Abstract
The cervical spinal cords of 2 horses with equine degenerative myeloencephalopathy (EDM) were evaluated for evidence of oxidative damage to the central nervous system (CNS) using immunohistochemical staining for 3-nitrotyrosine (3-NT) and 4-hydroxynonenol (4-HNE). Neurons of the CNS from horses with EDM had positive immunohistochemical staining, whereas control samples did not, thus supporting the theory that oxidative damage is a potential underlying factor in horses with EDM. In addition, serum vitamin E concentration was low in both EDM-affected horses, and vitamin E concentration was also deficient in the cerebrospinal fluid in 1 EDM horse, further supporting the association between low vitamin E concentrations and oxidative damage to the CNS. Continued research is necessary to further define the pathophysiologic mechanisms of EDM.
Equine degenerative myeloencephalopathy (EDM), typically characterized by symmetric ataxia, proprioceptive deficits, and spasticity, is a chronic and progressive noncompressive spinal cord disease predominately observed in young horses of multiple breeds. 13,17,18 Definitive antemortem diagnosis is not possible but is suspected based on signalment, clinical signs, and the presence of low serum concentrations of vitamin E and/or selenium. 17 Definitive diagnosis is based on histopathologic findings of diffuse neuronal fiber degeneration of the white matter of the spinal cord and caudal brainstem. 14 Although clinical signs and microscopic findings of EDM have been well described, the pathophysiologic mechanisms of disease still remain obscure. Proposed theories include familial/genetic predisposition, toxicity, and oxidative injury to the spinal cord, but the definitive cause remains to be elucidated. 5,6 The report here describes 2 cases of EDM in which further diagnostic tests and immunohistochemical staining were evaluated in an attempt to identify if oxidative damage to the spinal cord contributes to the pathophysiologic mechanisms of EDM.
Clinical History and Diagnostic Evaluation
Horse No. 1 was a 1-year-old Quarter Horse filly that presented to the Lloyd Veterinary Medical Center at Iowa State University with a 4-month history of repetitive stumbling, prolonged recumbency, and difficulty rising. General physical examination was unremarkable, but neurologically, the filly had severe (grade III/IV) ataxia of all limbs. Both the serum vitamin E (0.3 ppm; reference interval, 2–10 ppm) and selenium (67 ppb; reference range, 140–250 ppb) concentrations were deficient.
Horse No. 2 was a 2-year-old Quarter Horse filly presented for a 10-month history of progressively worsening incoordination. Vital parameters and general physical examination were within normal limits, but abnormal findings on neurologic examination included severe (grade III/IV) weakness and symmetric ataxia of the hindlimbs along with a hypometric forelimb gait, especially when the head was elevated. The cerebrospinal fluid (CSF) protein was 44.8 mg/dl (reference interval, 47–69 mg/dl) with a nucleated cell count of 4 cells/μl (reference interval, 0–4 cells/μl); no cytologic abnormalities were noted in the CSF. Serum vitamin E concentration was low (1.2 ppm), whereas serum selenium concentration was marginally low (133 ppb) and serum copper was within reference ranges (1.1 ppm; reference range, 0.65–2 ppm). The CSF vitamin E concentration also was low (2.0 ppb; reference range, 4.1–13.5 ppb). 11 Survey radiographs of the cervical vertebrae from both horse Nos. 1 and 2 were unremarkable. On the basis of the severity of clinical signs and poor prognosis, both horses were euthanized.
Necropsy and Histologic Findings
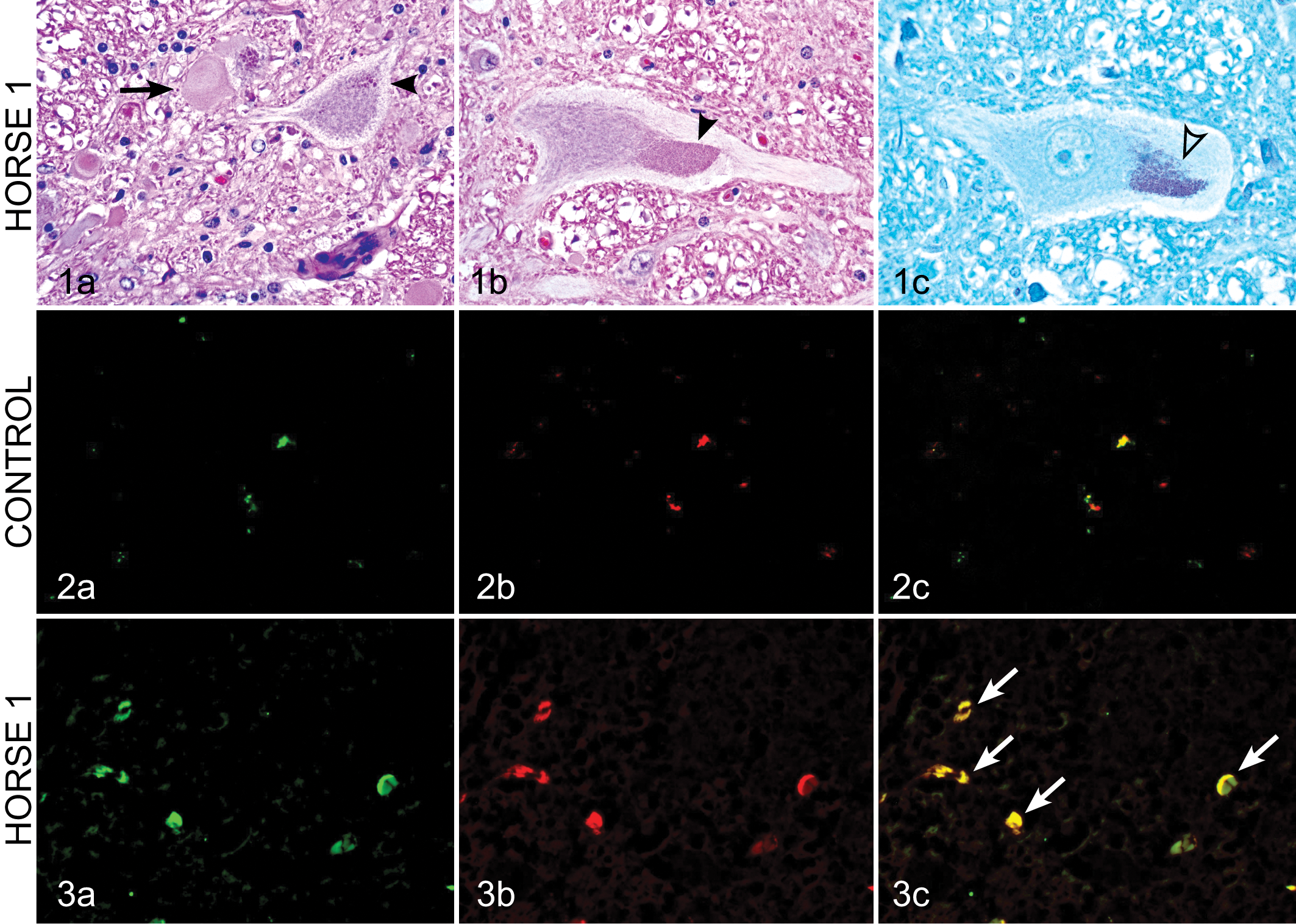
Gross necropsy findings were unremarkable in both cases except for evidence of arteritis of the cranial mesenteric artery secondary to nematodes in horse No. 2. Histologic examination of both cases confirmed the diagnosis of EDM (Fig. 1). Horse No. 1 was characterized by moderate numbers of dilated axon sheaths, often lacking axons, of the dorsal aspect of the lateral funiculi (dorsal spinocerebellar tracts) involving the cervical, thoracic, and lumbar spinal cord. Less frequently, axons were swollen and hypereosinophilic (spheroids) or were replaced by cellular debris admixed with gitter cells (Wallerian degeneration). In addition, within these tracts, there were fewer axons than normal, and the intervening neuropil was sclerotic and contained slightly excessive numbers of fibrous astrocytes and other glial cells. A few swollen axons and dilated axon sheaths were present within the ventral funiculi as well. Within the gray matter, particularly adjacent to the central canal (nucleus thoracicus), many neurons contained multiple, golden brown, periodic acid-Schiff (PAS)–positive intracytoplasmic granules consistent with lipofuscin (Fig. 1). The only other significant microscopic finding was the presence of mild eosinophilic pneumonia indicative of parasitic larval migration.
In horse No. 2, microscopic examination of the cervical spinal cord identified gitter cells, spheroids, and dilation of axon sheaths in the descending ventromedial funiculi of the cranial cervical segments and cranial to mid-thoracic segments of the thoracic spinal cord. In addition, there was acute and subacute multifocal Wallerian degeneration of the ventrolateral funiculi. In the brainstem, there were degenerative neurons observed within the cuneate nucleus on the left and right sides. Liver vitamin E (2.4 ppm; reference range, 2.5–7.1 ppm) and copper (3 ppm; reference range, 4–7.5 ppm) concentrations were slightly low.
Immunohistochemistry
Samples were compared from sections of cervical spinal cord from the 2 horses with EDM and an age-matched (2-year-old) control horse that was euthanized for non-neurologic disease. Formalin-fixed, paraffin-embedded tissues were sectioned at 5 μm. Paraffin-embedded sections were then deparaffinized in xylene followed by ethanol. Antigen retrieval was accomplished using 10 mM sodium citrate buffer (pH 8.5) for 30 minutes to expose antibody binding sites. Nonspecific sites were blocked with 2% bovine serum albumin, 0.5% Triton X-100, and 0.05% Tween-20 in phosphate-buffered saline (PBS) for 1 hour. To evaluate for oxidative stress, sections were incubated with anti–3-nitrotyrosine (3-NT) antibody (rabbit polyclonal; Chemicon; 1:500) and anti–4-hydroxynonenol (4-HNE) antibody (mouse monoclonal; R&D 1:500) overnight at 4°C. Sections were then incubated in appropriate secondary antibodies (Alexa Fluor 488 and 555, Invitrogen; 1:1500) for 2 hours and then washed with PBS followed by dehydration steps using ethanol. Sections were viewed under an inverted fluorescence microscope (model TE-2000U; Nikon). Positive staining for 3-NT and 4-HNE was negligible in the cervical spine of the control horse (Fig. 2), whereas strong positive reactivity for 3-NT and 4-HNE was demonstrated in neurons of numerous sections of cervical spinal cord from both horses with EDM (Fig. 3).
Quantitative analysis of immunofluorescent reactivity was subsequently accomplished by measuring the optical densities from the region of interest using image analysis software (MetaMorph 5.07). Briefly, images were first calibrated and then optical density was measured using an integrated morphometric analysis function. Optical density of 3-NT and 4-HNE was counted at 10 different regions from 2 different slides of the cervical spine from each horse with EDM as well as the control horse. The relative 3-NT and 4-HNE mean optical density for horse Nos. 1 and 2 was statistically compared to the control horse using 1-way analysis of variance (ANOVA) with Tukey’s multiple comparison testing (Prism 4.0 Software, GraphPad) with statistical significance defined as P < .05. The optical density for both 3-NT and 4-HNE in horses with EDM was statistically higher (P < .01) than that of the control (Figs. 4 and 5).
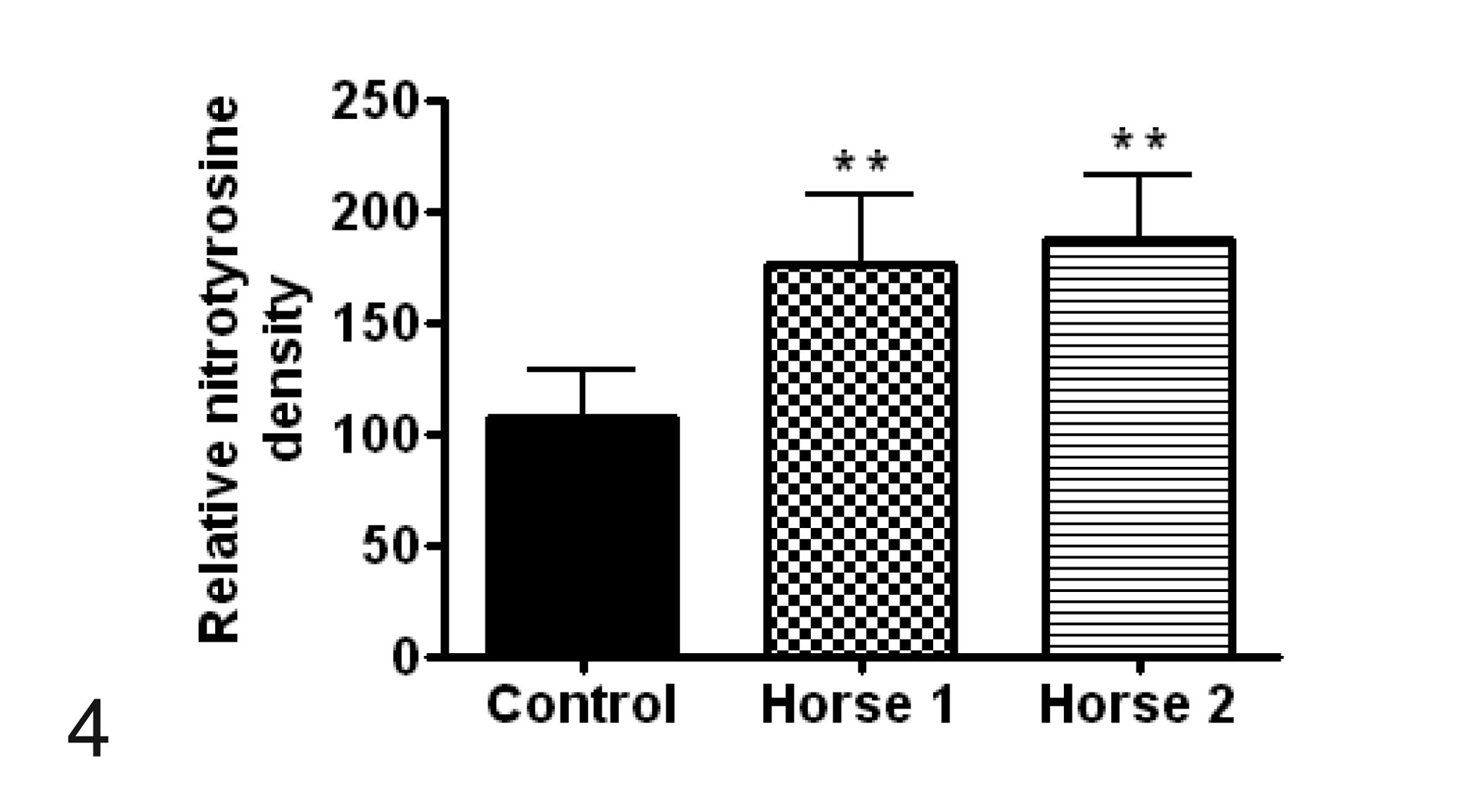
Comparison of optical density, representing mean ± SD relative immunoreactive density, of 3-nitrotyrosine (3-NT) positive cells between control samples and horse Nos. 1 and 2 with equine degenerative myeloencephalopathy. **P < .01.
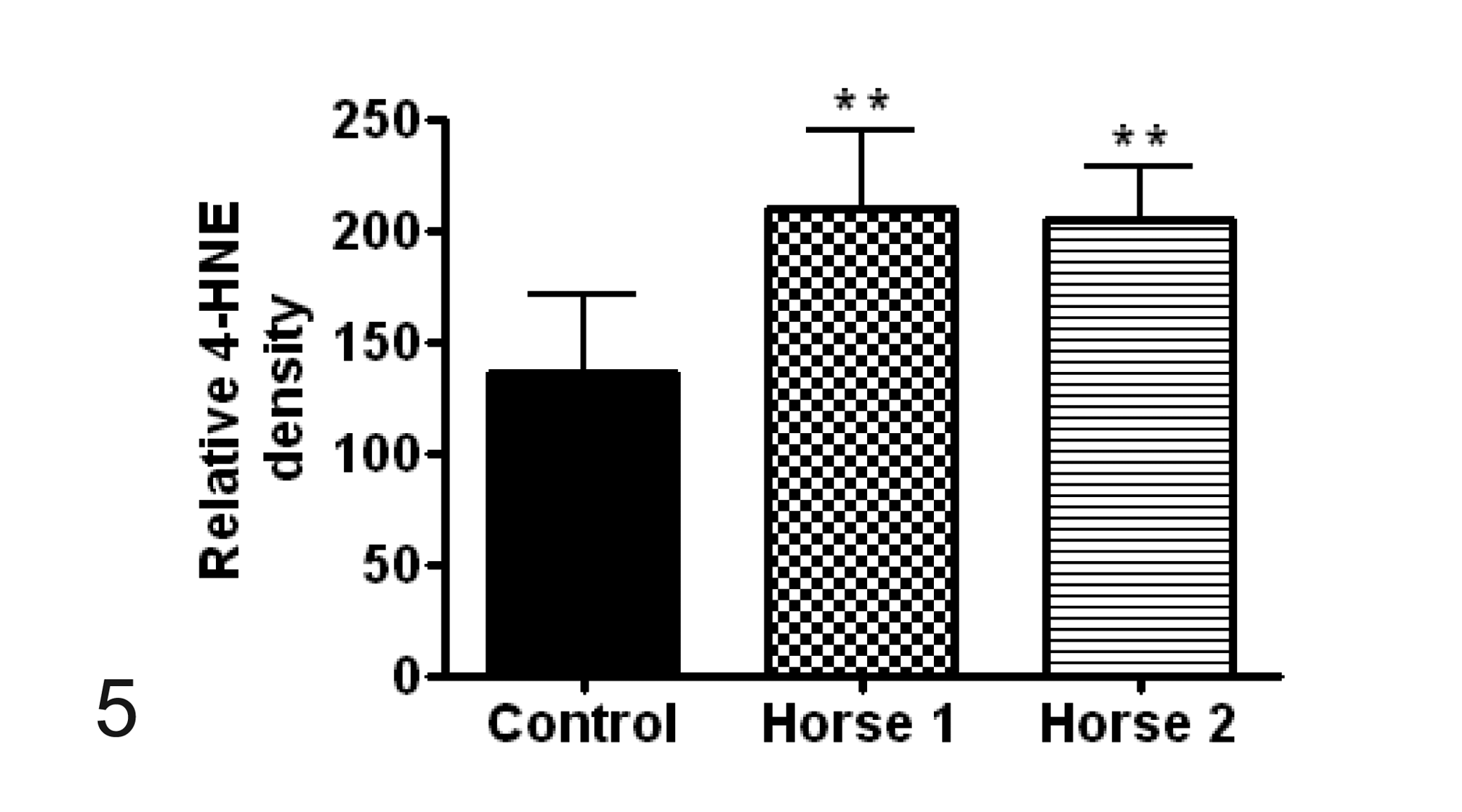
Comparison of optical density, representing mean ± SD relative immunoreactive density, of 4-hydroxynonenol (4-HNE) positive cells between control samples and horse Nos. 1 and 2 with equine degenerative myeloencephalopathy. **P < .01.
Discussion
In human conditions involving oxidative stress, peroxynitrite (3-NT) and unsaturated aldehyde (4-HNE) concentrations markedly increase in neurons and mediate neuronal apoptosis. 1 In this report, positive immunoreactivity for 3-NT and 4-HNE was detected in the neurons of 2 horses with EDM, thus supporting the theory that EDM may be associated with oxidative damage to the CNS. EDM is a common neurologic disorder in the United States, but little advancement in pathophysiologic mechanisms has been identified. 6,14 A familial predisposition in some breeds has been determined in Morgans, 2 Appaloosas, 4 Paso Finos, and Standardbreds 15 by pedigree analysis, whereas risk factors for EDM include exposure to overcrowded grassless paddocks or toxins such as insecticides or wood preservatives and vitamin E or copper deficiency. 6,8 To date, vitamin E deficiency and oxidative damage to the CNS is a widely accepted theory based on low serum vitamin E concentrations and the presence of lipofuscin within the CNS of horses with EDM. 3 –5,8,15 Supplemental administration of vitamin E in farms with a history of EDM appears to decrease the disease incidence. 5,8 Information from the clinical report presented here provides further evidence that oxidative damage, identified through specific immunohistochemical stains, is present within the CNS of horses affected with EDM.
Reactive oxygen and nitrogen species are generated through physiologic processes but, when produced in excess, overwhelm endogenous antioxidant and antinitrosative agents, resulting in oxidative and nitrosative stress. 12 Some oxygen metabolites, such as superoxide radical, hydrogen peroxide, and hydroxyl radical, serve as potent oxidants capable of causing cellular injury and lipid peroxidation. 7 These reactive oxygen species are associated with local cellular injury; however, breakdown products produced during lipid peroxidation create lipid peroxides, such as 4-HNE, malonaldehyde, acrolein, and hexanal; diffuse from their site of origin; and may have toxic and reactive properties. 10 Hydroxynonenal is a major product of unsaturated aldehyde formed during oxidative stress due to lipid peroxidation and is widely used as a marker of membrane lipid peroxidation. 20 Hydroxynonenal is a cytotoxic product that can cause cross-linking of proteins, DNA fragmentation, and proteolysis, as well as modification of lipids’ biophysical properties. 10 In contrast, peroxynitrite is generated by reaction of superoxide with nitric oxide and is a reactive nitrogen species that selectively nitrates tyrosine residue, thus generating 3-NT, and is associated with a variety of neurodegenerative diseases. 19,21 Peroxynitrite is cytotoxic and can cause direct tissue damage via lipid peroxidation, protein nitration, and DNA damage, or it can decompose to form hydroxyl radicals and nitrogen dioxide, also resulting in tissue injury. 7 Because peroxynitrite has the ability to cause protein modification, including the formation of 3-NT, detection, via immunohistochemical staining, of 3-NT has attracted considerable interest as a biomarker of peroxynitrite formation and nitric oxide–dependent oxidative stress. 22
In this clinical report, 4-HNE and 3-NT were used immunohistochemically to identify oxidative damage within the CNS of 2 horses with EDM. Subjectively, an obvious increased immunopositive staining of neurons within the CNS of diseased horses with EDM was present, when compared with an age-matched horse without neurologic disease, and identified the presence of an increase in oxidative stress and injury. Although only 2 cases of EDM are described here, statistical comparisons of the optical densities of 4-HNE and 3-NT immunoreactivity between affected horses and control samples demonstrated a significantly higher reactivity in the horses with EDM. In addition, the serum concentration of vitamin E was deficient in both cases described here and was consistent with other cases of EDM. Furthermore, in horse No. 2, low CSF vitamin E concentration also was reported. Vitamin E is a potent antioxidant that is embedded within cell membranes and scavenges free radicals, converting them to stable compounds and thereby attenuating the process of lipid peroxidation that is associated with free radicals. Although larger numbers of horses with EDM need to be examined, this information suggests that peroxynitrite-induced tissue damage to the CNS may be involved in the pathogenesis of EDM and that lipid peroxidation and the formation of 4-HNE of the neurons may also contribute to the development of EDM. Furthermore, oxidative changes may be associated with low vitamin E concentrations in the serum and CSF.
Interestingly, many neurodegenerative diseases in domestic animals are clinically and morphologically similar to human counterparts, and a tremendous amount of research has been directed at elucidating pathophysiologic processes in these various disorders. In the cases reported here, increased immunoreactivity to 3-NT and 4-HNE was observed in the CNS of EDM-affected horses, suggesting oxidative injury may potentially contribute to the development of disease. However, further investigation into many of human and veterinary neurodegenerative diseases is necessary to determine the exact mechanisms of disease. For example, a possible explanation for the development of another equine disorder, pituitary pars intermedia dysfunction, is oxidative injury to this region, supported by the fact that histological examination of this area in affected horses had a 16-fold increase in 3-NT in the nerve terminals of the periventricular neurons and abundant lipofuscin accumulation in pituitary neurons compared with healthy horses. 9,16 Unfortunately, large gaps in knowledge with regard to equine neurodegenerative diseases exist and await further investigation.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
