited limited viral labeling in PI alpacas and there was significantly less extensive antigen distribution detected in alpaca gastrointestinal and nervous tissues. Exceptions to this occurred in skin and endocrine tissues, in which BVDV distribution was similar between the 2 species. Widespread antigen distribution occurred in respiratory, digestive, and cutaneous sites, and these may serve as sources of virus for transmission; thus, PI alpacas appear well suited to serve as a reservoir for BVDV transmission. The uniform prominent staining of cutaneous sites implies that skin biopsy can be a useful screening sample for PI alpacas, as it is in cattle.
We report the occurrence of ocular dysplasia in 1 PI alpaca associated with BVDV infection; BVDV antigen was localized in the lesion by IHC. Retinal dysplasia and cerebellar hypoplasia have been reported in other BVDV infected ruminants. 30 This is the first report of congenital ocular lesions in a PI alpaca.
Clinically, alpacas PI with BVDV are reported to develop ill-thrift, diarrhea, upper respiratory disease, and inappetance.3,12,18,19,26 A single alpaca in our study had a history of growth retardation, whereas the others had lesions of concurrent infection including pneumonia, hepatitis, glossitis, tonsillitis, and meningoencephalitis. It would appear that PI status affects susceptibility to secondary infections as it does in cattle through immunosuppression. Cattle in this study were identified in screening programs to detect and remove carriers and thus were less likely to have secondary lesions compared with alpacas tested for diagnosis of illness. Thymic atrophy noted in alpacas, characterized grossly by decreased size and microscopically by replacement with adipose tissue, may represent age-related involution or stress, as lymphoid depletion did not occur in PI alpacas.
There were profound differences in the bone marrow between the 2 species, with infrequent or minimal antigen labeling in alpacas compared with calves. Antigen labeling in bovine progenitor cells, including megakaryocytes and myeloid cells, has been reported and may play a prominent role in pathogenesis of BVDV infection in cattle.25,32
A recent study examining BVDV antigen labeling in bovine fetal brains after experimental infection found labeling in neurons, pericytes, perivascular macrophages, and glial cells consistent with astrocytes. 29 BVDV antigen is frequently present in neurons of the cerebral cortex and hippocampus of cattle, 17 which is consistent with our observations of distribution in PI alpacas. Antigen labeling in reproductive organs including, testis, ovaries, prostate, and uterus of PI alpacas was similar to the findings in cattle.8,14,20 This suggests that PI status may affect fertility and that the virus might be spread through reproductive tract fluids during breeding.
In summary, this study details comparable tissue distribution of BVDV antigen labeling in alpacas and calves PI with BVDV. We found increased labeling in the brain and occasionally the spinal cord in this population of PI alpacas compared with previous reports. 12 The study is also the first to report the presence of congenital ocular disease in an alpaca PI with BVDV as well as limited antigen labeling in the bone marrow of PI alpacas. The data suggest that the sequelae of enhanced secondary infection and ill thrift and the importance of PI alpacas as reservoirs of infection are comparable to those of PI cattle. Skin is likely to be a useful diagnostic sample for screening live animals in addition to blood. Skin, C3 compartment of the stomach, cerebrum, parotid and submandibular salivary glands, thyroid, thymus, esophagus, nasal turbinate, and lung tissue stained consistently for BVDV antigen and will be useful to detect PI alpacas at necropsy.
Footnotes
Acknowledgements
We thank Dr. Ed Dubovi for identification of the PI alpacas, Dr. Stephen Ferree for performing necropsies on 2 of the calves, and Mal Hoover for assistance on revisions to the images.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University of Nebraska Agricultural Research Division [grant no. NEB 14-126] from funds provided through Animal Health, USDA and by the Mid-Atlantic Alpaca Association through the Alpaca Research Foundation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
