Abstract
The phenotype of genetically engineered mice is a combination of both genetic and environmental factors that include the microflora of the mouse. The impact a particular microbe has on a mouse reflects the host–microbe interaction within the context of the mouse genotype and environment. Although often considered a confounding variable, many host–microbe interactions have resulted in the generation of novel model systems and characterization of new microbial agents. Microbes associated with overt disease in mice have been the historical focus of the laboratory animal medical and pathology community and literature. The advent of genetic engineering and the complex of mouse models have revealed previously unknown or disregarded agents that now oblige the attention of the biomedical research community. The purpose of this article is to describe and illustrate how phenotypes can be affected by microflora by focusing on the infectious diseases present in genetically engineered mouse (GEM) colonies of our collective institutions and by reviewing important agents that are rarely seen in most research facilities today. The goal is to introduce the concept of the role of microflora on phenotypes and in translational research using GEM models.
Keywords
The impact a particular microbe has on a genetically engineered mouse (GEM) reflects the host–microbe interaction within the context of the mouse genotype and environment. The underlying supposition is that any GEM, regardless of the genetic modification, should be considered “immunovague” at best; vigilance on the part of veterinary pathologists and scientists is essential to identify and characterize emerging infections or outbreaks that may be responsible for or may alter phenotypes. 41 Alterations in the microbial susceptibilities of GEM may be an anticipated effect (eg, in targeted disruption of key cytokines) or an unanticipated primary or secondary effect (eg, alterations in mucociliary clearance in the lung). Infectious agents that do not result in disease or persistent shedding in “normal” (inbred and outbred) mice can cause illness in GEM.
Microflora can affect translational research through overt disease and death; however, more often the resident microflora can result in subclinical disease and alterations in physiological function (immune response), microscopic changes (increased extramedullary hematopoiesis in the spleen 98 ), or altered study end points (xenograft tumor kinetics, 81 gene expression). 91
As research colonies become more stringent in defining specific pathogen-free requirements, most pathogenic agents such as Sendai virus have become significantly less prevalent. As such, new agents or those previously considered incidental or commensal are now being brought into the focus of the laboratory animal medicine and pathology community, particularly as the use of GEM increase. These microbial “emerging diseases” are defined as infectious agents that have been recently discovered (eg, mouse norovirus), infectious agents that have increased incidence (eg, mouse parvovirus), or microbes with newly identified roles on a specific phenotype (eg, Helicobacter spp) (C. Clifford, unpublished data, 2011). Most of these emerging agents were discovered or recognized in parallel with the tremendously increased use of genetically modified mice. Although attention has focused on the interaction of primary or opportunistic pathogens with a mouse host (see references 2, 41, and 98 for detailed reviews), the role of normal flora or variations in normal flora should not be ignored. The mouse microbiome (defined as the microbial communities and their genetic elements in an environment such as gastrointestinal tract, see Table 1 ), is poorly understood but is increasingly thought to play a role in phenotype development as mammals and their microbiota form a symbiotic relationship. 32,113,118,119 Although often considered a confounding variable, naturally occurring alterations in microflora (such as infection with an opportunistic bacteria) have resulted in the generation of novel model systems 39 and characterization of new microbial agents. 57
Glossary of Terms Related to Genetically Engineered Mice

SPF, specific pathogen free.
Although the bulk of this article primarily focuses on infections with a single viral or bacterial agent, an increasing body of work recognizes the multifactorial nature of disease. 11,22,65 GEM are often carriers of multiple infectious agents and have a unique enteric microbiome 29,113 that may variably affect the mouse depending on the genetic manipulation. Infection with 1 virus or bacterial species may alter the outcome of infection with another virus or bacterial species. 22,65,74 In this review, we provide a glossary of terms related to GEM, their microbes, and microbial status specifically (Table 1). Although most of these terms will be familiar to readers, certain concepts such as the different definitions of specific pathogen free between facilities, germ free, defined flora, and conventional mice are important to understand. The tables within this article (Table 1, glossary and definitions in the context of mice; Table 2 , summary of agents by primary disease type) and in supplemental materials (Supplemental Tables 1, viral agents; 2, bacterial agents; and 3, eukaryotic agents) list additional important agents, and the reader is referred to the references for further details (for supplemental materials, please visit http://vet.sagepub.com/supplemental).
Agents by Primary Disease Phenotype When There Are Pathology Findings or Clinical Disease in Mice a
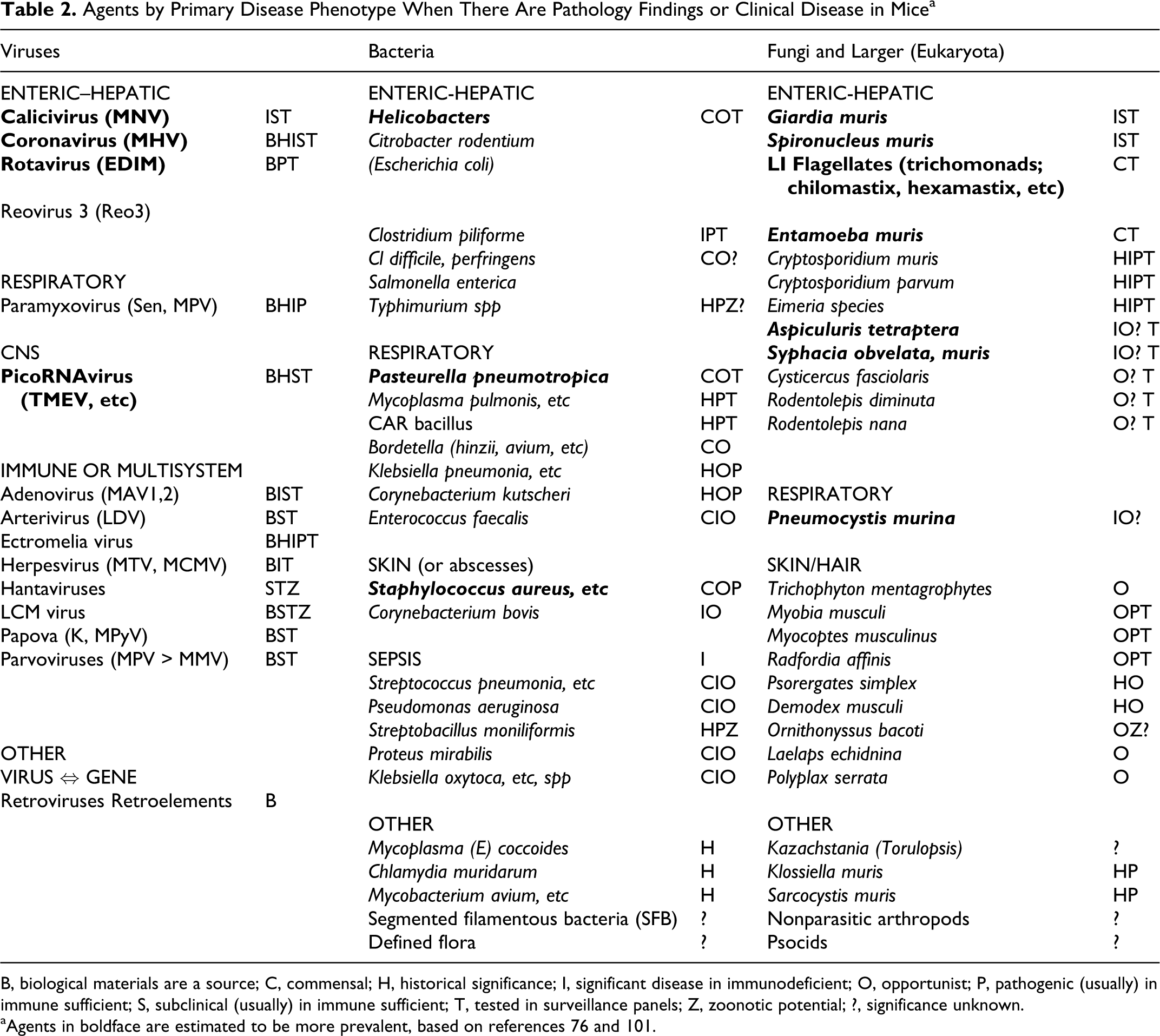
B, biological materials are a source; C, commensal; H, historical significance; I, significant disease in immunodeficient; O, opportunist; P, pathogenic (usually) in immune sufficient; S, subclinical (usually) in immune sufficient; T, tested in surveillance panels; Z, zoonotic potential; ?, significance unknown.
aAgents in boldface are estimated to be more prevalent, based on references 76 and 101.
General Concepts and Definitions
Mice and other macroorganisms have broad and uncharacterized flora. Although mice can be maintained germ-free or with defined flora, most facilities will house mice with undefined flora. Many institutions exclude certain agents from their colonies and house mice under specific pathogen free (SPF) conditions usually with some sort of barrier housing. SPF mice are maintained free of excluded agents by rigorous testing of sentinel mice as part of the institution health monitoring programs (surveillance, discussed below). 23,21,69 Each institution, and occasionally individual mouse rooms within an institution, will have a unique list of excluded agents. Most modern institutions exclude the historically important and usually pathogenic agents, for example, mouse hepatitis virus (MHV), Sendai virus, Citrobacter rodentium, Clostridium piliforme, pinworms, and mites, among others. Selection of microbes to exclude is usually based on criteria such as zoonotic potential (ie, lymphocytic choriomeningitis virus, Hantavirus, Salmonella spp), pathogens that are fatal to most mice (ie, MHV, ectromelia virus), pathogens that are not fatal but cause overt disease (ie, Pasteurella spp), and opportunists (Pneumocystis murina, Staphylococcus spp). 120 Infectious agents can damage cells, tissues, or organs directly by killing cells and indirectly by immune responses that damage cells or impair normal function (eg, inflammatory cell infiltrates in lungs that impede gas exchange or infiltrates or exudates that obstruct airways). Less obvious, but equally important, is the impact of infectious agents on physiological parameters including immune responses, cancer development, 104 and the generation of context-specific phenotypes (ie, disappearing phenotypes, see below). The widespread sharing of GEM between laboratories and institutions increases the potential for alterations in the mouse microbiome as well as microbial contamination. When mice become unintentionally contaminated with an agent, many parameters may be altered, often in a nonuniform fashion. Even if 100% of the mice are eventually infected, it will have been at different ages, with different infecting doses and slight individual variations in host response, even in genetically uniform mice, resulting in the stochastic effects of an uncontrolled variable. There are reports of “disappearing phenotypes” once mice are rederived into more stringent SPF facilities. 4 For example, Smad3-deficient mice maintained free of the gram-negative enterohepatic Helicobacter spp for up to 9 months did not develop colon cancer as initially reported. Subsequent infection of Smad3-deficient mice with Helicobacter restored the colon cancer phenotype. 75 The SMAD-deficient Helicobacter model is now used to explore inflammation-driven colon cancer, highlighting the utility of the interactions between microflora and GEM as a research tool.
Sources of Infection
Sources of infection may be other mice, fomites (eg, bedding and experimental equipment), human caretakers, feed, and water. Biological materials can serve as a source of infection. Cell lines, antibodies, conditioned media, and serum have all been implicated as potential source of contamination (discussed in detail in the section “Biological Materials”). Autoclaving of cages, food, and water or irradiation of diets is performed to reduce the chance of contamination through husbandry and environmental sources, and these as well as barrier facilities are not infallible. Despite stringent barrier practices and protocols, clinical disease outbreaks and changes in the microflora that alter phenotypes will happen.
Contemporary mouse colony surveillance programs test for a diversity of microbial agents. Many of the tested agents are not common or likely in today’s relatively clean research mouse colonies, but they can still be found in pet rodents, wild rodents, or biological materials and may be zoonotic. Surveillance programs aim to detect these before they compromise a colony or project. Detailed discussion of all of the agents tested (>30 in some comprehensive screens) is beyond the scope of this review, but references and resources for 30+ viral, bacterial, protozoan, and metazoan agents are summarized here in table format.
Surveillance programs vary in their level of scrutiny, that is, how many cages or animals are surveyed by a sentinel animal, how sentinels are exposed, how many tests are done, and how often. Academic mouse-based research occurs in diverse facility and institutional environments. Replication of results depends on details of housing and husbandry conditions in a particular research setting, information that is unfortunately lacking from many reports. This oversight is being addressed with the proposed Animals in Research: Reporting In Vivo Experiments (ARRIVE) guidelines, 59 which require more stringent reporting of animal care conditions. In pharmaceutical settings, environmental conditions tend to be more tightly controlled in accordance with Good Laboratory Practice requirements and anticipated scrutiny from the US Food and Drug Administration.
The production of novel GEM is expanding exponentially with input from both enormous international initiatives (such as the Knock-out Mouse Project [KOMP] and others) and individual laboratories. As such, there is frequent sharing of these novel mice (and their microbiomes) across the globe. Importation of rodents into facilities is usually overseen by the rodent health monitoring or attending veterinarian as part of the institutional surveillance program. 69 Steps to prevent outbreaks of excluded agents usually include review of the home institution’s health reports, quarantine, and possible prophylactic treatments (eg, medicated chow) upon receiving the mice. Quarantine of incoming rodents is a keystone of surveillance programs as an attempt to reduce contamination via newly introduced mice. 105 With the global nature of research and the interinstitutional sharing of mice, contamination of transferred rodents during shipment is a reality. Exposure of transport boxes to wild rodents should be expected, and thorough decontamination and housing procedures are required to prevent contamination.
Prevalent and Emerging Infectious Agents and Phenotypes in GEM
Detailed descriptions of infectious diseases in mice have been discussed at length in books and review papers, 2,37,41,78,98 including clinical signs and impact on research. 52 The agents we cover in detail were selected as those that in our collective experiences are increasingly diagnosed in GEM models. We will focus on specific examples of viruses (eg, mouse norovirus [MNV], mouse parvovirus [MPV], MHV, bacteria (eg, Helicobacter spp; Pasteurella pneumotropica; staphylococcal, enterococcal, and streptococcal species, Corynebacterium bovis; and Sphingomonas paucimobilis), and internal and external parasites and fungi (Pneumocystis murina and gastric yeast) for which the effects may be extrapolated to variety of other specific agents.
Viral Agents
Viral agents can cause overt clinical diseases and even death. However, subclinical infection is the norm and affects research primarily through alterations in the immune response. By far the most prevalent viruses identified in routine serological tests are the mouse MNVs (calicivirus), with 33% of externally sourced samples testing seropositive at a large rodent diagnostic laboratory, 101 although higher incidences may be encountered in some local populations. 14 Antibodies to MHV and MPV are detected in fewer serum samples, approximately 1%–2% each, 14,101 but these viruses are present in a high percentage of academic institutions and can have significant effects on research. 14,52
Mouse norovirus
Although there are many serotypes of MNV, by far the most studied is MNV1. 48 Most mouse vendors are negative for MNV; thus, the virus is surviving and perpetuating within research institutions. There is evidence that MNVs may undergo homologous recombination events, creating viruses different from the parental strain. 80 Serologic cross-reaction occurs for most of the strains, but MNV-1 may be distinct. Infection with one strain may not provide cross-protection against other strains. 89 Although most serotypes cause persistent infection, MNV-1 and perhaps others may be cleared within several weeks. In typical mouse strains without genetic modification, MNV-associated lesions have not been identified. 121 The method of transmission is thought to be fecal–oral. As a nonenveloped virus, MNV has the potential to remain active in the environment. However, MNV is readily disinfected by chlorine or ozone in drinking water and, thus, is susceptible to inactivation by oxidizing disinfectants. 28,66
MNV has not been associated with clinical disease in immune-competent mice. However, oral inoculation with a culture-adapted strain of MNV-1 (10 7 plaque-forming units) into 129S6/SvEv inbred mice results in very slight and transient granulocytic infiltration of the lamina propria of the duodenum at 24 hours post infection (without apoptosis of enterocytes) as well as mild increases in extramedullary hematopoiesis and lymphoid hyperplasia in the spleen. 90,121 MNV1 has tropism for macrophage and dendritic cells, which are essential mediators of the innate immune system. 123 In dendritic cells, the cytoplasmic helicase protein MDA5 (melanoma-differentiation-associated gene 5) has been demonstrated to be important in cell sensing of the virus. 83 Resistance to infection with MNV1 has been found to rely on the transcription factor STAT-1 and interferon (IFN)-αβ receptors for resistance to infection. Thus, Stat1 and Ifra1 (IFNαβR) mutant mice will develop clinical disease and death after infection with MNV-1, characterized by severe pneumonia, vasculitis, meningoencephalitis, hepatitis/necrosis, peritonitis, and pleuritis. 57,121
Although the innate immune system is essential to protect against lethal infection by MNV, the acquired immune system is important in clearing MNV. MNV does not cause clinical disease in mice with deficits in the acquired immune system, such as Rag1- and Rag2-deficient mice; however, they fail to clear the virus, which results in prolonged infection 121 (thus serving as virus reservoirs) that may affect immunology studies. In addition, coinfection of MNV with Helicobacter spp may accelerate the progression of Helicobacter-associated IBD in the Mdr1a-null mouse, despite the lack of clinical disease with MNV alone. 65 MNV has been shown to increase atherosclerotic lesion size and macrophage number in a C57BL/6 model of diet-induced obesity. 97 In addition, it has been demonstrated that BALB/c and C57BL/6 mice coinfected with MPV and MNV have prolonged shedding of MPV. 25 In contrast to these studies in which a consequence of MNV infection was reported, no effect of MNV infection was found in immunological response to vaccinia or influenza A virus, 49 in immune response to Friend retrovirus infection, 1 or in body weight gain, adiposity, triglycerides, or glucose metabolism in a study of diet-induced obesity in C57BL6/J mice. 96 The impacts of MNV on biomedical research are just beginning to be studied, and publications demonstrating effects on certain disease models and phenotypes will surely expand, suggesting that MNV will be added the excluded agent lists of numerous academic institutions. It should be noted, however, that these few publications on MNV already outnumber those for MPV, indicating not only that more study of MPV is needed but that the argument for exclusion of MNV from research facilities is at least as well-supported as for exclusion of MPV.
Mouse hepatitis virus
MHV refers to myriad strains of an enveloped ss-RNA coronavirus with significant variation in cellular tropism, pathogenicity, and even cell receptor affinities. MHV remains one of the most common viruses of mice in contemporary research facilities. 14,70,101 Common features shared among MHV strains include susceptibility to desiccation and detergent-inactivation (characteristics conferred by the lipid envelope) and a high degree of species specificity. All strains induce antibodies in immunocompetent mice that are sufficiently cross-reactive as to facilitate serological screening of mouse colonies although insufficient to prevent reinfection of mice with different MHV strains. 5 Broadly speaking, MHV strains can be further classified into those strains in which infection of immunocompetent mice is limited to the intestinal tract (ie, enterotropic strains) and those strains that initially infect the respiratory tract but may then disseminate widely (ie, respiratory or polytropic strains), although strains with intermediate tropism also exist. 50 Respiratory strains use CEACAM1 as the cell receptor. Mice such as the SJL strain with mutant CEACAM1 on their cell surface are highly resistant to respiratory strains of MHV, although they are fully susceptible to infection with enterotropic MHV. 50 In addition to using different receptors, respiratory strains also differ from enterotropic strains in being more pathogenic and being shed in much smaller quantities. 5 Perhaps because of decreased shedding, respiratory MHV appears to be much less prevalent than enterotropic MHV. 50 However, cell-culture–adapted laboratory strains of respiratory MHV such as JHM and A59 account for almost all of the MHV literature; relatively few studies have examined enterotropic field strains of MHV. Because respiratory MHV differs so much from enterotropic MHV, caution should be exercised in using the MHV literature to evaluate the potential for a given MHV outbreak to confound research results.
As respiratory MHV is less common, readers are referred to an excellent review 5 for a more complete discussion of that disease. In general, however, respiratory MHV causes foci of necrosis associated with syncytia and sometimes a mononuclear cell inflammatory response in many different tissues, including nasal epithelium, lung, liver (Figs. 1, 2), lymphoid organs, and sometimes the central nervous system. 98
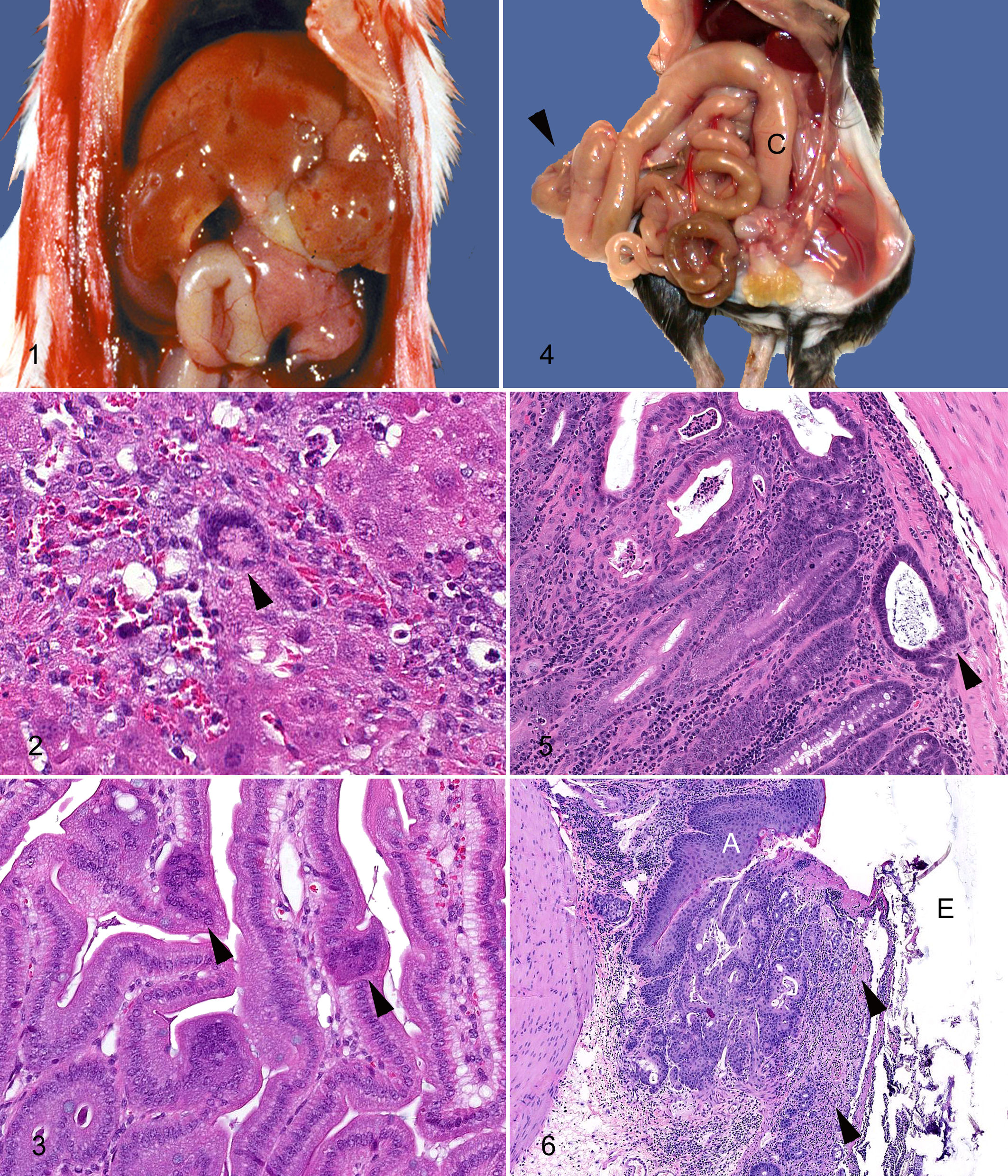
Enterotropic MHV infection rarely causes clinical disease in postweanling immunocompetent mice, but it is an important differential diagnosis for wasting disease in immunocompromised mice. 22,48 However, even in immunocompromised mice it may not always disseminate outside of the intestinal tract. 23,50 In suckling mice not protected by maternal antibodies, enterotropic MHV may cause diarrhea and mortality, thus the old acronym LIVIM (lethal intestinal virus of infant mice). The morbidity and mortality in infant mice probably result from slower cell turnover rates of the intestinal epithelium, with less ability than older mice to replace damaged cells. Grossly, infant mice may be dehydrated and the intestines filled with yellowish liquid and gas, changes similar to those described for group A rotavirus infection of infant mice. 98 Microscopically, syncytia of apical epithelial cells are likely to be the only finding (Fig. 3 ), although syncytia may also be observed in mesenteric lymph nodes. 5
Enterotropic MHV is much more contagious than respiratory MHV, being shed for longer periods of time and in greater amounts. Transgenic mice with immunological deficits may shed MHV for extended periods of up to 2 years, in the case of mice with T-cell deficiencies. 108 A controlled study with enterotropic MHV-Y found that BALB/c mice shed infectious amounts of virus for longer (4 weeks) than C57BL/6 mice (2 weeks). B-cell–deficient mice shed virus for more than 3 months but had no histological lesions. 23,24 In contrast, T-cell–deficient mice developed severe wasting disease after 2 weeks, requiring euthanasia at 1 month; MHV was still being shed in the feces at that time. 24
Enterotropic MHV can be a copathogen with Helicobacter hepaticus. 22 One study reported that γIFN-deficient mice with dual infections had less severe MHV-induced lesions during the first week (ie, acute phase) of infection but greater mortality and more severe lesions during a more chronic phase at 28 days post infection. 22 Such studies of disease and transmission of infection in mice of different strains, different immune status, and different microbial health status emphasize that the typically subclinical enterotropic strains of MHV carry the potential to alter research outcomes.
Parvoviruses
Parvoviruses are small-nonenveloped, ss-DNA viruses that remain among the most common exogenous viruses in laboratory mice. 14,70,101 Currently known parvoviruses of mice are divided into 2 groups, minute virus of mice (MVM) and MPV, the latter of which is subdivided into further serotypes, currently MPV1 through MPV5. 27 Both MVM and MPV share a high degree of resistance to desiccation, heating, and solvents. In addition, natural infection of immunocompetent mice with any of the parvoviruses is clinically silent, as is natural infection of most immunodeficient mice. 55 Disease was reported, however, in a line of mice with a B-lymphocyte maturational defect, NOD.h-2H4 homozygous for a targeted null mutation of immunoglobulin heavy chain, infected with a field strain of MVM, MVMm. In these mice, slow growth, deaths, and reduced fecundity were observed. Histologically, there was lymphohistiocytic inflammation in the kidneys and viral inclusions in splenic red pulp cells resembling hematopoietic progenitor cells. 8,94 Experimentally, disease can be produced by intracerebral inoculation of suckling mice with the more commonly studied immunosuppressive (MVMi) or prototype (MVMp) culture-adapted strains. 54
All parvoviruses of mice share several characteristics that make even subclinical, relatively nonpathogenic infections worrisome. First, like parvoviruses of other species, murine parvoviruses can only replicate in actively dividing cells. Because productive infection is cytolytic, there is concern as to interference of murine parvoviruses with oncology research or research in other areas in which active cell replication is an important feature, and another rodent parvovirus, H-1 parvovirus of rats, has been investigated as a potential anticancer agent. 87 Murine parvoviruses cause further concern because they are lymphocytotropic. Concern regarding long-term influence on the immune system is amplified because both MVM and MPV are shed in the feces for long periods of time by immunodeficient or infant mice, and viral DNA can be found indefinitely in mesenteric lymph nodes and spleen. However, both MVM and MPV are very difficult to grow in culture, so most work has been done with culture-adapted strains that may behave differently than field strains. Two studies in the 1990s found altered immune function in mice experimentally infected with MPV1a, a culture-adapted strain discovered because of its effects on T-lymphocyte cultures. 85,84 In these studies, BALB/c mice were inoculated with 10 6 TCID intraperitoneally and 10 5 TCID oronasally. Infected mice had accelerated rejection of tumor allografts but decreased killing by T cells. 84 Infected mice also had decreased lymphoproliferative responses in spleen and popliteal lymph node but increased proliferative responses in mesenteric lymph nodes. In a related study of BALB/c mice experimentally infected with MPV1a, infected mice had increased rejection of both allogeneic and syngeneic skin grafts, but the allogeneic-reactive T cells had decreased proliferative capacity. 85 No subsequent reports have corroborated these findings with any culture adapted or field strain of MPV. At this time, no long-term immunological effects have been shown for natural infection of mice with any field strain of MVM. No alteration of tumor development or growth has been shown for any field strain of MVM or MPV. In the only study to document a consequence of infection with a field strain of MPV, the authors conducted gel electrophoresis on serum from BALB/c mice (n = 3/time point) orally inoculated with 100 ID50 MPV1e or 1000 ID50 MPV5 from naturally infected mice. On day 7, the MPV1e infected mice had slightly decreased serum albumin, and on day 14 mice infected with either MPV1e or MPV5 had increased serum γ-globulin. On day 28, serum α1, α2, and β globulins were increased, and albumin–globulin ratios were decreased in the MPV5 but not the MPV1e group. These changes were generally mild, and no significant changes in specific acute phase proteins were detected by ELISA. 27
Because of the sparse evidence of research interference by MPV and the lack of overt disease or reproductive effects, the overall significance of MPV infection is unclear. Further complicating laboratory animal management decisions when MPV is detected in a facility is that even within MPV-positive vivaria, as few as 1% of cages may be infected if mice are housed in individually ventilated caging. 73,72
Approaches to control of MVM or MPV within mouse research facilities have ranged from euthanasia of all mice with cleaning of the entire facility followed by disinfection with an oxidizing agent 55 to more measured campaigns of testing and culling of infected cages or groups of cages while imposing careful biocontainment of suspect areas. 73
Bacterial Agents
Helicobacter spp, P. pneumotropica, and Staphylococcus aureus are the most prevalent bacterial agents isolated in laboratory mice. 14,101,116 Our personal experiences reflect this, and in addition we have noted increases in isolation of Staphylococcus xylosus, Enterococcus spp, and opportunistic agents such as S. paucimobilis (usually found in soils and water 51 ) with the explosive use of novel and usually immunodeficient (or immunovague) GEM at our collective institutions.
Helicobacter spp
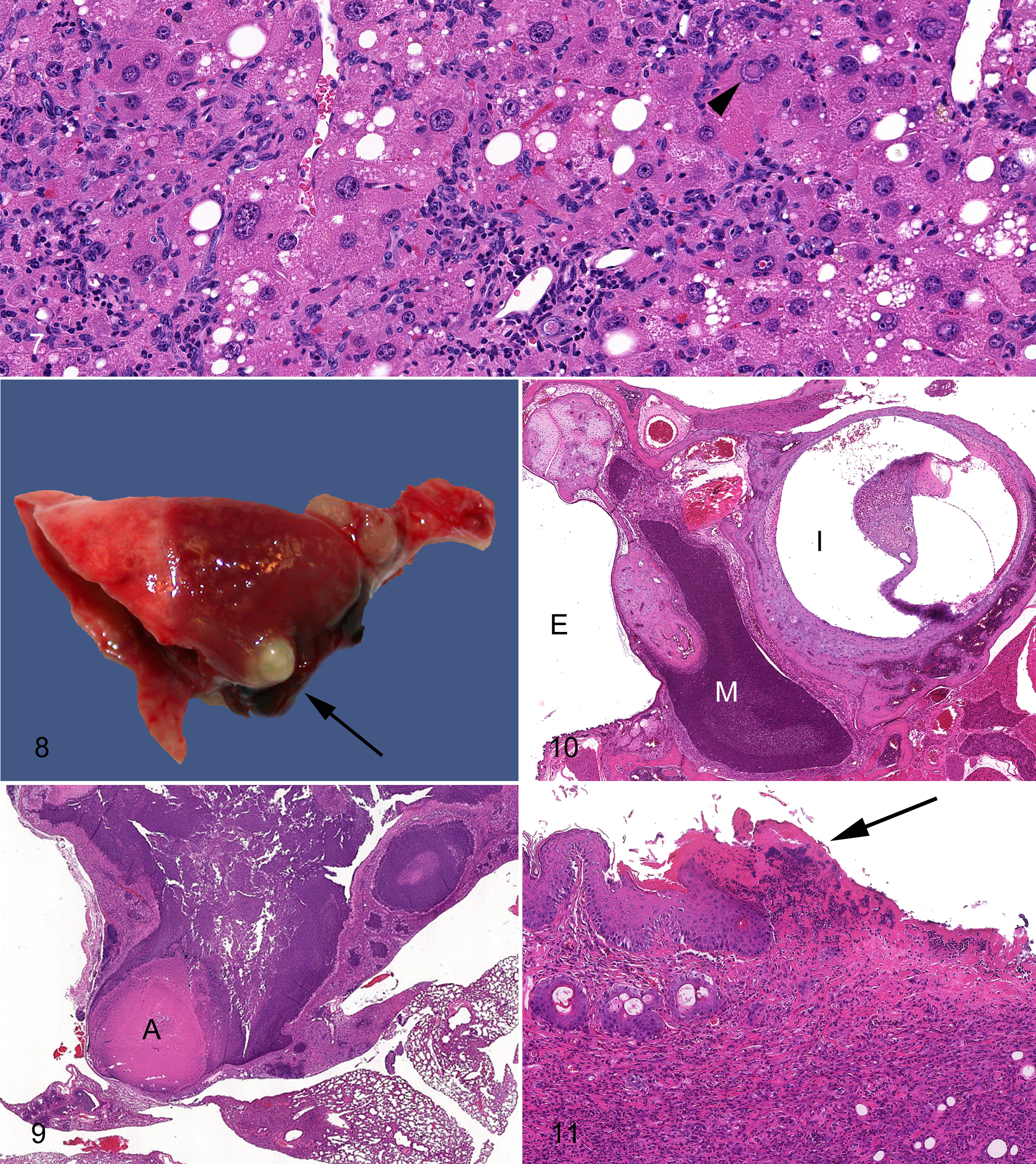
Disruptions in either the innate or the acquired immune system may result in Helicobacter-associated disease. Nine species of Helicobacter have been formally identified in mice, of which H. hepaticus, infecting the liver, biliary tree, colon and cecum, and H. bilis, infecting the liver, cecum, colon, and biliary tree, have gained the most attention. 39 In susceptible mice (see below and references 39 and 108) the common enterohepatic Helicobacter clinical signs include weight loss, diarrhea (manifesting commonly as sticky stools), and rectal prolapse. Grossly, opaque and thickened large bowel (Fig. 4 ) is present in mice with proliferative typhlocolitis. Gross liver lesions are variable depending on disease chronicity and host genotype. Similarly, the histological manifestations of typhlocolitis (Figs. 5, 6) and hepatitis (Fig. 7 ) depend on the infecting Helicobacter species and mouse genotype.
Helicobacter spp have been identified worldwide in both academic and commercial mice and are not excluded from most mouse barrier facilities. Helicobacter spp have been identified as a cause for typhlocolitis (In some cases termed inflammatory bowel disease—IBD) and hepatitis as well as carcinomas in the intestine, liver, and other sites. 33,38,39,92,103,111,116,122 Certain strains of immunomodified mice are most susceptible as well as spontaneously immunodeficient mice such as Rag1/Rag2 and Pkrdcscid mice. Helicobacter spp elaborate toxins, such as a cytolethal distending toxin (CDT). 43 The Helicobacter CDT has been demonstrated to induce dsDNA breakage and induce apoptosis, which may be important in mediating its carcinogenic effects. 43,71
The immune response to Helicobacter spp is initiated by the innate immune system, for which TLRs, NF-κβ, and MyD88 signaling appears to play an important role. 35,79 Th1 and Th17 responses have been identified as important in the development of colitis. Th17 cells are essential in promoting inflammation and host defenses against microbial pathogens, whereas suppression of inflammation is mediated by CD24+CD25+ regulatory T-cells. 117 Th17 cells are activated by interleukin (IL)-23 and produce numerous cytokines, including IL-17, IL-17f, IL-6, IFNγ, and tumor necrosis factor alpha (TNFα). 9,46 Studies have suggested that IL-23, IL-17, and IFNγ may play an important role in the development of Helicobacter spp colitis. 61
Many genetically engineered immune modified mice are susceptible to Helicobacter-induced typhlocolitis (comprehensively reviewed in reference 39; also see tables). Critically, a background strain of mice may have a significant impact on the interaction of Helicobacter with the targeted mutation; for example, Il-10–deficient mice had more severe disease when on a 129 background compared to severity on a C57BL/6 background. 6
P. pneumotropica
P. pneumotropica typically considered an opportunistic pathogen and is highly prevalent in mouse research colonies and wild mice. Transmission occurs through direct contact, likely fomites and in utero. 98 Lesions in susceptible mice include pulmonary (Fig. 8 ) and other soft tissue abscesses, pneumonia (Fig. 9, abscessation of retrobulbar tissues, rhinitis, sinusitis, conjunctivitis, blepharitis, and otitis (Fig. 10) and should be suspected in any purulent processes in susceptible mice. Outbreaks of P. pneumotropica have been reported in numerous GEM and spontaneous immunodeficient mice (reviewed in reference 41).
Staphylococcus spp
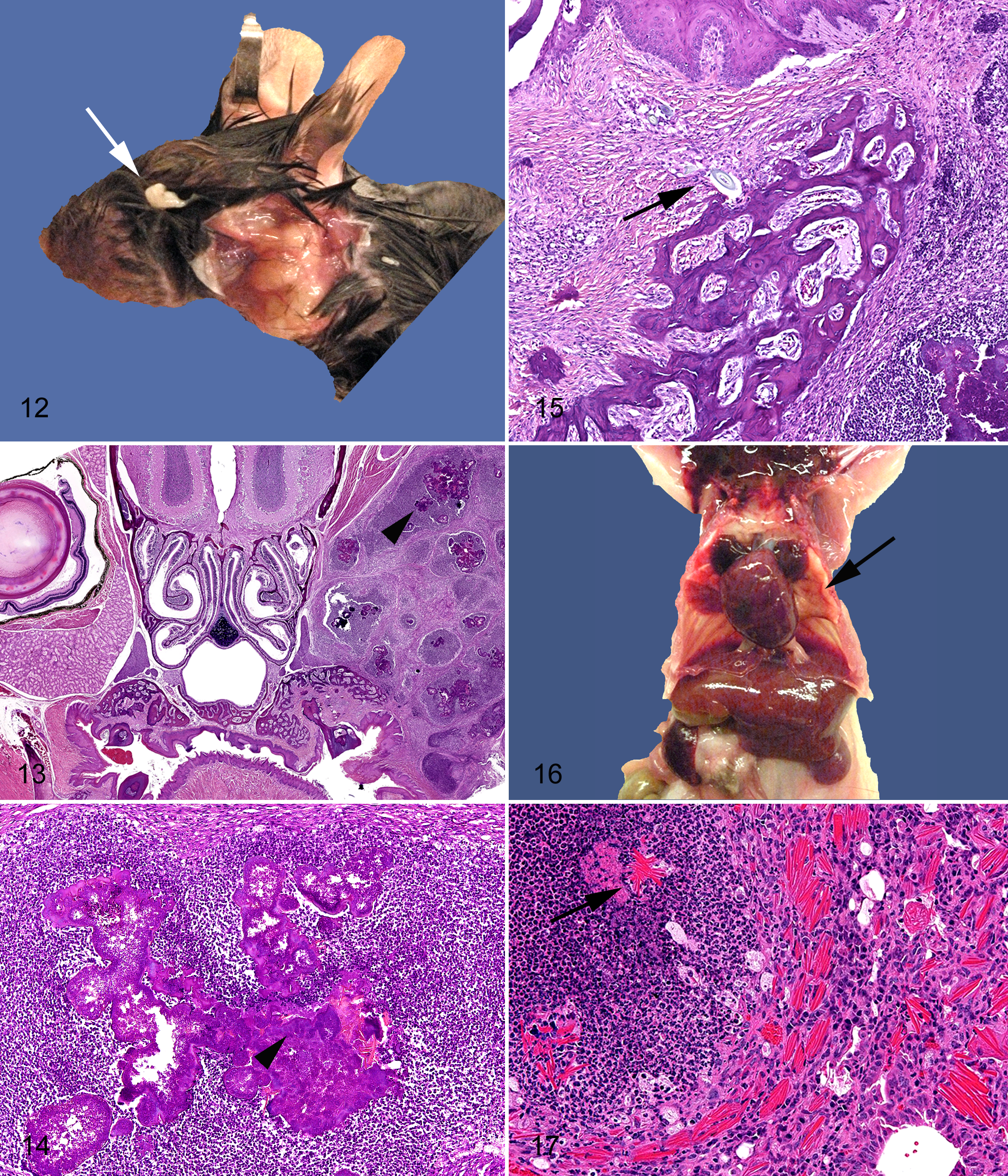
Although staphylococci are present on the skin and mucous membranes of most animals, most do not develop disease. Humans are a natural reservoir of S. aureus, and up to 20% of people may be persistently colonized. 53 S. xylosus may be found as an environmental contaminant, and thus elimination of staphylococci from mouse colonies is exceedingly difficult. S. aureus and S. xylosus are gram-positive bacteria that have been associated with spontaneous disease in immune-modified mice. Superficial suppurative lesions including dermatitis (Fig. 11 ), blepharitis (Fig. 12 ), conjunctivitis, otitis, sinusitis, and pneumonia 45 (unpublished observation, P.M.T.) and a necrotizing dermatitis (Fig. 11) are frequently noted. 98 Abscesses caused by S. aureus are sometimes termed botryomycotic granulomas, because of aggregates of bacteria that appear similar to Actinomyces spp sulfur granules, and often have deposition of Splendore-Hoeppli material at the center of these highly suppurative abscesses (Figs. 13, 14). 56 Most S. aureus lesions are thought to be opportunistic and secondary to trauma. For example, penetration of hair fragments into the periodontal tissues occurs frequently in mice, especially those prone to excessive grooming that introduces Staphylococcus and other oropharyngeal flora deep in the soft tissues (Fig. 15 ). Superficial excoriation of the skin, for example, through cage mate aggression or trichotillomania (excessive grooming) seen in some strains (eg, C57BL/6) or disruption of the corneal epithelial barrier, 60 can be a predisposing factors in colonized mice. However, in immunodeficient mice (Figs. 16,17), such as those with defects in NADPH oxidase and severe combined immune deficiency (SCID), severe pneumonia and spontaneous abscessation, particularly of the face, have been reported 45 (unpublished data, 2009-2011, P.M.T., R.S.S.) NOD/LtSz-scid Il2γ–null mice can develop facial abscesses as early as 7–9 weeks of age (unpublished data, 2009-2011, R.S.). Although S. aureus abscesses are apparently common in SCID mice in the research setting, reports of this disease are limited. 18,56,98
Staphylococci have a number of ways to avoid immune defenses. Some of the organisms may be resistant to eradication in neutrophils through charge neutralization of their anionic cell surfaces. 20 Expressions of a number of virulence factors as well as the host response to the organism play a role in the disease manifestation. In particular, the characteristic necrotizing dermatitis (Fig. 11) with superficial erosions to ulcerations and deep coagulative necrosis (burn like lesions) is due to the production of numerous proteins including exotoxins, hemolysins, proteases, collagenases, exfoliative toxins, and superantigens (enterotoxin A-C and toxic shock syndrome toxin 1) among others. 98
C. bovis
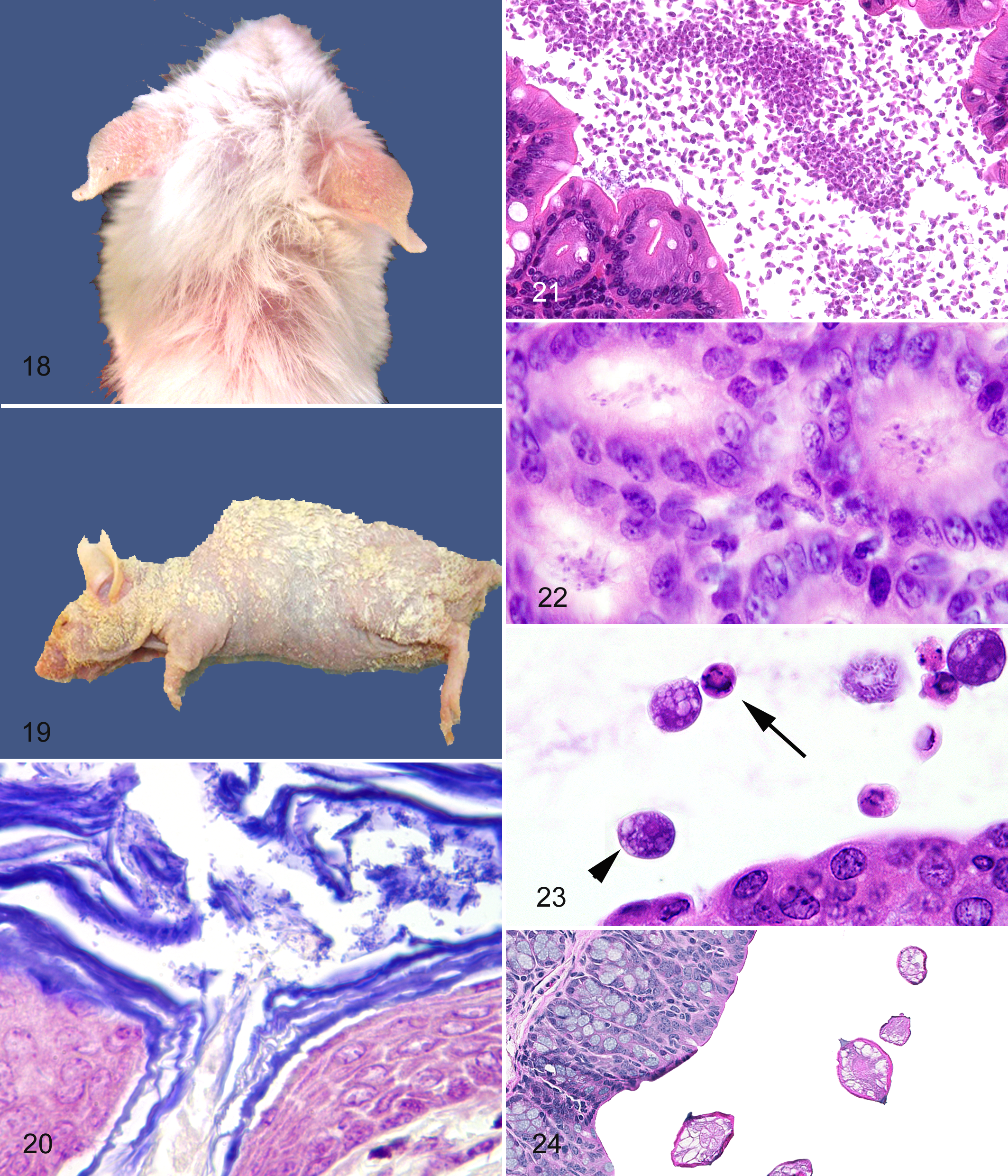
C. bovis is a gram-positive coryneform that has been associated with skin disease in mice with T-cell (Foxn1–/–, athymic nude) and T- and B-cell (Pkrdcscid ) immune deficits as well as in hairless (SKH1-Hrhr) mice with essentially normal immune function. 10,19,110 Clinical disease occurs with variable morbidity and low mortality approximately 7–10 days after infection. Gross lesions are hyperkeratosis and dehydration (shriveled appearance), which persist for approximately 7 days (Figs. 18, 19). Affected Pkrdcscid mice may also appear to have a slightly puffy appearance to the facial skin. Histological changes include orthokeratotic hyperkeratosis and widespread or diffuse acanthosis that persists after hyperkeratosis is no longer grossly visible. 19,110 A mild dermal mononuclear cell infiltration often accompanies the epidermal proliferation. Gram-positive coryneforms are usually readily detected in the stratum corneum and within the necks of hair follicles (Fig. 20). Infection persists indefinitely, perhaps lifelong, in the immunodeficient mice mentioned above. Immunocompetent hirsute mice, such as ICR stocks, have been reported to be transiently infected but perhaps eliminate the infection with time. Increased mortality among newborn athymic nude mice or in mice after treatment with chemotherapeutic agents has been reported. 19 Control is difficult as the organism may be widespread within contaminated facilities, including being detectable on cage surfaces, within hoods, and even on doorknobs and gloves and in tumor lines. 10,19,110
The mechanism by which C. bovis induces the epidermal proliferation is unknown, as is the mechanism by which the clinical signs are eventually diminished. However, whatever the mechanism, whether by defensins or other innate immune elements, mice deficient in those host defense components are likely to have increased disease.
Streptococcus and Enterococcus spp
Similar to the organisms described above, these organisms may infect and cause disease in GEM. More often seen in immunodeficient mice such as the Myd88 mutant, the organisms are increasingly cultured from mice with clinical disease (eg, abscesses, meningitis, pyelonephritis, disseminated infections, and bacteremia) (unpublished data, 2008, P.M.T.). 98 Historically, the pathogenic species use various proteins similar to Staphylococcus spp to impart virulence; however, in the immunodeficient mice the nonpathogenic α-hemolytic Streptococci are increasingly implicated. Typically, in the Myd88 mutant mice there is little to no host inflammatory response to a myriad of bacterial colonies that in severe cases efface tissue architecture (Supplementary Figs. S1–S3).
Parasites
External parasites such as mites (fur mites: Myobia musculi, Myocoptes musculinus; follicular mite: Demodex musculi; free living rat mite: Ornithonyssus bacoti) and lice (Polyplax serrata, the vector of Mycoplasma cocoides) are excluded from modern institutions. Outbreaks of fur mites still commonly occur, and some incoming rodent quarantines may automatically treat all mice for external and internal parasites as a result. 88,107 The principle impact mites have on GEM-based research is transfer of mice between institutions, causing pruritus, self-trauma, and secondary bacterial infection (see S. aureus section above), and may be a predisposing factor for the idiopathic dermatitis of C57BL/6 substrains. 2,98 Infection is via direct transfer and diagnosis is made by pelage examination, tape testing, and polymerase chain reaction (PCR).
Internal parasites (Figs. 21–24) include protozoa (Spironucleus muris, Tritrichomonas muris, Trichomonas minuta and wenyoni, Octomitus pulcher, Chilomastix bettencourt, Entamoeba muris, Cryptosporidium muris and parvum, Giardia muris, Klossiella muris, and several species of Eimeria among others), microsporidia (Encephalitozoon cuniculi, extremely rare), and helminths (Oxyurids: Syphacia obvelata, Aspiculuris tetraptera; adult and larval cestodes). 98 Helminths are excluded in facilities; however, as with mites, outbreaks of pinworms (oxyuriasis) are frequent enough to warrant automatic treatment of incoming rodents in many institutions because of the persistence of the eggs in the environment and the ease of transmission. Pinworms are relatively innocuous in most mice, with few reports of altered susceptibilities of GEM to parasite burden or infection with S. muris, the rat pinworm 41 ; however, they are a top differential diagnosis for rectal prolapse. Infection with helminths has experimentally been shown to alter immunophenotype, 64,47,112 and undetected natural infections could affect immunological studies. 52 Transmission is fecal–oral, and eggs are often aerosolized. 52 Diagnosis is via cecal or fecal examination, perineal tape tests, and PCR.
The intestinal protozoa, G. muris, Eimeria, and Cryptosporidium, are extremely rare in modern facilities. However, in our experience, blooms of intestinal flagellates are apparent in histological sections of gastrointestinal tracts of immunodeficient mice (Fig. 21 ). The impact that these blooms may have on the host is unclear, and most often there is no observable host response or tissue damage; rarely the protozoa appear to be adherent and possibly invading ulcerated intestinal mucosa, although this has yet to be fully characterized (P.M.T., unpublished data, 2009). S. muris may have clinical manifestations in young and immunosuppressed mice. 98,99 These various protozoa are definitively diagnosed by fecal wet mounts 36 ; on tissue section they are challenging to distinguish with the exception of relative size and locations within the gastrointestinal tract (Figs. 21–24), although they are readily observable. 99 The relatively large trichomonads usually are found in the colon and cecum (Fig. 23 ), whereas the small spironucleus trophozoites are usually found the small intestinal lumen or crypts of Lieberkuhn (Fig. 22 ).
P. murina is an opportunistic fungus transmitted by inhalation of infectious cysts that is adapted to live in the lungs of mice, even normal, immune-competent hosts. 16 Overt disease, however, usually manifests with immune suppression, either by genotype or drugs, and viral infections. 2 Clinical signs in immunosuppressed hosts include nonspecific sick rodent signs of weight loss and unkempt pelage and respiratory signs of dyspnea, less obvious cyanosis eventually leading to death. 98 The lungs at necropsy are characteristically pale and fail to completely collapse; they have a rubbery appearance (Fig. 25 ). Interstitial pneumonitis with the presence of eosinophilic, foamy to honeycombed material within the alveoli is rather diagnostic (Figs. 26a) but may be challenging to identify in the face of concurrent bacterial or acidophilic macrophage pneumonias that often accompany pneumocystosis. This material contains viable and dead organisms and surfactant. 17,98 Diagnosis in animal with clinical disease is possible using an impression smear at necropsy. The organisms can be visualized on a Wright’s stain, Romanowsky stain, or Grocott's methenamine silver stain (GMS) of the impression smear. Histological staining of the 3- to 5-μm cysts with methenamine silver (Fig. 26b) or periodic acid–Schiff aids the diagnosis. Subclinical infections and confirmation of histological findings can be made with PCR.
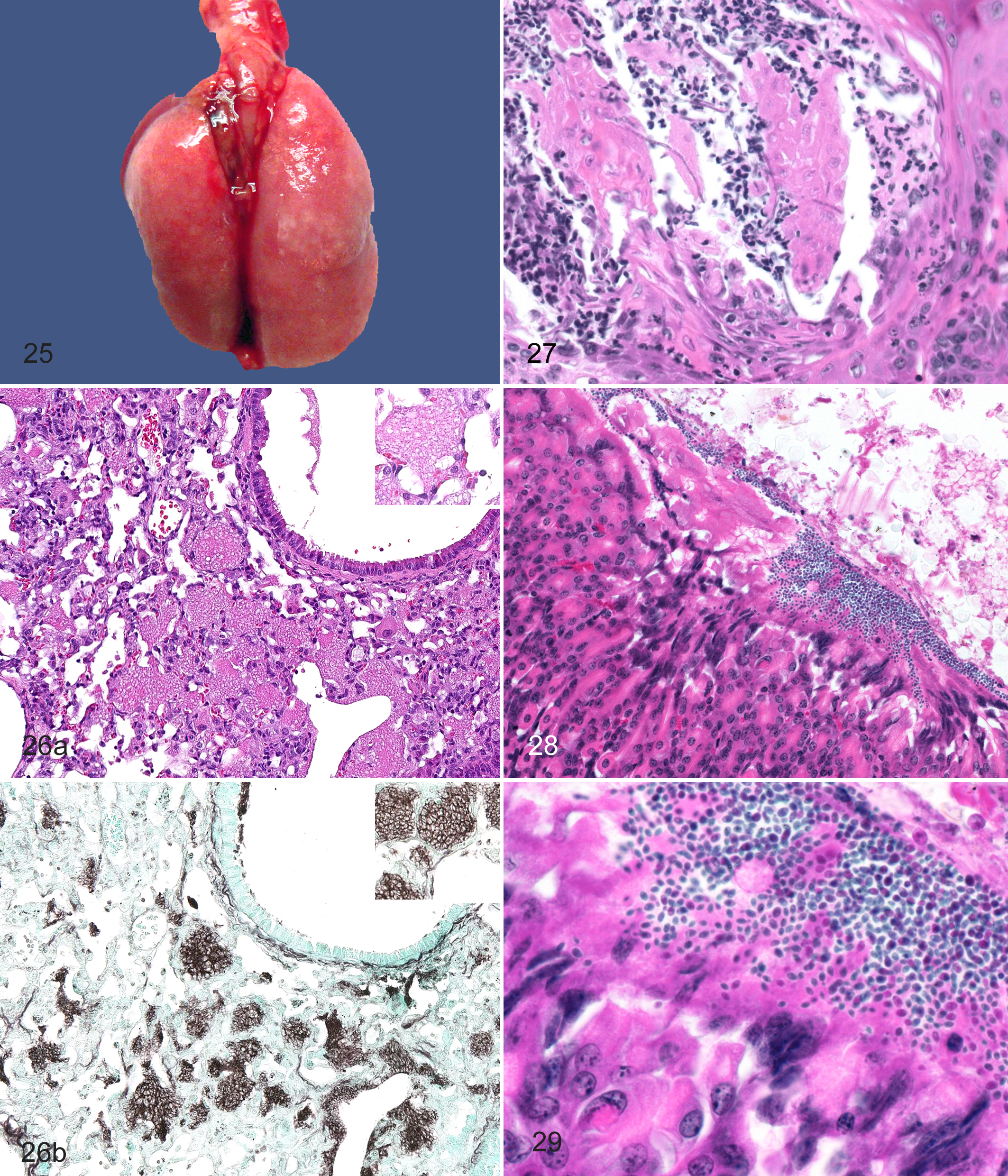
Infections with opportunistic fungi such as Paecilomyces spp and Aspergillus fumigatus, among others, are reported in mice with defects in NADPH oxidase developing pyogranulomata in multiple organs. 40,44 Infections of the upper digestive (esophageal or gastric) (Figs. 27–29) have been reported 98 and are infrequently diagnosed in our collective experience. However, bedding, feed, and possibly water may serve as sources of contamination 82 (see “Environmental Sources and Emerging Agents” below).
Biological Materials
In addition to the risks for disease introduction posed by incoming mice and husbandry materials, many research facilities import a wide range of cell culture lines or biologically derived reagents such as antibodies, conditioned media, and serum. These materials, loosely termed cell lines and biologicals, may be contaminated with viruses or Mycoplasma spp 93,100 and should be considered as additional sources of risk whenever mice are exposed to them or to materials that have come into contact with them. Such biological materials, whether cell lines or cell-free reagents, may be contaminated with agents indigenous to the species from which they are derived, such as human viruses in human cell lines or mouse viruses in mouse cell lines or serum. 67,68 They may also be contaminated by agents of other host species. For example, antibodies produced in rabbits may be contaminated with mouse viruses if the rabbit serum is purified on a column previously used for mouse serum. Similarly, human cell lines may be contaminated by mouse viruses if cultured on a feeder layer of mouse fibroblasts or if cultured with mouse serum or other biological material, for example, mouse-derived basement membrane matrix. Important considerations in assessing the level of risk posed by cell lines and biologicals may include whether they have been stored in a freezer from a time when adventitious infections were more prevalent 95 and whether the material has been previously tested. One should also recognize that at this time, the American Type Culture Collection (ATCC) does not routinely test cell cultures for rodent viruses.
Mycoplasma spp are the most commonly detected infectious agents in cell lines and biologicals. 15,34 Fortunately, it is rare to find any Mycoplasma capable of infecting mice and, specifically, Mycoplasma pulmonis has never been reported in a cell line. In fact, cell lines contaminated with nonmouse Mycoplasma spp may be decontaminated by passaging them through mice. 109 Recently, however, Mycoplasma arginini has been reported as a contaminant of cell lines and caused suppurative arthritis in Prkdcscid mice inoculated with the cells. 100,115
Infection risks for humans are appropriately a major concern. Testing of human cell lines for human-origin viruses at Charles River (unpublished data, Feb 2002 to April 2010) has detected hepatitis B, not surprisingly in hepatocarcinoma lines, as well as human papillomavirus-18. Zoonotic agents detected in rodent cell lines include lymphocytic choriomeningitis virus (LCMV), which has been detected in MaTu cells (known to carry LCMV) and in baby hamster kidney (BHK) cells.
Parvoviruses are among the most commonly detected endogenous rodent viruses in cell lines and biologicals, and they carry the potential to spread beyond the inoculated mice and contaminate an entire vivarium. Approximately 0.4% of cell lines and a similar percentage of other biologicals have been found to have parvoviruses, most often MPV and/or MVM. Note that any percentage prevalence cited in this article is not intended to imply a uniform risk across all cell lines or to establish an actual contamination rate for all cell lines and biologicals. However, finding a contamination in 1 out of every few hundred samples does establish that a risk of disease transmission exists and that if an institution imports large numbers of cell lines and other biologicals for use within a vivarium, the cumulative risk can be substantial. 63,67
The other common virus detected in cell lines and biologicals is lactate dehydrogenase elevating virus (LDV, LDHeV). 13 Of particular note with LDV is that this virus has not been routinely included in screening panels in the past. An enveloped arterivirus, LDV is primarily transmitted in the laboratory setting through infected tumors, cell lines, and other mouse-derived biological materials. Natural transmission usually occurs through bite wounds or sexual contact. This virus may also be transmitted transplacentally or via the milk. These latter 2 routes of infection are only clinically important if the infected animal is in the first week of infection, when viral shedding may occur via those routes. Animals remain persistently viremic after infection, but infection is unlikely to spread within most animal facilities. LDV infects macrophages and results in an increase in lactate dehydrogenase (LDH) within approximately 24 hours after infection. This increase in serum LDH persists and may be used as a screening tool for LDV infection, although LDH may also be elevated for many other reasons, including hemolysis. As viremia is persistent, PCR is often used on serum to confirm LDV infection in samples with elevated LDH. Recently, serology for LDV has been offered by rodent diagnostic laboratories. LDV infection may depress cellular immunity, increase cytokine activity, alter humoral immunity, reduce tumor growth, and alter immunity to copathogens, and cause hypergammaglobulinemia and autoantibody development. 3,98 In addition, some strains develop glomerulonephritis and/or polioencephalomyelitis. Polioencephalomyelitis, which may progress to paralysis and death, occurs in susceptible strains of mice, including AKR and C58, if immunosuppressed and coinfected with endogenous n-tropic mouse leukemia virus (MuLV). However, in a recent nationwide outbreak of LDV associated with a commercial biologically derived basement membrane matrix material, paralysis associated with LDV infection was also observed in Prkdcscid mice in which the MuLV status was not determined. 13
Occasional multicenter outbreaks of ectromelia, an orthopoxvirus, have been associated with use of commercial mouse serum in animal facilities. 63,68 Although an enveloped virus, ectromelia may spread widely via fomites or infected animals throughout a vivarium, and it is shed in feces, in respiratory secretions, and from the skin. Resistant strains, such as C57BL/6, C57BL/10, and AKR, may show no clinical signs but serve as a source of virus for the infection of other animals. In contrast, in susceptible strains such as A, CBA, C3H, BALB/c, and DBA/2, mortality may exceed 80%. 77 This mortality may occur with no other clinical signs, and animals often die before they shed virus. Susceptible mice have acute hepatocellular necrosis as well as necrosis of the spleen, Peyer’s patches, thymus, and lymph nodes. Hepatocellular necrosis may be seen as white spots on the liver. Clinical signs in strains of intermediate susceptibility may include ruffled fur, hunched posture, facial edema, swelling of the limbs, conjunctivitis, cutaneous pustules, ulceration of the muzzle, limbs, ears, and tail, and the lesion that gives the virus its name, ectromelia, or partial amputation of the limbs and tail.
Other viruses that have been detected in cell lines and biologicals include reovirus, polyomavirus (primarily in MethA and LLC cells), and cytomegalovirus. The presence of these uncommon viruses should serve as a reminder that although relatively few viruses appear to be circulating in contemporary populations of laboratory mice, cell lines and biologicals pose some risk for introducing other agents considered rare in modern facilities and for which testing is infrequently conducted.
Environmental Sources and Emerging Agents
Historically, watering systems have been associated with the ubiquitous Pseudomonas aeruginosa or Proteus mirabilis, and acidification or chlorination of systems is used to control but not eliminate growth. With the increased use of complex and severely immunodeficient mice, biofilm-related microflora within watering systems should be considered as a likely source of emerging agents for GEM. 7,26,86 With the concept that all GEM are of unknown immune status, pathologists must be alert for changes in phenotype or microflora in tissue sections that may indicate introduction of new environmental opportunistic pathogens. Recent examples from the University of Washington (UW) (P.M.T.) include a case of disseminated infection with S. paucimobilis, a gram-negative bacteria normally associated with water and soils and reported infrequently in human medical literature as a cause of nosocomial infections (abscesses, bacteremia) in immunocompromised patients. 30,42,51,58 We have cultured S. paucimobilis from case of severe necrosuppurative peritonitis in a C57BL/6Rag2tm1Cgn /J–background GEM (Figs. S4–S6). Bulk water cultures obtained from the automatic watering unit have detected S. paucimobilis, Brevundimonas vesicularis, Pseudomonas alcaligenes, Actinomycetes, Corynebacterium genitalium, Empedobacter brevis, and other unidentified bacteria and fungus. Additionally, gastric yeast morphologically similar to Kazachstania pintolopesii 62 was noted in stomach sections from numerous immunodeficient mice submitted to the UW diagnostic laboratory due to an investigator-observed increase in rectal prolapse within the colony (Figs. 28, 29). These particular mice had been housed in a Helicobacter spp–positive room with little notable clinical disease for a number of years. The gastric yeast has previously not been diagnosed in mice from the UW colonies. Although it is tempting to speculate that there are possible novel interactions between the host genotype and recently altered microflora in these anecdotal examples, the precise relationship between these potential pathogens requires further study to formally implicate these agents in altered phenotypes. 41 Fulfilling Koch’s postulates for newly recognized agents would be the next logical step in determining causation.
Conclusions
The complexity of the interactions between the microbiota (including pathogens) is increasingly recognized for its role in the development of immune competence and disease pathogenesis. 32,113,118,119 Although unrecognized infectious may result in confounding research outcomes, they may also result in new models of disease. Pathologists working with mouse models must be vigilant in unexpected changes in phenotypes that may relate to changes in pathogen status or microbiota and relate these findings to the mouse genotype as part of the process of mouse model validation. 12 The combination of mouse genetics, environmental factors (including infectious agents and “normal flora”—collectively the microbiome) must all be included in the evaluation of phenotype expression in GEM.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
