Abstract
Immunohistochemistry is commonly used to detect and characterize infectious agents in diagnostic pathology. The principal advantage of immunohistochemistry over other antigen detection techniques is the ability to identify antigen within the context of histologic lesions. Although epitope masking attributed to formalin fixation, especially prolonged fixation, has been considered a limiting factor in diagnostic immunohistochemistry, only a few studies have evaluated the immunohistochemical detection of infectious agents following prolonged formalin fixation. Therefore, the goal of this study was to evaluate the effects of prolonged formalin fixation on the immunohistochemical detection of 21 infectious agents. Tissue slices about 5 mm thick were fixed in 10% neutral-buffered formalin, processed, and paraffin embedded at day 1 or 2 and then at approximately weekly intervals. Three pathologists graded immunoreactivity according to a four-tier grading system: negative, weak, moderate, strong. Canine parvoviral immunoreactivity was markedly decreased following 2, 7, and 10 weeks of fixation in myocardium, small intestine, and spleen, respectively. Bovine respiratory syncytial virus immunoreactivity was markedly decreased following 7 weeks of fixation. Bartonella henselae had an abrupt loss of immunoreactivity following 9 weeks of fixation. Despite variation among time points, immunoreactivity remained moderate to strong throughout the study period for the other 18 antigens. These results suggest that prolonged formalin fixation of up to 7 weeks generally does not limit immunohistochemical detection of infectious agents. However, the effects of prolonged fixation depend on the targeted antigen and the selected antibody. The results of this study further validate the utility and reliability of immunohistochemistry in diagnostic pathology.
Immunohistochemistry (IHC) is routinely used in diagnostic pathology to detect and characterize infectious agents. 13 A principal advantage of IHC over other antigen detection techniques is that antigens are detected in the context of histologic lesions. This is especially important in diseases such as porcine circovirus infection, in which animals can be subclinical carriers and the virus may be present and detectable by IHC but not associated with clinical disease. 9 Additionally, because antigen detection is associated in the context of histologic lesions, nonspecific antibody binding can be identified and appropriately interpreted as a false-positive immunoreaction. A second advantage of IHC is that it is performed on tissues using routinely accepted methods of formalin fixation and paraffin embedding. This allows for detection of antigens in situations in which no fresh tissue is available and for detection of infectious agents that are not routinely cultured, such as Lawsonia intracellularis, Toxoplasma gondii, Sarcocystis neurona, or feline leukemia virus. Last, IHC is well suited for retrospective studies for which formalin-fixed, paraffin-embedded tissues are often the only tissues available.
Tissues used in diagnostic IHC are routinely fixed in 10% neutral-buffered formalin (4% formaldehyde buffered to pH 7.0). 10 Formalin is a cross-linking fixative that forms hydroxymethyl groups on amino acid side chains and subsequently cross-links amino acids via methylene bridges. 5,10 Formalin is an effective fixative that kills most infectious agents in tissue sections, inhibits cellular processes, and preserves tissue architecture. 5,10 However, one potential concern of formalin fixation is that formalin-induced protein cross-links could alter or hide epitopes (epitope masking) and therefore prevent immunohistochemical reactions. 1,21 Epitope masking is a concern when formalin fixation has been prolonged, because formalin fixation is a time-dependent process that continues until a point of equilibrium. 6 Studies have shown that formalin fixation, especially when prolonged, can result in decreased antigenicity 1 –3,18 ; however, antigen retrieval techniques, including proteinase digestion and heat-induced epitope retrieval (HIER), have improved antigen detection in formalin-fixed, paraffin-embedded tissues. 2,18
Few studies have evaluated the effects of prolonged formalin fixation on the immunohistochemical detection of infectious agents of veterinary interest in formalin-fixed, paraffin-embedded tissues. 8,19 Previous studies have evaluated single agents and have shown variable effects of formalin fixation depending on the antigen. Therefore, the goal of this study was to evaluate the effects of prolonged formalin fixation on the immunohistochemical detection of 21 infectious agents that are common animal pathogens.
Materials and Methods
Tissues and Formalin Fixation
Tissues were collected from samples submitted to the Purdue University Animal Disease Diagnostic Laboratory (PU-ADDL) for necropsy or histopathology examinations. Table 1 provides a complete list of infectious agents tested and the species and tissue that the agent was detected in. All tissues had characteristic histologic lesions for the identified infectious agent. Whenever possible, the presence of each infectious agent was confirmed by a second ancillary test (Table 1). Tissues were collected at necropsy or biopsy and immersion fixed in 10% neutral-buffered formalin. Following 24 to 48 hours of fixation, an initial 3- to 5-mm-thick tissue slice was routinely processed and paraffin embedded, and a 5-μm tissue section was stained with hematoxylin and eosin for the initial histologic diagnosis. Following the initial diagnosis, 3- to 5-mm thick serial tissue slices were trimmed, placed in tissue cassettes, and returned to formalin. Tissue cassettes were then processed into paraffin blocks at approximately 1-week intervals.
Antigens, Host Species, and Tissues Included in the Study and Confirmatory Ancillary Tests
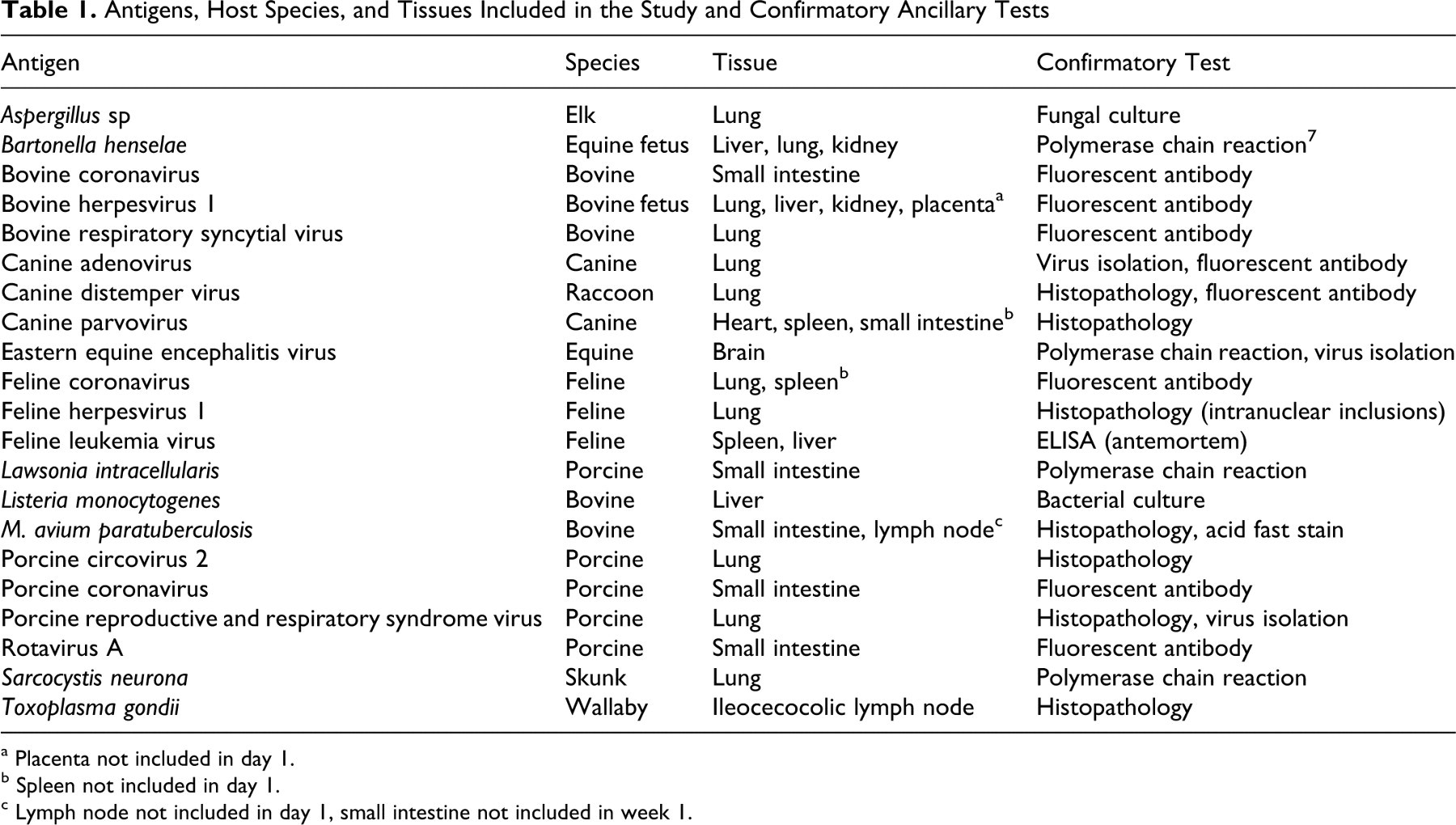
a Placenta not included in day 1.
b Spleen not included in day 1.
c Lymph node not included in day 1, small intestine not included in week 1.
Tissues from 2 pigs with porcine reproductive and respiratory syndrome (PRRS) viral pneumonia were included in the study. Two cases of canine parvovirus infection were also included. One case was canine parvoviral myocarditis, and the heart was tested. The second case was systemic canine parvovirus infection, and the spleen and small intestine were tested. Tissues from each PRRS virus–infected pig and parvovirus-infected dog were tested separately.
Immunohistochemistry
All paraffin blocks for a given antigen/tissue were sectioned on the same day, and IHC was performed simultaneously on all blocks. An exception to this was day 1 porcine coronavirus, type A rotavirus, and Aspergillus sp, which were run at a later date because of logistical errors. IHC of these time points were performed separately, but the two subsequent time points were rerun simultaneously to ensure that the IHC results between the two runs were comparable. All IHC was performed at PU-ADDL using a Dako autostainer (Dako, Carpineteria, CA) as described, 11 except for eastern equine encephalitis virus IHC, which was performed at the University of Pennsylvania New Bolton Center. 4 Table 2 lists information regarding all antibodies tested at PU-ADDL, including the antibody clone (if applicable), source, antigen retrieval technique, and detection system.
Antigens, Antibodies, and Immunohistochemical Protocolsa
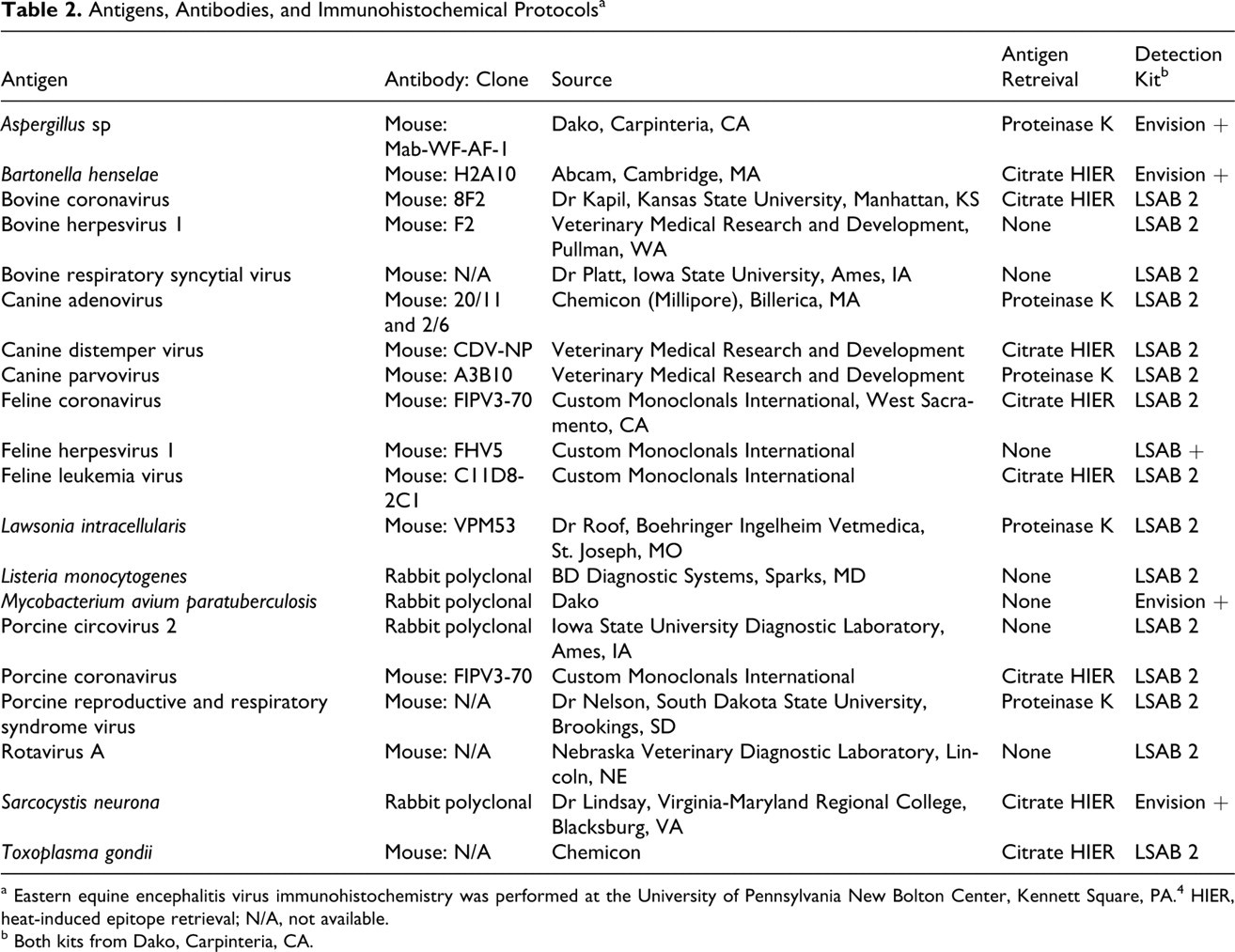
a Eastern equine encephalitis virus immunohistochemistry was performed at the University of Pennsylvania New Bolton Center, Kennett Square, PA. 4 HIER, heat-induced epitope retrieval; N/A, not available.
b Both kits from Dako, Carpinteria, CA.
Tissue sections (4 μm) were deparaffinized in xylene, rehydrated in graded alcohol, and rinsed in water. For antigens that required HIER, citrate buffer (pH 6.0; Dako) and EDTA (pH 9.0; Dako) HIER were performed in a decloaking chamber (Biocare Medical, Concord, CA) with concentrated solutions (Dako). Proteinase K antigen retrieval was performed by incubating slides with ready-to-use proteinase K (Dako) for 5 minutes at room temperature on the autostainer. Before incubation with primary antibodies, slides were incubated for 5 minutes with a ready-to-use endogenous peroxidase blocking reagent (Dako). Nonspecific antibody binding was blocked by 5-minute incubation with ready-to-use protein block reagent (Dako). All immunoreactions were detected with diaminobenzidine (Dako). Following IHC, slides were counterstained in Mayer’s hematoxylin, dehydrated in graded ethanol, cleared in xylene, and coverslipped.
IHC Evaluation
Immunohistochemical reactions were evaluated as described. 20 Three pathologists (J.D.W., M.A.M., J.R.V.) evaluated and graded all slides. Immunoreactions were evaluated on a four-tier grading system: 3+ (strong), 2+ (moderate), 1+ (weak), 0 (negative). All slides in a series were first evaluated to determine the range of immunohistochemical labeling. Slides with the most intense immunolabeling were scored 3+, and the other slides in the series were graded on the basis of that reference point. In cases of disagreement, a final grade was determined by reevaluation and mediation.
Results
Twenty-four tissues from 23 animals were independently evaluated. PRRS virus immunoreactivity was independently evaluated in tissues from 2 pigs. Canine parvovirus immunoreactivity was evaluated in 3 tissues (heart, spleen, and small intestine) from 2 dogs. Sections of spleen and small intestine were from the same dog and were evaluated in tandem on the same slide. For all other antigens, immunoreactions were evaluated in single tissues or in multiple tissues on single slides, but all tissues had similar immunoreactivity. All time point results that are not available (N/A) are due to the unavailability of tissues fixed for the given time point.
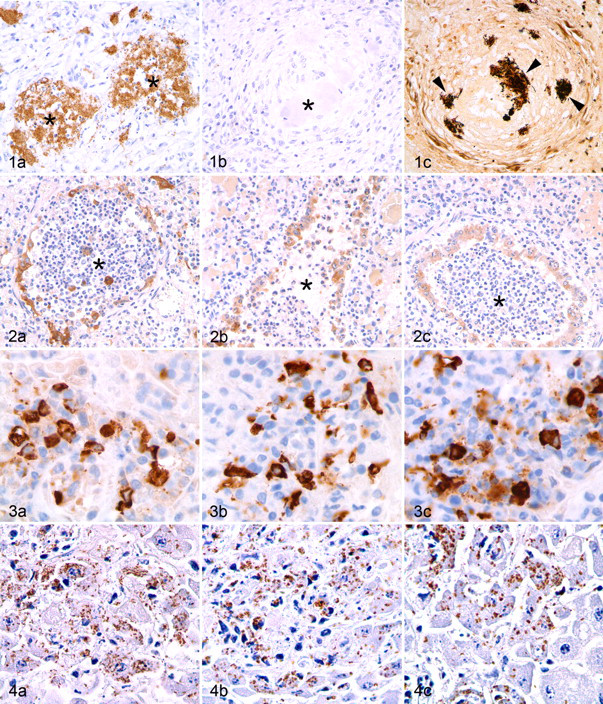
Antigens of interest for each infectious agent were detected in at least 67% of the time points evaluated (Table 3 ). Bartonella henselae, canine parvovirus, and bovine respiratory syncytial virus all had marked decreases in immunoreactivity following prolonged formalin fixation. Bartonella immunoreactivity was strong until 9 weeks of fixation, at which time there was complete loss of immunoreactivity (Fig. 1). In the heart, canine parvovirus had strong immunoreactivity on day 1, but at the next time point (week 2) and thereafter, immunoreactivity was weak and sparse. In the small intestine, canine parvovirus immunoreactivity was weak or absent following 7 weeks of fixation. Moderate to strong immunoreactivity was maintained in the spleen until week 10, when it became weak. Bovine respiratory syncytial virus had strong immunoreactivity at day 1 and week 1, moderate immunoreactivity between week 2 and week 6, but weak immunoreactivity following 7 weeks of fixation (Fig. 2). For all other antigens, there was moderate to strong immunoreactivity at all time points, although there was slide-to-slide variation in immunoreactivity for some antibodies (Figs. 3, 4) .
Consensus Immunohistochemistry Grades Among 3 Pathologists a
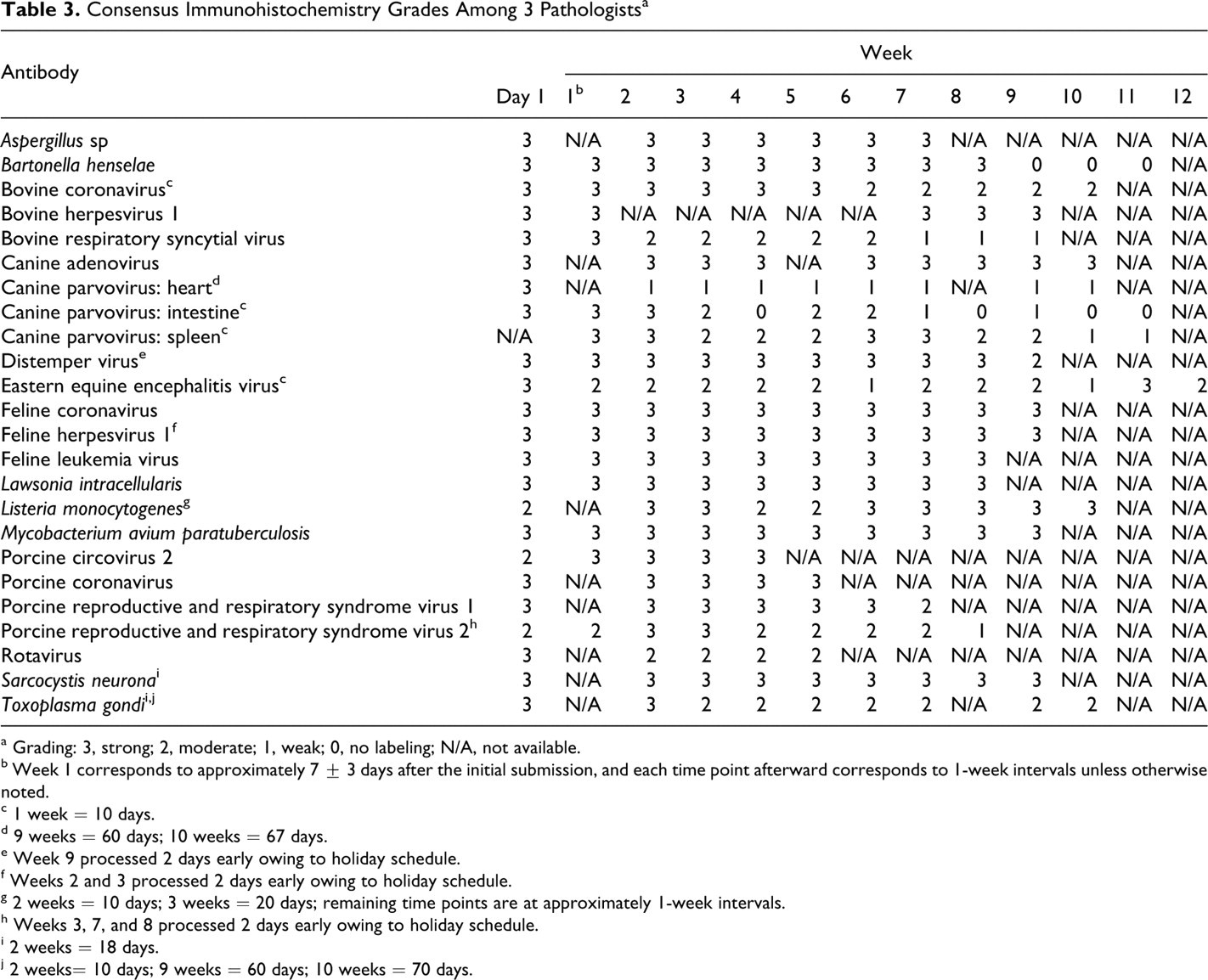
a Grading: 3, strong; 2, moderate; 1, weak; 0, no labeling; N/A, not available.
b Week 1 corresponds to approximately 7 ± 3 days after the initial submission, and each time point afterward corresponds to 1-week intervals unless otherwise noted.
c 1 week = 10 days.
d 9 weeks = 60 days; 10 weeks = 67 days.
e Week 9 processed 2 days early owing to holiday schedule.
f Weeks 2 and 3 processed 2 days early owing to holiday schedule.
g 2 weeks = 10 days; 3 weeks = 20 days; remaining time points are at approximately 1-week intervals.
h Weeks 3, 7, and 8 processed 2 days early owing to holiday schedule.
i 2 weeks = 18 days.
j 2 weeks= 10 days; 9 weeks = 60 days; 10 weeks = 70 days.
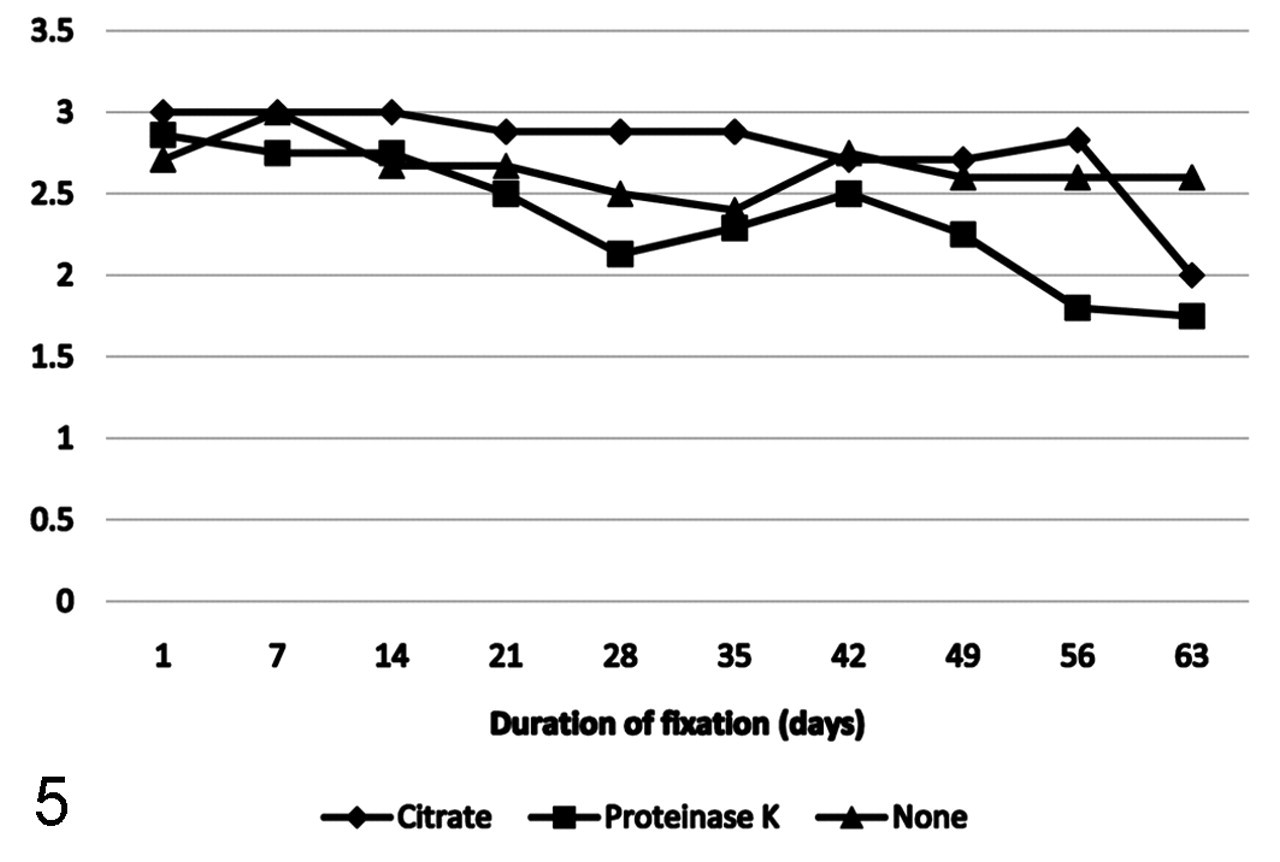
Mean immunoreactivity over time of antigens grouped by antigen retrieval protocol.
When grouped by antigen retrieval protocol, antigens that were retrieved with citrate buffer HIER maintained the strongest immunoreactivity up to 56 days following formalin fixation. Antigens retrieved with proteinase K had the most dramatic decline in immunoreactivity, which was first apparent at week 3 and continued through week 9, for a final mean score of 1.75. Antigens that did not require antigen retrieval tended to maintain moderate to strong immunoreactivity throughout the study; however, immunoreactivity did fluctuate among time points (Fig. 5).
Discussion
For most infectious agents in this study, moderate to strong immunoreactivity was maintained following prolonged formalin fixation. However, decreases in immunoreactivity were marked for some infectious agents, which suggest that the effects of prolonged formalin fixation on IHC are likely dependent on antigens, antibodies, and possibly tissues. These results are consistent with those of our previous studies that evaluated the effects of prolonged formalin fixation on the detection of cellular markers, 12,14 –16,20 suggesting that for most (but not all) antibodies and antigens, prolonged formalin fixation does not significantly inhibit immunoreactivity on paraffin sections.
Most studies evaluating the effects of prolonged formalin fixation on IHC have focused on cellular antigens, with few studies evaluating the detection of infectious agents. Bovine viral diarrhea virus immunoreactivity persisted through 36 days of fixation, but no antigen could be detected at 176 days. 8 Immunoreactivity of PRRS virus was decreased after 5 days of fixation with loss of immunoreactivity at all later time points. 19 In an earlier study, PRRS virus was detected after 7 days of fixation. 17 Because these studies had different results, we investigated PRRS virus immunoreactivity with tissues from 2 pigs. In each sample, there was moderate immunoreactivity following 42 days of fixation. Variations in the results of these studies probably reflect variations in the antigen retrieval protocols and immunodetection kits used in each study. The results of our study suggest that PRRS virus can be detected following prolonged formalin fixation, but this might depend on the protocol that is used.
An additional consideration when evaluating the immunoreactivity of PRRS virus and other infectious agents is that antigens might not be evenly distributed throughout the tissue. Uneven and sparse antigen distribution was mostly a problem for PRRS virus and intestinal parvovirus. When antigen was detected, it tended to be moderate to strong, but for some time points, antigen was detected in only a few cells and not in every section. In these cases, it was difficult to determine whether a lack of immunoreactivity was due to fixation or paucity of antigen. The amount and distribution of antigen should be considered in diagnostic cases. Lack of immunohistochemical reaction does not rule out the antigen’s presence in the tissue as a whole; it only indicates that the antigen is not present at detectable levels in the evaluated tissue section. 13
As in our previous study, antigens retrieved with citrate buffer HIER tended to maintain strong immunoreactivity, whereas proteinase K digestion was associated with the most pronounced decrease in immunoreactivity. 20 Interestingly, many antibodies that have been optimized for use without antigen retrieval effectively labeled antigens of interest following prolonged fixation. In these cases, it is likely that formalin fixation minimally cross-linked or masked the antigen of interest. These results suggest that one of the most important factors for IHC following prolonged formalin fixation is to have a protocol that has been optimized for a given antigen–antibody combination. Additionally, when optimizing IHC protocols, antibodies should be tested with tissues that have been fixed over multiple time points up to 2 months. 12 –16 This will facilitate interpretation of unexpected results and evaluation of IHC results in tissues that have been fixed for extended or unknown durations.
In summary, the results of this study suggest that for most antibody–antigen combinations, formalin fixation, up to 7 weeks or more, does not limit the use of IHC. IHC has several advantages over other diagnostic tests, including the ability to colocalize antigens with histologic lesions; detect antigens in formalin-fixed, paraffin-embedded tissues; and detect pathogens for which routine isolation is impractical or dangerous. These findings further validate the utility of IHC in diagnostic pathology.
Footnotes
Acknowledgments
Part of this study was presented as a poster at the 52nd annual meeting of the American Association of Laboratory Diagnosticians in San Diego, CA, October 2009. We thank Dr Del Piero for performing the eastern equine encephalitis virus immunohistochemistry and the Animal Disease Diagnostic Laboratory staff for technical assistance.
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article.
