Abstract
Canine cutaneous mast cell tumors (MCT) of different histological grades have distinct biological behaviors. However, little is known about underlying molecular mechanisms that lead to tumor development and increasing malignancy with higher tumor grade. Recent studies have identified the interleukin-2 receptor (IL-2R) subunits CD25 and CD2 as markers that distinguish nonneoplastic from neoplastic mast cells in human systemic mastocytosis. In this study, their potential as a marker for canine MCT and their possible impact on MCT carcinogenesis were evaluated. mRNA expression levels of both genes were compared between grade 1 (n = 12) and grade 3 (n = 8) MCT, and protein expression levels of CD25 were compared in 90 MCT of different tumor grades. mRNA expression levels of both CD25 and CD2 were upregulated in grade 3 MCT. In contrast, CD25 protein was expressed by fewer tumor cells and at decreased levels in grade 3 tumors, while most grade 1 MCT had strong CD25 protein expression. Moreover, CD25 was not expressed by nonneoplastic, resting cutaneous mast cells, while few presumably activated mast cells in tissue samples from dogs with allergic dermatitis had weak CD25 expression. Taken together, these findings suggest that CD25 may play a critical role in early MCT development and may be a stimulatory factor in grade 1 MCT, while grade 3 MCT seem to be less dependent on CD25. Because of the low number of CD25-positive tumor cells in high-grade tumors, the usefulness of CD25 as a tumor marker is, however, questionable.
Keywords
CD25 (IL-2Rα) is the α-subunit of the membrane-bound interleukin-2 receptor (IL-2R), which is mainly expressed by regulatory T cells. 25,28 In addition, CD25 expression was also identified on natural killer cells, activated B and T cells and, some dendritic cells. 4,28,37 Functionally, CD25 is essential for IL-2R signaling by binding the ligand interleukin-2 (IL-2). 39 Stimulation of IL-2R promotes T-cell proliferation, differentiation, and cell survival. 25,28
Recently, CD25 has been proposed as a reliable marker for distinguishing neoplastic from nonneoplastic human mast cells in patients with systemic mastocytosis (SM). 7,23,34 In fact, CD25 is expressed by neoplastic bone marrow, gastrointestinal, and cutaneous mast cells from SM patients but not by normal and reactive mast cells in corresponding tissues. 6,10,11 Similarly, the T-cell adhesion molecule CD2 is also expressed by neoplastic mast cells from most SM patients but not by nonneoplastic cells. 7,16 Accordingly, CD25 and/or CD2 expression is currently defined by the World Health Organization as a minor diagnostic criterion for human systemic mastocytosis. 13,35
In dogs, no specific diagnostic markers are available that discriminate between neoplastic and nonneoplastic mast cells. c-KIT mutations have been proposed as a suitable marker for canine cutaneous mast cell tumors (MCT), and they are significantly associated with higher tumor grades and increased malignancy. 42,46 However, several studies have revealed that only 9%–17% of canine MCT carry relevant c-KIT mutations. 24,27,43,46 Aberrant cytoplasmic protein expression of the commonly plasma membrane-bound KIT is also associated with malignant MCT behavior but not present in all tumors. 41,42
These restrictions in the availability of MCT markers besides histological grading are paralleled by the large unclear molecular mechanisms of MCT development and malignant progression. Although KIT signaling of mutated c-KIT appears to be a contributor to MCT growth, 43 the low incidence of c-KIT mutations indicates that further molecular mechanisms contribute to MCT carcinogenesis and malignancy.
In the present study, we hypothesized that CD25 is expressed by canine MCT and is involved in MCT development and carcinogenesis. We further speculated on whether CD25 may serve as a tumor marker that distinguishes neoplastic from nonneoplastic canine mast cells. To this end, the CD25 mRNA and protein expression levels of different tumor grades were analyzed in canine MCT and compared with nonneoplastic resting, and activated connective tissue mast cells.
Materials and Methods
Case Selection
Ninety cutaneous MCT from 90 dogs submitted to the Department of Veterinary Pathology, Freie Universität Berlin, Germany, were included in this study. All cases were graded according to the Patnaik grading system 31 and the Kiupel grading system. 18 Clinical data of the dogs included breed, age, sex, and number as well as location of tumors. All samples were formalin-fixed and paraffin-embedded (FFPE) and stained with hematoxylin and eosin (HE) according to routine diagnostic procedures. In questionable cases, additional mast cell-specific toluidine staining was performed to confirm the diagnosis. Additional fresh-frozen material was obtained from 20 of the tumors and stored at –80°C.
The study was approved by the local animal welfare officer. Surgical resection of the tumors was part of the therapy according to the welfare of the animals and to the state of the art of medical science under full anesthesia. The study therefore had no influence on the common diagnostic or therapeutic measures usually applied on animals with MCT, and inclusion into the study did not induce any additional treatments, pain, or discomfort-inducing manipulations during the entire study.
Extraction of DNA and mRNA
Eight-micrometer sections were cut from fresh-frozen material and HE stained to ensure a high content of tumor cells. Subsequently, 100–150 sections of 8 μm each were cut from the center of the tumor and used for DNA (NucleoSpin Tissue; Macherey-Nagel, Düren, Germany) or mRNA (modified NucleoSpin RNA II; Macherey-Nagel) extraction. Likewise, 100 sections of 2 μm each were collected from FFPE tissue for DNA and mRNA extraction according to a standard protocol. 44 cDNA was generated from 100 ng of mRNA using the iScript cDNA synthesis kit (BioRad, Hercules, CA).
c-KIT PCR Exon 11
Polymerase chain reaction (PCR) amplification for the detection of c-KIT mutations was performed using a previously described primer pair for c-KIT Exon 11 (Table 1). 15 A total PCR reaction volume of 50 μl was prepared, containing 5 μl cDNA, respectively, DNA, 20 pmol of each primer, 1.25 U of GoTaq polymerase (Promega, Madison, WI), 10 μl Green GoTaq Flexi Buffer (Promega), 5 μl of 2 mM dNTP-Mix (Fermentas, Burlington, Ontario, Canada) and 3 μl of 25 mM MgCl (Promega). Cycling conditions were 95°C for 5 minutes followed by 40 cycles at 94°C for 1 minute, 52°C for 2 minutes, 72°C for 2 minutes, and a final extension of 72°C for 5 minutes (modified from Webster et al 42 ). Amplified PCR products were size fractionated by agarose gel electrophoresis on a 2.5% ethidium bromide-stained agarose gel.
Primer Sequences Used for PCR and qPCR

Quantitative Real-Time Reverse Transcriptase PCR
The mRNA expression levels of CD25 and CD2 were compared between 12 grade 1 and 8 grade 3 MCT and 13 low-grade (LG) and 7 high-grade (HG) MCT, respectively. Specific primer pairs were designed for both target genes listed above and the 3 housekeeping (HK) genes ATP-synthase subunit 5B (A5B), hypoxanthine-phosphoribosyl transferase (HPRT), and ribosomal protein L32 (RP32), as shown in Table 1. 19 Quantitative real-time reverse transcriptase PCR (RT-PCR) and data analyses were performed using 96-well plates with triplicates of each cDNA sample and additional no-template controls with water instead of cDNA and were run on an MX3000P thermocycler (Stratagene, La Jolla, CA). The PCR assays had an efficiency of 95% to 101%, and specificity of amplification products was confirmed by sequence analysis. The total reaction volume of 15 μl contained 5 μl cDNA, 7.8 μl Maxima SYBR Green/ROX qPCR Master Mix (Fermentas), and 6 pmol of each primer. Cycling conditions were 95°C for 10 minutes followed by 40 cycles at 95°C for 30 seconds, 58°C for 1 minute, 72°C for 30 seconds, and a final extension of 73°C for 10 minutes. The gene of interest (GOI) mRNA expression levels were normalized to the geometric mean of the 3 HK genes and compared between grade 1 (GI) and grade 3 (GIII) MCT and LG and HG MCT, respectively, using the ▵Ct method. The calculated fold change (FC = 2⁁(GI – GIII); FC = 2⁁(LG – HG)) cutoff values were set at >1.5 for increased gene expression and <0.5 for decreased gene expression. Normal distribution and significance of differences in mRNA expression levels between tumor groups were confirmed by the Kolmogorov-Smirnov test and t-test.
Cloning of Canine CD25
Total mRNA extraction of fresh-frozen lymph node tissue and subsequent cDNA synthesis was performed as described above. Specific PCR primers were designed flanking the canine CD25 ORF (GenBank accession number AB501356; Table 1). Amplifying of PCR product was performed with a total reaction volume of 50 μl, containing 5 μl cDNA, 20 pmol of each primer, 1.25 U of High Fidelity PCR Enzyme Mix (Fermentas), 5 μl 10× High Fidelity PCR Buffer with 15 mM MgCl2 (Fermentas), and 5 μl of 2 mM dNTP-Mix (Fermentas). The PCR conditions were 94°C for 5 minutes followed by 35 cycles at 94°C for 30 seconds, 58°C for 30 seconds, 72°C for 1 minute, and a final extension of 72°C for 10 minutes. The PCR products were purified (Nucleo Spin Extract II; Macherey-Nagel) for gel electrophoresis on a 1.5% ethidium bromide-stained agarose gel and subsequent sequence analysis. The expression vector pcDNA3.1 was linearized by Eco32I-digestion for 5 minutes at 37°C. T-addition to 500 ng linearized vector was performed in a total reaction volume of 50 μl with 2.5 U GoTaq Flexi DNA Polymerase (Promega), 10 mM dTTP (Invitrogen, Carlsbad, CA), 5 μl 5× GoTaq Flexi Buffer (Promega), and 2 μl 25 mM MgCl2 for 30 minutes at 70°C. For A-addition, the purified PCR product was incubated with 5× Qiagen A-Addition Mix (Qiagen, Venlo, the Netherlands) for 30 minutes at 37°C. Ligation was performed with 1 U T4 DNA Ligase (Fermentas) for 1 hour at room temperature. Subsequently, 2 μl of ligation reaction mix was used to transform 50 μl of XL2-Blue Ultracompetent Cells (Stratagene) according to the manufacturer’s protocol. After incubation overnight at 37°C, colonies were harvested and subcultured, and plasmid DNA was isolated (NucleoBond Xtra Midi; Macherey-Nagel). Candidate clones were sequenced and aligned with a published CD25 sequence (GenBank accession number AB501356). For transfection experiments, HEK293 cells were grown on a 8-well chamber slide (Lab-Tek II Chamber Slide; Thermo Fisher, Waltham, MA) in DMEM high glucose with stable glutamine (Biowest, Logan, UT) supplemented with 10% fetal calf serum (Biowest) and kept at 5% CO2 and 37°C until 90% confluence. HEK293 cells were transfected with 500 ng CD25 ORF in pcDNA3.1 using TurboFect transfection reagent (Fermentas) according to the manufacturer’s protocol. Cells transfected with pcDNA3.1 vector alone served as negative controls. After 16 hours, cells were fixed with paraformaldehyde for 24 hours at 4°C for subsequent immunohistochemistry.
Immunohistochemistry
For immunohistochemical staining, 4- to 5-μm sections of FFPE tissue from 90 canine MCT were cut and deparaffinized in xylene and rehydrated in graded alcohols. Endogenous peroxidases were blocked by incubating sections in methanol with 3% hydrogen peroxide for 20 minutes. Antigen heat retrieval was performed in citric buffer for 12 minutes followed by a cooling period of 15 minutes. Slides were then incubated for 30 minutes with Roti-Immunoblock (Roth, Karlsruhe, Germany) and goat serum to block nonspecific antibody binding. Sections were incubated overnight in a humidity chamber at 4°C with one of the following antibodies: monoclonal mouse-anti-human CD25 (dilution 1:100, clone 4C9; Novocastra, Buffalo Grove, IL) or polyclonal rabbit-anti-human CD3 (dilution 1:800; Dako, Glostrup, Denmark). In each run, negative controls were incubated with irrelevant commercial mouse or rabbit immunoglobulins (BioGenex, Fremont, CA) instead of primary antibodies listed above. Two slides were incubated with CD25 antibody, which had been preincubated with 1 μg/100 μl of the corresponding peptide (IL-2 sRα; Sigma, St Louis, MO) for 30 minutes at room temperature as a preincubation control. Slides were then incubated with a biotinylated secondary goat-anti-mouse or goat-anti-rabbit antibody (dilution 1:200 each; Vector Laboratories, Burlingame, CA) for 30 minutes at room temperature. Immunolabeling was performed by an avidin-biotin-immunoperoxidase system (Vectastain Elite ABC Kit; Vector Laboratories). CD25 immunoreaction was visualized by HistoGreen-staining (Linaris) for 4 minutes at room temperature. Diaminobenzidine tetrahydrochloride (DAB; Merck, Darmstadt, Germany) was used for antigen visualization of CD3. Finally, all slides were counterstained with Mayer’s hematoxylin. To measure the number of CD25-positive cells within a single tumor, a semiquantitative evaluation system with 4 categories was established: less than 5% positive tumor cells, 5% to 39% positive tumor cells, 40% to 69% positive tumor cells, and 70% to 100% positive tumor cells. All slides were evaluated by 2 pathologists and classified into 1 of the groups. In case of diverse results, slides were reevaluated by both observers with a consent diagnosis. A statistical analysis of differences in semiquantitative protein expression between tumor groups was analyzed using the Kruskal-Wallis test.
Nine canine MCT FFPE-tissue samples (three grade 1, 2, and 3 each) were further investigated with a CD25/CD3 double immunolabeling. Sections of 4–5 μm were cut and pretreated as described above. Slides were incubated with a mixture of both primary antibodies, that is, monoclonal mouse-anti-human CD25 (dilution 1:100, clone 4C9; Novocastra) and polyclonal rabbit-anti-human CD3 (dilution 1:800; Dako), and incubated overnight at 4°C. Afterward, slides were first incubated with a biotinylated secondary goat-anti-rabbit antibody (dilution 1:200; Vector Laboratories) for 30 minutes at room temperature followed by 30 minutes of immunolabeling with an avidin-biotin-immunoperoxidase system (Vectastain Elite ABC Kit; Vector Laboratories). CD3 immunolabeling was then visualized by DAB. Subsequently, slides were incubated with a biotinylated secondary goat-anti-mouse antibody (dilution 1:200; Vector Laboratories) for 30 minutes at room temperature and immunolabeled with an avidin-biotin-alkaline phosphatase system (Vectastain ABC-AP Kit; Vector Laboratories) followed by Neufuchsin staining (Merck). Slides were finally counterstained with Mayer’s hematoxylin.
Chamber slides with CD25 and pcDNA3.1-transfected HEK293 cells were incubated with 1% TritonX in phosphate-buffered saline (PBS) for 5 minutes at room temperature. To prevent nonspecific antibody binding, slides were blocked with blocking buffer, containing PBS, goat serum, and 0.5% Tween 20 for 30 minutes at room temperature. Slides were incubated overnight at 4°C with monoclonal mouse-anti-human CD25 (dilution 1:100, clone 4C9; Novocastra). Afterward, slides were incubated with a biotinylated secondary goat-anti-mouse antibody (dilution 1:200; Vector Laboratories) for 30 minutes at room temperature. Subsequent immunolabeling was performed by an avidin-biotin-immunoperoxidase system (Vectastain Elite ABC Kit; Vector Laboratories), and immunoreaction was visualized by HistoGreen-staining (Linaris) for 4 minutes at room temperature and counterstaining with Mayer’s hematoxylin.
Immunofluorescence
Four representative canine MCT (two grade 1 and 2 each), 4 canine skin and 4 canine allergic dermatitis FFPE tissue samples were used for immunofluorescence double labeling to evaluate co-expression of KIT and CD25. Sections of 4–5 μm were cut and dewaxed in xylene followed by rehydration in graded alcohols. Blocking of endogenous peroxidases, antigen retrieval, and blocking of nonspecific antibody binding was performed as described above. Slides were then incubated overnight in a humidity chamber at 4°C with a mixture of both primary antibodies, that is, monoclonal mouse-anti-human CD25 (dilution 1:20, clone 4C9; Novocastra) and polyclonal rabbit-anti-human CD117 KIT (dilution 1:50; Dako). A mixture of irrelevant commercial mouse and rabbit immunoglobulins (BioGenex) instead of primary antibodies was used as negative control. For immunofluorescent detection, slides were incubated with an Alexa Fluor 488-labeled secondary goat anti-rabbit antibody (dilution 1:200; Invitrogen) for 1 hour at room temperature, washed with PBS, and then incubated with a Dylight 549-labeled secondary goat-anti-mouse antibody (dilution 1:200; Dianova, Barcelona, Spain) for 1 hour at room temperature. After a terminal wash, slides were covered with glycerin TBS.
Results
Comparison of MCT Grading Systems and Incidence of c-KIT Mutations
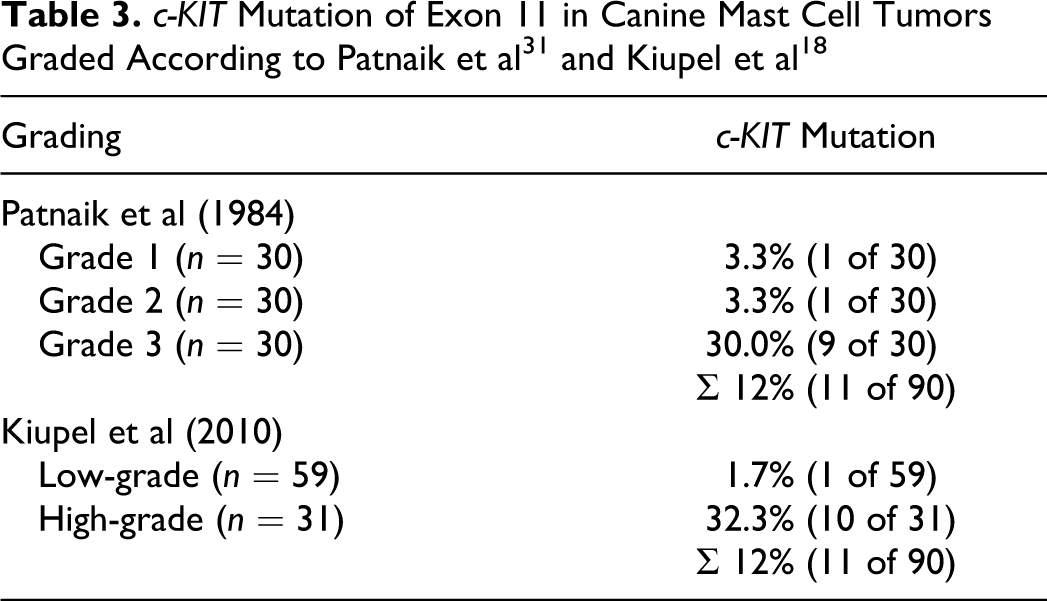
Ninety canine cutaneous MCT were included in this study, with 30 tumors of each Patnaik grade 1, 2, and 3. 31 All cases were also reexamined and graded according to the new 2-tier histologic grading system by Kiupel et al. 18 This reevaluation led to the diagnosis of 59 LG tumors (65.6%) and 31 (34.4%) HG tumors (Table 2). In detail, all 30 grade 1 tumors (100%), 25 grade 2 tumors (83.3%), and 4 grade 3 tumors (13.3%), according to the Patnaik grading system, represented LG tumors as evaluated by the Kiupel grading system. The HG tumor cohort according to the Kiupel grading system was composed of 5 Patnaik grade 2 (16.7%) and 26 Patnaik grade 3 (86.7%) MCT (Table 2).
The overall c-KIT mutation rate was 12%, with 11 of 90 tumors showing an internal tandem duplication in exon 11. c-KIT mutations were particularly prevalent in Patnaik grade 3 (n = 9; 30%) and Kiupel HG tumors (n = 10; 32.3%). c-KIT mutations were also present in 1 Patnaik grade 1 and 1 Patnaik grade 2 (each n = 1; 3.3%) MCT or 1 LG tumor (n = 1; 1.7%) following the Kiupel grading system (Table 3).
mRNA Expression Levels of CD2 and CD25
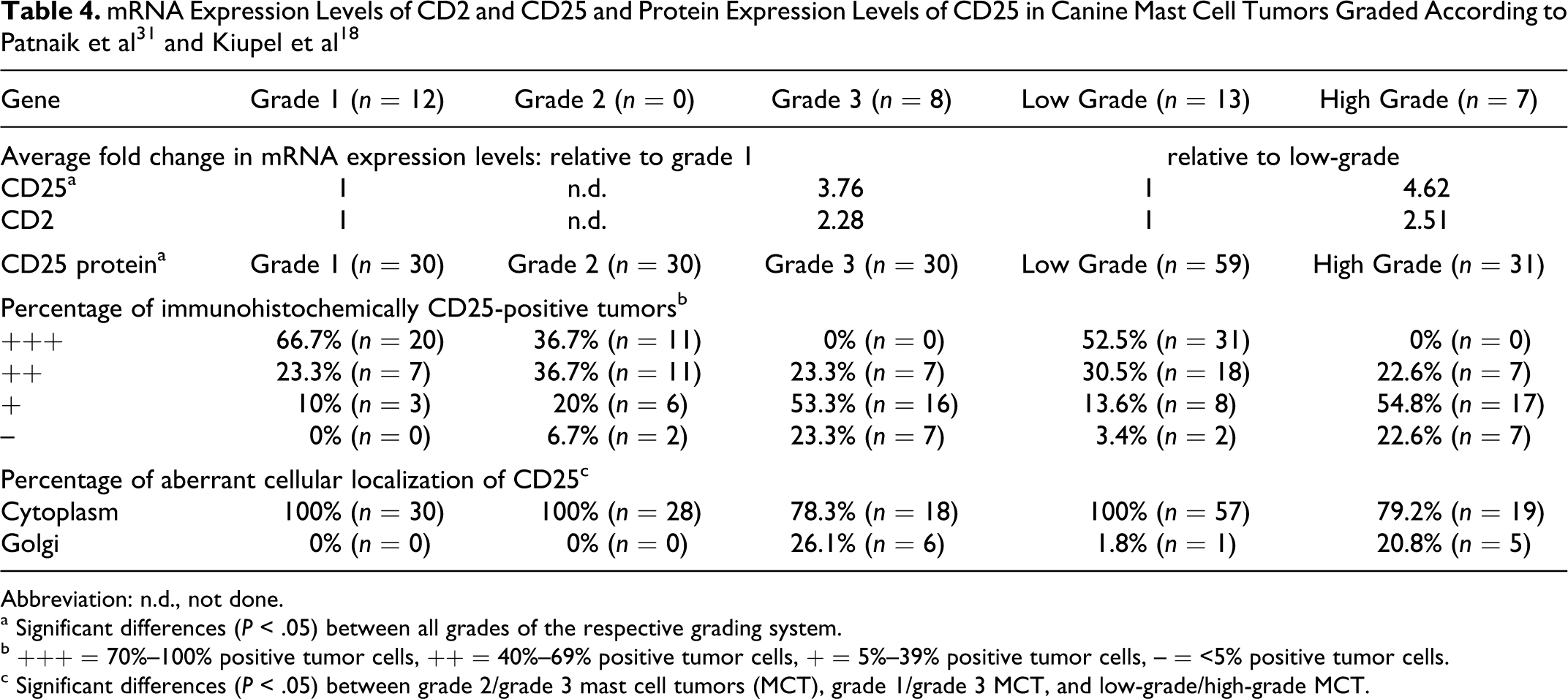
Using quantitative real-time RT-PCR, the median CD2 and CD25 mRNA expression levels of grade 1 and grade 3 MCT and HG and LG MCT, respectively, were compared. CD25 mRNA was significantly differentially expressed (P < .05) between Patnaik grade 1 and grade 3 MCT, with a 3.76-fold upregulation in grade 3 MCT (Mean (CtGOI – CtHK) = 7.75; SD = 1.48) compared with grade 1 MCT (Mean (CtGOI – CtHK) = 9.66; SD = 2.21). Similar significant differences (P < .05) were observed when comparing HG MCT (Mean (CtGOI – CtHK) = 7.46; SD = 1.33) and LG MCT (Mean (CtGOI – CtHK) = 9.66; SD = 2.12) with a 4.62-fold upregulation in high-grade tumors. The CD2 mRNA level was 2.28-fold upregulated in grade 3 MCT (Mean (CtGOI – CtHK) = 4.05; SD = 1.84) compared with grade 1 MCT (Mean (CtGOI – CtHK) = 5.24; SD = 1.87) and 2.51-fold upregulated in HG MCT (Mean (CtGOI – CtHK) = 3.90; SD = 1.94) compared with LG tumors (Mean (CtGOI – CtHK) = 5.23; SD = 1.79; Table 4). However, differences were not significant (P < .05).
Abbreviation: n.d., not done.
a Significant differences (P < .05) between all grades of the respective grading system.
b +++ = 70%–100% positive tumor cells, ++ = 40%–69% positive tumor cells, + = 5%–39% positive tumor cells, – = <5% positive tumor cells.
c Significant differences (P < .05) between grade 2/grade 3 mast cell tumors (MCT), grade 1/grade 3 MCT, and low-grade/high-grade MCT.
Protein Expression and Cellular Localization of CD25
Cross-reactivity of the applied mouse-anti-human CD25 antibody to canine CD25 was demonstrated by immunohistochemistry of canine CD25-transfected HEK293 cells. Canine CD25-transfected cells showed a distinct membrane staining of CD25 (Fig. 5a). In contrast, cells transfected with the empty pcDNA3.1 vector had no CD25 expression (Fig. 5b).
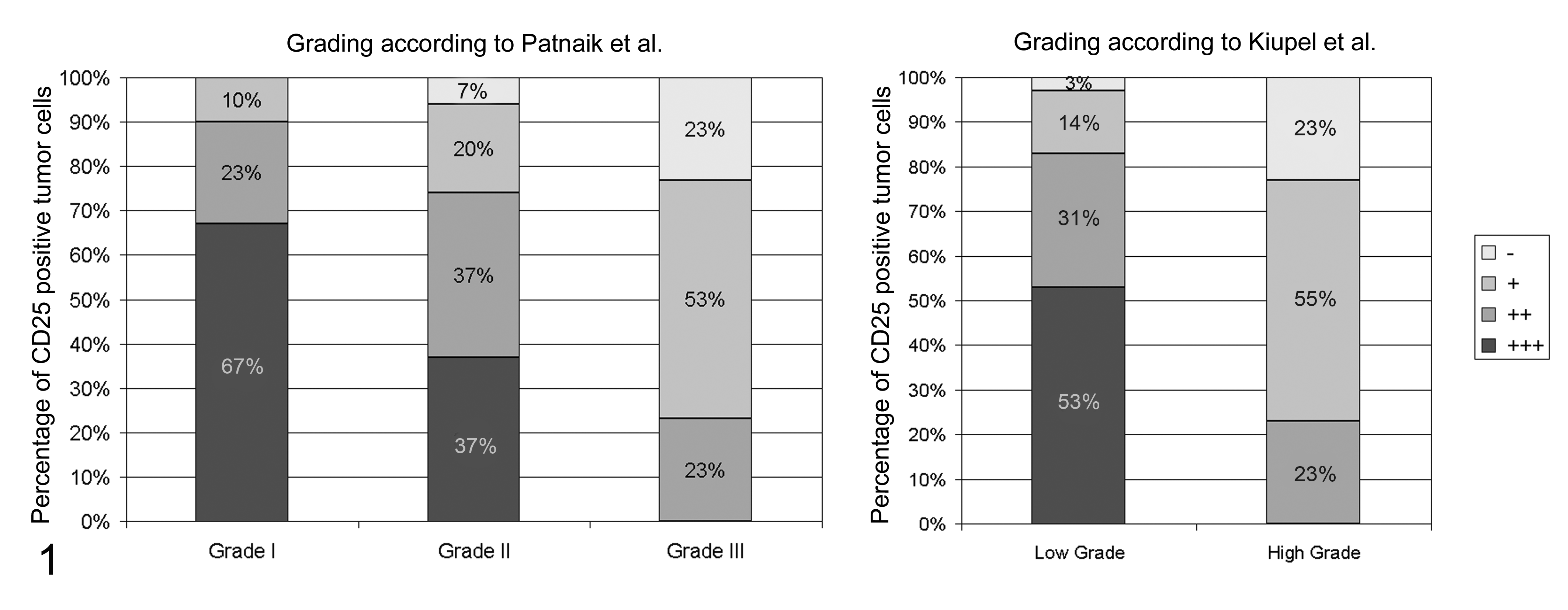
Percentage of immunohistochemically CD25 positive tumor cells in 90 canine cutaneous mast cell tumors according to the Patnaik and Kiupel grading systems. +++ = 70-100% positive tumor cells, ++ = 40-69% positive tumor cells, + = 5-39% positive tumor cells, − = < 5% positive tumor cells. The percentage of tumor cells expressing CD25 is negatively correlated with tumor grade in both grading systems.
Differences in CD25 protein expression levels and cellular localization between tumor grades were analyzed by immunohistochemistry in 90 canine cutaneous MCT (Table 4). The number of positive cells within a single tumor was measured semiquantitatively and categorized into 4 groups: less than 5% positive tumor cells, 5% to 39% positive tumor cells, 40% to 69% positive tumor cells, and 70% to 100% positive tumor cells. CD25 protein was expressed in 30 of 30 grade 1 MCT (100%), 28 of 30 grade 2 MCT (94%), and 23 of 30 grade 3 MCT (77%). Grade 1 tumors had the highest percentage of CD25-positive tumor cells with 20 of 30 grade 1 tumors having 70%–100% positive tumor cells, whereas 16 of 30 grade 3 tumors had only 5%–30% positive tumor cells. In comparison 11, grade 2 tumors each had 70%–100% and 40%–60% positive tumor cells (Table 4; Fig. 1). This decreased CD25 expression with increasing tumor grade was confirmed by comparing LG versus HG MCT. Accordingly, 31 of 59 LG tumors had 70%–100% positive tumor cells, whereas none of the 31 HG tumors had more than 60% positive tumor cells (Table 4; Fig. 1). These differential protein expression levels were significant (P < .05) between all tumor groups according to the Patnaik and the Kiupel grading systems. Interestingly, an aberrant cytoplasmic localization of CD25 in addition to the normal membrane-bound protein expression was observed to varying degrees in MCT of different grades. A diffuse cytoplasmic protein expression was detected in all grade 1, grade 2, and LG MCT (Table 4; Figs 2–3). A focal cytoplasmic staining pattern within the Golgi region was detected only in dedifferentiated MCT, with 26% grade 3 and 21% HG MCT, respectively (Table 4; Fig. 4).
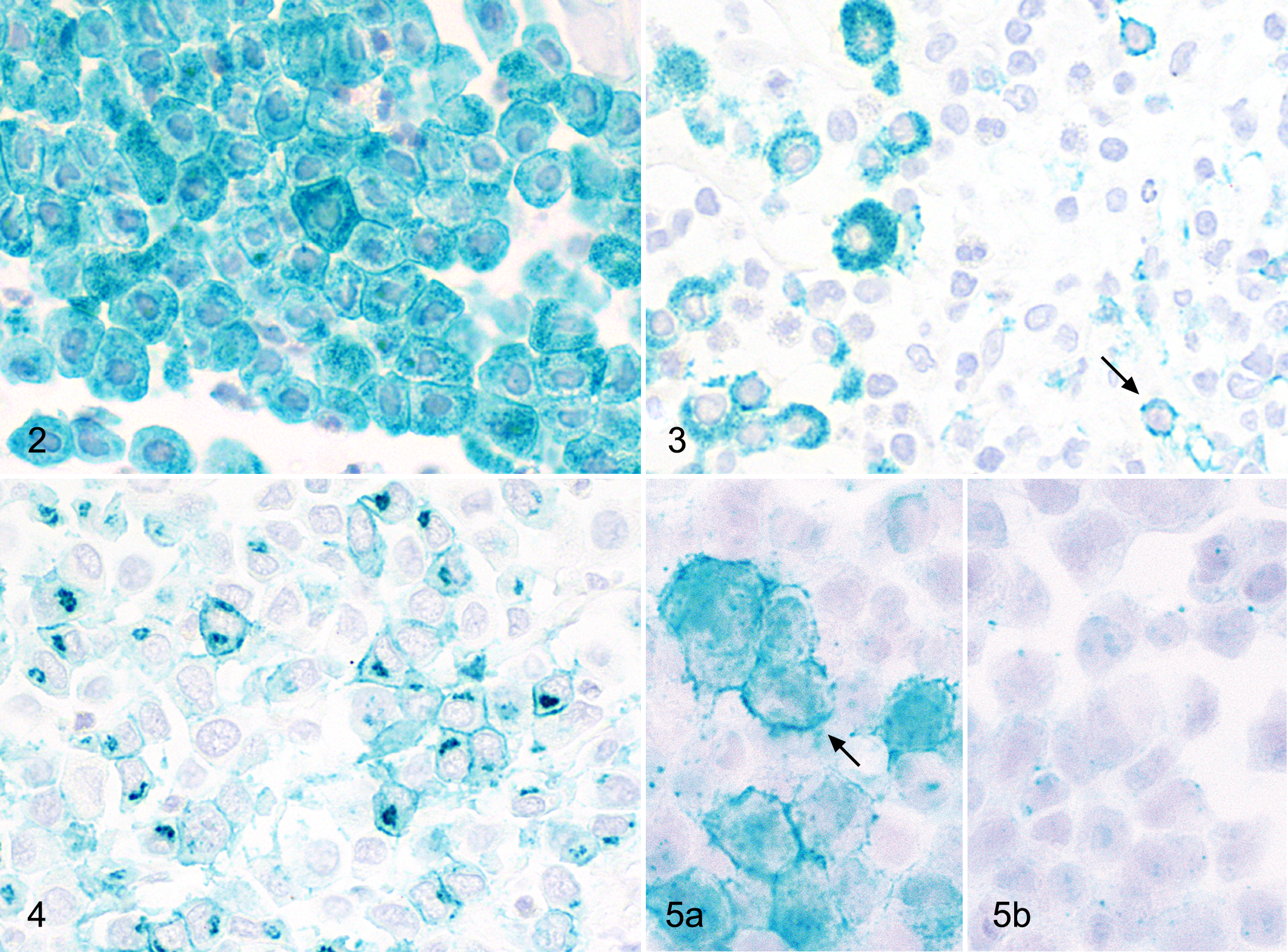
Mast cell tumor grade I, dog. Diffuse cytoplasmic and membrane-bound CD25 protein expression in 100% of tumor cells. Immunohistochemistry for CD25, chromogen HistoGreen, Hematoxylin counterstain.
CD25 Co-localization with CD3 and KIT
To test whether CD25 signals in MCT were due to infiltrating lymphocytes, all tumors were analyzed using CD3 immunolabeling. Grade 3 and HG tumors had a marked lymphocytic infiltration with up to 15% CD3-positive T cells within the tumor, whereas grade 1, grade 2, and LG tumors had less than 5% CD3-positive T cells. A CD25/CD3 double immunolabeling confirmed that virtually all CD25-positive cells did not co-express CD3, indicating that only a few CD25-positive T cells contribute to CD25 expression in MCT.
To test for expression of CD25 in nonneoplastic, resting, and reactive connective tissue mast cells, a CD25/KIT double immunofluorescence staining was performed on 4 tissue samples from canine healthy skin, 4 samples from canine allergic dermatitis, and 4 representative canine MCT (two grade 1 and 2 each). All MCT had a consistent co-expression of cytoplasmic CD25 and membrane-bound and/or Golgi KIT staining pattern (Fig. 6). In contrast, none of the nonneoplastic mast cells in the healthy skin specimens as identified by positive KIT staining had CD25 expression (Fig. 8). Interestingly, while most reactive mast cells in allergic dermatitis specimens had no CD25 labeling, up to 5% of reactive mast cells had CD25/KIT co-expression (Fig. 7).
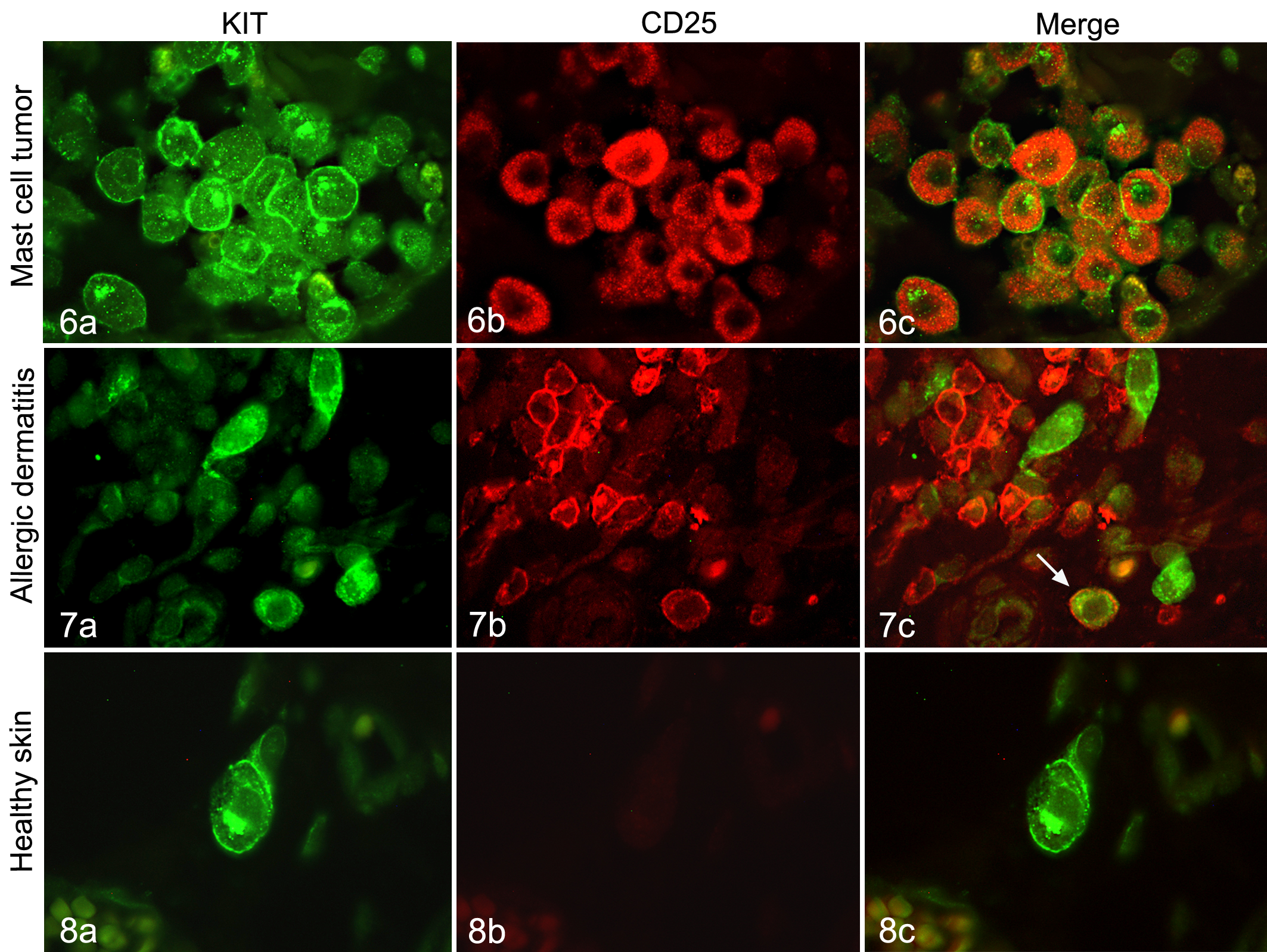
Mast cell tumor grade 1, dog. The KIT-positive (a; green) tumor cells co-express CD25 (b; red), shown as CD25/KIT double labeling (c). Immunofluorescence for CD25 and KIT.
Discussion
In the present study we hypothesized that the IL-2R subunit CD25 is expressed by canine MCT and may play a role in MCT development and malignancy and may also be a potential marker that distinguishes neoplastic from nonneoplastic canine mast cells.
The study identifies CD25 expression in canine cutaneous MCT depending on tumor grade, while nonneoplastic, resting connective tissue mast cells do not express CD25. These findings correlate in some aspects with recent findings in human medicine, where CD25 is a reliable marker for neoplastic mast cells in patients with SM, which is not expressed either in connective tissue or mucosal or bone marrow-derived, resting, or reactive mast cells. 10,23,34,36 The sensitivity to detect SM by CD25 expression was 80% to 100% and comparable to the present findings with 81 of 90 MCT being positive for CD25. 2,23,34 However, particularly grade 3 and HG canine MCT had partially only few tumor cells expressing CD25, a finding that was also described by Hollmann et al, 11 who found in all cases only a subset of dermal mast cells positive for CD25. In both human and canine tumors, this obviously reflects the heterogeneity of neoplastic cells within a single tumor.
An aberrant cytoplasmic protein expression in addition to the normal membrane-bound CD25 was observed in MCT of all grades. This expression pattern is uncommon but has been described previously in human lung cancer and lymphomas. 29 The reason for this aberrant distribution in human and canine tissues is unclear.
Of note, a gradual decrease and loss of CD25 expression was found in higher-grade and thus more dedifferentiated MCT. This suggests a role in initial MCT development from nonneoplastic to neoplastic mast cells rather than in malignant progression. A similar decreased receptor expression with increasing malignancy has been described in several canine tumors before, including the serotonin receptor on canine MCT and several growth factor receptors on metastatic canine mammary carcinomas. 8,20 –22 This down-regulation of mostly proliferative but also inhibitory receptors therefore seems to be a common mechanism in canine tumors and reflects the increasing independency of malignant tumors from external proliferation stimuli when compared with benign tumor variants or nonneoplastic cells.
On the other hand, CD25 expression may also be associated with general mast cell activation since up to 5% reactive mast cells in allergic dermatitis express CD25 as well. This hypothesis would also lead to the assumption that there could be a hypothetical link between mast cell activation and tumor development. The exact function and molecular mechanism by which CD25 is associated with carcinogenesis remains, however, unclear. High serum levels of the soluble form of CD25 (sCD25) have been associated with increased tumor activity, tumor aggressiveness, and also poor prognosis, mainly in human lymphoproliferative neoplasms but also in renal cell cancer and melanoma. 9,17,30 Furthermore, de novo CD25 expression was found on neoplastic lymphoid cells and to a lesser extent on nonlymphoid cancer cells such as melanoma and several carcinomas, but again, the function and impact on tumor development and behavior have not been clarified. 1,3,12,14,33,40,45
Apart from its potential role as a pro-proliferative factor, CD25 could also function as a modulator of immune-mediated tumor response. As mentioned before, CD25 is essential for IL-2 binding and subsequently leads to increased T-cell proliferation. Capturing of IL-2 by IL-2R-expressing MCT cells could therefore attenuate the antitumor immunoreactivity by suppressing T-cell proliferation and may function as a mechanism of tumor cells to escape from immune response. 5,32 Indeed, grade 1 and grade 2 MCT with high CD25 expression contained a lower number of tumor infiltrating T cells than did grade 3 tumors with low CD25 expression. In the context of immunomodulation, it is also noteworthy that CD25 expression can be induced on dendritic cells by a combination of tumor necrosis factor–α and prostaglandin E2 (PGE2), the latter of which is synthesized by mast cells. 26,38 It could be speculated that PGE2 production by mast cells contributes to their CD25 expression level.
Taken together, the present study identified CD25 as an MCT marker that is present on canine neoplastic mast cells but not on resting connective tissue mast cells and only rarely on activated mast cells in allergic dermatitis. Because its expression levels are negatively correlated with the histological grade of MCT, CD25 may rather have a potential impact on mast cell tumor biology during early tumor development but seems less necessary for malignant behavior. In addition, the decrease of CD25 expression in higher-grade MCT conflicts with its use as a positive malignancy marker. This study raises questions as to whether canine MCT do express the other IL-2R subunits and hence may be stimulated by binding of IL-2.
Footnotes
Acknowledgements
We thank Monika Schaerig and Michaela Dauer for excellent technical assistance. The present study is part of a PhD thesis and was supported by the Dahlem Research School.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.