Abstract
Coronary arterial disease (CAD) is the leading cause of death in the United States, the European Union, and Canada. Percutaneous coronary intervention (PCI) has revolutionized the treatment of CAD, and it is the advent of drug-eluting stent (DES) systems that has effectively allayed much of the challenge of restenosis that has plagued the success of PCI through its 30-year history. However, DES systems have not been a panacea: There yet remain the challenges associated with interventions involving bare metallic stents as well as newly arisen concerns related to the application of DES systems. To effectively address these novel and ongoing issues, animal models are relied on both to project the safety and efficacy of endovascular devices and to provide insight into the pathophysiology underlying the vascular response to injury and mechanisms of restenosis. In this review, preclinical models of restenosis are presented, and their application and limitation in the evaluation of device-based interventional technologies for the treatment of CAD are discussed.
Coronary artery disease (CAD) is the progressive accumulation of atherosclerotic plaque within the lumen of a coronary artery, with the typical result being luminal narrowing, reduced compliance of the vessel wall, and either a gradual reduction or a dramatic and sudden loss of blood supply to the myocardium. It is the leading cause of death in the United States, the European Union, and Canada, singly accounting for the death of 1 in 5 Americans. 117,185 In treatment options for symptomatic CAD, percutaneous coronary intervention (PCI) is the foremost modality, being based on the nonsurgical placement of a bare metallic scaffold, or stent, to prop open the artery to regain patency. However, there are drawbacks to coronary stenting, with restenosis being the Achilles' heel, accounting for a rate of failure up to 15 to 25%. 18,124 Drug-eluting stents (DESs) have revolutionized PCI by effectively reducing restenosis rates to the single digits over standard bare metallic stents.
DES systems combine mechanical and pharmacological approaches to assuage one or more events in the cascade that results in restenosis. These combination products consist of an intravascular scaffolding device that presents or releases single or multiple bioactive agents or pharmaceuticals into the bloodstream and arterial wall. Traditional DESs have consisted of a platform (stent), a carrier (usually a polymer), and an antirestenotic agent that is delivered locally to avert the need for high, systemically administered doses. The fundamental goal of the DES is to restore lumen patency while curtailing the diseased vessel’s innate and often exuberant response to the implanted foreign material (stent) and to stent-induced injury. Despite the success of current DESs at achieving these goals, concerns have surfaced with their accruing clinical use. Such concerns pertain to vascular healing and late stent thrombosis, biocompatibility, and the continued challenge of restenosis in select patient subsets. 82,114,173,175,191 Since the US approval of the first-generation DES in 2003, there has been development of many novel technologies that attempt to address these concerns. Because of the complex, multidisciplinary, and dynamic nature of this technology, thorough evaluation of DES systems in preclinical models is crucial for predicting clinical safety and efficacy as well as for providing details into the pathophysiology of the vascular response to injury and restenosis. This summary provides an overview of preclinical models of restenosis, outlines guiding principles for the preclinical evaluation of DES, and reviews the pathophysiology of in-stent restenosis (ISR) while drawing corollaries that may be deduced between preclinical models and the clinical setting. This article focuses on DES systems designed for coronary application, although the technology is being developed and has been applied in peripheral vasculature. Although coronary and peripheral applications share several conceptual similarities in their preclinical evaluation, there are special considerations for peripheral devices with regard to model, anatomic location, and clinical application, for which supplementary references are provided. 41,98,161,208 Moreover, there is increasing complexity in the design of DES systems and the development of alternative endovascular treatments for ISR. Given that no set program or means for preclinical safety and efficacy evaluation can encompass all these endovascular technologies, this review seeks simplicity by focusing on DES systems commercially available or with published preclinical results.
Animal Models of Restenosis
A variety of animal models have been used for evaluating the pathophysiology of ISR and endovascular devices for its prevention or treatment, including models based in rodents, rabbits, dogs, sheep, swine, and nonhuman primates. The variety of models exists because of the absence of an ideal; that is, each model features advantages and shortcomings. The underlying principle of preclinical models of ISR has been the induction of arteriosclerotic lesions through induced injury that provokes a localized proliferative healing response that mimics ISR. Although antecedent atherosclerotic lesions of human arteries are absent in standard preclinical models, some models may be compounded through the addition of spontaneous or induced atherosclerosis or a systemic condition that affects arterial healing, such as diabetes. Thus, preclinical models aptly suited for the evaluation of endovascular devices may be categorized as models of arteriosclerosis (vascular injury) or dual models combining vascular injury either with atherosclerosis or with diseases having cardiovascular influence. Given that restenosis and atherosclerosis are two separate though related pathologic events, this review focuses foremost on models of vascular injury and ISR and secondarily on models with the added complexity of atherosclerosis.
Rodents
Murine models of restenosis carry the distinctive advantages of ease of handling and housing, cost-effectiveness, high throughput, reproducibility, and a wide offering of molecular markers. In mice, technical challenges have historically limited studies on neointimal hyperplasia to those involving modified procedures of arterial injury, including Epon bead–induced endothelial denudation, 219 guidewire-induced injury, 109 arterial ligation, 100 electrical injury, 19 and periarterial placement of a nonconstrictive polyethylene cuff around the femoral or carotid arteries. 122 These models have limited application: There is little thrombus formation, and the induced neointimal hyperplasia tends to be smooth muscle cell rich with little resemblance to human pathology specimens. 160,205,215 Furthermore, in application to endovascular treatments, screening of antirestenotic compounds requires systemic administration. Recent mouse models suited for the evaluation of device-based therapies of ISR have been developed, including the use of a poly (ϵ-caprolactone) cuff capable of drug elution or the use of balloon injury and endovascular stenting. 3,143,144 Despite the procedural challenges, these two models, using inbred and genetically modified mouse models, can provide insight into the pathophysiology of atherosclerosis and mechanisms of neointimal hyperplasia and restenosis following vascular injury. 65,99,205
Rats have been used extensively since the 1960s as models for the evaluation of the vascular response to injury and smooth muscle cell proliferation using a variety of injurious stimuli. Although rat models have historically been limited by the same, size-related challenges as those encountered in mouse models, technical advances allow deployment of specially designed or commercially manufactured stents in the carotid artery or aorta, respectively (Fig. 1). 49,77,105,112,153 Similar vascular responses and rates of vascular healing have been reported for the carotid and aortic models. 49,77,105 The abdominal aortic model has the advantages of lesser technical difficulty and a reportedly greater proliferative neointimal response, as compared with the carotid arterial or thoracic aortic models. 105,153 Analogous to mouse models, the availability of antibodies to cellular proteins, as well as the availability of transgenic, diabetic, obese, and hypertensive strains, is an advantage to the use of rats for endovascular studies of restenosis. Still, a degree of skepticism remains toward rat arterial injury models; that is, early investigations of antirestenotic compounds that yielded favorable results in this species failed to produce similarly positive results in other preclinical models or in clinical trials. 57,68,75,103,146 These may be due to differences in cellular and molecular mechanisms involved in response to arterial injury in rats, primates, and other species. 75,79,133 The rates of vascular healing in rat carotid arterial and aortic models appear to parallel those of models utilizing larger species qualitatively; however, the vascular response in the rat has similar salient features as mice, including only sparse thrombus formation and fibrin deposition, minimal inflammation, and a lack of remodeling. 11,77,104,105,186
Murine models have promise for elucidating pathophysiologic mechanisms related to atherosclerosis and ISR; nonetheless, the cumulative shortcomings of these models warrant their careful application and critical interpretation. First, murine arteries suitable for the evaluation of ISR are mostly elastic, which are less prone to injury and have only modest neointimal responses following vascular injury when compared to muscular arteries. 75,79 In addition, the flow dynamics and vasomotor function of elastic murine arteries is inherently incongruent with coronary arteries. This aspect is particularly important with respect to DES systems, given that these factors may affect stent dynamics, polymer bioabsorption, and elution profiles. Because of their size, defined endpoints for neointimal hyperplasia can only be morphometrically evaluated. This limitation can result in false positives because absolute changes in morphometric endpoints may not be readily visible in the angiographic endpoints that can be evaluated in large animal models and are the primary endpoint of clinical studies. 34,163 Finally, although the availability of proteins and genetic biomarkers is a notable advantage of murine models, these utilities have limited and as yet unclear translational value in the clinical setting.
Rabbit
Rabbits have the merits of being small and easy to handle and are relatively inexpensive. Common anatomical sites for inducing restenosis have included the aorta and carotid and iliofemoral arteries, which are of comparable size to human coronary arteries and thus allow for the evaluation of commercially manufactured devices to morphometric and angiographic endpoints (Fig. 2). The rabbit ear crush injury model also has been used for localized, nondevice-based studies on restenosis. 9 The rate and phases of vascular healing following stent-induced injury in the normocholesterolemic rabbit iliac arterial model parallel those of standardized porcine coronary arterial model. 162,199 Additionally, elution studies for current DES systems have shown comparable results between the two models, suggesting that the flow dynamics of rabbit iliac arteries are similar to porcine coronary arteries. 11 Despite these similarities, there are postulated shortcomings of rabbit arterial models. Foremost is the inability to directly assess cardiac events following stent implant. Second, although arterial responses are similar, the susceptibility of the elastic-natured aorta and iliac artery to injury is less when compared with a muscular coronary artery. 6,73 This ultimately can affect the degree of neointimal hyperplasia, thrombus formation, and inflammation, each of which may be intrinsically less in device studies using this model as compared with porcine coronary arteries. One characteristic attribute of the rabbit iliac arterial model is its tendency for delayed re-endothelial coverage postinjury, making this model more suitable for the comparative endoluminal evaluation of with respect to endothelial recovery and function. 83,129,141
Rabbits are readily susceptible to dietary-induced hypercholesterolemia, and there are strains of rabbits with a predisposition for developing hypercholesterolemia. 181,207 As such, rabbits have been used extensively for the study of atherosclerosis and ISR, which merits hypercholesterolemic rabbit models deserving of particular review. On a diet supplemented with cholesterol, fats, and/or casein, rabbits readily develop lipid and cholesterol deposits throughout the reticuloendothelial system, in resemblance to a lipid storage disease. The formation of arterial plaques rich in lipid-laden macrophages (foam cells) with abundant extracellular matrix can be readily and reproducibly induced, especially with the addition of targeted mechanical injury through air desiccation, endothelial denudation, or balloon injury (Fig. 3). The nature, time course, and sequencing of dietary alterations and mechanical injury can affect the character of the atheromatous lesions that develop. 24,31,35,36,47,140 This includes, but is not limited to, the extent of foam cell and leukocytic involvement, smooth muscle cell density, the amount and character of extracellular matrix, the degree of plaque concentricity, and the degree of lumen stenosis. Most commonly utilized models of ISR involve several weeks of a cholesterol-supplemented diet combined with balloon denudation or injury induced 1 to 2 weeks into the dietary phase (Fig. 3). 129,148,213 Lipid deposition occurs in the intimal plaque as well as in restenotic neointimal tissue, similar to humans; however, there are inherent differences in the histological composition and behavior of the atheromatous lesions and in the lipid metabolism between rabbits and humans. 17,30,103,155,213 These differences have raised reservations in the relevance of the hypercholesterolemic rabbit model and merit consideration when interpreting studies evaluating drug and device therapies.
Swine
The porcine coronary arterial model is the standard model for the preclinical evaluation of endovascular devices for ISR. The cardiovascular anatomy of swine is similar to humans with regard to size, morphology, relative collateral arterial supply, and the presence of a well-developed vasa vasorum; the general physiology and coagulation systems of this species closely parallel those of humans. 62,113 Large animal coronary vasculature models are suitable for the evaluation of performance of the stent, guide wire, and catheter in complex and tortuous configurations analogous to diseased human coronary arteries. Furthermore, the local flow dynamics of porcine coronary arteries closely parallels that of humans, allowing for greater confidence in the accuracy of pharmacokinetic studies. The neointimal response is of a similar histology to that in restenotic human coronary arteries; however, as with other preclinical models, the degree of restenosis often is not sufficient to be of clinical significance even with heightened injury through overstretch models (Fig. 4). However, as opposed to other preclinical models, negative or constrictive remodeling comparable to humans occurs more predictably following balloon angioplasty of porcine coronary arteries, resulting in stenotic lesions formed by neointimal hyperplasia and negative remodeling. 5,30 Although this remodeling may be of minor significance with respect to metallic stent systems relative to their structural integrity, negative remodeling can be a significant factor for nonmetallic or bioabsorbable endovascular treatments.
The disadvantages of animal and housing costs of large animal species are partially offset by the ability to implant 2 to 3 devices in coronary arteries, including single or overlapping stents. Devices also can be implanted in intramammary arteries in pharmacokinetic studies. A key limitation to the common use of domestic farm swine is their rapid growth rate and high body weight potential (> 400 kg), which imparts logistical difficulties in long-term studies, including limitations on handling and equipment. This has been overcome, in part, through the use of miniature swine, such as Yucatan, Sinclair, Göttingen, and Hanford strains, which maintain more modest body weights into adulthood. 125,155 Swine are susceptible to development of spontaneous and dietary-induced atherosclerosis, familial hypercholesterolemia, and pharmacologically induced diabetes; however, the added expense and time required for lesion development in these models have precluded their routine use in the evaluation of endovascular treatments for ISR. Nonetheless, these modified swine models can offer insight into vascular physiology that more closely parallel the clinical setting compared to other modified models in other species. 53,145
One drawback to the use of the porcine model, applicable to domestic farm and miniature breeds, is the propensity for this species to develop inflammation in relation to bare metallic, * polymeric or polymer-coated, 38,156,188,189 and DES 128,129,169,214 systems in coronary and peripheral applications. 41,129,156,190 This inflammation is of a foreign-body (granulomatous) character and often occurs at all implant sites within a host, indiscriminate of device type. In addition, there may be significant eosinophilic involvement. In consideration of this, hypersensitivity cannot be excluded in the etiology, although eosinophilic inflammation of this nature has not been observed in relation to comparable devices in other species, including rat, mouse, rabbit, canine, ovine, and nonhuman primate. † This suggests that inflammation with eosinophilic involvement in relation to these endovascular devices may be unique to the porcine. Because animal health and husbandry may be factors affecting the inflammatory predilection of this species, careful interpretation of results are warranted. 187 When such reactions arise, it may be prudent to utilize a supplementary animal model for thorough preclinical evaluation of DES.
Other Large Animal Models: Dogs, Sheep, and Nonhuman Primates
Dogs were once considered an attractive model for endovascular studies owing to size, ease of handling, cost, availability, and ability to easily deploy stents in coronary and peripheral arteries. However, multiple drawbacks make this model inappropriate for routine use in the study of ISR. First and foremost, dogs are considered “notoriously resistant” to neointimal formation following either balloon angioplasty or coronary stenting. 165 Even extensive deep medial laceration often produces minimal neointimal hyperplasia. Dogs also have high fibrinolytic activity and a coagulation system distinctive from that of humans. 119 Given that the amount of thrombus formation has been related to the neointimal formation, this high fibrinolytic activity of canines may predicate the meager neointimal formation observed in this species. Furthermore, dogs have a low potential for acute stent thrombosis when compared with humans. Indeed, a set of preclinical stent studies using this model failed to predict the thrombotic complications noted in early clinical trials. 157,174 Combined with the well-developed collateral arterial supply in this species (as compared to humans), the occurrence of sudden cardiac events may fail to be detected in this model. 61,71 Finally, as carnivores, dogs are inherently resistant to the development of atherosclerosis, precluding the production of dietary modified models. 125,155
Sheep are commonly overlooked as a candidate for the evaluation of ISR, despite the docile nature of this species, a coagulation and fibrinolytic system with more similarities to that of humans than other species, and a coronary anatomy that favors its use. 87 Although studies on ISR utilizing this model have been few, they have demonstrated that the responses to stent-induced injury in ovine coronary arteries parallel those of porcine coronary arteries (Fig. 5). 4,147,158 An added benefit to the ovine model, compared to the swine model, is the availability of appropriately sized bifurcations of the coronary vasculature, which may make this model more appropriate for the evaluation of bifurcation devices. Sheep feature advantages parallel to those of pigs for the evaluation of ISR, although the robustness relative to low-precedent application is a chief shortcoming.
Nonhuman primate models have the advantages of phylogenetic proximity, dietary and metabolic similarities, and disposition to develop metabolic disease, cardiovascular disease, and acute thrombosis following percutaneous interventions. However, ethical considerations, maintenance expenses, and the potential for harboring viral zoonoses outweigh these benefits in favor of other preclinical models. 11,155 Studies that have been conducted to date in normal and atherosclerotic nonhuman primate models have provided valuable insight with regard to vascular responses to injurious stimuli, atherosclerosis, and the influence of genetics on vascular pathophysiology. 30,57,192 Nonetheless, the paucity of studies in nonhuman primate models utilizing either balloon angioplasty or stent implantation and the relative inconsistencies among studies are further shortcomings. 64,186
Model Selection, Application, and Limitations
Each preclinical model of ISR has a unique set of attributes, assumptions, and limitations. Given that there is no single model that serves as a blanket ideal for investigations into ISR and drugs or devices designed for its treatment, each model discussed herein is appropriate in its own right for application to answer targeted questions relative to ISR. The logical and methodical formulation of study objectives should guide model selection. With specific regard to ISR, three categories of objectives should be considered:
Safety assessment of an endovascular therapy: The ideal models to satisfy this objective should most accurately mimic the clinical setting in design and execution, using standardized controls with known performance as a means for comparison. Evaluation methods should likewise mimic those of the clinical setting to evaluate toxicokinetics.
Evaluation of the efficacy of an endovascular therapy or proof of concept studies: “Efficacy” should be defined by specific endpoints, including but not limited to the reduction of neointimal hyperplasia and luminal stenosis, the reduction of inflammation or inflammatory markers, the reduction of thrombus formation, or the enhancement of reendothelialization and other features of vascular healing.
Study the pathogenesis of restenosis following arterial injury to identify new strategies for next-generation treatments: Models with atherosclerosis or systemic disorders support this objective because uncomplicated models utilizing healthy, naïve, and often juvenile arterial systems may not be able to elucidate novel target pathways relevant to diseased arteries.
The normocholesterolemic porcine coronary and rabbit iliac arterial models are the conventional models for nonclinical safety evaluation of coronary DES.
162,187
As such, the remainder of this review primarily references these two preclinical models. These models—in particular, the porcine coronary arterial model—have provided results that predict well the clinical device performance, and they have allowed for better understanding of the pathophysiology underlying the arterial response to injury.
81,113,162
However, these models are primarily predictive of safety and may not necessarily reflect the true efficacy of a test system at fulfilling the key clinical drivers of reducing ISR, reducing treatment-induced arterial or plaque injury, and improving reendothelialization and benign healing in an atherosclerotic, aged, human coronary artery.
33,162
Assessment of DES Safety
Representatives from interventional, regulatory, commercial, and scientific communities have composed an integrated set of recommended guidelines for the preclinical evaluation of coronary DES systems. 162 Additionally, under current revision is a guidance document for industry for conducting nonclinical and clinical studies as set forth by the Food and Drug Administration. 187 These documents provide valuable guidance for the design and conduct of preclinical safety studies, although the dynamic nature and multidimensional approaches of DES technologies oblige tailoring these recommendations to suit the individual device and application. Furthermore, these guidance documents do not fully address early proof of concept studies in which the application of animal models alternative to the standard porcine coronary and rabbit iliac arterial models may prove more valuable.
The fundamental preclinical studies for assaying DES safety include (1) in vitro and in vivo pharmacokinetics, (2) dose range evaluation, and (3) vascular response safety studies specifically designed to mimic the clinical applications, including the use and evaluation of single and overlapping stents, for example. For DES dosing and safety studies, involvement of the trained pathologist is essential. These studies are phased over multiple time points, including evaluations performed at acute and intervallic chronic time points that target particular phases of vascular healing, drug elution, and/or polymer bioabsorption and extend suitably beyond the projected time of complete drug elution and/or polymer bioabsorption to the end phase of vascular quiescence.
DES safety evaluations include clinical observations of morbidity and mortality; preterminal arteriography, hematology, and serum chemistry; and postlife analysis of the implanted arteries, heart, and select peripheral organs pertinent to drug metabolism or distal embolization. Given that many DES systems are based on a metallic stent platform, collected stented arteries are perfusion fixed in situ and processed for plastic or epoxy embedding and either ground and polished to histological grade or sectioned on a rotary microtome equipped with tungsten carbide blades. High-resolution radiography after perfusion fixation and/or of explanted stented arteries is performed to assess stent placement and evaluate for strut fractures. Plastic embedding also is recommended for most polymer-based DES systems because this method better preserves the relationship of the device within the tissue. A review of techniques and considerations in preparation of DES systems and related devices is available. 154 In addition, a subset of dedicated stented arteries may be sectioned longitudinally and processed for analysis by scanning electronic microscopy to further characterize reendothelialization, endothelial cell morphology, side branch patency, leukocytic and/or platelet adherence, mural thrombus formation, and other features of the local native and stented vascular lumen.
Quantitative and semiquantitative evaluation is performed on sections taken from each implanted artery. The recommended sampling for stents less that 15 mm includes cross sections that target the proximal, middle, and distal regions. Proximal and distal host (nonstented or native) arterial sections may be paraffin embedded, and they also are qualitatively evaluated to determine peri-stent effects, such as dissections, aneurysms, excess edge neointimal proliferation or injury, and upstream or downstream arterial toxicity. Quantitative histomorphometry on stented arterial sections targets the primary endpoints of percentage stenosis, mean neointimal thickness, neointimal area, medial area, and medial thickness. These endpoints are derived from measurements of the areas bounded by the external and internal elastic lamina (EEL and IEL) and the lumen area. Neointimal thickness may be measured over individual and between adjacent struts, and the distance between struts and the IEL may be approximated by calculation from the neointima area and vessel diameter. Any separation between strut and IEL, defined as radial gap, is quantified when observed. The commonly accepted method of semiquantitative injury scoring is the Schwartz method, 167 a schema of ordinal designations of disruptions to the IEL, media, and EEL at each individual strut in a section. However, this method is not a wholly accurate indicator of injury; that is, it primarily applies only to muscular arteries and not elastic arteries. This is relative to the anatomical differences between these 2 arteries and greater resistance of the elastic arteries to mechanical injury. The Schwartz method gives a good numerical representation of the depth of mural injury but does not account for its circumferential or longitudinal spread. Other semiquantitative analyses may be performed at each individual strut, as a quadrant-based approach, and/or for the section as a whole. These include but are not limited to endothelialization, fibrin deposition, inflammation, mineralization, hemorrhage, neovascualarization, and necrosis. Each parameter may be assessed collectively or separately for each mutual component (neointima, media, and adventitia). Finally, qualitative descriptions of the neointimal maturity, cellular and extracellular components, or infiltrates of the neointima, media, and adventitia are important for appropriate interpretation of a device’s performance.
The important safety features of DES should include, but are not limited to, those provided in Table 1. Multiple supplemental technologies currently used for investigational purposes or in the clinical setting also have been applied to preclinical models for their application to evaluating next-generation DESs for improved safety and efficacy. These include temporal quantitative en face endothelial analysis via scanning electronic microscopy, 83,128,141 confocal microscopy targeting endothelial and inflammatory markers, 83,128,141 alternative imaging systems (intravascular ultrasound, optical coherence tomography, micro–computed tomography), 27,59,106,183,201 and vasomotor function tests. 80,107 As experience is gained with these technologies, they may well become incorporated into a standard set of valuations in the assessment of DES safety.
Pathophysiology of ISR: Preclinical, Clinical, and the Influence of DES
ISR occurs with all revascularization strategies and therefore is used herein interchangeably with vascular healing. The pathophysiology of vascular healing is likened to that of wound healing, with both processes sharing many cellular and molecular mechanisms. 118,170 However, in the case of coronary stenting, many unique factors affect healing, such as the intravascular location, exposure of the device and wound to pulsating blood flow, local tissue motility around the device owing to vasomotion and heart rhythm, and long-term presence of a foreign material (stent). Further adding to the pathophysiologic complexity of healing relative to DES systems are the localized and temporal effects of the device and its components (eg, drug, polymer). Finally, because it is through the comparative evaluation of animal and human pathologic specimens that we have gained and continue to gain our greatest understanding of ISR and its potential treatment, the comparative vascular responses of normal (preclinical) and diseased (clinical) coronary arteries add further factors for consideration.
Vascular healing or ISR following implant of a metallic stent can be broken into the 6 fundamental stages outlined in Table 2. 32,160,199 These stages are consistent between species, although there is clear temporal delay in the atherosclerotic coronary arteries of humans 199 and manipulated animal models 141 Diseased human coronary arteries are reported to take at least 5 to 6 times longer to heal when compared to healthy porcine coronary arteries. The temporal ranges following metallic stent implant provided in Table 2 serve only as approximations; these become less concrete for DES systems owing to the potential for pharmacological delay in reendothelialization and healing, 82,83 modulation of inflammation, 52,97,134,197 and localized toxicity. 63,160,184
Six Fundamental Stages of Vascular Healing or In-Stent Restenosis a
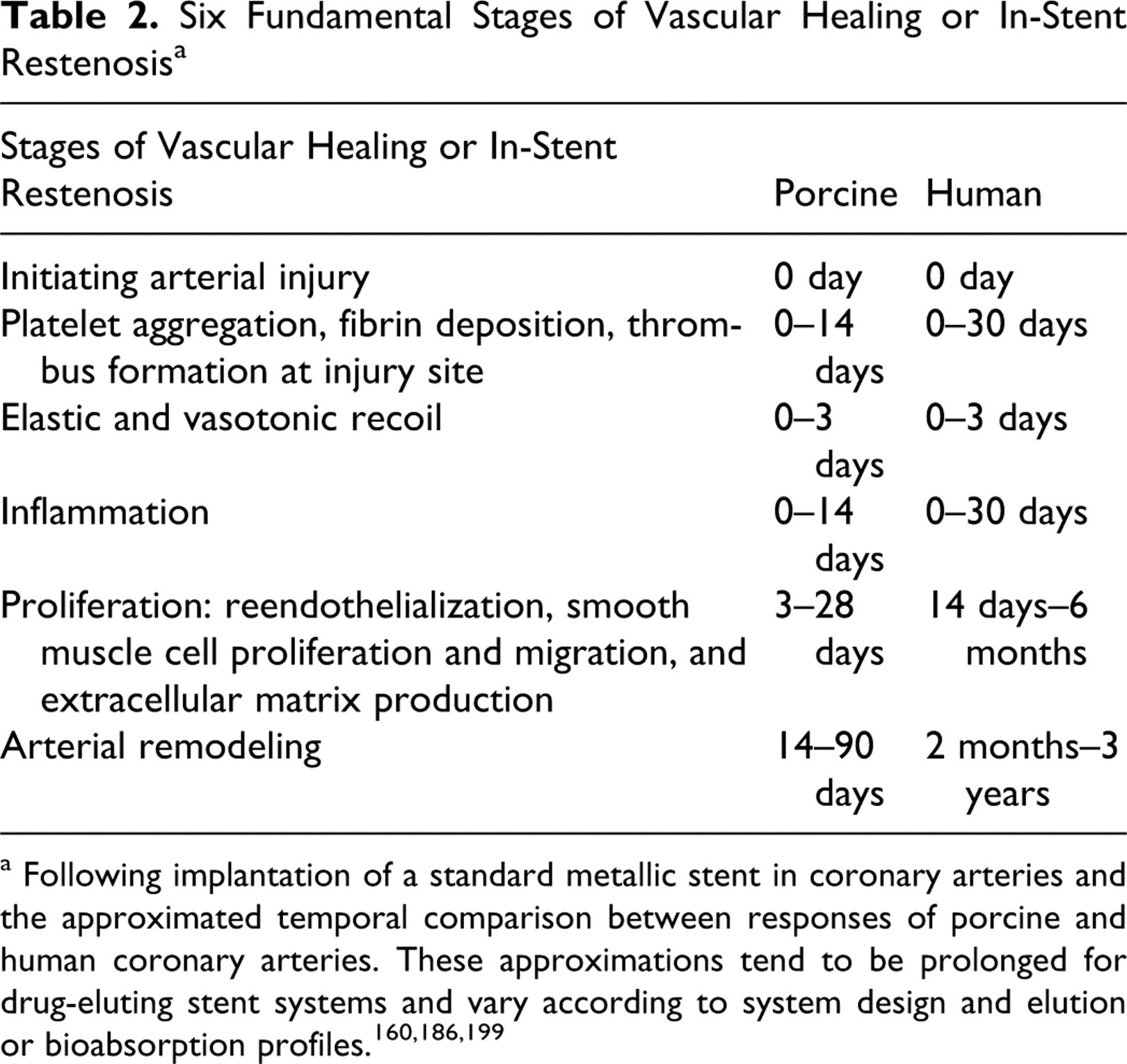
a Following implantation of a standard metallic stent in coronary arteries and the approximated temporal comparison between responses of porcine and human coronary arteries. These approximations tend to be prolonged for drug-eluting stent systems and vary according to system design and elution or bioabsorption profiles. 160,186,199
Although presented as discrete stages, the 6 stages of arterial healing following endovascular intervention actually form a coordinated continuum, with each stage influencing and having temporal overlap with others. Simplistically, these stages can be divided into early and late phases. Whereas the early phase occurs in weeks to months and includes injury, thrombus, inflammation, and proliferation, the late phase involves the chronic mechanisms that lead to increased lumen size, a process known as remodeling. 92 The cumulative result of ISR is dependent on two main contributors: neointimal hyperplasia and arterial remodeling. Given that the arterial wall is an adaptable tissue capable of reshaping in response to hemodynamic, mechanical, and biomechanical forces, there exist additional contributing factors that bear on ISR. These include, but are not limited to, the vascular location, local hemodynamics, 48,92,95,110,167 lesion and stent length, 92 underlying arterial or systemic disease, 1,88,92,166,193 and, for stent-based systems, the implant procedure. 8
For all revascularization strategies, injury to the arterial wall is the initiating event in the ensuing orchestrated response. The degree of arterial stretch and disruption to the arterial wall relates proportionately to neointimal hyperplasia in both porcine and human coronary arteries, although this relationship is less direct in other animal models, such as the rabbit, dog, and baboon. 49,105,165 The presence and character of underlying atherosclerotic plaque is a contributory factor in vessel injury, given that compression and/or strut penetration of the plaque has been documented in up to 90% of angioplasties, which can further the potential for subsequent inflammation and cellular proliferation. 44,45
Concurrent with the initiating injury and in the following hours to days, thrombus formation occurs where there is endothelial denudation, exposed subintimal components, and exposed surfaces of the device. This thrombus serves as the scaffold for the upcoming stage of proliferation by providing a surface for reendothelialization as well as anchorage for smooth muscle cell migration and proliferation. With this and the contributing release of multiple growth and migratory-promoting factors from activated platelets, the amount of mural thrombus has been found to be proportionate to the subsequent degree of neointimal hyperplasia. 26,94,159,164 In comparison of preclinical and clinical settings, the amount of thrombosis is not only dependent on the amount of injury to the arterial wall but also is affected by species-dependent platelet and coagulation factors. 163,165 For endovascular devices, fundamental attributes, including material, design, and thickness, can affect thrombus formation, and for current DES systems, the drug and polymer surfaces can have a positive or negative effect on platelet function and deposition and thrombus formation. For example, both rapamycin and paclitaxel—antimitotic pharmaceuticals utilized in two first-generation DES systems—have been shown to promote endothelial tissue factor expression that theoretically could result in a localized prothrombogenic effect. 176 Conversely, novel endovascular stent systems incorporating alternative designs, biocompatible or thromboresistant materials, and bioactive coatings (eg, heparin, nitric oxide generators) continue to be developed. 7,72,84,89 Thrombus formation, acute (< 48 hours) and subacute (2 to 30 days), is a significant clinical concern for metallic stents and DESs. DES systems also have been implicated in a slight numerical increase in the incidence of late stent thrombosis (30 days to 1 year) and very late stent thrombosis (> 1 year) postulated to be related to delayed healing. 72,82 Although thrombus herein is discussed in the acute phase of vascular healing, the clinical application of DES systems denotes that thrombosis, an often catastrophic event, can be an indolent concern for which appropriate preclinical models are lacking.
Reactive vasoconstriction or elastic recoil occurs conjointly with mural thrombosis in the acute phase of overstretch injury. For balloon angioplasty, elastic recoil is a significant contributor to acute lumen loss. 54,66,170 Metallic stents were introduced to counteract this acute reactive elastic and vasotonic recoil. 127,160 Yet, acute angiographic recoil of up to 20 to 30% has been documented with balloon-expandable metallic stents 14,20 and may be even more be substantial for bioabsorbable devices with radial strengths less than that of currently approved metallic stents. 69,70,202,203 Distinct from the reactive recoil of the arterial wall, stent recoil can be a contributing factor in both acute and long-term arterial lumen loss, especially for bioabsorbable devices that progressively lose radial strength with degradation. This has been reflected in clinical 115,180 and preclinical 70,203 studies. Therefore, whereas device-based strategies may overcome much of the acute arterial recoil reaction, their mechanical design and structural integrity remain significant contributing factors to consider in restenosis.
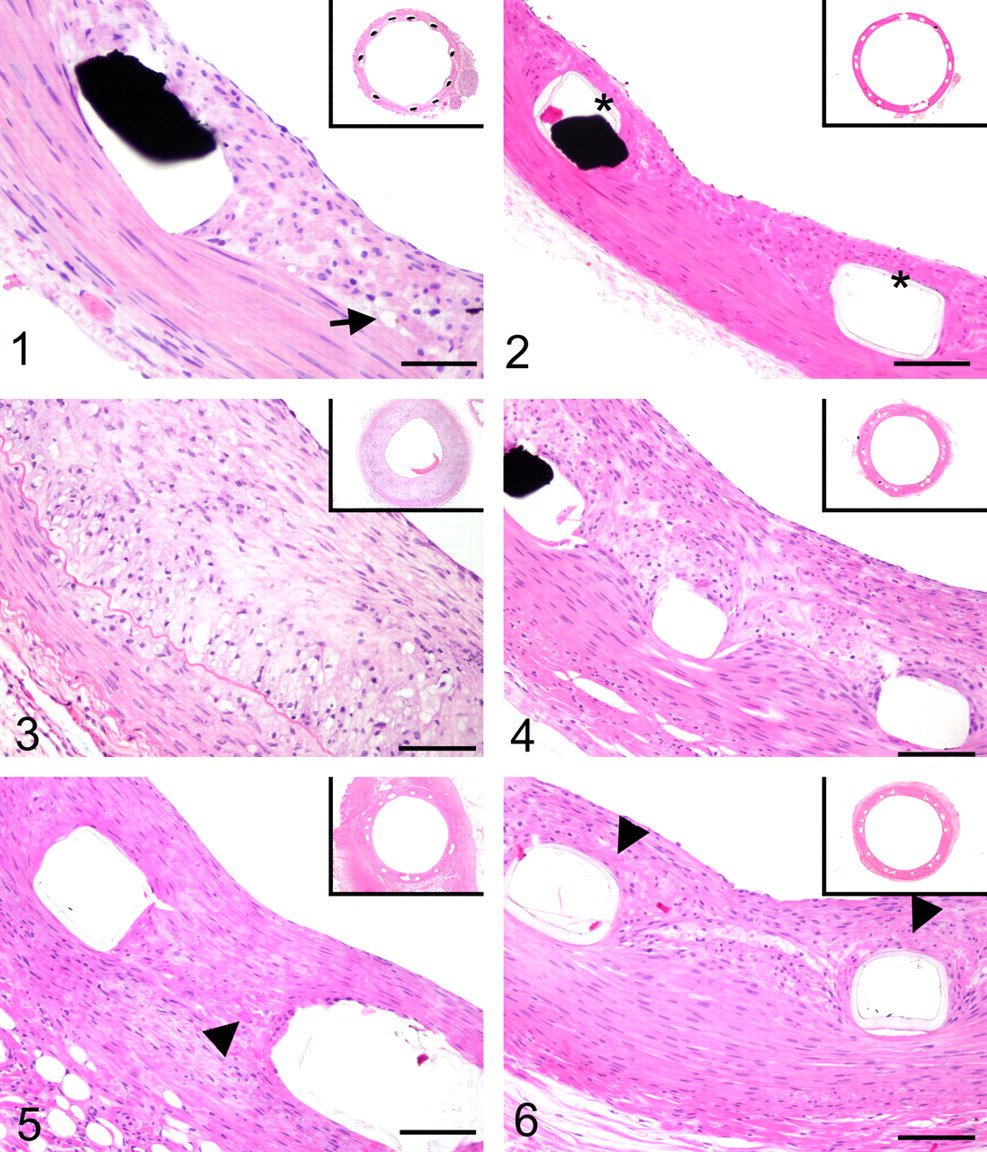
Following closely the stages of injury, thrombus, and recoil is the fourth stage, inflammation, which remains as an active and evolving process throughout much of vascular healing. Dominated initially by surface-adherent neutrophils and monocyte, days to weeks postimplant of a metallic stent, this acute inflammation subsides and is replaced by chronic inflammation composed of macrophages and T lymphocytes, with giant cell formation being common immediately around struts (Fig. 4). 44,199 Multiple intrinsic and extrinsic aspects modulate the extent and character of inflammation through the course of vascular healing following implant of a metallic stent or DES system. These include the microenvironment in which struts localize within an atherosclerotic plaque; 45,82 the biocompatibility of the stent system, its subcomponents, leachables and degradables; 15,38,39,189 stent surface characteristics over time (protein absorption); 15 polymer integrity, durability, and elution rate; 142 and the mechanisms of action and elution rate of the drugs. 56,111,126 As with injury and thrombus formation, studies in animal models and on human autopsy specimens show a correlate between the type and amount of inflammation and the subsequent amount of neointimal hyperplasia and resultant restenosis. 45,164,193,210 The induction of inflammation relative to certain polymers and metals in animal models has likewise confirmed that chronic active inflammation exacerbates neointimal proliferation, resulting in increased restenosis and remodeling. 32,37,95,96,189 Moreover, the nature of inflammation is an important feature affecting vascular healing because hypersensitivity responses featuring eosinophilic infiltrates are reported clinically in association with metallic stents, polymer-coated metallic stents, and DES systems. 97,134,197 With this, inflammation has been cited as playing a paradoxical role in inhibiting certain aspects of vascular healing, the clinical result being late to very late thrombosis. 51,52,82,108,197 Unfortunately, preclinical models to evaluate the potential for stent-related hypersensitivity or late stent thrombosis observed in the clinical setting are as yet lacking. Finally, in addition to inflammation induced by implantation, the amount of macrophages in preexistent atherosclerotic lesions has been noted as a predictor for restenosis and thus may be a compounding factor in atherosclerotic animal models. 121 Inflammation undoubtedly plays an important role in ISR, and although DES systems seem ideal vehicles to modulate and mitigate inflammation’s influence, biocompatibility challenges remain.
Stage 5 of vascular healing, the proliferative stage, occurs hand-in-hand with inflammation. In both animal and human arteries, this stage is marked by a granulation tissue response with reendothelialization, neovascularization, smooth muscle cell migration and proliferation, and deposition of extracellular matrix. 25,44 Proliferating cells are located deep within the neointima adjacent to stent struts, suggesting that proliferation is a chromic low-grade reaction to the stent. 218 Histological distinctions are apparent between preclinical and clinical specimens during this stage. In animal models following metallic stent implantation, the neointima is typically dominated by hyperplastic smooth muscle cells with minimal proteoglycan or collagenous extracellular matrix. 30 Conversely, in stented human coronary arteries, the neointima is less cellular, and the extracellular matrix may account for more than 50% of the total volume of neointima. 28,55,166,212 Although much of vascular healing at this stage occurs in the neointima, responses in the media and adventitia—including cellular proliferation, extracellular matrix deposition, inflammation, and neovascularization—play contributing roles in ISR. 10,45,91,102,116,138 Adventitial hypercellularity owing to fibroblast/myofibroblast proliferation occurs within 3 to 7 days postangioplasty, and collagen deposition follows, resulting in substantial adventitial thickening. 172 The deposition and maturation of adventitial collagen become a significant factor in the subsequent remodeling phase. 195 Current generations of DES systems target primarily the proliferative phase of vascular healing, mainly by impairing smooth muscle cell migration and proliferation. 176 This localized effect of DES systems can be morphologically identified as localized retention of fibrin derived from residual thrombus, intraintimal collections of plasma and/or erythrocytes, and/or increased circumstrut extracellular matrix deposition, each of which is directly reflective of delayed arterial healing (Fig. 6). 82,131,200 Pharmacological suppression of smooth muscle cell proliferation is readily evident in autopsy specimens, being rarely seen until 3 to 6 months postimplant of a DES and even persisting to 1 year with select DES systems. 131 Current DES systems also ultimately have a nonselective effect on reendothelialization by inhibiting endothelial cell migration and proliferation, inhibiting the homing, proliferation, and differentiation of endothelial progenitor cells, and hindering proper endothelial cell function. 176 The delay in re-endothelialization caused by current DES systems is of clinical concern; it translates to balancing DES safety with efficacy. 182,198 Herein lies a critical difference between preclinical and clinical settings: Contrary to arteries of swine and rabbits that completely reendothelialize within 14 to 28 days, incomplete reendothelialization has been documented time and again in human coronary arteries implanted with selected DES systems, even with implant durations greater than 1 year. 50,60,82,130,211 Preclinical models to assess poststent reendothelialization and endothelial function are under development, although their clinical relevance is as yet to be established. 83
The final stage of vascular healing and contributor to ISR is that of remodeling. The definition of remodeling varies by setting, in the strictest sense referring to a change in size or structure. In the vasculature, there are two forms of remodeling: geometric or adaptive remodeling, referring to a change in vessel size or diameter, 46 and arterial remodeling, referring to chronic structural changes in the arterial wall that may or may not affect vessel diameter. 58
Geometric remodeling is a compensatory phenomenon “characterized in a continuous spectrum by any change in vascular dimension,” 13 involving either arterial expansion (positive remodeling) or arterial contracture (negative remodeling). 13,32,168,209 More specifically, it is the relative change in vessel size or the cross-sectional area within the confines of the EEL. 168,206 Synonymous terms for positive remodeling and negative remodeling include expansive or outward remodeling and constrictive or inward remodeling, respectively. Given that geometric remodeling is a chronic event occurring from 1 to 6 months following balloon angioplasty, it is distinct from acute arterial elastic recoil that occurs within minutes to days of intervention, even though both processes involve a change in the EEL area. The type of geometric remodeling and the role that it plays in ISR vary by animal model and the type of initiating injury. 30,46,71,168 This highlights the importance of model selection and means of evaluation in chronic studies of endovascular devices.
In a diseased artery or following balloon angioplasty, positive remodeling may compensate for plaque burden or lumen loss relative to neointimal hyperplasia, or, at the extreme, it may result in the detrimental outcome of aneurysmal dilatation. 168,206 Paradoxically, whereas positive remodeling may seem a favorable clinical event compensating for luminal plaque burden, the consistency of associated heightened inflammation, expression of proinflammatory cytokines, and increased protease activity contributes to plaque instability and an enhanced potential for acute coronary events. 136,137 In arteries implanted with metallic devices, positive remodeling is unfavorable, clinically translating to stent malapposition and preclinically being measured quantitatively as radial gap. Stent malapposition is indicative of a localized adverse effect of the device. 2,197 There are two underlying, though not mutually exclusive, pathophysiologies for positive remodeling in stented arteries. First, it may be the result of localized toxic mural necrosis, being evidenced by the presence of fibrin and/or cellular debris filling the remnant void of cellular loss (Fig. 7). This type of toxicity has been observed in association with DES eluting cytotoxic drugs, as well as with radioactive stents. ‡ Second, positive remodeling may be caused by inflammation that, in the porcine model, is often characterized by granulocytic (neutrophils, eosinophils) involvement and is affiliated with hypersensitivity reactions in autopsy specimens. 129,197 Again, these two modalities of positive remodeling are not mutually exclusive; that is, a localized toxic effect often is associated with a brisk inflammatory response. 195 The time of occurrence and the nature of positive remodeling observed in preclinical models can serve as a useful indicator as to the DES-driven etiology by correlating histological observations with the profiles of drug elution and/or polymer degradation or integrity.
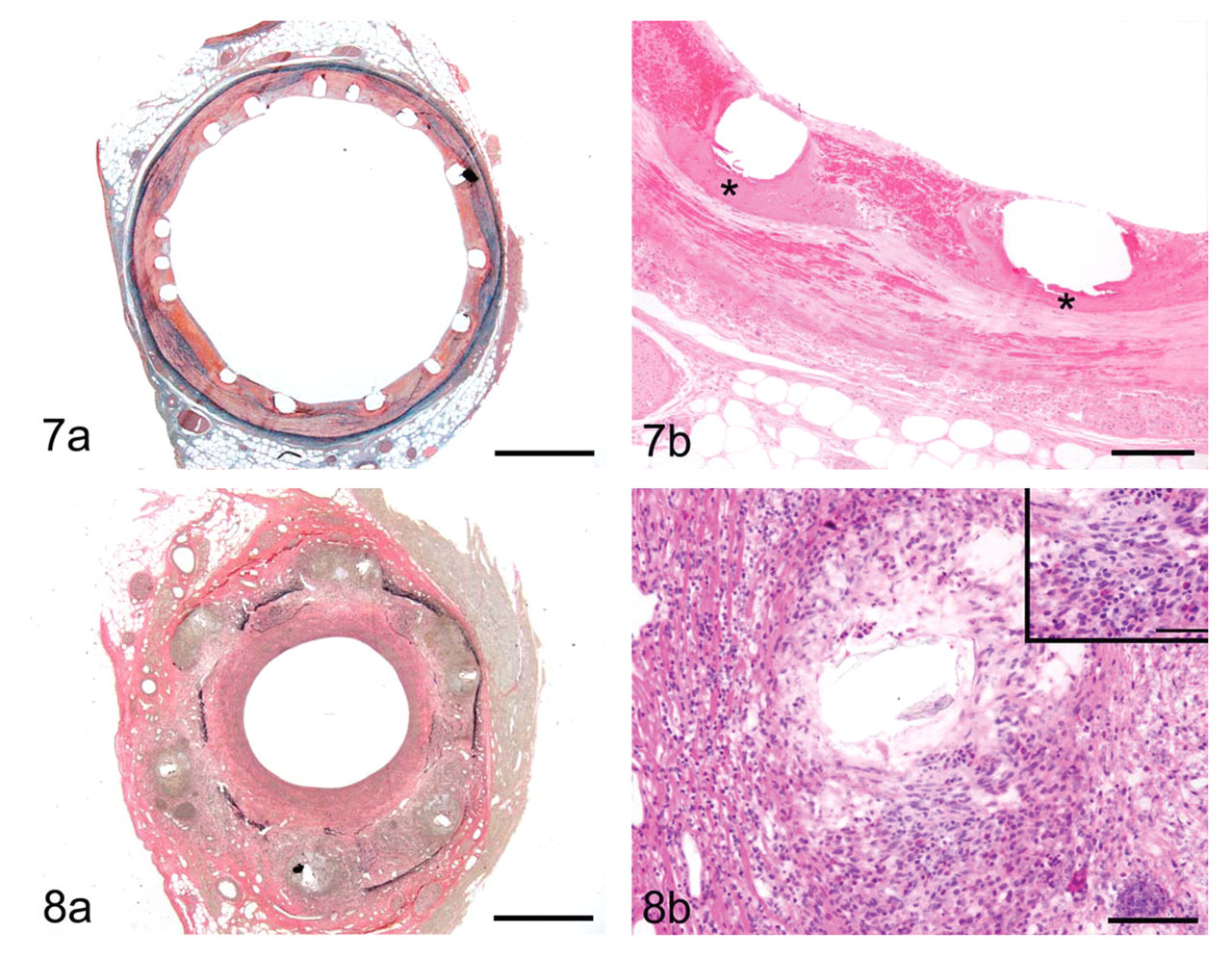
In contrast to positive remodeling, negative remodeling is defined by the restenotic loss of lumen area. Metallic stents were introduced to prevent this common reason for failure following balloon angioplasty and intravascular brachytherapy, 5,85,90,120,194 Even despite the structural support of a metallic stent, there is circumferential contracture of the fibrotic neointima and adventitia. Histologically this is evidenced by disruption of the IEL and EEL, fibrotic replacement of the media, and ultimately greater lumen loss (Fig. 8). Additionally, stented arteries with initial positive remodeling eventually heal with negative remodeling because progressive adventitial fibrosis, exuberant neointimal proliferation, and arterial contracture compensate for the loss of mural integrity. 78,196 Negative remodeling may also be a factor affecting bioabsorbable or other nonpermanent devices as they gradually lose radial strength and succumb to the contractile forces of negative remodeling. 37,69,115,203 Mechanistically, for chronically implanted bioabsorbable devices, negative remodeling may be distinguished from stent recoil histologically by medial integrity and the presence (remodeling) or absence (recoil) of medial and adventitial fibrosis. In this application, correlating the preclinical angiographic and histological observations with bench and in vivo data on stent radial strength and degradation can offer valuable insight.
In addition to positive and negative remodeling, the more encompassing definition of arterial remodeling includes the progressive modifications of the neointima and arterial wall that occur until vascular quiescence at the site of endovascular intervention is achieved. Within this final phase, neointimal regression is the paramount event. 198 This has been documented in preclinical and clinical settings, although as with other stages of vascular healing, there is a significant temporal difference between human and preclinical models in its onset and duration. 49,93,101,157,199 Contributing factors in neointimal regression include changes in proteoglycans and replacement of type III collagen with type I collagen; 25,149,178,216 furthermore, in autopsy specimens, a progressive loss of smooth muscle cells is noted. 43 All together, the resulting qualitative and quantitative changes in the neointima, media, and adventitia determine ISR. 46,116 Relatively small changes in vessel diameter or neointimal thickness can make for major changes in lumen diameter, especially for arteries with a thick vascular wall relative to the vessel diameter, whether mural thickening is due to atherosclerotic plaque and/or restenosis. 85
Although many of the stages of vascular wound healing are comparable to basic wound healing typified by uncomplicated dermal lesions, there is substantial temporal difference between the two. 118 Uncomplicated dermal healing is largely complete by 2 weeks, whereas complete healing following coronary stenting with a metallic stent may take up to 18 months (Table 2). Additional delays may be imparted by the foreign material (stent) and underlying atherosclerotic lesions. The localized effects of the drug and/or polymer for DES systems may result in even further delay. 43,170,199 Eventually, however, the site of uncomplicated vascular injury becomes quiescent and stable, which contrasts the periods of activity and instability that complicate the life history of atherosclerosis. 135 For DES systems, determining the time point at which this vascular quiescence is reached remains elusive in the clinical setting and has been established for only one DES system in the preclinical setting. 139
Current Generation DES: Correlating Preclinical Studies and Clinical Performance
Currently in the USA, there are 4 approved DESs—CYPHER, TAXUS, ENDEAVOR, and XIENCE V—with innumerable other DESs engaged in international clinical trials. Correlates between the published preclinical and clinical performance of approved and investigational DES systems offer insights into the impact of stent design, polymer biocompatibility, and toxicokinetics on vascular response and healing and may provide further insight into attributes or handicaps unique to each DES system. Regardless of the DES system, it is foremost to recognize the consistent trend for DES systems to fail to demonstrate efficacy in long-term studies (> 30 days) in standard preclinical models as measured by the reduction of neointimal hyperplasia (ISR), even though efficacy may be shown as a short-term effect. 22,23,29,179,214 This contrasts the clinical efficacy demonstrated with approved DES systems in pivotal clinical trails. 12,123,171,177 The dichotomy between preclinical and clinical performance of DES systems reiterates the need for caution in the interpretation of quantitative data from animal models and their extrapolation to humans, given that study endpoints are not the same (eg, histomorphometric in animals versus angiographic in humans). Current preclinical models should not be expected to offer an indication of long-term clinical efficacy based on the current methods of evaluation.
Standard preclinical models are predictive of overall DES safety, in contrast to efficacy—with adverse events and observations such as high mortality, myocardial infarction, medial necrosis, excess fibrin deposition, strut malapposition (elevated radial gap width), and positive remodeling being accurate indicators of localized vascular toxicity (Fig. 7). 82,131 These effects have been observed principally in association with DES systems eluting cytotoxic drugs. 67,78,151,195,214 Other projected indicators of DES safety—such as inflammation, rate of reendothelialization, mineralization, intraintimal fibrin and/or hemorrhage, and stent or subcomponent integrity—do not have as clearly defined acceptance criteria, although these changes have been observed with current DES systems. 22,51,130,142,169 Because of the inherent limitations of the current standard preclinical models, there is a drive to develop next-generation models that can better assess the performance of next-generation endovascular devices or can offer additional insights into the performance of current approved devices. Atherosclerotic and diabetic models are on the forefront of development, becoming more desirable for evaluating DES performance, although their standardization is lacking. Despite the inherent lesion variability that occurs with these models, 140,186 it is of value to evaluate DESs in an environment in which the primary cause for the vascular intervention, the underlying atherosclerotic lesion, is present. Furthermore, DES systems may affect not only the development of ISR but they also may impact the formation or progression of the local atherosclerotic lesion and, thereby, long-term device efficacy. 21,132,143 This impact has been alluded to clinically, with some current DESs being associated with accelerated development of localized atherosclerosis, as compared to metallic stents. 132
Conclusion
A variety of small and large animal models have been used to study ISR and systemic or local device-based therapies aimed at its prevention. The number of preclinical models reflects the inability of any particular model to fulfill all three objectives for the evaluation of endovascular treatments for ISR: device safety, device efficacy, and pathophysiology of atherosclerosis and vascular healing. Although small animals are low cost, readily available, easy to handle, and genetically well defined, results of studies evaluating agents or devices for reducing ISR in these models have offered only limited predictive clinical value. This is especially true for murine models. Conversely, large animal models—namely, the rabbit iliac and porcine coronary arterial models—have proven to be more predictive of ISR inhibition in humans, although their predictive value is not absolute. Animal models of ISR have proven useful for the assessment of local toxicity and for the determination of the margin of safety despite the intrinsic differences noted between animal and human studies.
DES systems are novel and highly dynamic treatment modalities. As data from clinical trials and standardized preclinical studies accrue, so will our understanding of the predictive value of preclinical models in assessing long-term safety and potential efficacy. At the current time, a well-designed preclinical program for DESs and related endovascular technologies may best utilize small animal models or diseased animal models for early investigations into pathophysiology and efficacy screening of proposed agents or device systems, whereas development studies should focus utilization within large animal models—namely, the porcine coronary arterial model—for its robustness and standardization. To derive the maximum benefit from preclinical investigations into the safety and efficacy of endovascular devices, appropriate model application, respect and understanding of model limitations, and appropriate interpretation of the study results in light of model limitations are critical. Moreover, continued development and characterization of suitable preclinical models is necessary to match the demands of interventional cardiology as next-generation DES systems further evolve.
DES systems are not unique: The research and development on combination devices, defined as consisting of 2 or more regulated components (such as a device with a drug and/or biologic), is ever increasing. Combination devices are being developed for a range of applications beyond coronary and peripheral arterial diseases, including orthopedics, catheters, dental implants, and wound dressings. 76 Given that these products are more complex than the sum of their parts, veterinary pathologists play a critical role in evaluating the safety and efficacy of combination devices in their ever-increasing multitude of applications.
Footnotes
Acknowledgements
I thank Dr Igor Polyakov and Katrin Boeke-Purkis at Abbott Vascular and Dr Leslie Coleman at eValve for review of this article. In addition, I recognize the members of the preclinical research department of Abbott Vascular and Dr Renu Virmani and her staff at CVPath Institute, Inc., for their continued contributions to advance our understanding of coronary arterial disease and the development of related interventional treatments.
The author declared no conflicts of interests with respect to the authorship or the publication of this article.
The author received financial support for the research and authorship of this article from Abbott Vascular, Santa Clara, CA.

