Abstract
The objective of this report was to characterize the enhanced clinical disease and lung lesions observed in pigs vaccinated with inactivated H1N2 swine δ-cluster influenza A virus and challenged with pandemic 2009 A/H1N1 human influenza virus. Eighty-four, 6-week-old, cross-bred pigs were randomly allocated into 3 groups of 28 pigs to represent vaccinated/challenged (V/C), non-vaccinated/challenged (NV/C), and non-vaccinated/non-challenged (NV/NC) control groups. Pigs were intratracheally inoculated with pH1N1and euthanized at 1, 2, 5, and 21 days post inoculation (dpi). Macroscopically, V/C pigs demonstrated greater percentages of pneumonia compared to NV/C pigs. Histologically, V/C pigs demonstrated severe bronchointerstitial pneumonia with necrotizing bronchiolitis accompanied by interlobular and alveolar edema and hemorrhage at 1 and 2 dpi. The magnitude of peribronchiolar lymphocytic cuffing was greater in V/C pigs by 5 dpi. Microscopic lung lesion scores were significantly higher in the V/C pigs at 2 and 5 dpi compared to NV/C and NV/NC pigs. Elevated TNF-α, IL-1β, IL-6, and IL-8 were detected in bronchoalveolar lavage fluid at all time points in V/C pigs compared to NV/C pigs. These data suggest H1 inactivated vaccines followed by heterologous challenge resulted in potentiated clinical signs and enhanced pulmonary lesions and correlated with an elevated proinflammatory cytokine response in the lung. The lung alterations and host immune response are consistent with the vaccine-associated enhanced respiratory disease (VAERD) clinical outcome observed reproducibly in this swine model.
Influenza A virus (IAV) is the cause of an acute respiratory disease in swine characterized by anorexia, fever, dyspnea, coughing, and nasal discharge. 30 Influenza A virus is a member of the Orthomyxoviridae family, with segmented, negative sense RNA genomes that permit the generation of novel, antigenically distinct viruses. The swine respiratory tract contains receptors for both avian and human IAVs, which may increase susceptibility to interspecies transmission and facilitate genetic diversity through reassortment. 13 Influenza infection in naive swine is typically associated with high morbidity and low mortality, with some variation in virulence between strains. In the Northcentral United States, disease incidence peaks during the fall; however, epizootics may occur that do not follow this once typical seasonality. 1
The stability of classical H1N1 (cH1N1) IAV in North American swine persisted for over 70 years, ending in 1998 with the introduction of the triple reassortant H3N2 that contained genes of human-, avian-, and swine-virus lineages. 47,48 Influenza A viruses that are currently endemic in North American swine are all triple reassortants that contain the triple reassortant internal gene (TRIG) cassette, which consists of human-IAV lineage polymerase basic 1 (PB1), avian-IAV lineage polymerase basic 2 (PB2), polymerase acidic (PA) and swine-IAV lineage nucleoprotein (NP), matrix (M), and nonstructural (NS) genes. 20,41 The apparent ability of the TRIG cassette to exchange gene segments for the surface glycoproteins hemagglutinin (HA) and neuraminidase (NA) has further increased the generation of reassortant IAVs with marked antigenic diversity. 40 In addition, drift variants within the three major subtypes, H1N1, H1N2, and H3N2, have also emerged. 3,19,24,29,40,44
Currently, four H1 phylogenetic clusters, α, β, γ, and δ, are endemic in US swine, acquired through interspecies transmission, reassortment events, and antigenic drift. 40,42 Unlike IAV variants isolated before 1998, which demonstrated more consistent cross-reactivity in the hemagglutination inhibition (HI) assay, contemporary viruses exhibit limited HI cross-reactivity between different clusters, in spite of the similarity in subtype. 39,40 Additionally, 2 subclusters have emerged within the H1 δ-cluster from two apparently independent introductions of a human seasonal HA into IAVs in swine and have been designated δ-1 and δ-2. 18 Collectively, the emergence of the pandemic 2009 H1N1 virus, the introduction of human seasonal influenza viruses in swine, and the generation of antigenic variants within a subtype with less consistent serological cross-reactivity provides substantial evidence of the increasing heterogeneity among IAVs in swine.
The use of inactivated IAV vaccines in swine is common in the US, and such vaccines effectively reduce clinical disease and lesion induction by homologous viruses, 38 although protection against heterologous infection has been inconsistent. 2,21,36,38 However, inactivated influenza vaccines also have been implicated in vaccine-associated enhanced respiratory disease (VAERD) in swine. 8,12,16,38 Recently, a vaccination-heterologous-challenge model was described in which naïve pigs vaccinated with an inactivated, adjuvanted H1N2 swine δ-cluster IAV and challenged with pandemic 2009 A/H1N1 human influenza virus demonstrated severe clinical disease, enhanced lung consolidation, and potentiated microscopic lesions compared to nonvaccinated and challenged pigs. 8 The vaccine did not prevent infection, and hemagglutination inhibition and serum neutralizing (SN) antibodies did not cross-react with the challenge virus. The inactivated vaccine was decidedly associated with VAERD, although the mechanism remains unclear at this time. Due to the emergence of novel IAVs in swine and a complex number of cocirculating subtypes and variants, VAERD could occur in field situations where pigs may be vaccinated with farm-specific vaccines and then subsequently break with antigenically diverse IAVs. The vaccine-associated enhancement of disease phenomenon is not exclusive to swine and inactivated IAV vaccines. Nonprotective, low avidity antibodies elicited by respiratory syncytial virus and measles formalin-inactivated vaccines have also been implicated in causing enhanced respiratory disease in human infants. 4,26
The objective of this study was to characterize the enhanced macroscopic and microscopic lung lesions in pigs administered an inactivated δ-cluster H1N2 IAV and challenged with pandemic 2009 A/H1N1 (pH1N1) as compared to nonvaccinated/challenged pigs using a previously described model. 8 Macroscopic and microscopic lung lesions, virus distribution in the lungs, and local inflammatory responses were evaluated at 1, 2, 5, and 21 days post inoculation (dpi) to elucidate the onset and development of VAERD in pigs challenged with pH1N1 IAV.
Materials and Methods
Experimental Design
Eighty-four, 3-week-old cross-bred pigs were obtained from a herd free of IAV and porcine reproductive and respiratory syndrome virus (PRRSV) and treated with ceftiofur (Pfizer Animal Health, New York, NY) according to label directions to reduce respiratory bacterial pathogens prior to the start of the study. Pigs were housed in biosafety level 2 (BSL2) containment during the vaccine phase of the study. Fifty-six pigs were transferred to ABSL3 containment 1 week prior to challenge for the remainder of the experiment. Pigs were cared for in compliance with the Institutional Animal Care and Use Committee of the National Animal Disease Center. The experimental design is described in Table 1.
Experimental Design
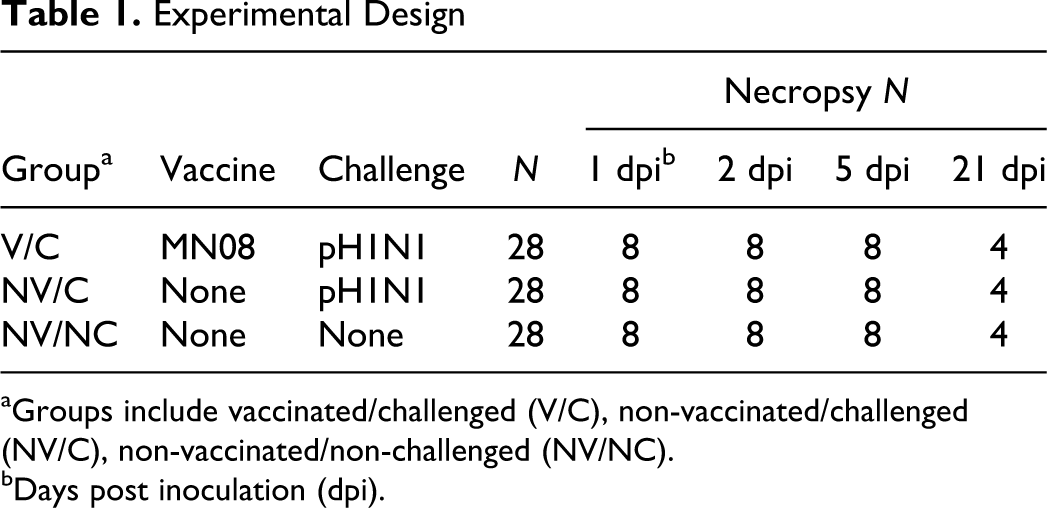
aGroups include vaccinated/challenged (V/C), non-vaccinated/challenged (NV/C), non-vaccinated/non-challenged (NV/NC).
bDays post inoculation (dpi).
Influenza A virus vaccine was prepared with a δ-1 cluster A/Sw/MN/02011/2008 H1N2 (MN08) at approximately 32 HA units or 105.7 50% tissue culture infectious dose (TCID50) that was inactivated by ultraviolet irradiation and mixed with an emulsified oil-in-water adjuvant (Emulsigen®-D, MVP Technologies, Omaha, NE) at a v:v ratio of 4:1 virus to adjuvant. Pigs were vaccinated with 2 ml by the intramuscular route at approximately 6 and 9 weeks of age, and challenged at 13 weeks of age. For viral challenge, pigs were inoculated intratracheally with 2 ml of 1 × 105 TCID50 of pandemic A/CA/04/2009 H1N1 (pH1N1) propagated in Madin-Darby Canine Kidney (MDCK) cells, as previously described. 42 Pigs were observed daily for signs of clinical disease, and rectal temperatures were taken on –1, 0, 1, 2, 3, 4, and 5 days dpi. Nasal swabs (Fisherbrand Dacron swabs, Fisher Scientific, Pittsburg, PA) were taken on 0, 1, 2, 3, 5, and 21 dpi to evaluate nasal virus shedding as previously described. 39 After serum collection by venipuncture, pigs were humanely euthanized with a lethal dose of pentobarbital (Sleepaway, Fort Dodge Animal Health, Fort Dodge, IA) on 1, 2, 5, or 21 dpi to evaluate lesions, cytokine concentrations, and virus titers in the lungs. Postmortem samples included bronchoalveolar lavage fluid (BALF), lung, and trachea.
Pathologic Examination of Lungs
At necropsy, lungs were removed and evaluated for the percentage of the lung affected with purple-red consolidation typical of IAV in swine. The percentage of the surface area affected with pneumonia was visually estimated for each lung lobe, and total percentage for the entire lung was calculated based on weighted proportions of each lobe to the total lung volume. 11 Tissue samples from the trachea and right middle or affected lung lobe were fixed in 10% buffered formalin for histopathologic examination. Tissues were processed by routine histopathologic procedures and slides stained with hematoxylin and eosin. Microscopic lesions were evaluated by a veterinary pathologist blinded to treatment groups. Individual scores were assigned to each of 4 parameters to adequately reflect the contribution of each lesion associated with VAERD: bronchial and bronchiolar epithelial necrosis or proliferation, suppurative bronchitis or bronchiolitis, peribronchiolar lymphocytic cuffing, and alveolar septal thickening with inflammatory cells (interstitial pneumonia). The first 2 scores focused on the intrapulmonary airways: (1) percentage of bronchi and bronchioles affected with epithelial lesions (necrotizing or proliferative bronchitis and bronchiolitis) and (2) percentage of bronchi and bronchioles that contained purulent exudate (suppurative bronchitis or bronchiolitis). Two additional lung lesion scores were based on the following: (3) magnitude of peribronchiolar lymphocytic cuffing and (4) presence and severity of alveolar septal inflammation (interstitial pneumonia). A composite score was computed using the sum of the 4 individual scores. The average group composite score was used for statistical analysis. Scoring parameters for lung and trachea are described in Table 2.
Scoring Parameters Used for Evaluation of Hematoxylin/Eosin (H&E) Microscopic Lesions and Immunohistochemistry (IHC) Signals in Lung and Trachea (Scores Range From 0 to 3)
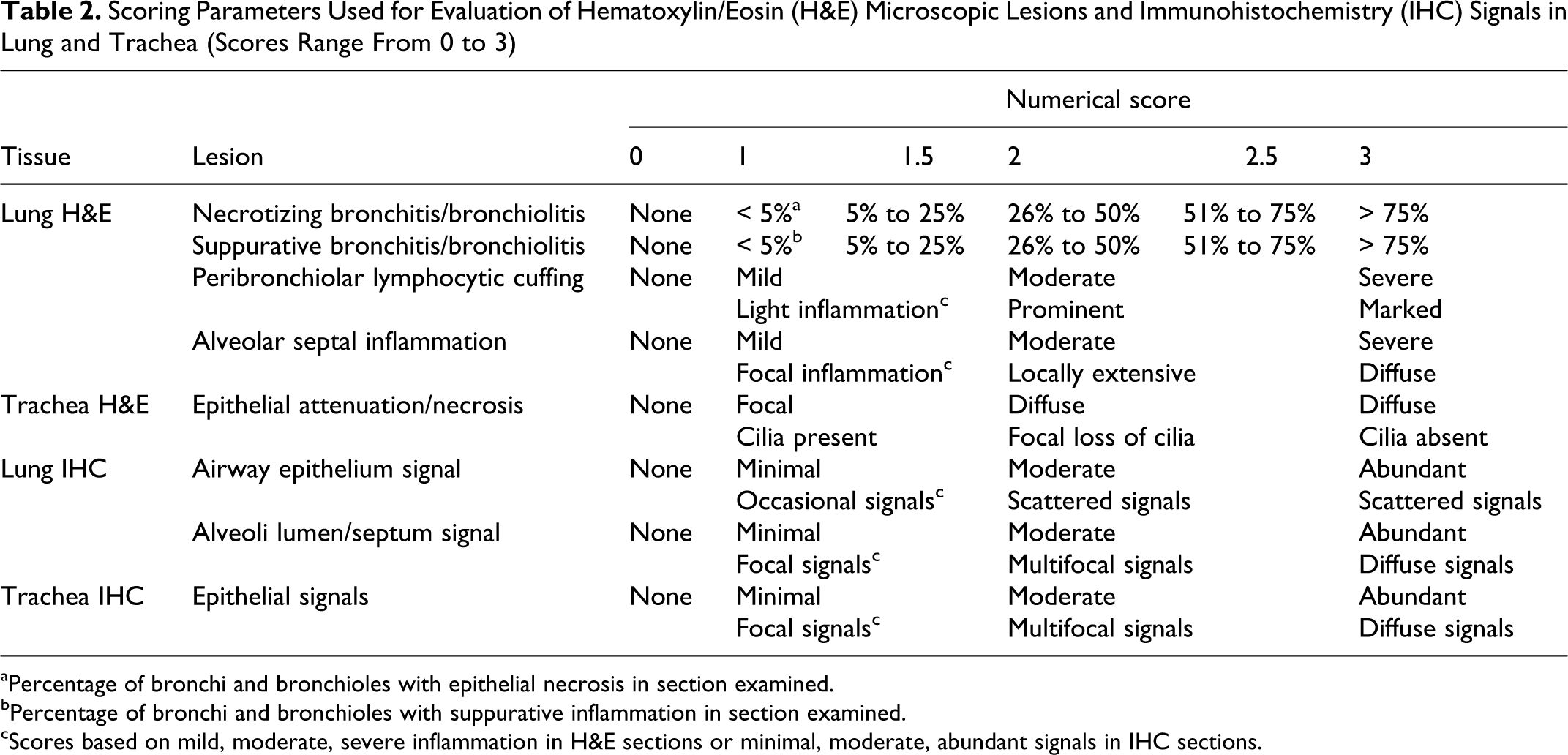
aPercentage of bronchi and bronchioles with epithelial necrosis in section examined.
bPercentage of bronchi and bronchioles with suppurative inflammation in section examined.
cScores based on mild, moderate, severe inflammation in H&E sections or minimal, moderate, abundant signals in IHC sections.
Influenza virus Type A-specific antigen was detected in lung and trachea using a previously described immunohistochemical (IHC) method with minor modification. 38,43 Modifications include treating tissue sections in 0.05% protease for 2 min. Influenza A virus-specific monoclonal antibody (MAb) HB65 (ATCC, Manassas, VA) was applied at 1:100 dilution. Bound MAbs were stained with peroxidase-labeled anti-mouse IgG followed by chromogen using the DAKO LSAB2-HRP Detection System (DAKO, Carpinteria, CA) according to the manufacturer’s instructions. Antigen detection in lung was given 2 scores: (1) airway epithelial signals and (2) alveolar septa/lumen signals. Both scores were based only on affected lobules. IHC slides were evaluated by a veterinary pathologist blinded to treatment groups. Scoring parameters are described in Table 2.
Diagnostic Microbiology
All pigs were screened for antibody against influenza A NP by ELISA (MultiS ELISA, IDEXX, Westbrook, ME) according to the manufacturer’s recommendation upon arrival to ensure absence of existing immunity from prior exposure or maternal derived antibody and at 0 dpi to detect vaccine-induced NP antibodies. BALF samples from 1, 2, 5, and 21 dpi were cultured for aerobic bacteria on blood agar and Casmin (NAD enriched) plates. Polymerase chain reaction (PCR) assays for porcine circovirus type 2 (PCV2), 25 Mycoplasma hyopneumoniae, 32 and PRRSV also were conducted on nucleic acid extracts from BALF collected at each necropsy.
Viral Replication and Shedding
Nasal swabs and BALF were processed as previously described. 38,39 MDCK-inoculated monolayers were evaluated for cytopathic effect (CPE) between 48 and 72 h postinfection and subsequently fixed with 4% phosphate-buffered formalin and stained using immunocytochemistry with an anti-influenza A nucleoprotein monoclonal antibody as previously described. 15 A TCID50 titer was calculated for each sample using the method of Reed and Muench. 28
Antibody Detection Assays
Hemagglutination inhibition assays were conducted as previously described. 38 The HI assays were performed with MN08 or pH1N1 virus as antigen and turkey red blood cells (RBCs) as indicators using standard techniques. 45 Reciprocal titers were divided by 10 and log2 transformed, analyzed, and reported as the geometric mean.
Enzyme-linked immunosorbent assays (ELISA) to detect total IgG and IgA antibodies against whole virus preparations of MN08 and pH1N1 present in serum were performed as previously described. 38 Independent assays were conducted using concentrated MN08 or pH1N1 as ELISA antigen. The optical density (OD) was measured at 405 nm wavelength with an automated ELISA reader. Antibody levels were reported as the mean OD and the mean OD of each treatment group were compared.
For the SN assay, sera were heat inactivated at 56°C for 30 min, then 2-fold serially diluted from 1:10 to 1:20, 480 in 96-well plates, using 50 µl serum diluted in serum-free MEM supplemented with L-1-tosylamido-2-phenylethyl chloromethyl ketone (TPCK) trypsin and antibiotics. Fifty microliters of SIV diluted to 103.3 TCID50 /ml was added to each dilution and incubated at 37°C for 1 hr. Post incubation, 100 µl of the serum and virus mixture was added to each well of confluent MDCK cells prepared in 96-well plates 48 hours in advance and washed twice in sterile PBS. Plates were incubated for 48 hours or until the presence of cytopathic effect (CPE). Presence of influenza virus was verified by immunocytochemistry for all plates, and titers were recorded as the highest dilution negative for virus or CPE. Reciprocal titers were log2 transformed, analyzed, and reported as the geometric mean.
Cytokine Assays
A 5 ml aliquot of BALF was centrifuged at 400 × g for 15 min at 4°C to pellet cellular debris. The cell-free BALF was stored at –80° C. Cytokine concentrations in BALF were determined by multiplex ELISA performed according to the manufacturer’s recommendations (SearchLight, Aushon Biosystems). The average of duplicate wells for each sample was used for statistical analysis.
Statistical Analysis
Macroscopic pneumonia scores, microscopic pneumonia scores, log10 transformed BALF and nasal swab virus titers, log2 transformations of HI and SN reciprocal titers, mean OD ELISA reading, and cytokine data were analyzed using analysis of variance (ANOVA) with a P value ≤ .05 considered significant (JMP, SAS Institute, Cary, NC; GraphPad Prism Version 5.00, San Diego, CA). Response variables shown to have a significant effect by treatment group were subjected to pairwise comparisons using the Tukey-Kramer test. Rectal temperature data were analyzed using a mixed linear model for repeated measures. Linear combinations of the least squares means estimates were used in a priori contrasts after testing for a significant (P < .05) treatment group effect of vaccination status. Comparisons were made between each group at each time-point using a 5% level of significance (P < .05) to assess statistical differences.
Results
Clinical Disease
Mild lethargy was observed at 1 dpi in the non-vaccinated/challenged (NV/C) pigs with activity returning to normal by 2 dpi. Coughing or dyspnea was not observed in the NV/C pigs. In contrast, vaccinated/challenged (V/C) pigs were lethargic and anorexic for 48 hours, and 20% of the V/C pigs exhibited coughing and/or dyspnea until 3 dpi. Challenge with pH1N1 virus induced a significant (P ≤ .05) febrile response in both V/C and NV/C groups at 24 and 48 hours post infection compared to non-vaccinated/non-challenged (NV/NC) pigs. This initial febrile response in the challenged pigs subsided by 3 dpi. However, a significantly different febrile response was not detected between V/C and NV/C pigs on any sampling post challenge. NV/NC pigs did not demonstrate clinical signs or a febrile response during the trial.
Viral and Microbiological Assays
All pigs were free of IAV antibodies prior to the start of the experiment. No PCV2, PRRSV, or M. hyopneumoniae nucleic acid was detected in BALF in any pigs. Routine aerobic bacterial cultures of BALF isolated Bordetella bronchiseptica from one pig in the NV/C group. Bacteria were not isolated from the lungs of any other pigs during the trial.
Macroscopic Pneumonia
Pigs challenged with pH1N1 displayed purple-colored, multifocal to coalescing consolidation of the cranioventral regions of lung typical of IAV infection in swine. Lung lesions were of variable extent in both challenge groups and were predominantly located in the left and right cranial and middle lung lobes. Lesions were also observed in the cranial portion of the diaphragmatic lobes in some of the V/C pigs but not in the NV/C pigs. The V/C group had a significantly greater (P ≤ .001) percentage of lung consolidation compared to the NV/C pigs at 1, 2, 5, and 21 dpi (Table 3). The average percentage of lung consolidation increased at each acute time point in V/C pigs from approximately 10% at 1 dpi (Fig. 1) to >20% at 5 dpi (Fig. 2). In the NV/C pigs, only slight increases in percentage of lung consolidation were observed between 1 dpi (Fig. 3) and 5 dpi (Fig. 4) and never exceeded 7%. Macroscopic lung lesions were absent or minimal in pigs in the NV/NC group.
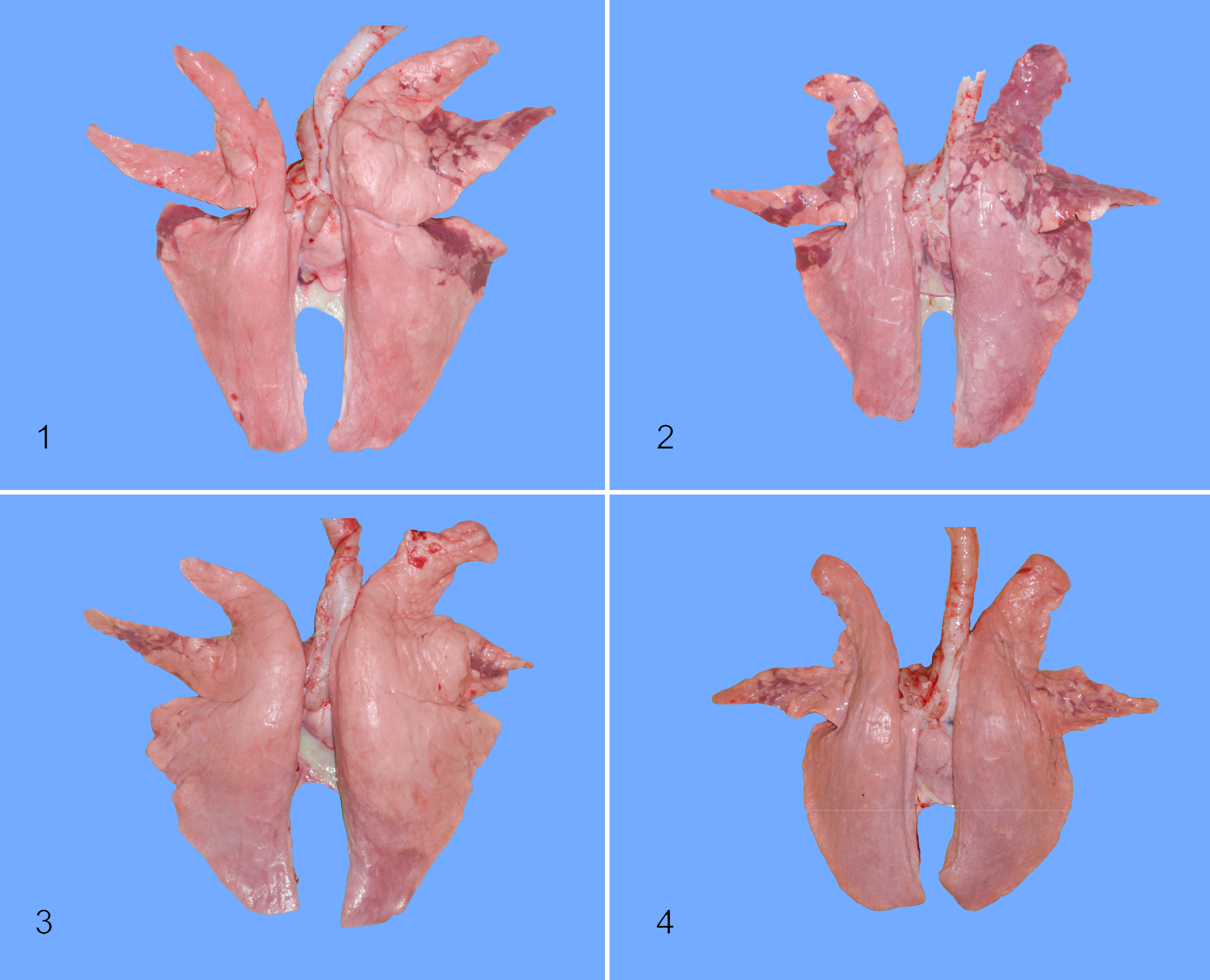
Lung, pig No. 112, vaccinated/challenged (V/C) group. Note the multifocal, purple-colored cranioventral and diaphragmatic pulmonary consolidation at 1 day post infection (dpi) in a pig with vaccine-associated enhanced respiratory disease (VAERD).
Comparison of the Mean Percentage Lung Lesions (Purple-Red Consolidation) and Mean Microscopic Lesion Score (Necrotizing Bronchiolitis, Suppurative Bronchiolitis/Bronchitis, Peribronchiolar Lymphocytic Cuffing, Alveolar Septal Thickening With Mononuclear Inflammation) in Vaccinated/Challenged (V/C), Non-Vaccinated/Challenged (NV/C) and Non-Vaccinated/Non-Challenged (NV/NC) Pigs at 1, 2, 5, and 21 Days After pH1N1 Inoculation (dpi; Data Presented as the Mean Percentage Consolidation or Mean Composite Lesion Score ± Standard Error of the Mean)
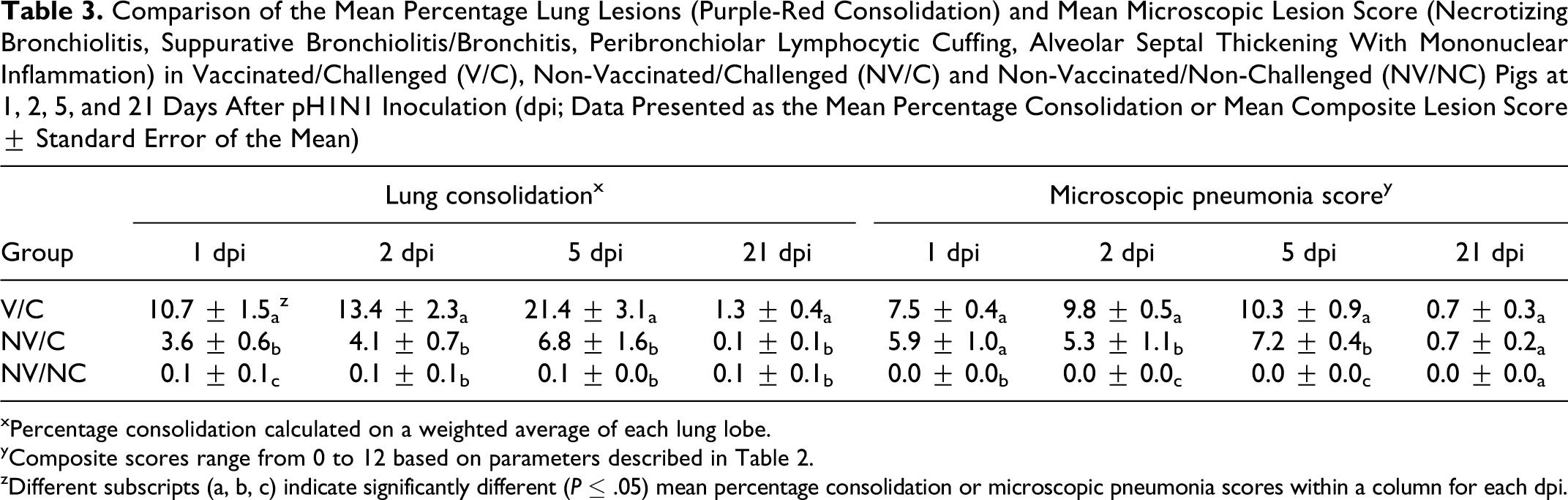
xPercentage consolidation calculated on a weighted average of each lung lobe.
yComposite scores range from 0 to 12 based on parameters described in Table 2.
zDifferent subscripts (a, b, c) indicate significantly different (P ≤ .05) mean percentage consolidation or microscopic pneumonia scores within a column for each dpi.
Microscopic Pneumonia
The average composite microscopic lung lesion score was significantly higher (P ≤ .01) in the V/C pigs at 2 and 5 dpi compared to NV/C pigs (Table 3). The average composite score was also higher in the V/C pigs at 1 dpi compared to the NV/C group, although a statistically significant difference was not observed. Microscopic lesions were not evident in lung in the NV/NC pigs at any time during the study.
NV/C pigs exhibited microscopic lung lesions consistent with uncomplicated influenza virus infection, demonstrating mild airway epithelial attenuation with concurrent suppurative bronchitis and/or bronchiolitis and mild bronchointerstitial pneumonia at 1 dpi (Fig. 5). Multifocal bronchi, bronchioles, and alveolar lumina contained low to moderate numbers of neutrophils and scattered macrophages. Necrotizing bronchiolitis became more pronounced by 2 dpi, although suppurative inflammation in bronchi, bronchioles, and particularly the alveoli had almost completely subsided by this time. At 5 dpi, NV/C pigs demonstrated necrotizing or proliferative bronchitis and bronchiolitis with light, peribronchiolar lymphocytic cuffing (Fig. 6). Multifocally, alveolar septa were expanded by mild to moderate numbers of lymphocytes and macrophages.
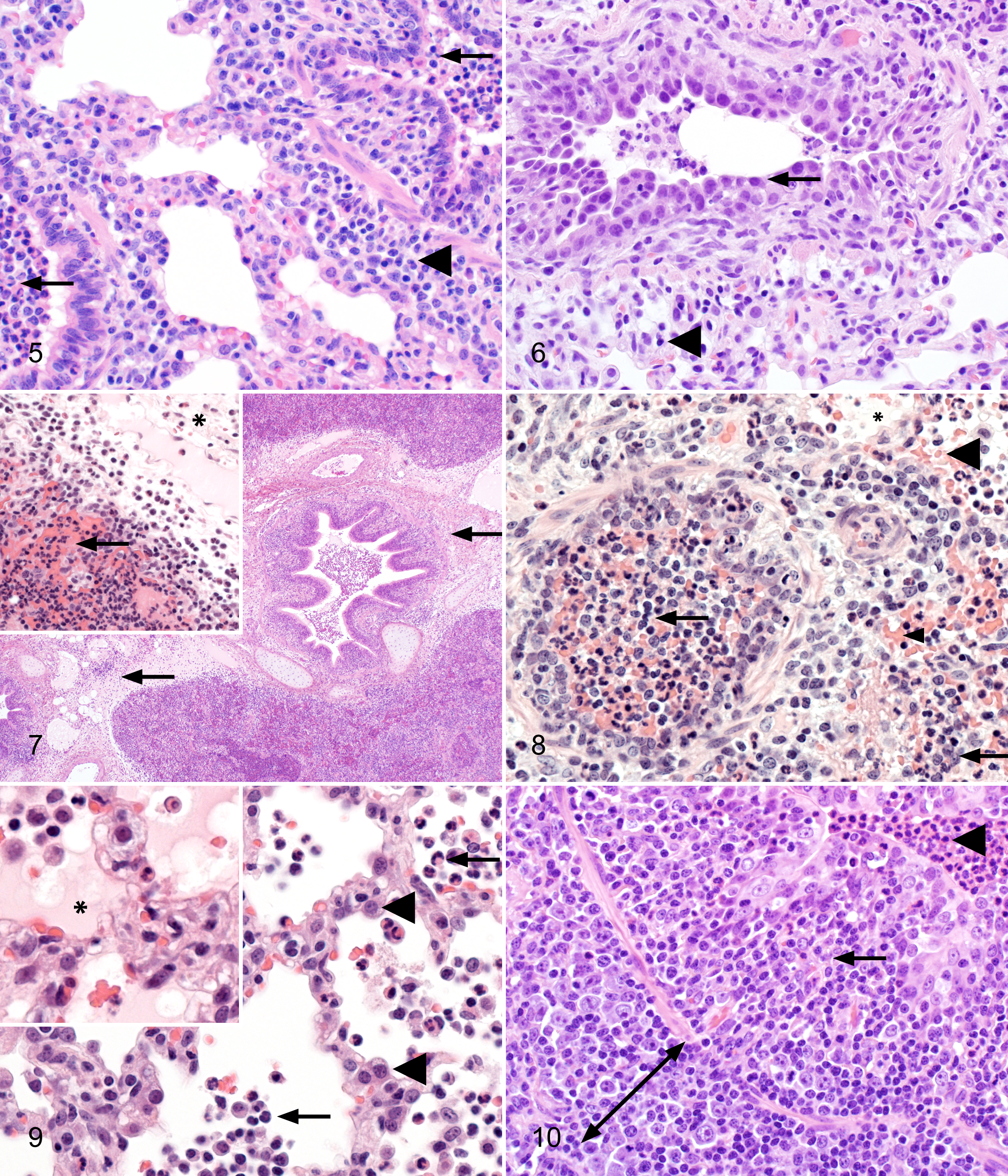
Lung, pig No. 135, non-vaccinated/challenged (NV/C) group. Mild bronchiolar epithelial attenuation and suppurative bronchiolitis (arrow) were observed at 1 day post infection (dpi) with mild alveolar septal thickening and light peribronchiolar cuffing (arrowhead). Hematoxylin and eosin, 400×.
In contrast to the mild microscopic lung lesions demonstrated in the NV/C pigs, lung lesion profiles were notably more widespread and severe in the V/C pigs with VAERD occurring as rapidly as 1 dpi. Alveolar lumina were flooded by large amounts of eosinophilic proteinaceous material (edema) and hemorrhage (Fig. 7) that was not observed in the NV/C group. Marked interlobular edema was also demonstrated exclusively in the V/C pigs (Fig. 7), and an abundant, suppurative inflammatory infiltrate was observed in a larger percentage of bronchi and bronchioles in the VAERD-affected pigs compared to the NV/C group. Alveolar lumina bordering affected bronchioles and extending into adjacent lobules were expanded or occluded by large numbers of neutrophils and scattered macrophages in V/C pigs (Fig. 8), contrasting the mild suppurative inflammatory response in alveolar lumina of NV/C pigs (Fig. 5). However, VAERD-affected pigs demonstrated mild bronchi and bronchiolar epithelial necrosis that was similar to the NV/C group at 1 dpi.
By 2 dpi, composite microscopic lesion scores increased in the V/C group (Table 3) due to a moderate elevation in alveolar septal mononuclear inflammation and abundant neutrophils, macrophages, edema, and hemorrhage that persisted in alveolar lumina of affected lobules (Fig. 9). Suppurative inflammation present in the NV/C pigs at 1 dpi noticeably diminished by 2 dpi. Vaccinated/challenged pigs also demonstrated prominent peribronchiolar lymphocytic cuffing in contrast to light peribronchiolar cuffing observed in the NV/C group. Both challenge groups exhibited more severe epithelial necrosis of the bronchioles and occasional bronchi at 2 dpi.
Microscopic lung lesion profiles were characterized by increased lymphocytic inflammation in the V/C group at 5 dpi. The alveolar luminal edema and neutrophils decreased or were replaced by large numbers of alveolar macrophages, sloughed pneumocytes, and cellular debris. Alveolar lumina were often collapsed by marked, widespread alveolar septal thickening by lymphocytes, macrophages, and type II pneumocyte hypertrophy and hyperplasia that was less prominent in the NV/C pigs at 5 dpi. Only the VAERD-affected pigs demonstrated broad peribronchiolar cuffs of densely packed lymphocytes (Fig. 10). Large numbers of lymphocytes expanded the bronchiolar lamina propria, displacing the affected epithelium and occasionally narrowing the bronchiolar lumen (Fig. 10). Neutrophils persisted in the lumen of a larger percentage of bronchi and bronchioles in the V/C pigs (Fig. 10) compared to the NV/C group, with occasional neutrophil exocytosis within bronchiolar epithelium. Both challenge groups demonstrated similar levels of bronchi and bronchiolar epithelial necrosis, which was often replaced by proliferative, columnar epithelium by 5 dpi.
Microscopic Trachea Lesions
Average trachea lesion scores in the V/C group were significantly higher (P ≤ .001) at 1, 2, and 5 dpi compared to NV/C pigs. Average trachea lesion scores for NV/C pigs were 0.16, 0.13, and 0.63 at 1, 2, and 5 dpi, respectively. NV/C pigs had minimal to mild attenuation of the tracheal epithelium with mild to moderate, multifocal, lymphocytic tracheitis at 1 and 2 dpi that became more pronounced at 5 dpi. In contrast, average trachea lesion scores for V/C pigs were 1.41, 1.88, and 1.84 at 1, 2, and 5 dpi, respectively. Vaccinated/challenged pigs demonstrated multifocal regions of trachea epithelial attenuation with moderate lymphocytic and suppurative infiltrates at 1 dpi. At 2 and 5 dpi, tracheal lesions became more severe and widespread to include locally extensive, lymphoplasmacytic and necrosuppurative tracheitis with regional loss of cilia and severe lymphoplasmacytic infiltration of tracheal submucosa (Fig. 11). Occasionally, aggregates of neutrophils were apparent in trachea epithelium in the V/C pigs. Lesions were similarly minimal in both challenge groups at 21 dpi. Microscopic lesions were not evident in trachea in the NV/NC pigs at any time during the study.
Trachea, pig No. 117, vaccinated/challenged (V/C) group. Pigs with vaccine-associated enhanced respiratory disease (VAERD) demonstrated diffuse necrotizing and lymphocytic tracheitis (arrows). Note the marked, subepithelial lymphocytic infiltration and epithelial sloughing (asterisk). Hematoxylin and eosin, 400×.
Lung Immunohistochemistry
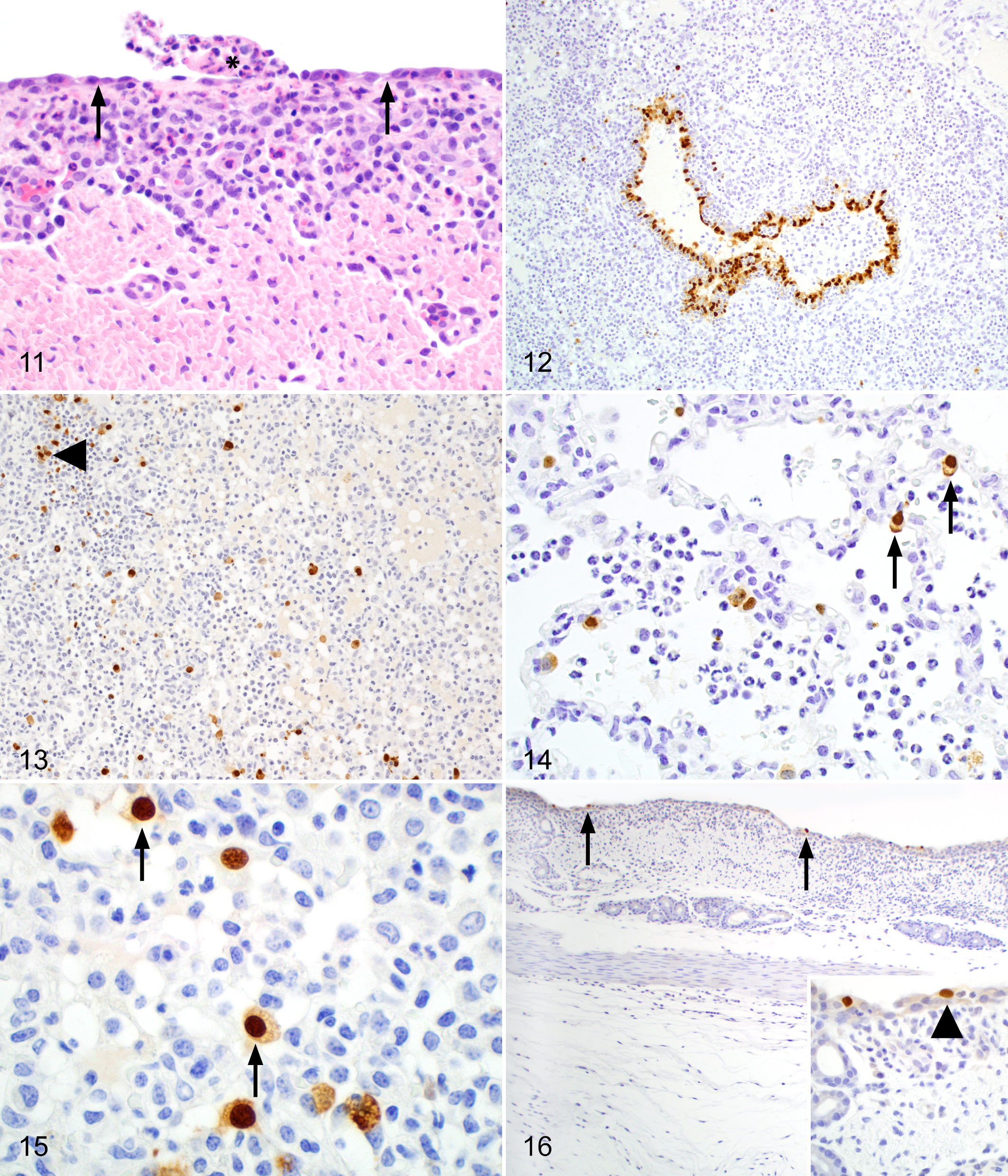
Influenza A virus antigen was detected by IHC as brown, granular, nuclear, and cytoplasmic signals in all pigs challenged with pH1N1 at 1 dpi (Fig. 12). At 2 dpi, IAV signals were detected in 8 of 8 V/C pigs and in 6 of 8 NV/C pigs. In contrast, at 5 dpi, IAV antigen was detected in 6 of 8 V/C pigs and in 8 of 8 NV/C pigs. Influenza A virus signals in bronchioles of affected lung tissue from both challenge groups consisted of aggregates of immunoreactive epithelial cells that were attenuated, necrotic, or occasionally morphologically unaffected. In addition, rare individual immunoreactive cells were identified in the epithelium of bronchi. Scores for airway epithelial IAV signals did not differ significantly between the challenge groups at 1 dpi (data not shown). However, the average score for airway epithelial signals in the V/C group was significantly higher (P ≤ .01) than the score for the NV/C pigs at 2 dpi. Conversely, the average score was significantly higher (P ≤ .05) in the NV/C pigs at 5 dpi compared to the V/C group.
IAV signals in the alveolar septa or lumen, although present in both challenge groups, were more widely distributed in the V/C pigs at 1 and 2 dpi (Fig. 13), in which average IHC scores specifically for alveolar signals were significantly higher (P ≤ .05) (data not shown). Similar levels of alveolar septa and lumen signals were observed in both challenge groups by 5 dpi. No influenza antigen was detected in either challenge group at 21 dpi or in the NV/NC pigs at any time during the trial. Influenza A virus antigen was identified as cytoplasmic or nuclear signals in hypertrophic and hyperplastic type II pneumocytes in the alveolar septum (Fig. 14) or within individual cells confined to the alveolar lumen and interpreted as alveolar macrophages or sloughed pneumocytes (Fig. 15).
Trachea Immunohistochemistry
Moderately abundant immunoreactive trachea epithelial cells contained brown cytoplasmic or nuclear signals observed in necrotic (Fig. 16) or histopathologically unaffected epithelium. IAV antigen was not detected in the submucosa. Scores for IAV signals in trachea were significantly higher in the V/C pigs at 2 dpi but similar to the NV/C pigs at both 1 and 5 dpi (data not shown).
Virus Levels in Lung and Nasal Secretions
No IAV was detected in nasal secretions from challenged pigs in either group at 0 and 1 dpi. At 2 dpi, 7 of 8 V/C pigs and 3 of 8 NV/C pigs were shedding virus. At 3 and 5 dpi, all remaining pigs challenged with pH1N1 were shedding virus in nasal secretions. Virus titers from nasal swabs at 2 and 3 dpi were not significantly different between the V/C and NV/C pigs. In contrast, at 5 dpi, titers in swabs from the NV/C pigs were significantly higher (P ≤ .01) than from V/C pigs (Table 4). Virus was not being shed in nasal secretions from any challenged pig remaining at 21 dpi.
Comparison of pH1N1 Virus Titers in Bronchoalveolar Lavage Fluid (BALF) and Nasal Swabs in Vaccinated/Challenged (V/C), Non-Vaccinated/Challenged (NV/C), and Non-Vaccinated/Non-Challenged (NV/NC) Pigs at 1, 2, and 5 Days After pH1N1 Inoculation (dpi; Data Presented as the Mean Virus Titer ± Standard Error of the Mean)
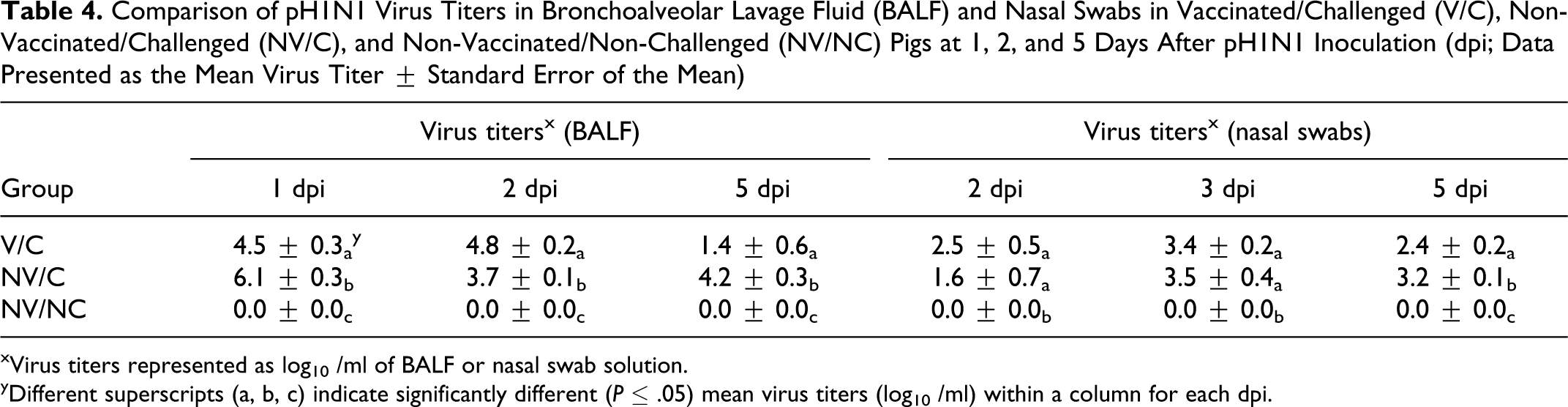
xVirus titers represented as log10 /ml of BALF or nasal swab solution.
yDifferent superscripts (a, b, c) indicate significantly different (P ≤ .05) mean virus titers (log10 /ml) within a column for each dpi.
Influenza A virus was isolated from the BALF of all challenged pigs at 1 and 2 dpi and from 8 of 8 NV/C pigs and 4 of 8 V/C pigs at 5 dpi. Virus was not detected in any pigs remaining from either group at 21 dpi. Mean virus titers in the BALF were significantly higher (P ≤ .05) in the NV/C pigs than in V/C pigs at 1 and 5 dpi. In contrast, mean BALF virus titers were significantly higher (P ≤ .0001) in the V/C pigs compared to NV/C pigs at 2 dpi (Table 4). Virus was not detected at any time in BALF or nasal swabs in the NV/NC pigs.
Nucleoprotein ELISA, Hemagglutination-Inhibition, and Serum Neutralization Tests
Pigs in the NV/NC group remained seronegative throughout the study. Anti-nucleoprotein ELISA (data not shown) and anti-MN08 HI antibody titers were detected in serum exclusively in the V/C pigs on the day of challenge (0 dpi). The geometric mean reciprocal HI titer was 37.5 (range, 10-160). Unvaccinated pigs did not demonstrate detectable anti-MN08 HI antibodies. None of the pigs demonstrated detectable anti-pH1N1 HI antibodies at 0 dpi. Serum neutralization titers against MN08 virus were only observed in the V/C pigs. The geometric mean reciprocal titer was 951.0 at 0 dpi (range, 80-5,120). Serum neutralization titers were not observed against the pH1N1 virus in the V/C, NV/C, or NV/NC groups at 0 dpi.
IgG Antibody Response to Whole Virus MN08 and pH1N1
Serum anti-MN08 and cross-reacting anti-pH1N1 IgG antibodies were detected in V/C pigs at 0 dpi, the day of challenge (data not shown). Vaccinated pig serum anti-MN08 and –pH1N1 IgG OD levels were significantly higher (P ≤ .0001) than the NV/C and NV/NC IgG OD levels. Serum IgG OD levels to either antigen were not significantly different between NV/C and NV/NC pigs.
Local Anti-Viral and Pro-Inflammatory Cytokine Response
Cytokine concentrations in BALF were evaluated on days 1, 2, 5, and 21 post challenge. At 1 dpi, concentrations of IL-1β, IL-8, and TNF-α were significantly higher in the V/C pigs compared to the NV/NC pigs (Figs. 17, 18). This trend continued on dpi 2, although not all concentrations in V/C pigs were significantly greater than NV/C or NV/NC pigs. Pigs in the V/C group had significantly (P < .05) higher concentrations of all cytokines at 5 dpi compared to NV/C and NV/NC pigs (Figs. 17, 18). At 21 dpi, concentrations of proinflammatory cytokines were considerably lower in all groups compared to dpi 5 and were comparable to dpi 1 concentrations.
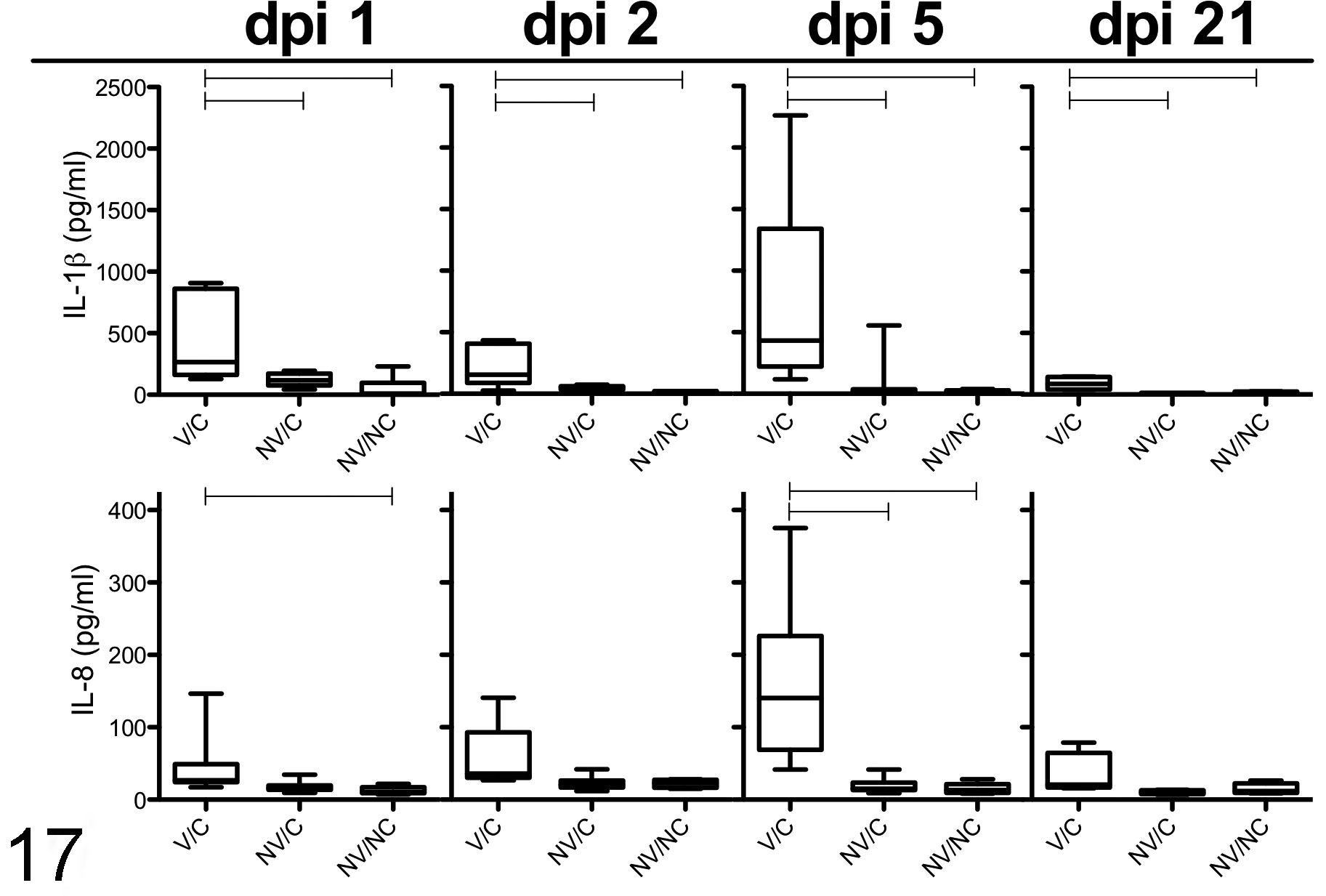
Cytokine IL-1β and IL-8 protein concentrations in bronchoalveolar lavage fluid (BALF) from vaccinated/challenged (V/C), non-vaccinated/challenged (NV/C), and non-vaccinated/non-challenged (NV/NC) pigs at 1, 2, 5, and 21 days after pH1N1 inoculation. Data presented as box plots demonstrating the median cytokine concentration (pg/ml), 5th and 95th percentile and standard deviation per group. Connecting horizontal lines are significantly different (P < .05).
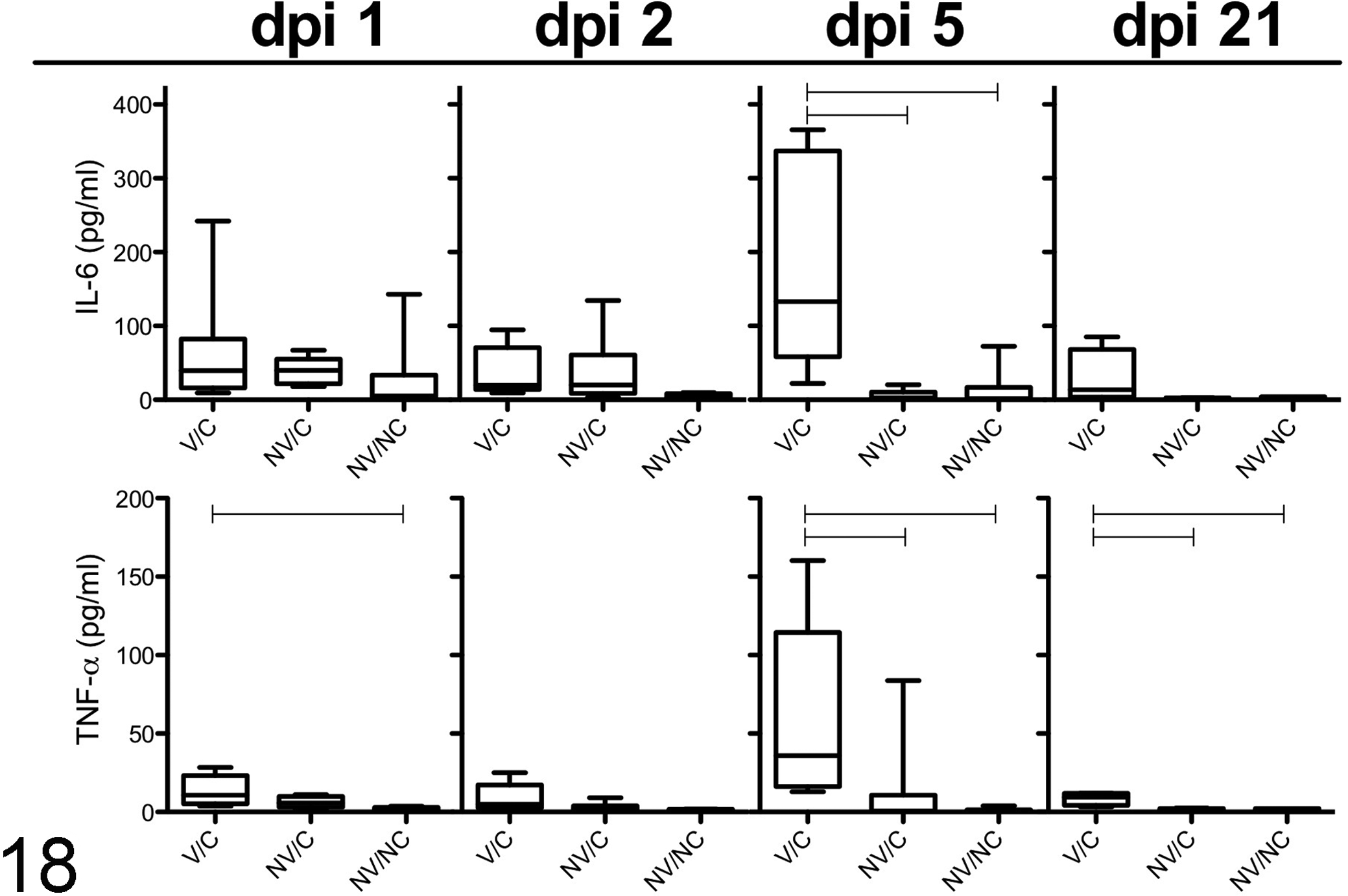
Cytokine IL-6 and TNF-α protein concentration in bronchoalveolar lavage fluid (BALF) of vaccinated/challenged (V/C), non-vaccinated/challenged (NV/C), and non-vaccinated/non-challenged (NV/NC) pigs at 1, 2, 5, and 21 days after pH1N1 inoculation. Data presented as box plots demonstrating the median cytokine concentration (pg/ml) per group with the 5th and 95th percentile and standard deviation. Connecting horizontal lines are significantly different (P < .05).
Discussion
Vaccine-associated enhanced respiratory disease was demonstrated in pigs administered a δ-cluster inactivated adjuvanted IAV vaccine followed by challenge with pH1N1, a strain in which the HA is genetically related to the γ-cluster H1 swine viruses. 6,37 Vaccine-associated enhanced respiratory disease is a reproducible phenomenon characterized by an acute onset of severe respiratory disease reported previously in studies with vaccinated and challenged cross-bred pigs. 8,12,16,38 However, to further understand the pathogenesis and elucidate potential mechanisms associated with VAERD, it was necessary to evaluate the development of macroscopic and microscopic lung lesions at multiple times during the acute post-challenge phase of the inflammatory response.
Microscopically, uncomplicated influenza infection in swine affects the intrapulmonary epithelium causing a necrotizing bronchiolitis. 9,10 During the acute phase of infection, a transient neutrophil response to the epithelial necrosis is replaced by peribronchiolar and interstitial lymphocytes similar to what we describe in the NV/C pigs in this report. 23,29,34,46 However, V/C pigs demonstrated more severe microscopic lesions in sharp contrast to the NV/C group that we interpret as features exclusive to VAERD. Widespread interlobular and alveolar edema with marked hemorrhage were observed as early as 1 dpi, suggesting VAERD is an acute inflammatory reaction associated with the immune response to the vaccine (Figs. 7, 8). Interlobular edema and hemorrhage have been previously described for other porcine respiratory viruses such as PCV2 7 and PRRSV. 11 To our knowledge, the enhanced pulmonary lesions we observed in VAERD-affected pigs are unique compared to lesions associated with acute influenza virus infection described previously in naïve pigs 34 and may be a component of the pathogenesis of VAERD. In addition, a marked suppurative inflammatory response in the V/C pigs extended beyond affected bronchi and bronchioles to include widespread regions of the alveoli in affected lobules at 1 dpi (Fig. 7). The hemorrhage and edema persisted at 2 dpi in the V/C group with a persistent neutrophil response that had subsided as expected in the NV/C pigs, suggesting another difference between VAERD-affected and non-vaccinated/challenged pigs (Fig. 9). It is unknown whether the elevated lung consolidation in the V/C pigs may have been elicited by the abundant neutrophil response in both intrapulmonary airways and alveoli, although they have been implicated as contributors to the clinicopathologic effects of influenza virus infection. 17,33
At 2 and 5 dpi, microscopic lung lesions continued to increase in severity in the V/C pigs and correlated with the elevated macroscopic lung consolidation (Fig. 2). Widespread alveolar septal thickening was characterized by intense lymphocyte and macrophage infiltration of alveolar septa and lumina, prominent type II pneumocyte hypertrophy and hyperplasia, and variable numbers of neutrophils (Fig. 9). Additional lesions characteristic of VAERD included thick peribronchiolar lymphocytic cuffs and marked infiltration of lymphocytes in the bronchiolar lamina propria (Fig. 10). The pronounced lymphocyte inflammatory response in the lung appears to be a consistent lesion associated with VAERD in swine. Our data also suggest the trachea undergoes an enhanced lymphocytic inflammatory response similar to the lungs representing another component of VAERD. Interestingly, V/C and NV/C pigs demonstrated similar levels of necrotizing bronchitis and bronchiolitis throughout the study, reflecting the effects of influenza virus replication in respiratory epithelium. This is consistent with the lack of cross-protective vaccine immunity observed in the V/C pigs. However, viral titers in BALF or nasal swabs were not correlated with enhanced lesions demonstrated in the V/C pigs as the acute, post-inoculation period progressed and elevated viral replication does not explain the pathogenesis of VAERD.
Pro-inflammatory cytokines play an important role in the pathogenesis of influenza infection in swine and have been correlated with viral replication and clinical signs 35 as well as increased lung lesions and neutrophil infiltration. 34 In this report, TNF-α, IL-1β, IL-6, and IL-8 BALF concentrations were elevated in the V/C pigs compared to the NV/C pigs. A similar pro-inflammatory cytokine response was previously described by our group, 8 suggesting elevated pro-inflammatory cytokines may be characteristic of VAERD and contribute to the development of acute lesions observed in the V/C pigs. Elevated IL-8, a neutrophil chemotactic cytokine, may have contributed to the prominent and prolonged suppurative response in alveoli, bronchi, and bronchioles in V/C pigs. In addition, previous reports have shown a positive correlation between elevated TNF-α and IL-1β with neutrophil recruitment to the lungs, suggesting both cytokines may have been involved in the neutrophil response described in this report. 34 A specific mechanism, such as Fc-receptor or complement mediated activation, may have contributed to the exaggerated cytokine response detected in the V/C pigs. Mechanisms associated with VAERD remain unknown but under investigation at this time.
VAERD in swine has been reproduced under experimental conditions although it may remain undiagnosed in field situations where mixed viral and bacterial respiratory infections are common. However, an antigenically diverse population of IAVs combined with increased IAV vaccine usage in swine creates optimal conditions for vaccine-virus mismatch and potential VAERD. Seronegative children or mice administered an inactivated RSV vaccine followed by exposure to wild type RSV demonstrated enhanced respiratory disease similar to what we describe for VAERD in swine. 4,14,27 Preexisting vaccine antibodies may enhance other viral infections in humans. Atypical measles resulted from the use of formalin-inactivated vaccines administered to infants and children who later developed severe forms of the disease when exposed to wild type virus, suggesting a similar mechanism to enhanced disease associated with vaccine immunity to RSV. 5
Our data suggest VAERD results from a multifactorial mechanism that includes cross-reactive antibodies (IgG isotype) to conserved or non-neutralizing epitopes elicited by inactivated vaccines in the absence of HI or neutralizing antibodies. Non-neutralizing antibodies induced by the vaccine may play a role in antibody dependent cell-mediated cytotoxicity or antibody dependent enhancement. Antibodies involved in antigen-antibody complexes or complement fixation may have also contributed to the clinical disease and lung lesions we observed in this study. A previous study in vaccinated swine described an association between non-neutralizing M2 antibodies and an NP-specific lymphoproliferative response with enhanced clinical disease. 12 The data suggested the cellular immune response (lymphocytes), in addition to antibodies, may have contributed to disease enhancement. The V/C pigs in the current study also demonstrated an NP antibody response to the inactivated vaccine, although the role for NP-induced antibodies or cellular responses in VAERD requires further characterization.
A recent epidemiological report from Canada described an association between prior immunization with the 2008-2009 trivalent inactivated seasonal influenza vaccine and an increased risk of medically attended pH1N1 illness in humans. 31 Although differences exist in the conditions involved in the human and swine diseases, data from human subjects suggest mechanisms that may correspond to VAERD in swine. 22 A recent report described a correlation between severe pandemic 2009 H1N1 respiratory disease in middle-aged adults and the presence of preexisting serum antibodies that cross-reacted with the virus but did not confer protection. 22 The report described the presence of pulmonary immune complexes consisting of low avidity antibodies to the 2009 pH1N1 antigen in association with high levels of activated complement in people with severe respiratory disease. A detailed description of potential VAERD mechanisms is beyond the scope of this report; however, studies are in progress to determine the pathogenesis of VAERD in swine.
In summary, this report demonstrates an enhanced disease process in vaccinated pigs challenged with a heterologous influenza A virus resulting in more severe macroscopic and microscopic lung lesions compared to pigs that did not receive prior vaccination but challenged with the same virus. Cross-reactive antibodies that lack neutralizing capabilities but have the ability to activate complement may play an initiating role in the process since features of the phenomenon occur very rapidly, as early as 1 dpi. However, multiple contributing factors, including dysregulation of proinflammatory cytokines and the various immune cell types identified in the cellular infiltrates in VAERD-affected lungs, need further investigating to determine the cause of the immunopathology associated with VAERD in swine.
Footnotes
Acknowledgements
The authors thank Michelle Harland, Gwen Nordholm, and Hillary Horst for technical assistance; Brian Pottebaum, Jason Huegel, and Jason Crabtree for assistance with animal studies; and Ann Greazel for assistance with graphic design. Assistance with microbiologic screening from Dr. Susan Brockmeier is appreciated. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Financial support for this study was provided by USDA-ARS.
