Abstract
Over 85% of people with lung cancer eventually succumb to this disease, largely because current chemotherapies are ineffective. The testing and validation of promising new approaches generally rely on achieving responses with cell lines in vitro or in tumor xenografts in nude mice. However, quite often the results seen with these models are not recapitulated in the clinic, thus necessitating the need for better animal models of lung cancer for preclinical testing of new therapies. One promising model is that of orthotopic lung cancer, where xenografts of human lung cancer are established in lungs of immunodeficient rodents. The problems associated with this model include poor rates of engraftment, limited tumor multiplicity, and a heightened risk for surgical trauma. The purpose of our study was to develop an efficient approach to engraftment of orthotopic tumors throughout the lungs of the Rowett nude rat. Initially, we augmented immunosuppression in the rats with whole-body X-irradiation and then used orotracheal cannulas to intratracheally instill human cancer cells from the Calu-6 cell line. This protocol produced a low rate of engraftment and low tumor multiplicity. The hypothesis that slight disruption of the pulmonary epithelium or the surfactant layer would allow better tumor engraftment was tested by coadministration of either pancreatic elastase or ethylenediaminetetraacetic acid (EDTA) along with the cell instillations. Lung tumor engraftment was evaluated 8 weeks after instillation. The inclusion of elastase or EDTA with the Calu-6 cells resulted in an 80–100% engraftment rate, respectively. Coadministration of EDTA resulted in significantly larger and greater numbers of tumors/lung than those in elastase-treated animals. Temporal studies demonstrated that small nodules were scattered throughout the lung parenchyma 5 weeks after instilling Calu-6 cells and EDTA. These nodules grew to coalesce and form large masses that effaced >75% of the parenchyma at 9 weeks postinstillation. The refinements made through our studies have led to the development of an orthotopic lung cancer model that should facilitate the evaluation of novel therapies designed to treat or impede lung cancer development.
Lung cancer is the leading cause of cancer-related death in the United States. 3 Paramount to this problem is the fact that approximately 85% of diagnoses are made when the tumor can no longer be removed surgically, and the median survival for these people is 13 months due to a lack of effective therapies. 28 The development of novel, strongly efficacious therapeutic approaches for lung cancer is an urgent priority for the oncology community.
Cancer cell lines have been propagated in immunodeficient rodents to study tumor behavior such as growth rates and metastasis. Palpable subcutaneous neoplastic xenografts established by injection into SCID or nude mice are also amenable to measurement of tumor shrinkage following oral and parenteral treatments with chemotherapeutic agents. Positive treatment results when using this animal model are perceived as evidence of chemotherapeutic efficacy in vivo. The validity of such results is questionable, given the pharmacodynamic differences between treatment of subcutaneous xenografts and treatment of neoplasms situated in organs from which they would normally arise. Indeed, tumor chemosensitivity to anticancer drugs depends on the anatomic site of xenografts in recipient animals. 18,19,21,34 Cancer models of orthotopic xenografts (accordingly, xenografts produced in the same organ or tissue from which the donor tumor arose) mimic the human pathophysiologic condition including tumor expansion, vascularization, and tissue destruction. Thus, evaluating cancer therapies using an orthotopic lung cancer model in the nude rat should better predict the responses that will be obtained when treating human patients. 18
Tumor xenografts have been produced in the lungs of immunosuppressed animals by intravenous injection of cultured human and rodent cancer cell suspensions. 11 Such tumors usually resemble metastatic disease and have been produced by injection of cells derived from melanoma, 9,20 fibrosarcoma, 25,27 and various nonpulmonary carcinomas. 10,22,32 Orthotopic models of primary lung cancer have also been produced by this method 21,36 as well as by surgical implantation of cells or tumor fragments via thoracotomy. 7,16
Several investigators have created orthotopic pulmonary xenografts by instilling tumor cells into the upper airways of SCID mice, nude mice, or nude rats. 12,13,17,23,24,37 However, many have relied on surgical exteriorization of the trachea for placement of cannulas and instillation of cells. Depending on the cell type, the tumors that develop from surgical instillation are often localized to a single lung lobe. 17,23,24 The purpose of our study was to develop an efficient and effective approach to engraft orthotopic tumors throughout the lungs of the Rowett nude rat. Because of the size differences between the mouse and rat, orthotopic lung cancer in the nude rat is a better model for testing the efficacy of drug or gene therapy delivered directly to the lungs via inhalation or direct instillation.
Materials and Methods
Animals and cell lines
Male and female Rowett nude rats (Cr:NIH-rnu), 8–10 weeks old, were obtained from Frederick Cancer Research and Development Center (Frederick, MD). Lung tumor-derived cell lines were obtained from original stocks of the American Type Culture Collection (Manassas, VA). The cell lines used included Calu-6 cells (anaplastic carcinoma), A549 cells (adenocarcinoma), and NCI-H292 cells (mucoepidermoid carcinoma). Cell lines were grown in RPMI medium with 10% fetal bovine serum, L-glutamine, and gentamicin (Gibco BRL, Gaithersburg, MD). Cells were harvested by trypsin and were washed in HEPES. Depending on the experiment, 1 to 20 × 106 cells were resuspended in 0.2–0.5 ml of RPMI containing 10% fetal bovine serum for intratracheal instillation into individual animals.
Tumor cell implantation
To further suppress their immune system prior to instillation of tumors, 17 rats were X-irradiated, whole-body, for varying lengths of time using a Phillips RT 250 X-ray therapy unit (Phillips Medical Systems, Shelton, CT) set at 250 kVp, 15 mA, and a source-to-object distance of 100 cm. Single-dose regimens of X-irradiation included either 450 or 500 rads. Rats were given amoxicillin-clavulanate (Augmentin®; SmithKline Beecham Pharmaceuticals, Philadelphia, PA) as a suspension in the drinking water at 0.35 mg/ml 17 or enrofloxacin (Baytril® Injectable Solution; Miles Animal Health, Shawnee Mission, KS) in the drinking water at 0.64 mg/ml. 14 Animals were anesthetized with Halothane, placed head up in dorsal recumbency on a 45° incline, and intratracheally instilled with human lung cancer cells using transdermal laryngotracheal illumination and modified 16-gauge intravenous catheters (Critikon, Inc., Tampa, FL) for orotracheal intubation. 4
Porcine pancreatic elastase (8.5–12.0 IU; Calbiochem, San Diego, CA) or ethylenediaminetetraacetic acid (EDTA, 5–7 µmole; Sigma, St. Louis, MO) were included in the cell instillates in some experiments. Elastase, when added to a suspension of Calu-6 cells (1.4 × 106 cells/ml) at up to 43 IU/ml (incubated on ice for 60 minutes and then warmed to room temperature for 30 minutes), had no untoward effect on cell viability as determined by trypan blue dye exclusion. Calu-6 cell suspensions also tolerated up to 20 mM EDTA. The elastase dose chosen for intratracheal instillation was approximately 0.05 IU/g body weight (BW) and was not expected to induce the severe parenchymal damage that occurs with emphysema models in rats. 8 EDTA in RPMI media was administered by intratracheal instillation to male Fischer 344 rats (mean BW 145.5 g) at up to 5 µmole/0.5 ml; these animals exhibited no clinical signs and no histologic pulmonary abnormalities.
Histopathology
The rats were killed 3–9 weeks postinstillation with an i.p. overdose of pentobarbital and were evaluated for gross and histologic evidence of pulmonary tumor formation and metastasis. Lungs were inflated via tracheal cannulas with either 10% neutral-buffered formalin or buffered 4% paraformaldehyde until the pleura was smooth and then were fixed further by immersion in fixative for at least 24 hours. Individual lobes were trimmed longitudinally to include sections of the axial airway, and tissue blocks were processed routinely and embedded in paraffin. Tumors that were not included in the axial sections were trimmed separately. Sections from the blocks were stained with hematoxylin and eosin (HE). In one experiment, two dimensions of the tumors in the HE-stained sections were measured, and an approximation of the original tumor volume was calculated for spherical or prolate ellipsoid shapes. 33 Statistical differences in tumor volumes and lung weights between treatment groups were determined by Pearson's correlation and analysis of variance (ANOVA) on rank-transformed data.
Results
Effect of cell number on tumor growth
Male and female rats were X-irradiated once with 450 rads and started on a 10-day treatment regimen of amoxicillin-clavulanate in their drinking water. Cells in 0.2 ml of media (1 or 5 × 106 Calu-6 cells, 1 or 2 × 106 A549 cells, 1 or 1.5 × 106 NCI-H292 cells) were intratracheally instilled into the rats (six per group) 4 hours after irradiation. The males were killed at 3 weeks and the females at 8 weeks postinstillation. None of the rats had grossly evident pulmonary or nonpulmonary tumors except for one male animal (1.5 × 106 NCI-H292 cells) that developed a tumor within the midcervical fascia encompassing the trachea and esophagus. The lungs of one male rat (2 × 106 A549 cells) contained histologic evidence of tumor formation, in which a small nodule approximately 10 alveoli in diameter effaced parenchymal tissue near the hilus within the right caudal lung lobe (not shown). Thus, 2 of 36 rats in this experiment developed tumors (one pulmonary, one nonpulmonary). The lack of xenografts in this experiment suggested that either the cancer cell dose or the radiation-induced immune suppression was insufficient.
Effect of EDTA or elastase coadministration with Calu-6 cells on tumor engraftment and growth
Slight disruption of the pulmonary parenchymal epithelium and/or surfactant layer by coadministration of either elastase or EDTA was postulated to produce a better environment for growth of the tumor cells. Ten male and 10 female nude rats were given enrofloxacin in the drinking water starting 3 days before immune suppression by X-irradiation. Animals were maintained on the antibiotic for the duration of the experiment. A one-time X-ray exposure of 500 rads was administered to the rats. The intratracheal dose of cells, EDTA, and/or elastase was determined by mean BW. On the day following irradiation, the males (mean BW 239.1 g, SD = 14.2) received 20 × 106 Calu-6 cells in 0.5 ml of RPMI media containing either 12 IU elastase (n = 5) or 7 µmole EDTA (n = 5). The females (mean BW 165.2 g, SD = 10.7) received 15 × 106 Calu-6 cells in 0.5 ml of media containing either 8.5 IU elastase (n = 5) or 5 µmole EDTA (n = 5).
Body weights in all groups increased from the start of the experiment to 4–6 weeks postinstillation. After 6 weeks in males and 4 weeks in females, coadministration of EDTA was associated with decreased body weight, while the weights of the elastase-treated animals were maintained to the final time point (data not shown). At necropsy (8 weeks postinstillation), eight of the 10 elastase-treated animals had tumors. In most specimens, the tumors were relatively small (<5 mm in diameter) and occurred multifocally in both peripheral and central regions of the lung parenchyma. All 10 of the EDTA-treated animals had large coalesced and contiguous masses. In three of the five EDTA-treated male rats, the tumors were restricted to the right lung lobes and were distributed primarily in the hilar regions or juxtaposed to the axial airway; occasional peripheral nodules were noted. In addition to tumors in the right lung lobes, the remaining EDTA-treated male rats also had large hilar tumors in the left lung. Likewise, in the EDTA-treated females, the right lung lobes were preferentially affected by large coalesced hilar tumors; one female rat lacked tumors in its left lung, while left lungs of other rats contained small nodules in both hilar and peripheral regions. The distribution was further varied, as evidenced by the lungs of one female where only a single mass was present in the dorsal region of the left lung. The lung weights of both female and male EDTA-treated animals were significantly greater than those of the elastase-treated animals (P < 0.0001, Table 1). The tumor volumes, approximated from dimensions on HE-stained sections and summed for individual specimens, correlated highly with the lung weights obtained at necropsy (P < 0.0001, r = 0.99, Pearson's correlation).
Summary of lung weights and approximate tumor volumes. ∗
∗ All values are the mean ± standard deviation.
† Body and lung weights are for all animals in the first experiment and for only those animals with lung tumors in the second and third experiments.
‡ NA = not applicable; PI = postinstillation with Calu-6 tumor cells; ND = not determined.
§ Two-way ANOVA on rank-transformed data in this experiment showed a statistically significant effect of elastase or EDTA coadministration (P < 0.0001) but no effect of gender or of an interaction on lung weights.
‖ Two-way ANOVA on rank-transformed data in this experiment showed a statistically significant effect of time (P < 0.0001) and gender (P = 0.0001) but no effect of an interaction on lung weights.
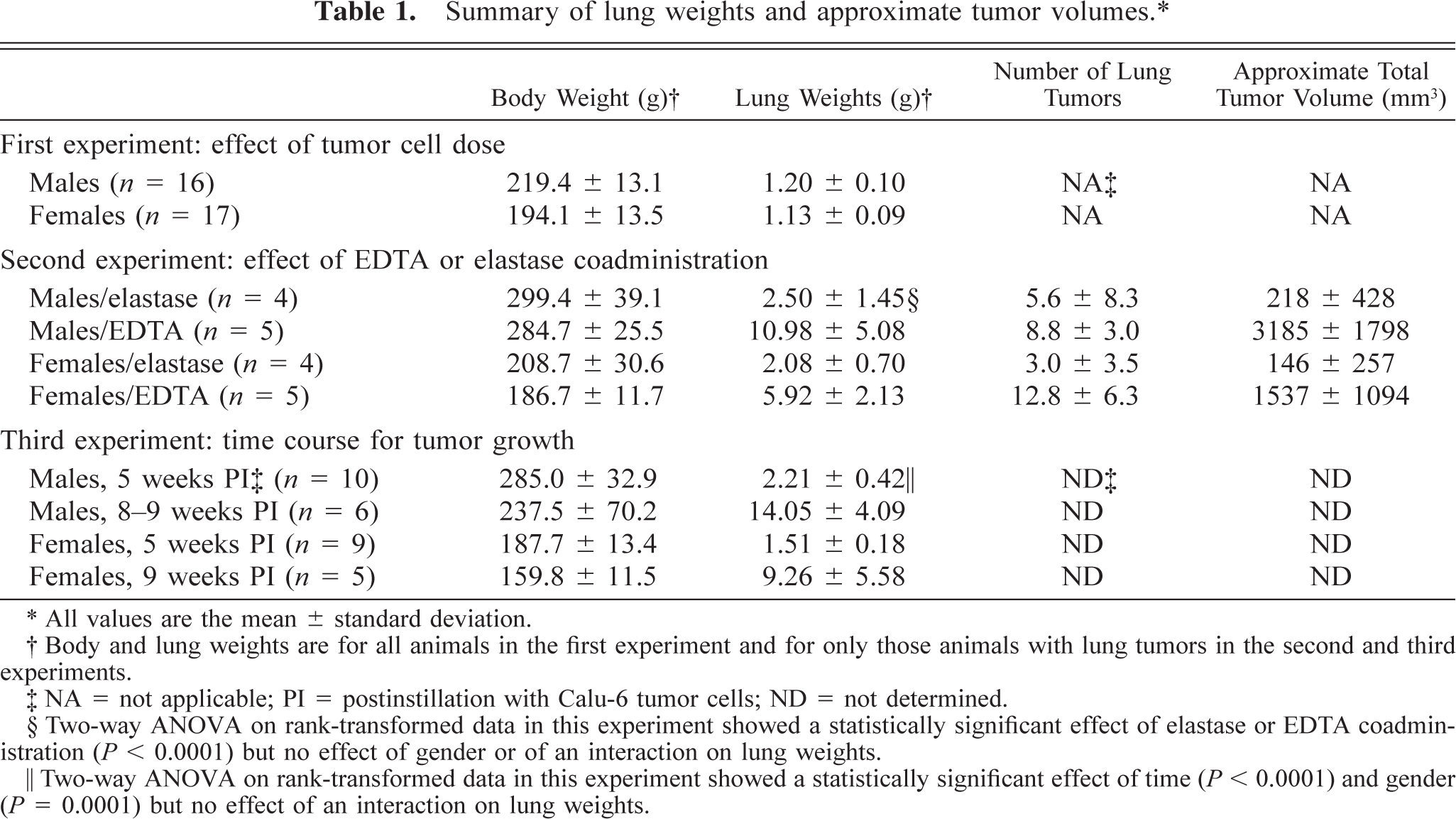
The tumors were typical of anaplastic solid carcinomas with effacement of the parenchyma and many of the smaller airways and blood vessels (Fig. 1). The largest masses effaced more than 75% of the parenchyma in a section. Parenchymal necrosis, inflammation, and hemorrhage were present in the periphery of some affected lobes, and central necrosis of larger masses was common. The cells were arranged in cords and nests that were sometimes bounded by remnants of alveolar septa. The cells had variably distinct margins, were disoriented, and varied in morphology. In some tumors, neoplastic cells had small to moderate amounts of pale basophilic cytoplasm, the nuclei were ovoid and mildly vesicular, and anisokaryosis was moderate. In other foci of the same tumors or in different tumors, anisokaryosis was moderate to marked and the cells had moderately large amounts of pale amphophilic cytoplasm with fine vacuoles that resembled mucoid vesicles. Mitotic figures and apoptotic bodies were often numerous.
Lung, male nude rat, 8 weeks after intratracheal instillation of 20 × 106 Calu-6 cells and EDTA. Four convergent masses in the right caudal lobe are composed of solid nests and cords of pleomorphic neoplastic cells. One mass encompasses and partially effaces a small airway (arrow). HE. Bar = 275 µm.
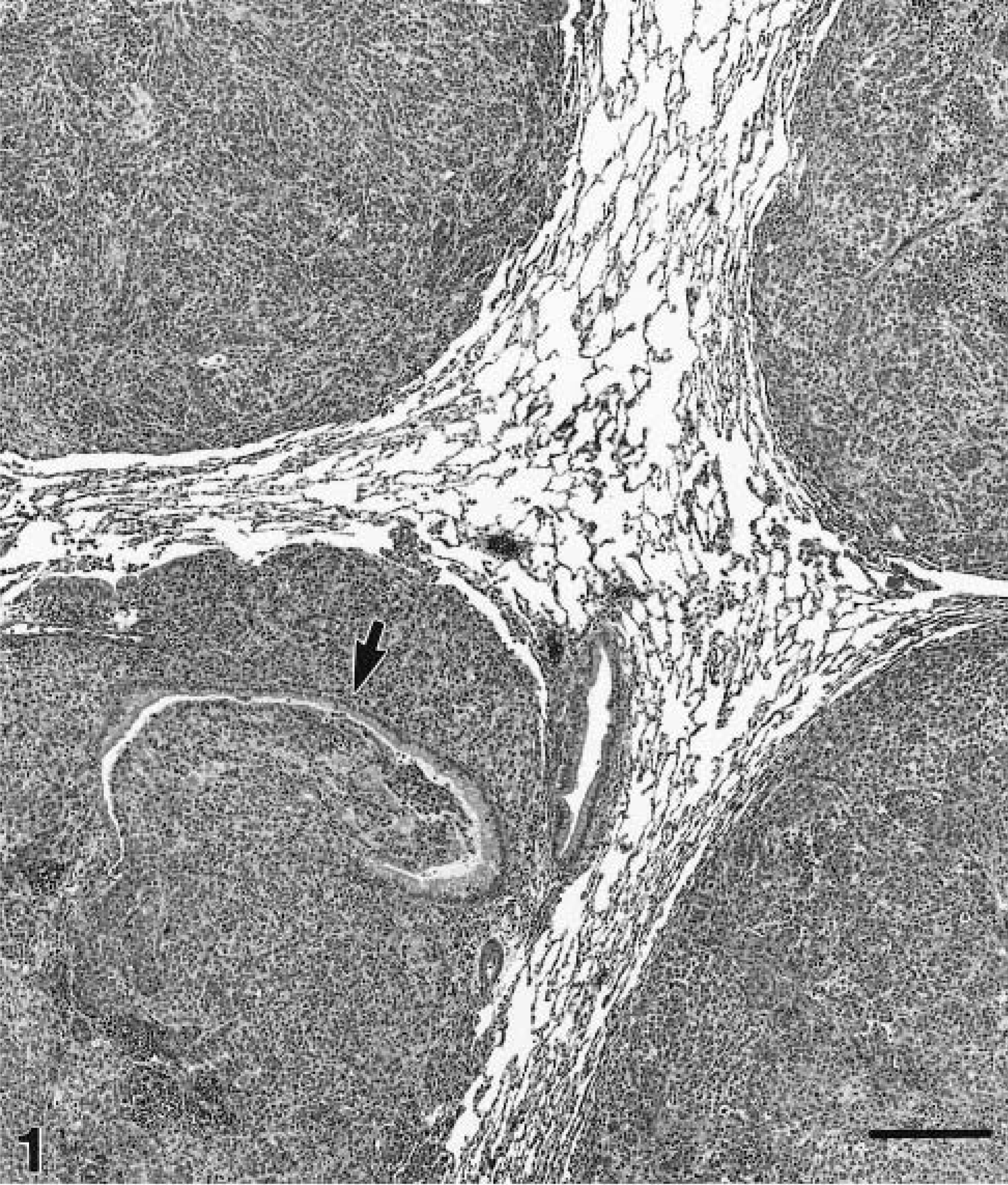
Vascular invasion by neoplastic tissue was evident only in lungs from four rats (one male, three females) given EDTA (Fig. 2). Despite these histologic findings and the fact that EDTA coadministration caused growth of large tumors that effaced large portions of lung lobes, metastatic lesions were found in only two rats. In one of these rats, neoplastic emboli caused infarction of the kidney. Neoplastic cells grew in a subcapsular sinus of a mediastinal lymph node of the other. Two animals had fibrous adhesions between lung tumors and the parietal pleura covering the diaphragm and dorsal thorax. Neither of these adhesions was associated with invasion into underlying musculature.
Lung, female nude rat, 8 weeks after intratracheal instillation of 15 × 106 Calu-6 cells and EDTA. A large mass has invaded the pulmonary vein at the hilus of the right caudal lung lobe. The intravascular extension of the mass is partially necrotic (∗), and the mass has destroyed part of the vessel wall (arrows). This animal had renal infarcts from tumor emboli. HE. Bar = 135 µm.
Time course for orthotopic lung tumor growth
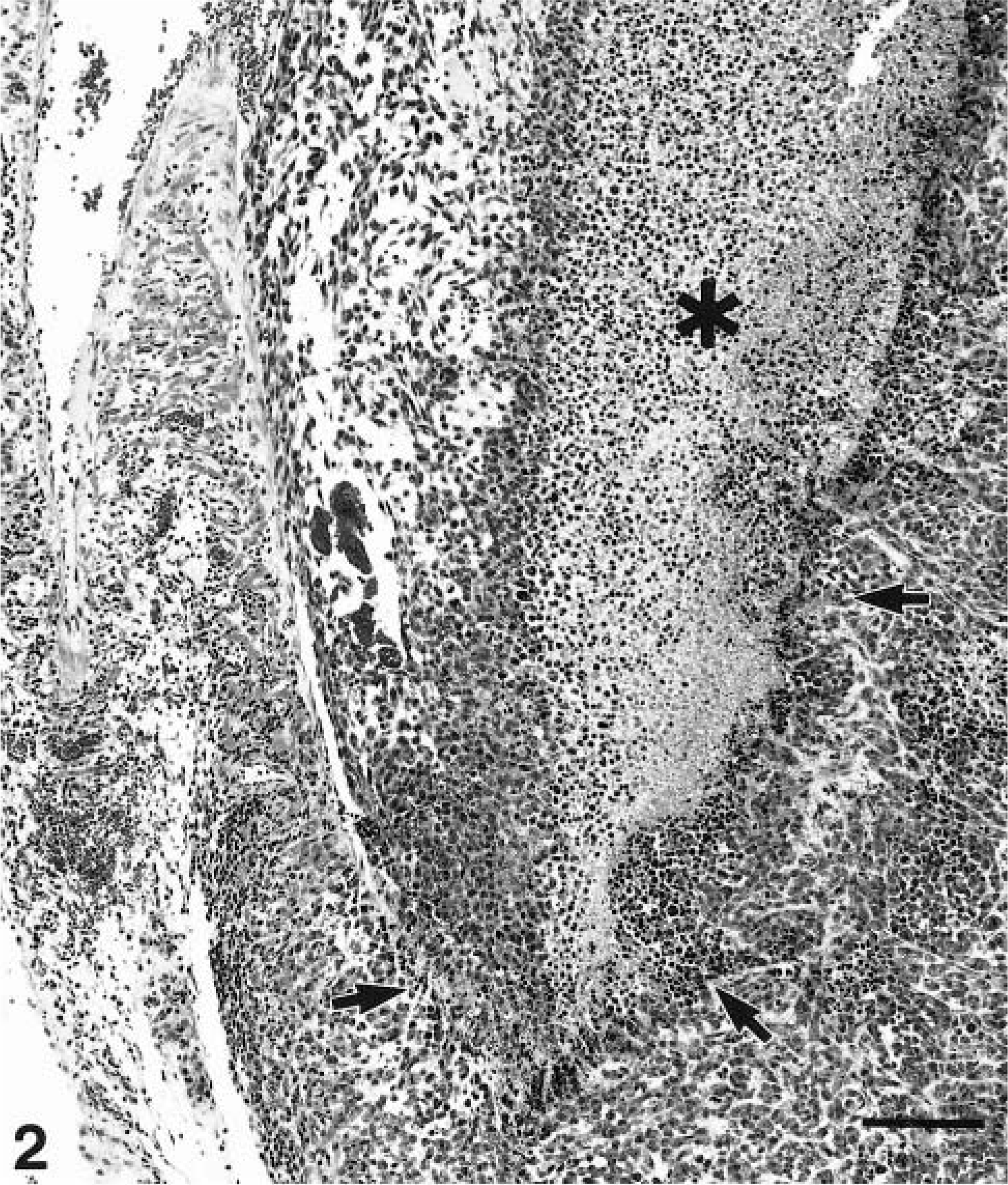
Thirty-two nude rats were X-irradiated with 500 rads. The following day, Calu-6 cells and EDTA were intratracheally instilled (16 males, 20 × 106 cells and 7 µmole EDTA each; 16 females, 15 × 106 cells and 5 µmole EDTA each). Animals were maintained on enrofloxacin antibiotic in the drinking water and were killed either at 5 or 9 weeks postinstillation for evaluation of xenograft formation. At week 5, 10 male and 10 female rats were killed. All males and nine females had lung tumors. The nodular xenografts were numerous, small (<1–3 mm diameter), and scattered primarily in the central regions of the parenchyma of most lung lobes (Fig. 3). For the remaining rats, one male died, and another was euthanized at week 8 postinstillation. The lungs of both animals contained large coalesced masses in all lobes. At week 9, the remaining six female and four male rats were killed. All males and five females had lung tumors. They were large (up to 3–4 cm in diameter), often effacing >75% of the parenchyma, and were composed of contiguous and coalesced masses (Fig. 4).
Lung, male nude rat, 5 weeks after intratracheal instillation of 20 × 106 Calu-6 cells and EDTA. Neoplastic nodules are scattered primarily in the central parenchyma of the right caudal lung lobe. HE. Bar = 670 µm.
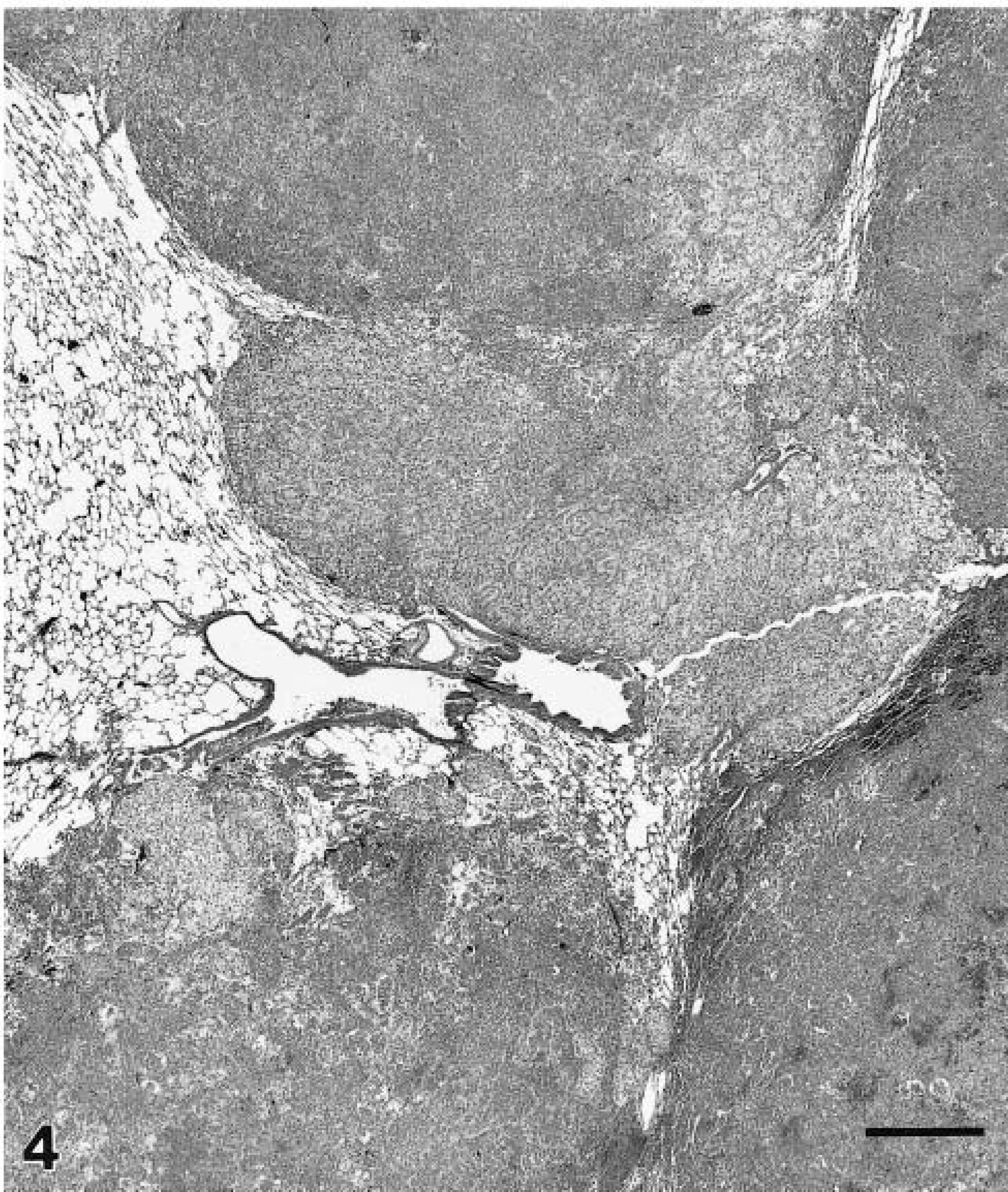
Lung, male nude rat, 9 weeks after intratracheal instillation of 20 × 106 Calu-6 cells and EDTA. Large coalesced and contiguous tumors are near the periphery in the right caudal lung lobe. The masses have slightly differing morphologic features having arisen from individual nodules. HE. Bar = 670 µm.
The weights of tumor-bearing lungs from rats of the later time points were significantly greater than those of the rats from week 5 (Table 1). The mean lung weight for male rats at later time points was 6.4 times that of the lungs of male rats at week 5, while the mean weight of tumor-bearing lungs from the female rats at week 9 was 5.4-fold greater than that at week 5. ANOVA indicated that there was a significant effect of gender (P = 0.0001) and of time (P < 0.0001) on lung weight, but no statistically significant interaction between gender and time was noted. Thus, tumors were larger in males, as indicated by lung weight, but time-dependent tumor growth was not significantly influenced by gender.
Discussion
Our studies have led to the development of a rapid and reproducible model of orthotopic lung cancer in which human lung tumor-derived cell lines can be engrafted throughout the pulmonary parenchyma. Key to the success of this model is the inclusion of EDTA with the instilled cells and the orotracheal method of intubation. The use of the nude rat for this orthotopic model allows the evaluation of promising therapeutic and prevention drugs for respiratory carcinogenesis using both systemic delivery of compounds and respiratory tract deposition of aerosols or instillations.
Orthotopic lung neoplasms were produced in virtually 100% of the animals when suspensions of Calu-6 tumor cells were coadministered with EDTA in intratracheal instillations. The EDTA coadministration was also associated with more numerous engrafted tumors. These tumors were dispersed throughout the lung parenchyma (primarily in the central regions) and grew to form large, coalesced masses that destroyed the majority of the lung structure. The improved engraftment in the elastase/EDTA coadministration experiment was partly due to the increased dose of instilled cancer cells in comparison with the first experiment. However, female rats in this experiment received only three times and males four times as many cells as in the first experiment, and the elastase coadministration was associated with much smaller and fewer tumors than those associated with EDTA coadministration. By disrupting calcium-dependent, cadherin-mediated, cell-cell adhesion, 31 EDTA may have exposed the parenchymal interstitial matrix and thus allowed better attachment and less subsequent clearance of the transplanted cells. Alternatively, EDTA may have affected cadherins of the instilled cancer cells to create a more invasive phenotype. 29 The EDTA may also have disrupted pulmonary surfactant by interfering with the calcium-dependent binding of surfactant proteins and lipids 30 or disrupted clearance of the cancer cells by alveolar macrophages and the mucociliary apparatus. Elastase lyses cadherins and matrix proteins of basement membranes, 5 so the dissimilarity between the results of elastase and EDTA coadministration is not understood. The elastase dose may have been insufficient, or inflammation typically associated with elastase instillation may have interfered with tumor cell attachment or may have enhanced tumor cell clearance.
The use of a modified intravenous catheter for orotracheal intubation and instillation of cells into the upper airways negated the surgical trauma induced by exteriorization of the trachea. With practice, the procedure is simple and efficient. In our studies, 20 rats were anesthetized, intubated, and instilled with cells within an hour without any associated mortality. Surgical approaches for transtracheal instillation of cells have produced engraftment rates in 80–100% of animals, 12,13,17 but such manipulations have also produced mortality of 5%. 17,24 These surgical approaches to instillation have had the goal of producing xenografts localized to a specific lung lobe (usually right caudal) to allow study of intrapulmonary metastasis. 17 However, such xenografts frequently remain localized and would not be appropriate for studying the effects of therapies on widespread pulmonary neoplasms.
Some studies have demonstrated that the inoculum dose of cancer cells is a determinant of the engraftment rate and subsequent tumor-related mortality. 24 This observation was supported by our studies where xenografts were not produced in animals receiving 5 × 106 cells or less. However, the enhanced engraftment of tumors in animals treated with EDTA indicates that the inoculum dose is not the only dependent variable. Cell type and cell-dependent time for tumor development may also determine the success of engraftment. 17 However, we have achieved a very high efficiency for engraftment of Calu-6 cells, as described here, and of A549 and Calu-3 cells as well (unpublished observations) when instilled with EDTA into the lungs of nude rats. Thus, orthotopic tumors can be established by our procedure independent of tumor phenotype and genetic alterations (e.g., p53, K-ras, or p16 status). This should greatly facilitate the ability to assess the responsiveness of genetically different lung tumors to cancer therapy.
The growth of scattered xenograft nodules throughout the lung parenchyma into large coalesced masses (Figs. 3 and 4) may lend itself to studies of cancer chemopreventive agents. Field cancerization, a condition that reflects the exposure of the whole lung to inhaled carcinogens within tobacco smoke, predicts that multifocal sites of independent initiation should be present throughout the lungs of people at highest risk for lung cancer. This concept has been validated through the identification of chromosome losses and gains both in preinvasive lesions and nonmalignant tissues adjacent or peripheral to primary tumors. 6,26,35 The impact of field cancerization is best appreciated by the fact that 25–40% of persons who have had resections for stage I lung cancer develop a second primary tumor. 2,15 The multifocal xenografts in the model of orthotopic lung cancer in the nude rat may mimic this human condition. Currently, the most popular model for evaluating field cancerization and lung tumor development is the A/J mouse lung. This model allows the assessment of intervention therapy on histologic and genetic progression of adenocarcinoma; however, the development of malignant tumors takes 32–40 weeks following carcinogen exposure. 1 One advantage of the orthotopic model in the nude rat is that the xenografts probably start to grow within days after instillation and then develop rapidly over 5–9 weeks. This should enable relatively quick assessment of the effects of therapeutic intervention administered prior to and soon after tumor cell instillation. Thus, this orthotopic model of human lung cancer offers an additional approach to evaluate novel therapeutics in the management of human respiratory carcinogenesis. The results with this orthotopic model may also better predict the responses for human clinical trials.
Footnotes
Acknowledgements
This work was supported by NIH grants P50 CA58184 and R01 ES08801; by National Research Service Award F32 CA88431 from the National Cancer Institute (PGT); by the Oxnard Foundation of Irvine, California; and by the Office of Biological and Environmental Research, U.S. Department of Energy under Cooperative Agreement DE-FC04–96AL 76406. The work was performed in facilities fully accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International. We thank Dolores Esparza, Sally Winters, Marcie Grimes, and personnel in Small Animal Care, Necropsy, and Histopathology at LRRI for expert technical assistance, Dr. Fletcher Hahn for a helpful review, and members of the Institute's Technical Communications Unit for editorial assistance and preparation of this manuscript.
