Abstract
Migration of Parelaphostrongylus spp. has been documented to cause central nervous system damage in a number of aberrant host species but appears to be uncommon in cattle. The current report describes the clinical and laboratory findings, antemortem and definitive diagnosis, and response to treatment of Parelaphostrongylus spp. infection in five 3–7- month-old Limousin calves from 2 farms. All calves had signs of acute (n = 2) and chronic (n = 3) progressive spinal cord dysfunction. Cerebrospinal fluid analysis revealed a marked eosinophilic (acute cases) or lymphocytic (chronic cases) pleocytosis and elevated protein in all calves. A necropsy and histopathologic evaluation was performed on 2 euthanized calves, and histopathology revealed lymphoplasmacytic and eosinophilic meningomyelitis with multiple intradural and intramedullary expansile hyperplastic lymphoid nodules containing germinal centers and nematode fragments. DNA sequencing was performed on nested polymerase chain reaction products amplified with parasite-specific primers obtained from formalin-fixed and frozen spinal cord; PCR products from these 2 calves were 100% identical to Parelaphostrongylus species on DNA sequencing, confirming the diagnosis. Surviving calves rapidly improved following treatment with anthelmintics and corticosteroids. This case series identified Parelaphostrongylus spp. (likely P. tenuis) as a cause of spinal cord disease in calves and highlights the need for vigilance against aberrant parasite migration in calves grazing wet, snail-infested pastures. Cerebrospinal fluid eosinophilia is useful for supporting an antemortem diagnosis of Parelaphostrongylus in calves with acute neurologic disease; however, a lymphocytosis is observed in chronic or treated cases.
Introduction
Parelaphostrongylus tenuis is a common, predominately nonpathogenic, nematode parasite of white-tailed deer (Odocoileus virginianus) with a complex life cycle involving intermediate terrestrial gastropod hosts.2,13 Naturally occurring aberrant migration of P. tenuis causes neurological disease in various mammals, including camelids, sheep, goats, horses, cervids, and a number of other cloven-hoofed animals.1,2,10,12-14,22,23 However, there are only 2 individual pathology reports of presumptive P. tenuis migration causing neurological disease in cattle5,25 with the diagnosis made on morphological characteristics of nematodes within spinal cord segments. To date, there is no commercially available antemortem test to confirm Parelaphostrongylus spp. infection in ruminants, with a presumptive diagnosis of P. tenuis often made in small ruminants and camelids following detection of cerebrospinal fluid (CSF) eosinophilic pleocytosis, combined with the characteristic neurological findings of asymmetrical limb weakness and ataxia and a documented clinical response to anthelmintic treatment.14,15,24 A more definitive postmortem diagnosis has been historically made by histopathological identification of nematode segments within the spinal cord or brain.1,12,23 Recently, a nested polymerase chain reaction (nPCR) assay has been developed and used for detection of Parelaphostrongylus spp. DNA in a horse.7,21 In the current study, presumptive P. tenuis infection was diagnosed antemortem in 5 calves based upon history, clinical signs, laboratory results, and response to treatment for aberrant larval migration. Parelaphostrongylus spp. infection was confirmed in 2 calves on postmortem examination by the presence of nematode fragments in the spinal cord with subsequent sequencing of parasitic DNA fragments amplified by nPCR.
Materials and methods
Affected calves resided on 2 farms separated geographically by 15 miles in upstate New York. Each farm had approximately 30 head of cattle (adult brood cows with nursing calves). The calves were evaluated between September and October 2009 at Cornell University’s Equine and Farm Animal Hospital (Ithaca, NY) where physical and neurological examinations, CSF collection, and analysis and necropsy studies were performed.
Cerebrospinal fluid collection and analysis
CSF was collected into ethylenediamine tetra-acetic acid–anticoagulated tubes from the lumbosacral space in all 6 calves. In calf 3, CSF was also collected from the atlanto-occipital space after sedation. Analysis consisted of assessment of gross appearance (volume, color, and turbidity), total nucleated cell (TNC), red blood cell count, and cytological examination (including 100 differential cell counts) of Wright-stained cytospin smears by a clinical pathologist.
Necropsy
A complete necropsy was performed following euthanasia of the 2 most severely affected calves. Tissue samples were fixed in 10% buffered formalin and routinely processed for histopathologic assessment. Samples of formalin-fixed (calves 3 and 5) and fresh frozen (calf 5) spinal cord from lesional areas were submitted to the University of Georgia (Athens, GA) for PCR to identify Parelaphostrongylus spp. DNA.
Polymerase chain reaction amplification and sequencing of parasite DNA
DNA was extracted a according to the manufacturer’s instructions. A nPCR reaction was used to amplify a portion of the second internal transcribed spacer region of the ribosomal RNA (rRNA) gene in Parelaphostrongylus spp. using the following primers: 1) primary reaction—forward or sense (5’-CCGTCGAATACATGTCATCC-3’) and reverse (5’-TCGTCAAGACGATGATTCCC-3’) 6 with a predicted 228–base pair (bp), and 2) secondary reaction—forward (5’-AGAATTACGACAATGGCAAC-3’) and reverse (5’-ATGATACCCATTGATAATC-3’) with a predicted 110-bp product. Polymerase chain reaction components and primary reaction cycling parameters were performed as previously described10,21 with the exception of a 43°C annealing temperature, which was used for the secondary reaction. Amplicon visualization, purification, and nucleotide sequencing was done as previously described. 10
Results
Antemortem findings
Five Limousin calves between 3 and 7 months old from 2 herds were examined. The initial clinical signs were stumbling in the hind limbs and difficulty rising. Two animals had shown clinical signs for 7–10 days, whereas the remaining 3 animals had displayed clinical signs for at least 1 month (Table 1). Both farms had experienced abnormally high summer rainfall and reported seeing higher than normal numbers of slugs and snails around rubber feeding buckets and water troughs prior to the onset of clinical signs. Large numbers of white-tailed deer had been commonly observed in the pasture with the beef cattle. The calves had been treated on the farm with vitamin E and selenium supplementation, a moxidectin, b and fenbendazole c 1 week prior to hospital admission. The clinical signs had transiently improved in all calves following treatment.
Signalment and summary of cerebrospinal fluid (CSF) findings in calves with Parelaphostrongylus tenuis.*
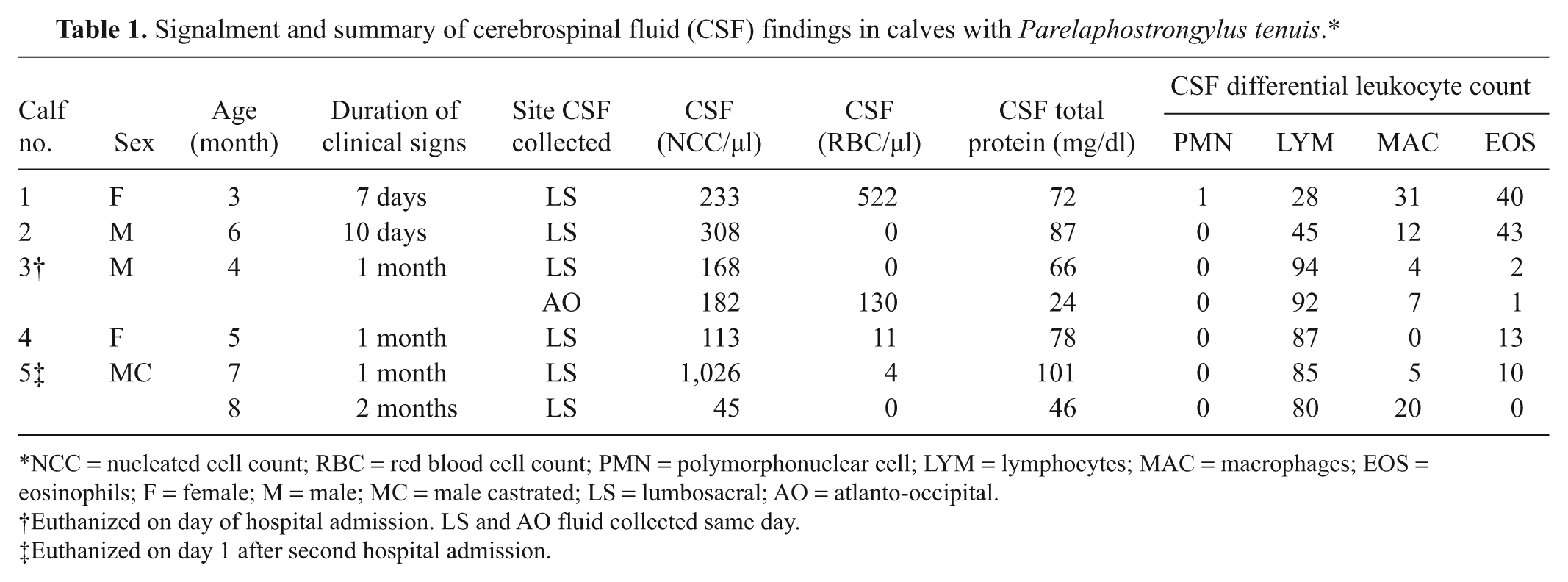
NCC = nucleated cell count; RBC = red blood cell count; PMN = polymorphonuclear cell; LYM = lymphocytes; MAC = macrophages; EOS = eosinophils; F = female; M = male; MC = male castrated; LS = lumbosacral; AO = atlanto-occipital.
Euthanized on day of hospital admission. LS and AO fluid collected same day.
Euthanized on day 1 after second hospital admission.
On hospital presentation, all calves were in excellent body condition and were bright and alert with a normal appetite. A complete neurological examination revealed signs of hind limb ataxia and paresis consistent with upper motor neuron spinal cord disease in all 5 calves. Signs of lower motor neuron disease (hyporeflexia of the limbs, loss of tail tone, and dribbling urine) were seen in 3 calves. One calf had forelimb ataxia and paresis and needed considerable assistance to rise. The CSF analysis showed marked pleocytosis (mean: 369 TNC/µl, range: 113–1026 TNC/µl, reference interval: <10 TNC/µl) and increased total protein (mean: 81 mg/dl, range: 66–101 mg/dl, reference interval: <66 mg/dl) in all calves (Table 1). More acute cases showed an eosinophilic (>20% eosinophils) pleocytosis, whereas chronic cases had a lymphocytic (>70% lymphocytes) pleocytosis with a lower percentage (2–10%) of eosinophils. Results of CSF collected from the atlanto-occipital region in the most severely affected calf were similar to that collected on the same day from the lumbosacral space.
The most severely affected calf was euthanized due to the severity of clinical signs after 1 day of hospitalization. Based on CSF fluid analysis and preliminary necropsy findings, the remaining calves were treated for presumptive parasite migration with ivermectin d (0.2 mg/kg subcutaneously for 5 days), fenbendazole (10 mg/kg orally for 5 days), and dexamethasone e (0.05 mg/kg intramuscularly, 2 doses on days 1 and 3). Management changes were also implemented on the farm to decrease snail and slug exposure, and a deworming protocol (ivermectin 0.2 mg/kg subcutaneously every 30 days until after the first frost) was added for all cattle on the farms. Three of the 4 remaining and affected calves responded rapidly to treatment and were asymptomatic 2 months after discharge. One treated calf (calf 5) did not respond completely to treatment and continued to have difficulty rising. On a second hospital examination 1 month later, calf 5 was still ataxic and paretic in the hind limbs, and continued to dribble urine. A lymphocytic pleocytosis was still present in the CSF, although the TNC counts were markedly decreased compared to the first sample collected 1 month earlier and eosinophil percentage in the CSF had decreased from 10% to 0% (Table 1). No other cases were identified on these 2 farms in 2009 or 2010 after implementation of the recommended management changes.
Postmortem findings
At necropsy, calves 3 and 5 were in fair body condition and weighted 128 and 298 kg, respectively. Macroscopic lesions included 3 (2 in calf 3 and 1 in calf 5) smooth, raised, round, tan or gray nodules that bulged on cut section within the ventral or dorsolateral aspects of the cervical and thoracic spinal cord (Fig. 1). On histologic examination of formalin-fixed tissues, there were varying degrees of lymphoplasmacytic and eosinophilic meningomyelitis, vacuolation, rarefaction, and swollen axons with gliosis and variable numbers of gitter cells (parasite migration tracts) found multifocally but especially in the white matter. There were also multifocal, large, dense lymphoid nodules containing regularly arranged follicles with germinal centers and high endothelial venules (Fig. 2A, 2B) in cervical, thoracic, and lumbar regions of the spinal cord. These lymphoid follicles were intramedullary in one of the calves and extramedullary in the other calf. Degenerate nematode remnants were observed at various segment levels within the parenchyma of the spinal cord, randomly in the white matter or within lymphoid nodules. The remnants ranged in size from 50 to 150 µm in diameter and had a thin amphophilic smooth to undulating cuticle and polymyarian coelomyarian musculature (Fig. 3). Nematode remnants in calf 5 were all partially mineralized (Fig. 4A, Fig. 4B), and all were closely associated or within the lymphoid nodules. This calf also had a mild lymphoplasmacytic meningoencephalitis and scattered Wallerian degeneration in the caudal brain stem. Amplicons of 108 bp (calf 3) and 102 bp (calf 5) were obtained after the nPCR reaction. Nucleotide sequence analysis of the secondary reaction revealed a 100% identity to both P. tenuis and P. andersoni as compared to the GenBank database (accession nos. GU122925 and EU020134).
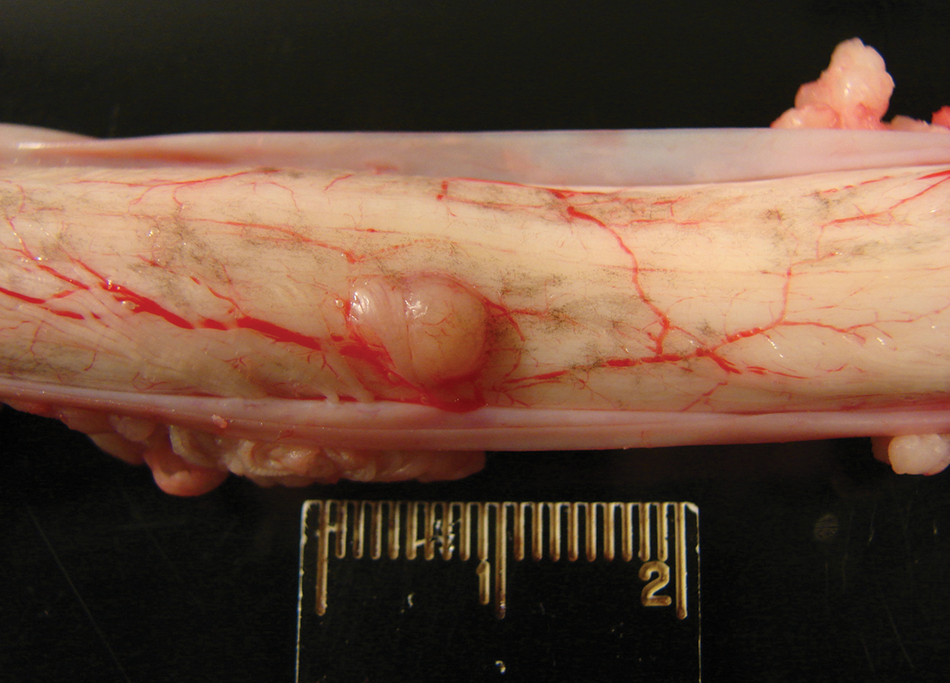
Calf no. 3. Raised nodule in the spinal cord at the level of the 4th thoracic vertebra.
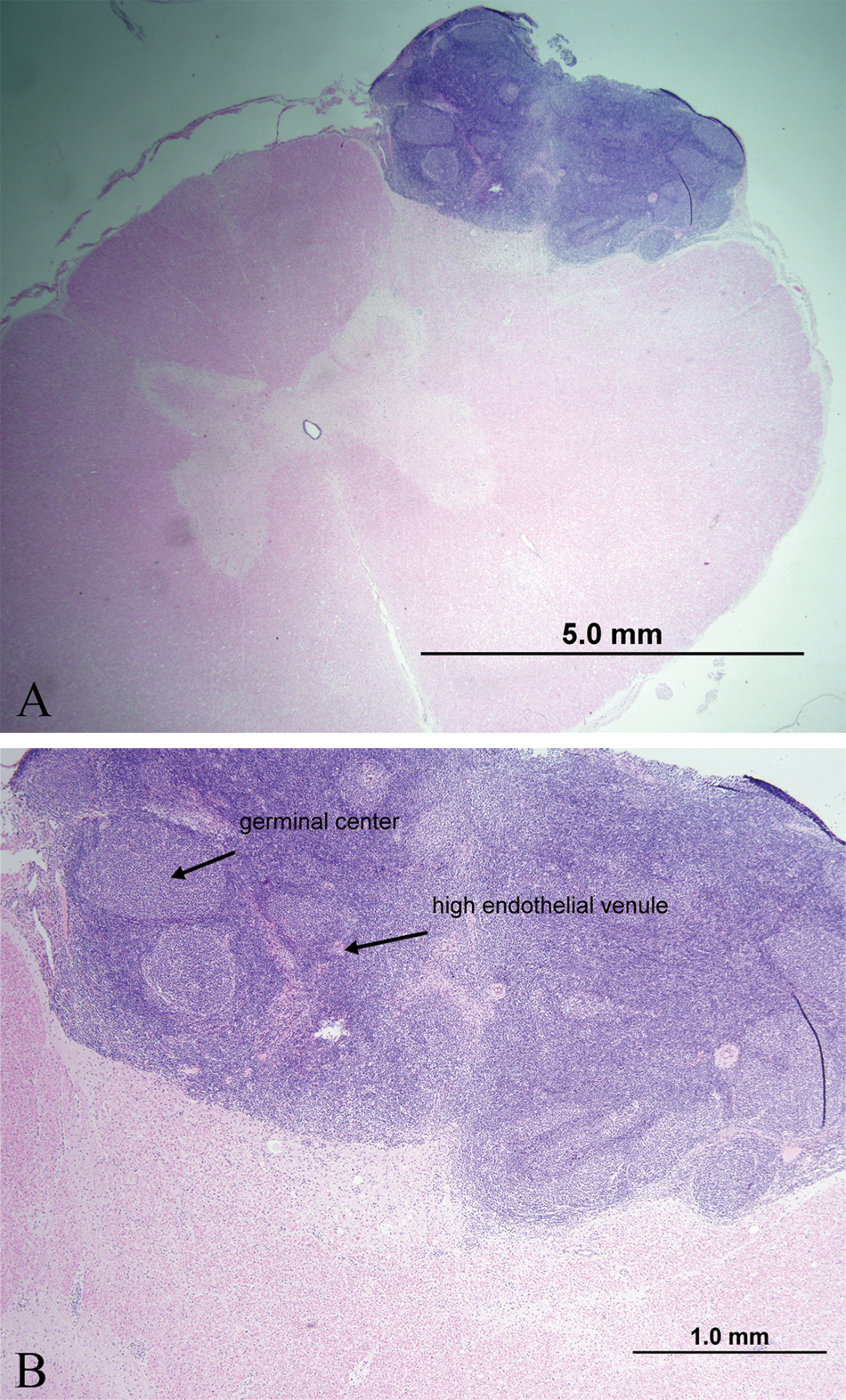
Calf no. 3.
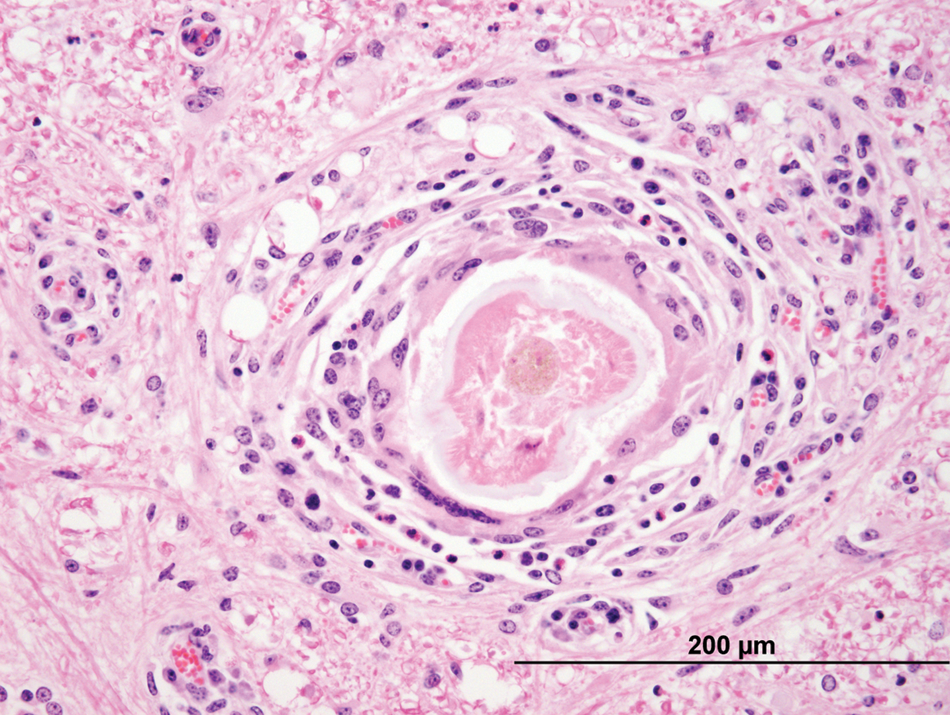
Calf no. 3. Cross-section of degenerate nematode within the white matter of the spinal cord at the level of the 7th cervical vertebra. The organism is approximately 100 µm in diameter and has coelomyarian musculature and a thick amphophilic cuticle. It is surrounded by moderate numbers of macrophages, multinucleated giant cells, eosinophils, and smaller numbers of lymphocytes. There is also mild lymphoplasmacytic perivascular cuffing in the surrounding white matter. Hematoxylin and eosin. Bar = 200 µm.
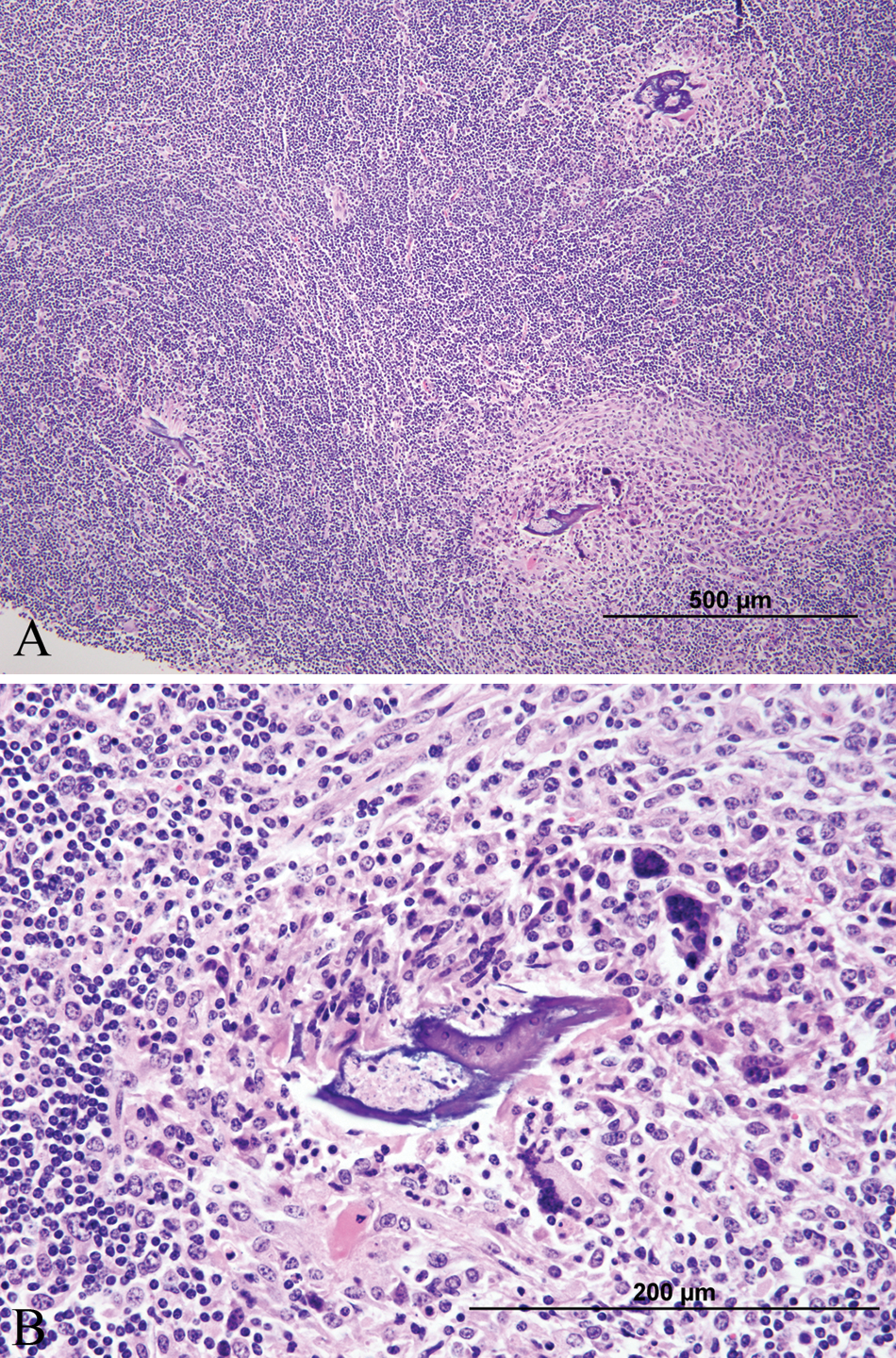
Calf no. 5.
Discussion
The current case series identifies Parelaphostrongylus spp. as a cause of spinal cord disease with ataxia and paresis in 5 calves. A definitive diagnosis of Parelaphostrongylus spp. infection was obtained in 2 calves following amplification and sequencing of parasitic DNA within the spinal cord tissue. A presumptive diagnosis was reached in the 3 remaining calves based on similarities in neurological examination, CSF analysis, and response to treatment for aberrant parasite migration within the spinal cord.
Clinical signs of P. tenuis infection are well documented in other species, with the classic development of progressive, asymmetrical hind limb weakness, and ataxia.1,12-15,19,24 More advanced cases may show fore limb involvement.17,19 Uncommonly, clinical signs of scoliosis and central brain disease are reported.15,22,24 The signs of upper motor and lower motor neuron dysfunction correlate to the migrating tracts of the parasite through both white and gray matter of the spinal cord. Occasionally extramedullary intradural lymphoid aggregates at the sites of parasite migration are also seen, which likely contribute to spinal cord compression at these sites.11,17 To date, no definitive antemortem test for Parelaphostrongylus spp. exists in ruminants. Antemortem diagnosis of aberrant migration of P. tenuis is based on history, clinical signs, and abnormal CSF results.14,15,24 The history of the 2 affected farms experiencing a wetter than normal summer and fall with large numbers of slugs and snails is consistent with previous reports of outbreaks involving aberrant hosts becoming infected with P. tenuis.12,13 Warm moist environmental conditions appear to favor survival and higher densities of the intermediate hosts. 13 Currently, very little is known about the pathogenesis of Parelaphostrongylus spp. infection in cattle. The affected calves in the current study are believed to have developed clinical signs of Parelaphostrongylus spp. infection due to the ingestion of large numbers of infective larvae (based on the snail burden around feed and water troughs), although snails and slugs were not tested for the presence of larvae. The infective dose of P. tenuis depends on the aberrant host species, with camelids appearing more sensitive to infection than other species.6,17,19 A larger infective dose results in quicker onset of clinical signs.6,17,19 Young calves may be more susceptible to Parelaphostrongylus spp. infection since no adult cows were observed to have clinical signs on the 2 farms in the current report. Since these 5 cases, in the fall of 2010, 2 additional 3- and 5-month-old angus calves from separate farms were presumptively diagnosed as having Parelaphostrongylus spp. infection of the spinal cord based upon the similarity of their clinical signs, response to treatment, and CSF findings to the calves in the present report or finding nematode fragments in the spinal cord at necropsy of one of the calves. No adult cattle were believed to have clinical signs on the 2 farms, which further supports the possibility that young growing calves are most susceptible to clinical disease resulting from aberrant migration of Parelaphostrongylus spp.
Abnormal CSF findings have been documented in small ruminants, llamas, and alpacas following infection with P. tenuis.4,12,14,19 Classically, these animals develop an eosinophilic pleocytosis in the CSF following infection with P. tenuis larvae, although the timing between infection and documentation of CSF eosinophilia can vary.15,16,19 In the current case series, eosinophilic pleocytosis (40–43% eosinophils) was seen in acutely affected animals (<10 days of clinical signs). Eosinophilic CSF pleocytosis is rarely observed in the CSF in cattle with previously reported neurologic disorders. 20 A mild eosinophilia (2–8% eosinophils) has been reported in the CSF from cattle with brain or spinal cord abscesses, polioencephalomalacia, neonatal meningitis, or spinal cord lymphoma, with a report of >40% eosinophils in a cow with Hypoderma bovis infection in the spinal cord. 20 Hypoderma bovis was, on admission, considered an unlikely cause of the neurologic disorder in these calves given the large number of calves affected and earlier season of the year than would be expected for Hypoderma bovis–induced spinal cord disease. 18 In contrast to the eosinophilic pleocytosis seen in the acute cases of parelaphostrongylosis in the current study, a lymphocytic pleocytosis (80–94% lymphocytes, with only 0–10% eosinophils) was seen in the CSF from more chronic cases or treated cases (clinical signs present for at least 1 month). These results suggest that an eosinophilic pleocytosis in CSF might be present only during the acute stages of parasite migration in calves and that the pleocytosis becomes more lymphocytic with chronicity or following treatment. Changes in CSF with chronicity or treatment (predominantly a transition from neutrophilic to monocytic pleocytosis) have been seen in cattle with other infectious disorders such as neonatal meningitis, listeriosis, or otitis meningitis. 20 The lymphocytic pleocytosis was quite marked in the calves in the current report and was higher than that seen in most other bovine neurologic disorders. 20 This is an important finding because a CSF lymphocytosis is generally not considered a typical finding in animals with parasitic diseases of the nervous system, and Parelaphostrongylus spp. infection may be mistakenly excluded from the differential diagnosis list in cattle if CSF is performed during the more chronic stages of infection. The CSF lymphocytosis in the spinal cord of the 2 necropsied calves in the present study seemed to correspond to the inflammatory lesions, which were predominantly lymphocytic with large lymphoid nodules. The nodular lymphoid hyperplasia seen in the spinal cord in both calves in the present report was previously reported in 1 presumptive case of bovine parelaphostrongylosis in an angus calf. 5 Bovine parelaphostrongylosis is an unusual lesion that has not been reported in other species with aberrant central nervous system P. tenuis migration other than a 1977 report in fallow deer (Dama dama). 16 In the case of the previously reported angus calf, 5 nematodes were not present within the lymphoid nodules; however, degenerate nematodes were present in the lymphoid nodules of the fallow deer 16 as they were in the 2 necropsied calves reported herein. This type of hyperplastic lymphoid tissue with germinal centers suggests a marked response to chronic antigenic stimulation driven by the presence of the nematodes. It has been suggested this lesion may represent a difference in species immune response to P. tenuis or may be due to the duration of infection prior to presentation and necropsy. 5 Nematodes were degenerate and mineralized in both of the necropsied calves in the current study suggesting some level of chronicity. A presumptive diagnosis of P. tenuis infection was initially made on identification of the parasite fragments on histopathological evaluation of spinal cord sections in calves 3 and 5. The anatomical location of the nematode in the spinal cord is consistent with P. tenuis or Elaphostrongylus rangiferi in other species. 13 The parasite was definitively confirmed to be Parelaphostrongylus by DNA amplification and sequencing of parasitic DNA. Both PCR sequences had 100% identity with P. tenuis or P. andersoni (GenBank accession nos. EU020134 and GU122925). Differentiation between P. tenuis and P. andersoni was not possible based on the length of the PCR products detected. However, P. andersoni infection was considered unlikely, as this is mostly a skeletal muscle parasite of caribou and reindeer in North America and Europe. Parelaphostrongylus andersoni has been found in isolated white-tailed deer populations in North America, and although there are no reports of P. andersoni infections in animals in New York State, the possibility of infection must still be considered.3,8,13 Parelaphostrongylus andersoni can infect incidental hosts and is found in skeletal muscle, lung parenchyma, fat, extradural spinal cord, and lymph nodes but not in the spinal cord or the brain. 13
There are several clinical reports of various treatment protocols for small ruminants and camelids including ivermectin, fenbendazole, diethylcarbamazine, levamisole, and moxidectin.13-15,24 After the presumptive diagnosis of aberrant migration of P. tenuis was made based on CSF analysis and preliminary necropsy findings, the 4 surviving animals in the current study were treated with a combination of anthelmintics and with dexamethasone, using protocols extrapolated for treating suspected P. tenuis infections in small ruminants and camelids.13-15,24 There are no controlled clinical studies to confirm an increased efficacy of using a combined anthelmintic treatment, although this is commonly recommended in both camelids and small ruminants with P. tenuis14,15 and was, therefore, recommended for treatment of these calves. All treated animals responded quickly to treatment; however, some neurologic signs persisted in 1 calf, which was later euthanized. The reduced response in the euthanized calf could be related to the persistent inflammation associated with mineralized parasites, the large intramedullary lymphoid nodule at the level of T4 causing spinal cord compression or persistent damage due to parasite migration. Similar spinal cord abnormalities have been observed in other species that failed to recover completely after treatment for aberrant P. tenuis migration.11,15,24 The remaining 3 calves made a clinical recovery, as often reported for camelids and small ruminants following similar treatment.12,14,24
In other aberrant hosts, preventative management recommendations are based upon decreasing exposure to the definitive host (white-tailed deer) and the intermediate host (snails and slugs) in addition to properly timed anthelminthic administration for aberrant larval migration.15,24 Although a hypothesis exists that ingested larvae can be prevented from reaching the spinal cord in small ruminants by regular deworming with ivermectin or similar products at 4–6 week intervals during the spring to fall in herds with a high risk of infection, this has not been documented. 15 Additionally, it should be noted that frequent use of anthelmintics, either avermectins or benzimidazoles, can increase parasite resistance to the respective drugs. 9 Reduction of the ingested larval burden can be achieved by maintaining clean feeding areas and water troughs and avoiding grazing heavily slug- or snail-infested pasture. Preventing white-tailed deer from entering the property is time consuming and expensive and is not an economical solution in most situations. 15
In conclusion, the current report identifies aberrant migration of Parelaphostrongylus spp. as a cause of spinal cord dysfunction in calves. The findings highlight the need for vigilance against aberrant parasite migration in calves grazing wet, snail-infested pastures shared with high numbers of white-tailed deer. A presumptive antemortem diagnosis can be made based on the typical neurological signs of upper and lower motor neuron spinal cord disease and CSF analysis (eosinophilic pleocytosis in acute cases and lymphocytic pleocytosis in chronic cases) while PCR and sequencing of parasitic DNA retrieved from spinal cord tissue can provide a definitive diagnosis of Parelaphostrongylus spp. infection.
Footnotes
Acknowledgements
The authors thank Dr. Jodie Gerdin for her help with the necropsy and histopathology of calf no. 3. The abstract was presented by Dr. Mitchell at the annual meeting of the American College of Veterinary Internal Medicine, Anaheim, CA, 2010.
a.
QIAamp® DNA Mini kit, Qiagen Inc., Valencia, CA.
b.
Bo-Se® (vitamin E & selenium), Schering-Plough Animal Health Corp., Union, NJ.
c.
Cydectin® (moxidectin) oral drench 0.1%, Boehringer Ingelheim Vetmedica Animal Health, St. Joseph, MO.
d.
Safe-Guard® (fenbendazole), Intervet Schering-Plough Animal Health Corp., Union, NJ.
e.
Ivomec® (ivermectin) 1% sterile solution injection, Merial Limited, Duluth, GA.
f.
Dexamethasone (2 mg/ml sterile solution), MWI Veterinary Supply Inc., Boise, ID.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This project was supported by funds from The W. C. Rebhun Memorial Fund and by Dr. Fred Hess DVM of Amherst, MA.
