Abstract
Formalin fixation and paraffin embedding (FFPE) is the standard method for tissue storage in histopathology. However, FFPE has disadvantages in terms of user health, environment, and nucleic acid integrity. Weigners fixative has been suggested as an alternative for embalming cadavers in human and veterinary anatomy. The present study tested the applicability of Weigners for histology and immunohistochemistry and the preservation of nucleic acids. To this end, a set of organs was fixed for 2 days and up to 6 months in Weigners (WFPE) or formalin. WFPE tissues from the skin, brain, lymphatic tissues, liver, and muscle had good morphologic preservation, comparable to formalin fixation. The quality of kidney and lung samples was inferior to FFPE material due to less accentuated nuclear staining and retention of proteinaceous interstitial fluids. Azan, Turnbull blue, toluidin, and immunohistochemical stainings for CD79a, cytokeratin, vimentin, and von Willebrand factor led to comparable results with both fixates. Of note, immunohistochemical detection of CD3 was possible after 6 months in WFPE but not in FFPE tissues. mRNA, miRNA, and DNA from WFPE tissues had superior quality and allowed for amplification of miRNA, 400-bp-long mRNA, and 1000-bp-long DNA fragments after 6 months of fixation in WFPE. In summary, Weigners fixative is a nonhazardous alternative to formalin, which provides a good morphologic preservation of most organs, a similar sensitivity for protein detection, and a superior preservation of nucleic acids. Weigners may therefore be a promising alternative to cryopreservation and may be embraced by people affected by formalin allergies.
Formalin fixation and paraffin embedding (FFPE) is the most common method for long-term conservation of tissue samples in biomedical research and diagnostics. It preserves the morphological tissue architecture and allows for protein analysis by a wide variety of antibodies for FFPE tissues by immunohistochemistry. Formalin is, however, harmful to the environment and has potential toxic, carcinogenic, allergenic, and teratogenic effects on human health.6,18,19 In addition, formalin has other negative side effects that limit its use in molecular biology. Contemporary biomedical research and diagnostics depend on the quantification and sequence analysis of nucleic acids (ie, DNA, microRNA [miRNA], and messenger RNA [mRNA]). FFPE tissue samples are quite challenging in terms of preservation of nucleic acids. In particular, long-term storage may lead to severe degradation of nucleic acids and failure to amplify DNA and RNA fragments larger than approximately 200 bp.12,22,24
Several other fixatives have been tested as nonhazardous and nucleic-preserving alternatives, but none of them has displaced formalin as the standard fixative due to diverse reasons (reviewed in Klopfleisch et al 12 ). Recently, Weigners fixative (WF), a nitrite pickling salt solution with ethanol and Pluriol, has been introduced as an alternative to formalin for embalming of cadavers. 7 Weigners has not been safety tested in its present formulation, but all compounds have been tested separately. Pickling salt is recommended by the US Department of Agriculture (USDA) for preservation of food; 21 ethanol, although neurotoxic, is regularly consumed as an oral stimulant; and Pluriol has been tested as nontoxic and not irritating after a single ingestion or application. 2 Weigners offers a good preservation of whole cadavers for 12 months without macroscopical signs of autolysis. Microbial culturing has revealed significant reduction in bacterial load, similar to cadavers embalmed with formaldehyde. However, histomorphological preservation, the application of immunohistochemistry, and the preservation of nucleic acids, DNA, miRNA, and mRNA have not been conclusively tested in Weigners fixative paraffin-embedded (WFPE) tissues so far.
In the present study, tissues were fixed in formalin and Weigners for various periods for up to 6 months. The morphologic preservation, applicability of FFPE-adjusted antibodies, and integrity of DNA, miRNA, and mRNA were tested in FFPE and WFPE tissues. The results show that Weigners preserves morphological details in most tissues similar to FFPE and, moreover, allows similarly sensitive protein detection by immunohistochemistry assays with antibodies applicable to FFPE. In addition, the preservation of nucleic acids is significantly superior in WFPE tissues when compared with FFPE tissue samples.
Material and Methods
Tissues and Fixation
Several samples from brain, lung, liver, kidney, spleen, heart, skin, skeletal muscle, and jejunum from one 4-year-old medium-size mixed breed (dog No. 1) and a 9-year-old Golden Retriever (dog No. 2), which were euthanized due to trauma, and one 8-year-old cat, which was euthanized due to a recurring fibrosarcoma, were fixed in neutrally buffered 4% formaldehyde or Weigners fixative (23% pickling salt, Ehlert GmbH & Co. KG, Gütersloh, Germany; 30% ethanol, 20% Pluriol E400 BASF, Ludwigshafen, Germany) approximately 8 hours postmortem. Tissue samples were cut into pieces with an approximate side length of 1 cm, and consecutive pieces were embalmed in vessels of similar volume of either Weigners or formalin. Tissue samples were removed from the fixatives 2 days, 2 weeks, 4 weeks, 8 weeks, 3 months, and 6 months (only dog No. 1) later and routinely paraffin embedded.
The animals from which tissues were taken were submitted by a referring small animal clinic for standard necropsy. Inclusion in the study therefore had no influence on the common diagnostic or therapeutic measures usually applied on animals and did not induce any additional treatments or pain or discomfort-inducing manipulations during the entire study. The study was therefore performed in concordance with the national and European law and ethical standards.
Histology and Immunohistochemistry
Tissue sections (2 μm) of the paraffin-embedded tissue samples were cut and stained with hematoxylin and eosin (HE). Briefly, FFPE and WFPE tissue samples were incubated for 5 minutes in hematoxylin and 1 minute in eosin (staining protocol A). A second staining protocol (staining protocol B) with a shorter incubation time of 10 seconds in eosin and a longer incubation of 12 minutes in hematoxylin was used to stain WFPE brain, liver, spleen, and skin due to their general higher affinity to eosin staining.
Azan, toluidin blue, Periodic-acid shiff (PAS), Masson’s trichrome, and Turnbull blue stainings were performed according to standard protocols. CD3, CD79a, vimentin, cytokeratin, and von Willebrand factor protein immunostainings were performed using antibodies selected for application in FFPE tissues. Primary antibodies, their dilution, and required antigen retrieval are specified in Table 1. Briefly, protein expression was immunohistochemically detected using the ABC method for all antibodies. Primary antibodies were diluted in Tris-buffered saline (TBS, 50 mM, pH 7.6) and incubated at 4°C overnight after a blocking step with 50% goat serum in TBS (30 minutes at room temperature). 8 All antibodies required either antigen retrieval by heating in citrate buffer (10 mM, pH 6.0) for 10 minutes in a microwave oven (600 W) or proteinase treatment (0.1 g/100 ml phosphate-buffered saline [PBS]; BioChemica, AppliChem, Darmstadt, Germany) for 20 minutes at 37°C. Secondary antibodies were also diluted in TBS and incubated for 1 hour at room temperature. Diaminobenzidine tetrahydrochloride (Sigma Aldrich, St. Louis, MO) was used as chromogen, and slides were counterstained with hematoxylin (Merck GmbH, Darmstadt, Germany). For negative controls, sections were incubated without a primary antibody or with an irrelevant rabbit antimurine ClCa antiserum.
Antibodies Used for the Detection of Specific Proteins in Formalin- and Weigners-Fixed Tissues by Immunohistochemistry.
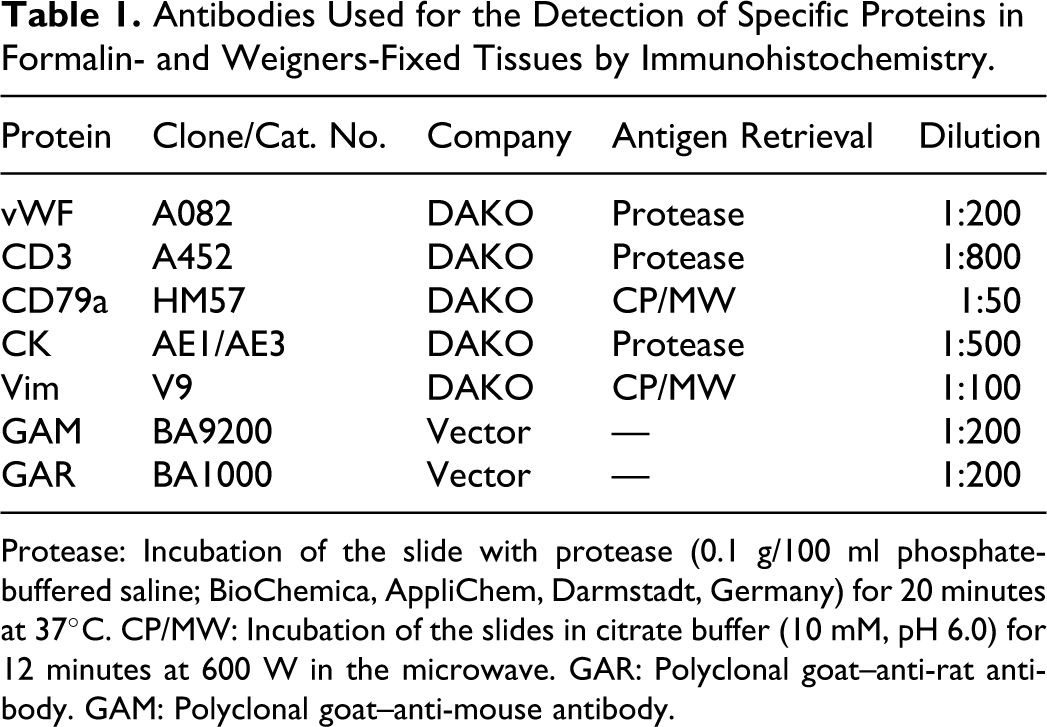
Protease: Incubation of the slide with protease (0.1 g/100 ml phosphate-buffered saline; BioChemica, AppliChem, Darmstadt, Germany) for 20 minutes at 37°C. CP/MW: Incubation of the slides in citrate buffer (10 mM, pH 6.0) for 12 minutes at 600 W in the microwave. GAR: Polyclonal goat–anti-rat antibody. GAM: Polyclonal goat–anti-mouse antibody.
DNA, miRNA, and mRNA Extraction From Formalin- and Weigners-Fixed Tissue Samples
DNA was extracted from FFPE and WFPE kidney samples of dog No. 1 as described before. 22 Kidney was selected because of its commonly high content of well-preserved nucleic acids.
In short: 20 to 40 paraffin sections of 2 μm were collected into a microcentrifuge tube. Samples were deparaffinized with xylene and resuspended in 300 μl Cell Lysis Solution (Gentra Puregene Tissue Kit; Qiagen, Hilden, Germany). The samples were heat pretreated with 65°C for 45 minutes followed by 98°C for 15 minutes. This was followed by proteinase K digestion for 48 hours, 3 hours, or 0 hours and RNase A digestion for 15 minutes.
Protein Precipitation Solution (Gentra Puregene Tissue Kit; Qiagen) was used to eliminate protein contamination, and DNA was precipitated by addition of 1 volume 99% isopropanol.
DNA pellets were washed twice in 70% ethanol, air-dried, and resuspended in 25 μl DNA Hydratation Solution (Gentra Puregene Tissue Kit; Qiagen). DNA concentration was determined spectrophotometrically (Nanodrop; Peqlab, Erlangen, Germany) and adjusted to 100 ng/μl by adding 5 mM Tris buffer, pH 7.4. Quality of genomic DNA was visualized by gel electrophoresis on 1% agarose gels.
Total RNA was also extracted from 20 to 40 FFPE and WFPE paraffin sections and deparaffinized with xylene and resuspended in 300 μl Cell Lysis Solution (Nucleospin FFPE RNA Kit, Macherey Nagel, Düren, Germany). This was followed by proteinase K digestion for 3 hours and incubation at 94°C for 10 minutes. Protein-DNA precipitation solution (4 M NaCl, 16 nM NaC6H5O7, 32 mM C6H8O7, pH 4.0) was used to eliminate protein contamination and DNA. RNA was precipitated by addition of 1 volume 99% isopropanol; RNA pellets were washed twice in 70% ethanol, air-dried, and resuspended in 30 μl water. RNA concentration was determined spectrophotometrically (Nanodrop; Peqlab) and adjusted to 100 ng/μl by adding aqua bidest. RNA quality extracted was controlled using the BioAnalyzer (Agilent Technologies, Santa Clara, CA).
Reverse Transcription of miRNA and mRNA
mRNA was reverse transcribed with the iScript cDNA Sythesis Kit (Bio-Rad Laboratories, Hercules, CA) according to the manufacturer’s instructions. miRNA complementary DNA (cDNA) synthesis required addition of a poly(A)-tail, which was added to the 3′ end of the miRNA (Poly(A) Polymerase Tailing Kit; EPICENTRE Biotechnologies, Madison, WI) according to the manufacturer’s instructions. An oligo-dT-adaptor was annealed to the poly(A) tail by incubating 5 minutes at 60°C and 2 minutes at 21°C. 3 miRNA was subsequently reverse transcribed with the RevertAidH Minus First Strand cDNA Synthesis Kit (Fermentas, Glen Burnie, MD) according to the manufacturer’s instructions.
Polymerase Chain Reaction Amplification of Genomic DNA and cDNA
Primers and polymerase chain reaction (PCR) conditions for amplification of ATP5B, RPL32, MIR29b, and the canine androgen receptor (AR) were applied as described before.9–11,22 PCR master mix consisted of 2.0 mM MgCl2, 0.2 mM of each dNTP, 0.2 μM of each primer, and 0.024 U/μl Taq DNA polymerase. Cycling conditions included initial denaturation at 94°C for 5 minutes, melting of DNA at 92°C for 30 seconds, annealing for 15 seconds at the respective annealing temperature (Table 2), amplification at 72°C for 80 seconds, and a final amplification at 72°C for 5 minutes. Primer sequences and annealing temperatures are listed in Table 2.
Primers Used for Detection of mRNA, miRNA, and Genomic DNA Fragments.

Results
Histology and Special Stains
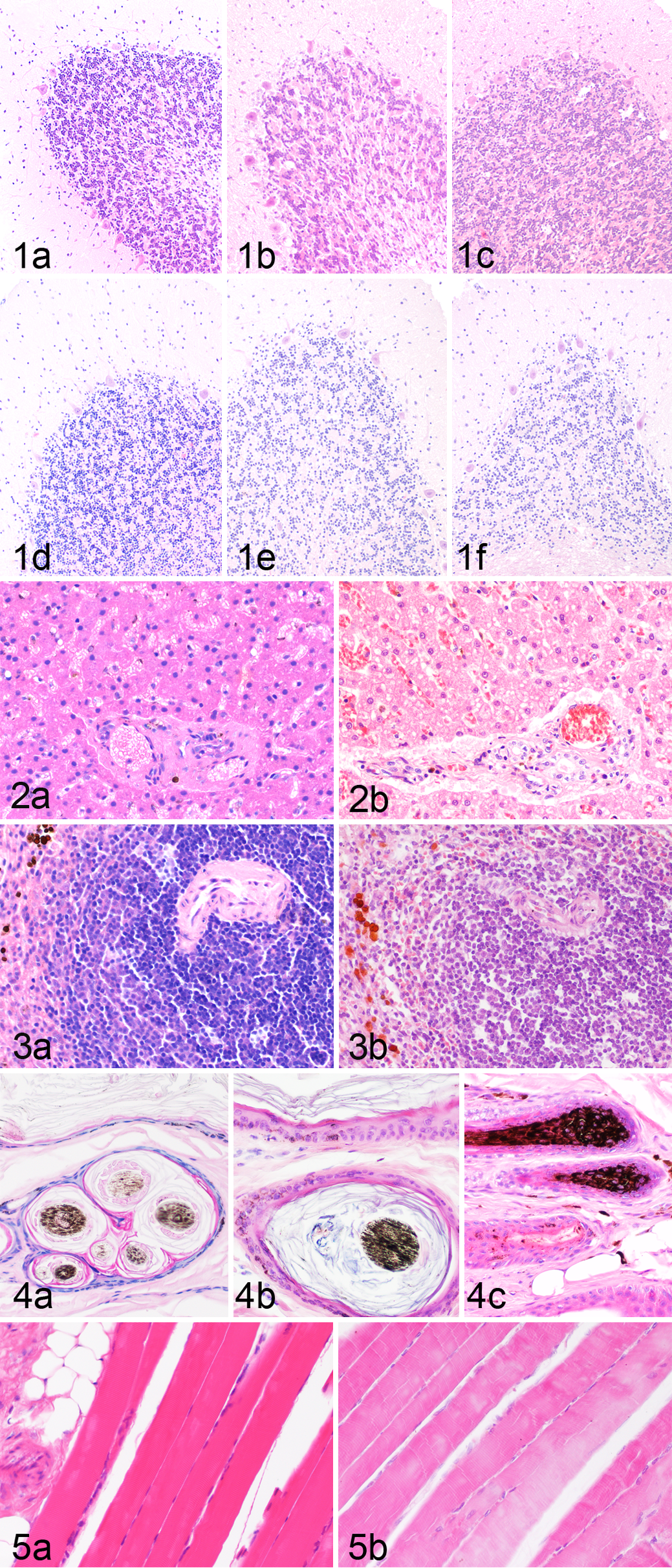
Histology of WFPE- and FFPE-fixed tissues was compared for all tissues and all time points. Generally, WFPE-fixed tissues showed less shrinkage of the cytoplasm and less coarse nuclear DNA staining when compared with FFPE. The direct comparison of HE-stained WFPE- and FFPE-fixed tissues led to variable results in the different organs analyzed (Table 3, Figs. 1–5). Short-term WFPE fixation of brain tissues led to slightly superior morphologic preservation with better preservation of cellular detail and stainability when compared with FFPE brain tissues (Table 3, Fig. 1a–f). However, after 2 weeks of fixation, both fixation methods led to similar results, and at 4 weeks, the effect reversed. At this time point, FFPE brain showed superior cellular detail and neuropil preservation when compared with WFPE brain. Liver fixation in Weigners led to similar morphologic structure when compared with FFPE liver samples (Fig. 2a, b). Nevertheless, as with most other organs, WFPE-fixed liver samples stained more intensive with eosin. Similarly, WFPE-fixed spleen (Fig. 3a, b) and skin (Fig. 4a–c) showed a morphologic perseveration similar to FFPE with slightly less accentuated nuclear details. Morphology of WFPE skeletal muscle was better preserved than FFPE muscle. WFPE skeletal muscle was characterized by constant and diffuse retention of striation and strong stainability (Fig. 5a). In addition, typical artifacts such as hypercontracted myofibers, myofiber cross fissures, and fragmentation seen in FFPE sections (Fig. 5b) were not seen. WFPE of myocardium led to similar results. Lung morphology of WFPE-fixed tissue sections was inferior due to the retention of proteinaceous exudates in areas with alveoli when compared with FFPE sections. Similarly, preservation of distal renal tubules was poor in WFPE of all 3 kidneys when compared with FFPE kidneys, whereas glomeruli and the proximal convoluted tubules had an acceptable morphologic preservation.
Morphologic Preservation of Tissues Fixed in Weigners Compared With Formalin-Fixed Tissues.

+, morphologic preservation with Weigners better than formalin fixation; –, morphologic preservation with Weigners less optimal than formalin fixation; =, morphologic preservation similar to formalin fixation. A, hematoxylin and eosin (HE) staining—1 minute eosin, 5 minutes hematoxylin; B, HE staining—10 seconds eosin, 12 minutes hematoxylin.
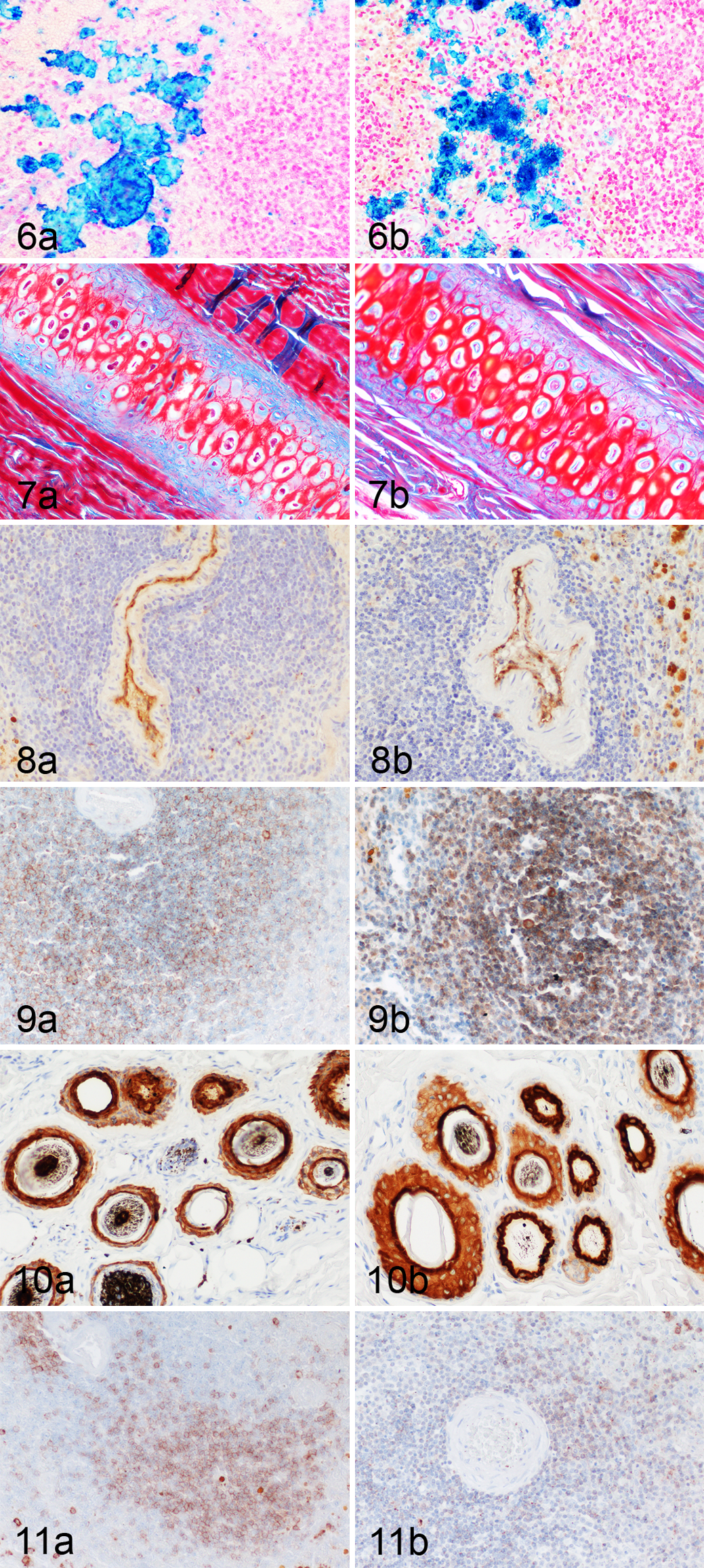
Azan and Turnbull blue staining led to similar results in WFPE and FFPE tissues. Extra- and intracellular Fe2+ ions were robustly stained by Turnbull blue staining in WFPE tissues (Fig. 6a, b). A similar distribution and intensity of stainable elements was observed in WFPE and FFPE sections stained with Azan (Fig. 7c, d). In contrast, WFPE tissues submitted for toluidin blue and Masson’s trichrome staining were severely overstained with the FFPE-adapted protocol, whereas PAS was less sensitive in WFPE than in FFPE tissues.
Immunohistochemistry
Immunohistochemistry with antibodies and protocols adapted to FFPE sections was used for the analysis of protein preservation in WFPE tissues. Protein expression was identified in WFPE tissues for all 5 antibodies: anti–pan-cytokeratin, vimentin, CD3, CD79a, and von Willebrand factor–related protein. Protein expression was identified after fixation for 2 days and 6 months for von Willebrand factor–related protein (Fig. 8a, b), CD79a (Fig. 9a, b), pan-cytokeratin (Fig. 10a, b) and vimentin with comparable intensity in WFPE and FFPE tissues. CD3 staining, however, was decreased in FFPE spleen after 6 months of formalin fixation but not in WFPE spleens after 6 months of Weigners fixation (Fig. 11a, b).
Comparison of DNA Integrity and Amplification From WFPE and FFPE Tissues
Genomic DNA (gDNA) was extracted from WFPE or FFPE kidney sections for all 6 time points following fixation. Three different extraction protocols were used that differed in the time of protease digestion of the dewaxed sections. Proteinase digestion for 48 hours allowed extraction of relatively intact gDNA from WFPE (Fig. 12). In contrast, intact gDNA was only extractable from FFPE fixed for 2 days, whereas later time points led to the accumulation of smaller gDNA fragments (Fig. 12). Duration of proteinase digestion had no influence on the amount of gDNA extracted from WFPE tissue samples (data not shown). In contrast, proteinase digestion of less than 48 hours reduced the amount of gDNA from FFPE tissue samples (data not shown).
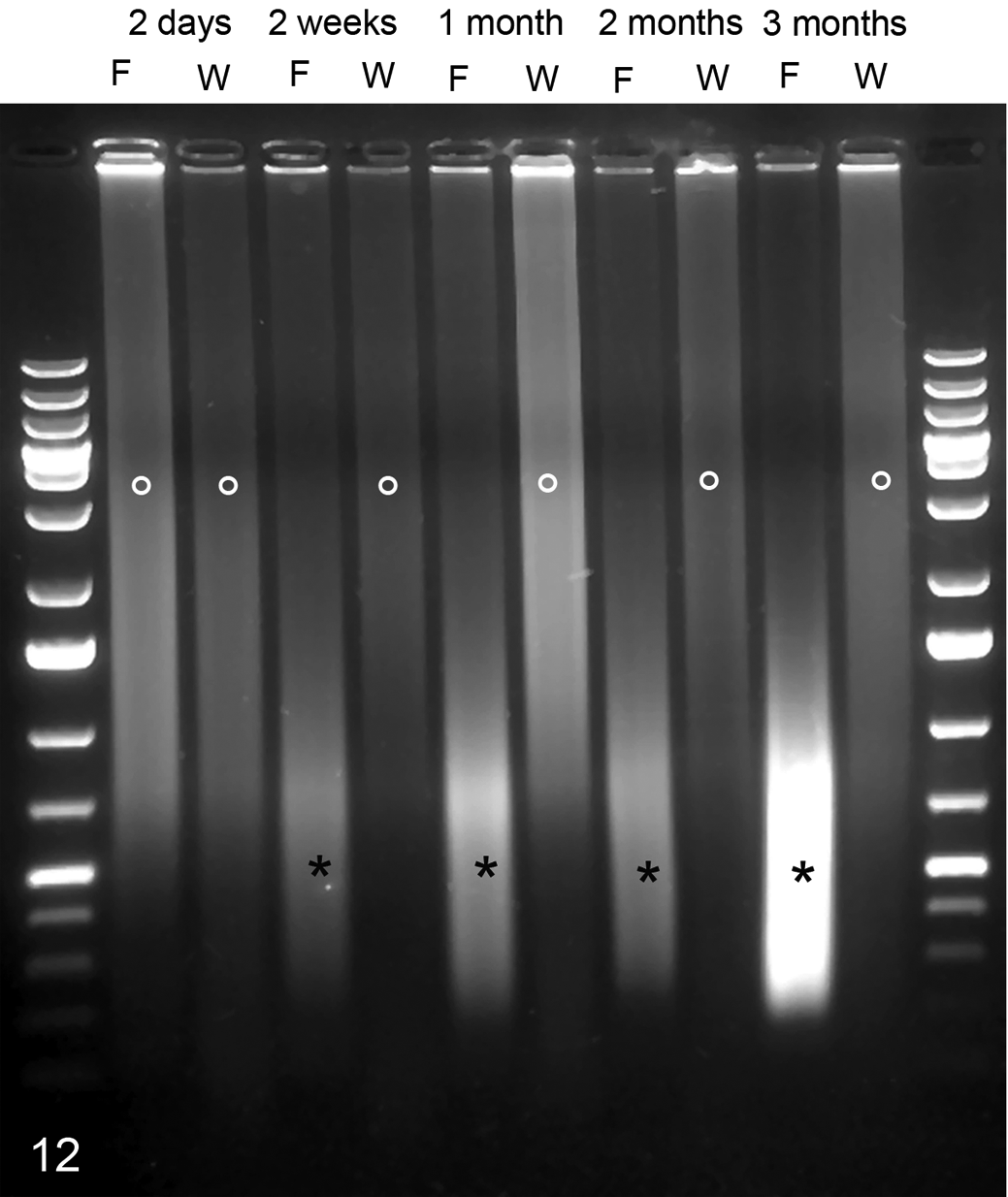
Integrity of genomic DNA after isolation from a kidney tissue sample fixed with Weigners (W) or formalin (F) for 6 months. A typical smear (°), indicative of good preservation of DNA integrity, was observed in tissues fixed for up to 6 months in Weigners but was only observable after 2 days of formalin fixation. Longer fixation with formalin led to progressive fragmentation of DNA into smaller fragments (*).
WFPE but not FFPE fixation allowed the amplification of a 1-kbp fragment of ATP5b (Fig. 13). Of note, prolonged digestion of WFPE tissues had a significant effect on the amount of amplifiable large gDNA fragments. FFPE allowed the amplification of gDNA fragments with a maximum size of 200 bp after up to 3 months of fixation but not after 6 months of fixation. Fragments of 400 bp were amplifiable from WFPE kidney at all time points but never from FFPE tissues.
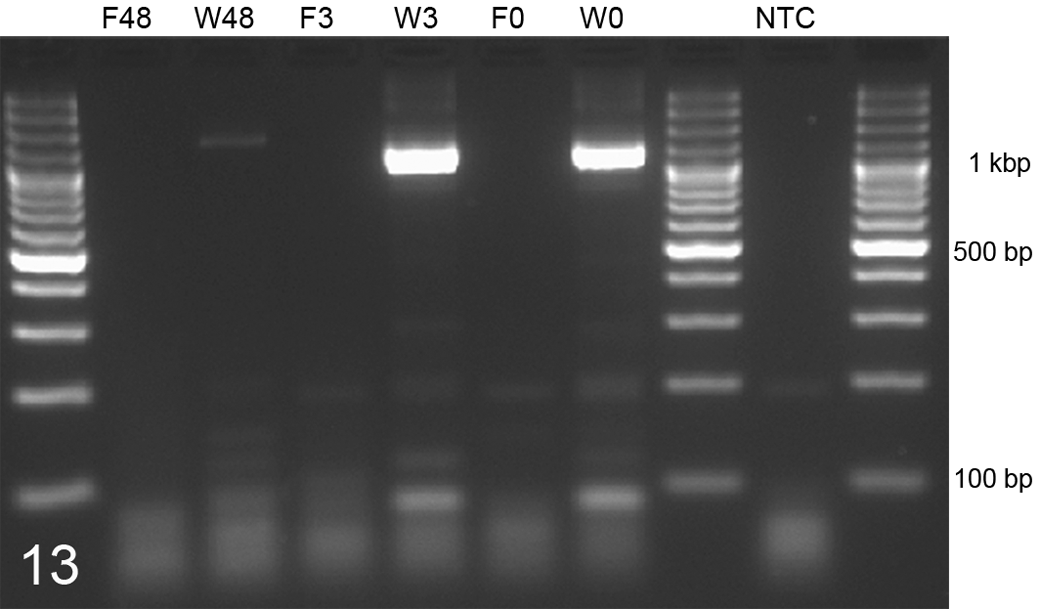
Comparison of maximal amplifiable DNA fragments in kidney tissue samples after 6 months’ fixation in Weigners (W) or formalin (F). Weigners allowed replication of DNA fragments of over 1000 bp after proteinase K digestion for 0 hours (W0), 3 hours (W3), and 48 hours (W48). However, proteinase digestion of 48 hours (W48) reduced the amount of amplifiable fragments in Weigners-fixed tissues. Formalin fixation hampers the amplification of 1000-bp DNA fragments after 0 hours (F0), 3 hours (F3), and 48 hours (F48). Polymerase chain reaction amplification of 1000-bp fragments of ATP5B. NTC, no template control.
Comparison of mRNA and miRNA Amplification From WFPE and FFPE Tissues
Total RNA was extracted from WFPE and FFPE kidney sections. WFPE kidney sections contained considerable amounts of mRNA, which allowed amplification of at least 400-bp-long cDNA fragments at all time points and had a RNA integrity number (RIN) of 8.2 at day 2, 7.5 at 2 weeks, and 7.0 after 3 and 6 months of Weigners fixation (Table 4, Fig. 14a, b). In contrast, mRNA fragments of a maximum of 180 bp were only amplifiable after 2 days and 2 weeks of formalin fixation but not at later time points (Fig. 14a, b). A RIN of 3.5 was measurable only after 2 days of formalin fixation but not at later time points due to severe fragmentation.
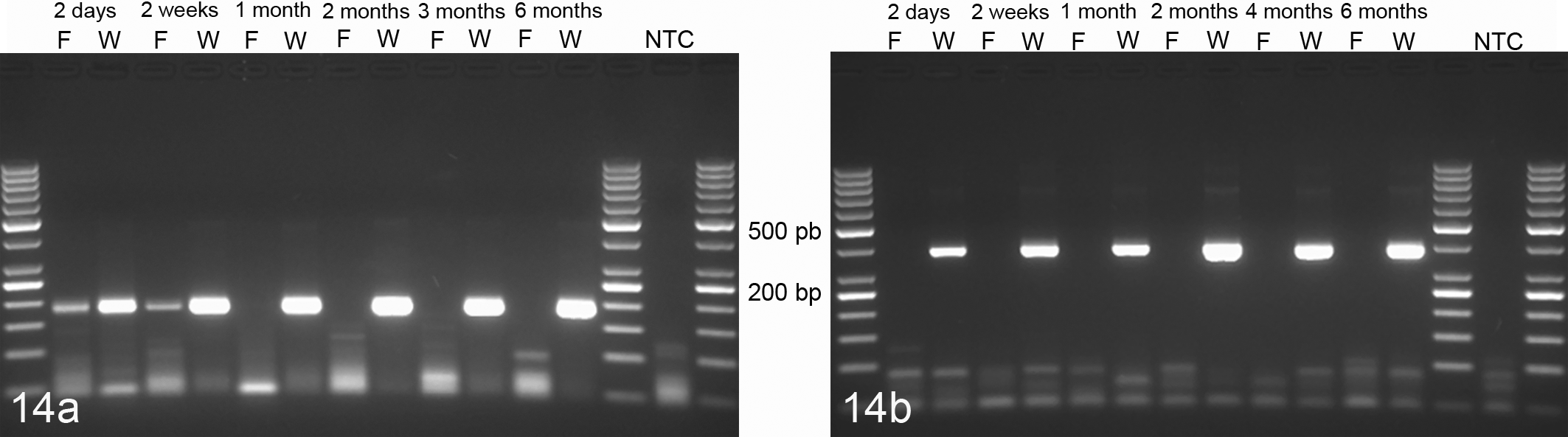
Amplification of transcribed RNA from tissues fixed for 6 months in Weigners (W) or formalin (F). Weigners fixation allowed amplification of 180-bp (a) and 400-bp mRNA fragments (b) for fixation times of up to 6 months. In contrast, formalin fixation allowed the amplification of a 180-bp fragment (a) only after 2 days and 2 weeks of fixation but not at later time points. Polymerase chain reaction amplification of a 180-bp fragment and a 400-bp fragment of RPL32. NTC, no template control.
Preservation of Nucleic Acids in Tissues Fixed in Weigners or Formalin.
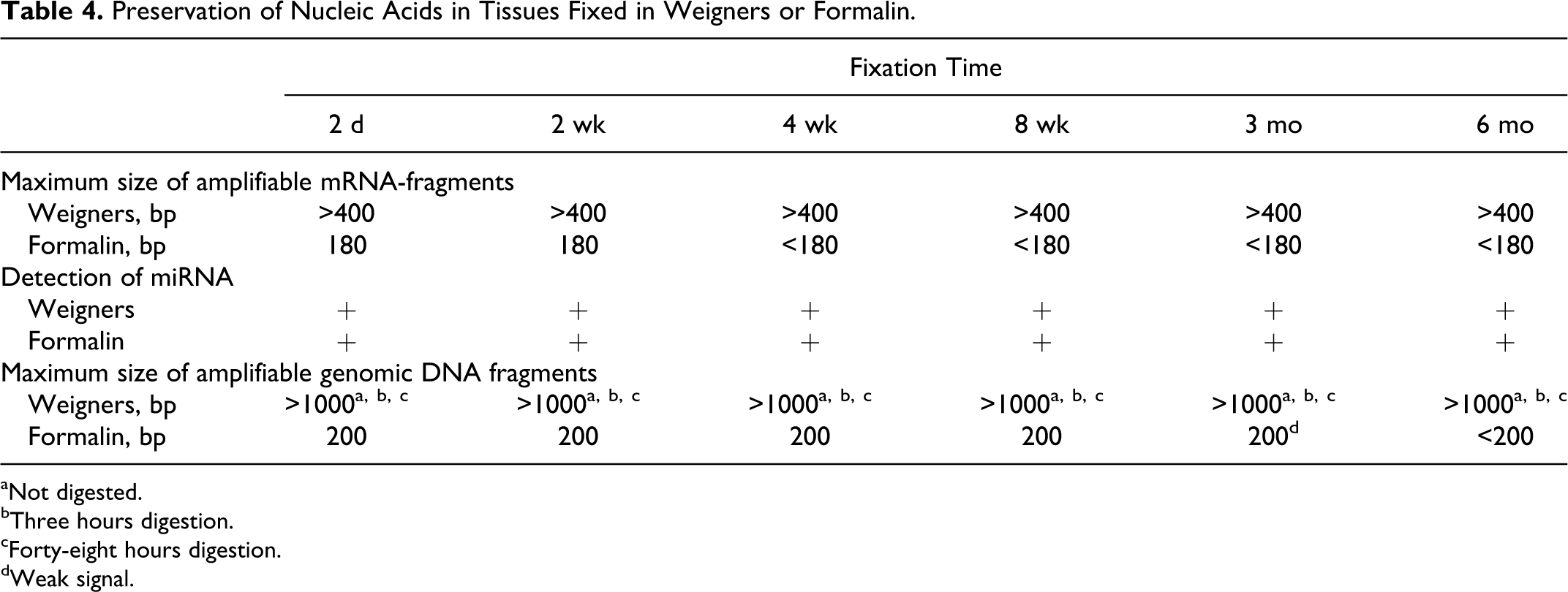
aNot digested.
bThree hours digestion.
cForty-eight hours digestion.
dWeak signal.
The fixative used did not significantly affect the extractability and amplifiability of miRNA from tissue sections. Similar amounts of miRNA were amplifiable from all WFPE and FFPE sections fixed for up to 6 months in Weigners or formalin (Table 4, Fig. 15).
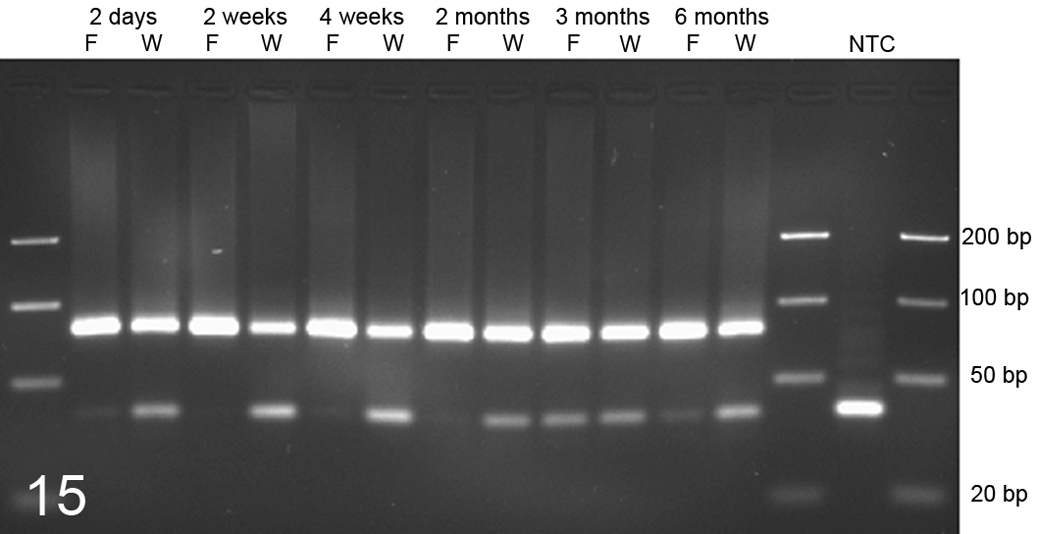
Amplification of MIR29b from kidney tissue samples fixed for 2 days, 2 and 4 weeks, and 2, 3, and 6 months in Weigners (W) or formalin (F). Both fixation methods allowed the extraction of amplifiable amounts of miR29b after up to 6 months of fixation. Polymerase chain reaction amplification of MIR29b. The lower band at approximately 40 bp represents primer dimers at varying concentrations. NTC, no template control.
Discussion
FFPE is the gold standard for tissue preservation for histologic and histopathologic analysis due to a good preservation of tissue architecture. This dominance is based on its low cost and serviceability in terms of biosecurity and preservation of morphologic details. 12 Formalin cross-links proteins by the formation of reversible methylol derivatives, Schiff bases, and stable methylene bridges between amino groups of the basic amino acids as well as other proximal nitrogen molecules.13,17 This protein denaturation directly preserves tissue morphology by fixation of structural proteins and the inhibition of intrinsic or microbial proteinolytic or nucleic acid–degrading enzymes, thereby preventing autolysis and heterolysis. 12 Despite these positive effects of formalin on morphologic integrity and decay prevention, it has adverse effects on the environment and health and causes a severe degradation of nucleic acids.4,14,15 These degrading effects are caused by a potentially low pH of unbuffered formaldehyde solutions 16 and by extensive cross-linking of nucleic acids with proteins. 15 This cross-linking leads to fragmentation of DNA and RNA and reduces their extractability. 15 This again leads to a failure to amplify RNA and DNA fragments longer than 200 bp, especially in nonbuffered formaldehyde solutions, although rarely DNA fragments of up to 600 bp in length have been reported.15,22–24 In contrast, the quality of miRNA seems to be less affected by formalin fixation. The general shortness of miRNA significantly weakens the degrading effect of formalin on miRNA. 12 Thus, several recent studies found that miRNA is apparently less affected by formalin fixation and well extractable from FFPE tissues. 20
The surprisingly good preservation of DNA and RNA from WFPE tissues indicates that similar degrading effects of Weigners on the nucleic acids are not present. The exact mechanism of nitrite pickling salt (Weigners) fixation on protein and nucleic acid fixation is unknown. However, it is conceivable that the main mechanism of fixation is based on the high pickling salt and ethanol concentration in the salt solution,5,7 and at least, the conserving effects of nitrite pickling salt have been used for food preservation for thousands of years. 1 Both mechanisms may contribute to the inhibition of intrinsic and extrinsic enzymes in the tissues without denaturation of proteins or cross-linking between proteins or proteins and nucleic acids. Moreover, Weigners seems to preserve nucleic acid integrity even after long-term storage without cutbacks in the size of amplifiable DNA or RNA fragments. Weigners may therefore be of interest for settings when cryopreservation of tissues is not possible or too expansive but well-preserved nucleic acids are needed.
The very good preservation of nucleic acids is accompanied by a good preservation of morphologic detail for most organs. Obviously, there are differences between both fixation methods. Although formalin fixation leads to superior preservation of nuclear details, there is a better preservation of cytoplasmic details with less shrinkage after Weigners fixation, although this “nonshrinkage” of the cytoplasm may challenge the eye of the formalin-adapted observer. In addition, erythrocytes seem to stain less intense after WFPE, and this difference in FFPE tissues is pronounced because of the necessary shorter eosin incubation time in some WFPE organs.
Weigners-fixed kidney tissue samples were an exception because they showed a loss of nuclear and cytoplasmic detail in distal tubular structures, although glomeruli and proximal contorted tubules were well preserved. The cause of this problem, exclusively seen in distal renal tubules, is unclear. Nevertheless, it can be speculated that the renal tubular enzymes are adapted to physiologically high salt concentrations that may lead to failure of enzyme activity inhibition by Weigners. The remaining activity of these enzymes may consecutively lead to increased autolysis in Weigners-fixed renal tissues and restricts the use of this fixative in its present form for the fixation of renal tissues. Nevertheless, the good preservation of morphologic detail of most tissues recommends Weigners for the submission of, for instance, skin samples to histopathology labs, especially by clinicians with a formalin allergy or during pregnancy when contact with formalin should be avoided.
Weigners fixation also allowed immunohistochemical analysis of protein expression similar to FFPE material. WFPE tissues were also superior for the detection of CD3 after long-term storage when compared with FFPE. Future experiments on the preservation of proteins will clarify if Weigners fixation also allows for Western blot or proteome analysis, methods that are only poorly accessible with formalin-fixed tissues.
In conclusion, Weigners fixative leads to suitable preservation of morphologic details in all organs except for kidney and a very good preservation of nucleic acid integrity. It therefore represents a valuable compromise between the nucleic acid–preserving properties of cryo-conservation and the morphology-preserving properties of formalin fixation. Its application is therefore conceivable in situations when cryo-conservation is impossible but intact nucleic acids are needed or when formalin fixation cannot be applied due to its allergenic or teratogenic effects or the need for preservation of nucleic acid integrity.
Footnotes
Acknowledgements
We thank M. Schaerig and A. Kmetsch for their excellent technical assistance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
