Abstract
The diagnosis of vascular neoplasms is often facilitated by the use of immunohistochemical markers such as factor VIII–related antigen, CD31, and CD34. However, the relative sensitivity and specificity of these markers have not been compared in cat vascular neoplasms. In this study, these 3 immunohistochemical markers were evaluated in 61 endothelial neoplasms (50 hemangiosarcomas and 11 hemangiomas) in 59 cats. All neoplasms were labeled by all 3 markers. CD34 had the highest average immunolabeling intensity in neoplastic endothelial cells. CD31 had the lowest average background labeling, followed by CD34 and factor VIII–related antigen, respectively. CD34 expression was also examined in 130 nonvascular neoplasms of cats; 14 of 62 epithelial neoplasms, 39 of 43 mesenchymal neoplasms, 8 of 23 leukocytic neoplasms, and 2 of 2 melanomas were positive. Given the broad expression of CD34 in mesenchymal neoplasms, this marker has limited diagnostic relevance for vascular neoplasms of cats.
Vascular neoplasms, including hemangioma and hemangiosarcoma, are uncommon in cats. 5,12,15,24 Vascular neoplasms are identified by the formation of blood-filled vascular spaces lined by neoplastic endothelial cells. 11 In well-differentiated vascular neoplasms, diagnosis is straightforward and seldom requires immunohistochemistry. However, hemangiosarcomas may regionally form solid sheets of spindloid neoplastic cells without formation of vascular spaces; poorly differentiated hemangiosarcomas may lack vascular channel formation altogether and morphologically mimic nonvascular spindle cell sarcomas. Telangiectatic osteosarcoma may grossly and histologically resemble osseous hemangiosarcoma and is included in the differential diagnosis for a lytic neoplasm of bone with blood-filled spaces. 10,11 Finally, an epithelioid phenotype of hemangioma and hemangiosarcoma, described in several domestic species, 30 further complicates morphological diagnosis. For these reasons, immunohistochemistry is commonly used to diagnose vascular neoplasms in domestic species.
Three well-established endothelial cell immunomarkers in human tissues are factor VIII–related antigen (FVIII-ra), CD31, and CD34. Factor VIII-ra is a protein packaged in endothelial cell Weibel-Palade bodies and is also expressed in megakaryocytes. 31 CD31, also known as platelet-endothelial cell adhesion molecule-1 (PECAM-1), is a transmembrane glycoprotein adhesion molecule expressed by platelets, megakaryocytes, and endothelial cells. 31 CD34 is a cell-surface marker expressed in endothelial cells and hematopoietic stem cells, as well as nerves, hair follicles, muscle bundles, and sweat glands. 14,31 Although the function of CD34 has not been fully elucidated, L-selectin has been identified as its ligand. CD34 may function in part as a cell trafficking molecule for leukocytes and hematopoietic stem cells. 18
The relative utility of FVIII-ra, CD31, and CD34 antibodies in the diagnosis of cat vascular neoplasms has not been evaluated. The aims of this retrospective study were (1) to evaluate and compare specific and background immunohistochemical labeling of vascular neoplasms of cats with anti-FVIII-ra, anti-CD31, and anti-CD34 antibodies and (2) to evaluate the labeling of anti-CD34 antibodies in nonvascular neoplasms as part of the validation of CD34 in the diagnosis of vascular neoplasms.
Methods
The Indiana Animal Disease Diagnostic Laboratory archives at Purdue University were searched for cases of vascular and nonvascular neoplasms in domestic cats. Cases included in the study were biopsy and necropsy specimens received from October 2000 to February 2010. All tissues had been fixed in 10% neutral buffered formalin, paraffin embedded, and stored in the dark at room temperature. For the vascular neoplasms, hematoxylin and eosin (HE)–stained sections were evaluated by all 3 authors, following established diagnostic criteria, 11 and a consensus diagnosis was attained. Vascular neoplasms were classified as hemangioma or hemangiosarcoma.
Immunohistochemistry was performed on all formalin-fixed, paraffin-embedded (FFPE) vascular neoplasms using antibodies to factor VIII–related antigen (FVIII-ra), CD31, and CD34 according to established protocols 21,23 with minor modifications (Table 1). Immunohistochemistry using anti-CD34 antibody was also performed on all FFPE nonvascular neoplasms. Immunohistochemistry was performed using a Dako autostainer; antibody incubations were at room temperature. Canine and feline lymph nodes and canine hemangiosarcoma samples were initially used as positive controls to standardize the immunohistochemical technique. The chromogen-substrate combination of 3,3′-diaminobenzidine-H2O2 was used to detect immunological labeling. Endothelial cells of normal tissue vessels were used as internal positive controls in all neoplasms. Normal lymphatic endothelial cells expressed the 3 endothelial markers examined in this study.
Immunohistochemical Techniques for Feline Vascular and Nonvascular Tumors
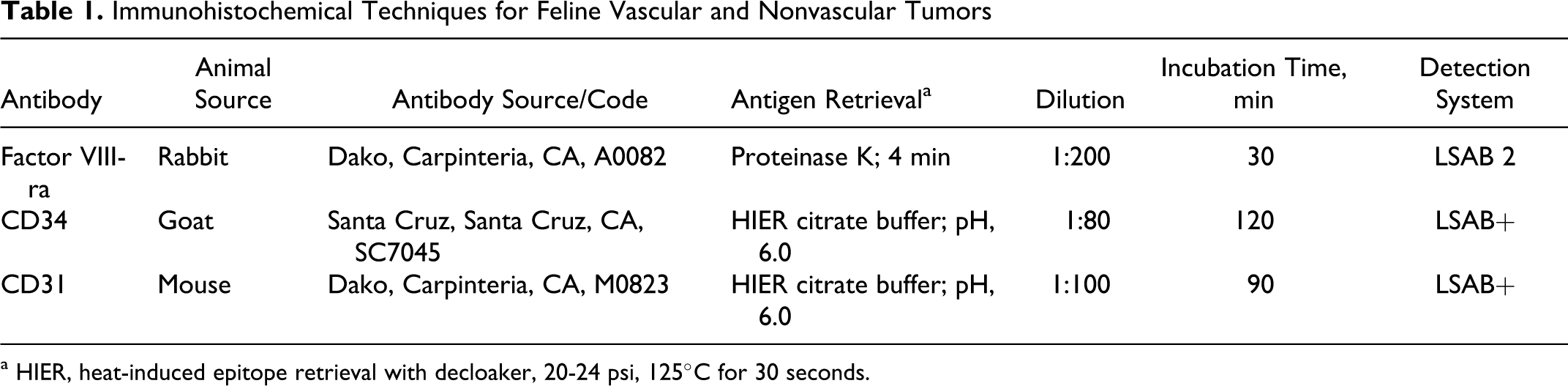
a HIER, heat-induced epitope retrieval with decloaker, 20-24 psi, 125°C for 30 seconds.
The authors independently evaluated the 61 vascular neoplasms for neoplastic cell immunohistochemical labeling intensity and nonspecific background labeling. Specific immunoreactivity and background were graded on a subjective scale of 0 to 3 (in increments of 0.5), in which 0, 1, 2, or 3 equaled absent, weak, intermediate, or strong labeling (or background), respectively. The mean immunoreactivity intensity index and background index were calculated for each vascular neoplasm and antibody. A value representing the difference between labeling intensity and background (labeling index) was calculated by subtracting the mean background index from the mean intensity index for each antibody. The labeling index for CD34 was compared with those of FVIII-ra and CD31 using a Student’s paired t-test (2-tailed, df = 60, P < .05).
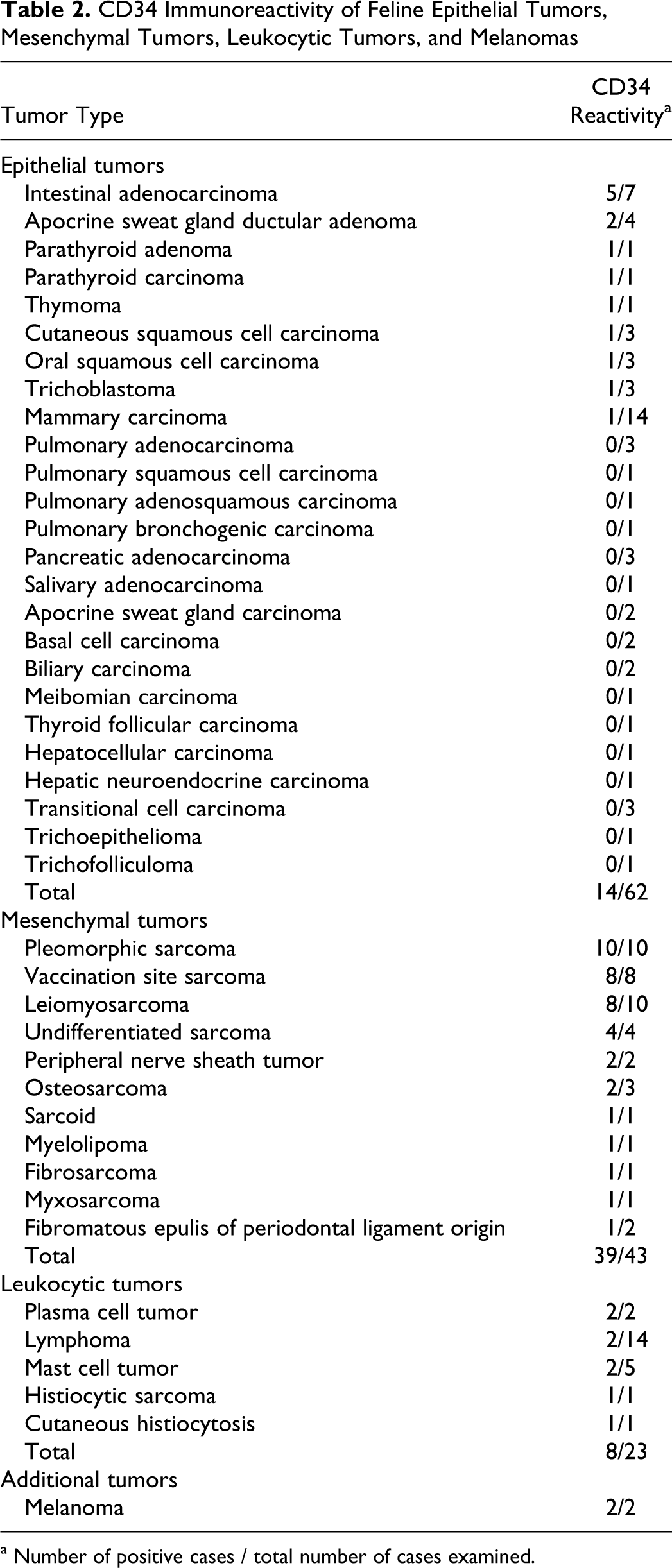
Two authors (RNJ and JAR) independently evaluated the immunoreactivity to CD34 in the nonvascular neoplasms of cats (Table 2), and a consensus diagnosis for immunoreactivity (positive or negative labeling) was made.
CD34 Immunoreactivity of Feline Epithelial Tumors, Mesenchymal Tumors, Leukocytic Tumors, and Melanomas
a Number of positive cases / total number of cases examined.
Results
Vascular Neoplasms
Sixty-one vascular neoplasms (50 hemangiosarcomas and 11 hemangiomas) from 59 cats were included in the study. Two of 48 hemangiosarcomas had metastasis (included in this study). Neoplasms of lymphatic origin were not available for this study. The sites of the neoplasms were skin (23), subcutis (21), lymph node (4), liver (3), mesentery (3), lung (2), colon (1), heart (1), tongue (1), gingiva (1), and 1 of unreported tissue origin. The endothelial cells of nonneoplastic vessels (internal positive control) within the histological sections were labeled with all 3 antibodies in all cases. All neoplasms were positive for factor VIII-ra, CD31, and CD34; however, variation among markers and neoplasms was marked (Figs. 1 –6). Immunolabeling for CD31 was predominantly membranous, with mild cytoplasmic labeling. CD34 and FVIII-ra had diffuse cytoplasmic labeling. FVIII-ra also had punctate, granular labeling in a few neoplastic cells. In summary, CD34 had the highest mean immunolabeling intensity (2.95), followed by factor VIII–related antigen (2.46) and CD31 (2.1). Factor VIII–related antigen had the highest mean background labeling (1.71), followed by CD34 (1.22) and CD31 (0.21). The average labeling intensity for FVIII-ra, CD31, and CD34 was 2.43, 2.27, and 2.94, respectively, in hemangiosarcomas, and 2.62, 1.5, and 2.98, respectively, in hemangiomas. Background labeling for factor VIII-ra was generally present within the neoplastic interstitium, in areas of necrosis, and within neoplastic vascular channels. Background labeling for CD34 was predominantly within the neoplastic interstitium and was less intense within vascular channels. Background labeling for CD31 was generally low, multifocal, and inconsistently present. The labeling index for factor VIII-ra, CD31, and CD34 was 0.75, 1.92, and 1.74, respectively. The labeling index for CD34 was significantly higher (P < .001) than that for FVIII-ra but not significantly different from that for CD31 (P = .115). The labeling index of the primary and metastatic neoplasms was similar.
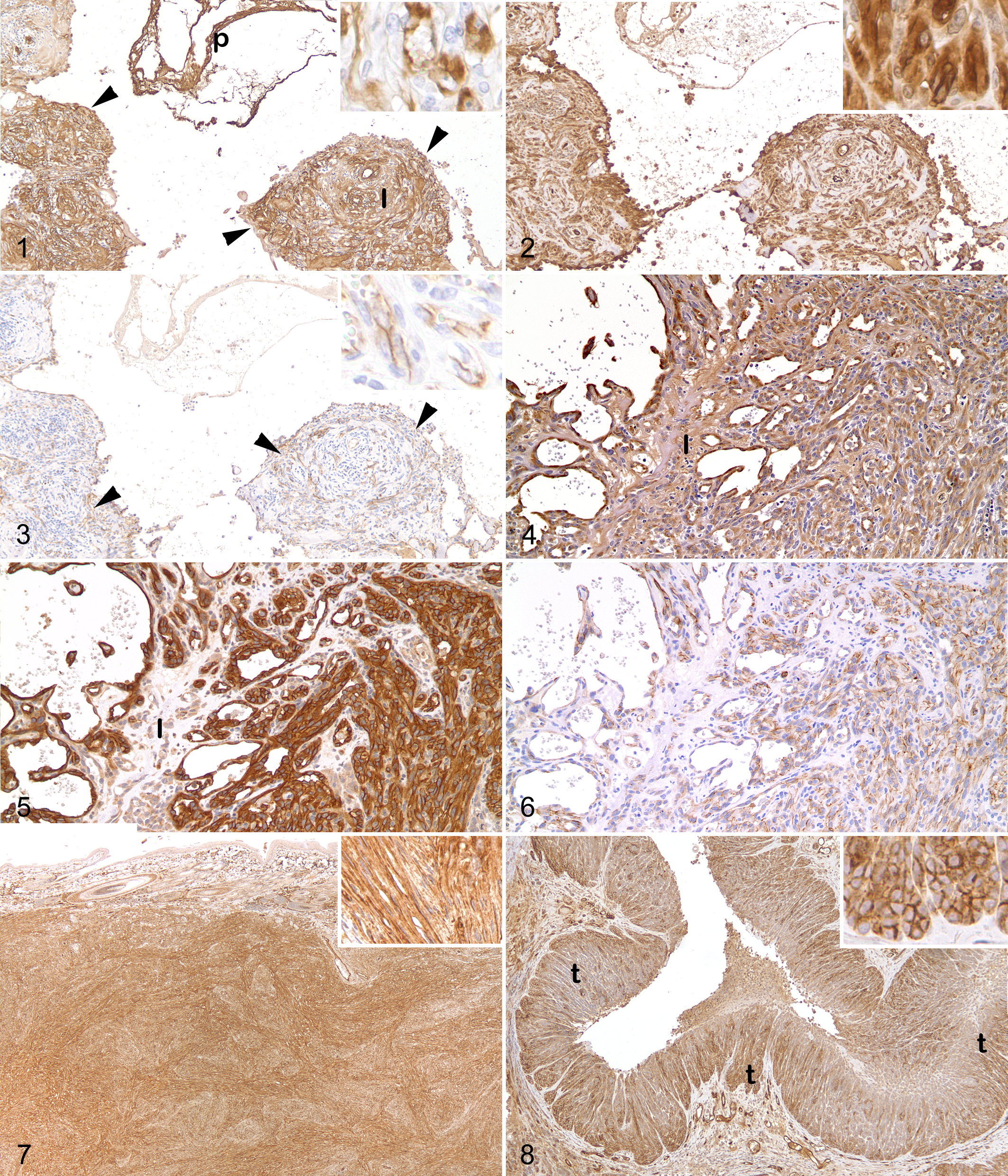
Lymph node; cat. Hemangiosarcoma. Aggregates of neoplastic endothelial cells (arrowheads) label strongly positive for factor VIII–related antigen, with moderate to marked background labeling of the tumor interstitium (I) and plasma (p). Inset: Detail of the cytoplasmic labeling. Immunoperoxidase with diaminobenzidine (DAB) chromogen and hematoxylin counterstain.
Nonvascular Neoplasms
One hundred thirty nonvascular neoplasms in cats were selected from the archives. Sixty-two were epithelial neoplasms, 43 were mesenchymal neoplasms, 23 were leukocytic neoplasms, and 2 were melanomas. The diagnosis of pleomorphic sarcoma was based on established criteria 6 and included heterogeneous neoplasms with mixtures of pleomorphic spindle-shaped cells, histiocytoid cells, and multinucleated giant cells. Details of the neoplasm types and CD34 reactivity are in Table 2. In summary, 14 of 62 epithelial neoplasms, 39 of 43 mesenchymal neoplasms, 8 of 23 leukocytic neoplasms, and 2 of 2 melanomas had CD34 immunoreactivity (Figs. 7, 8). In the sole thymoma, the neoplastic epithelial cells labeled strongly with CD34, whereas the benign lymphocytic component was CD34-negative. In the apocrine gland ductular adenoma, more basilar than luminal epithelial cells were positive for CD34, although this distinction was lost in more solid regions. Approximately 1% of the hematopoietic cells in the myelolipoma were positive for CD34 (and probably represented hematopoietic stem cells).
Discussion
Immunohistochemistry is useful in definitive diagnosis of vascular neoplasms in poorly differentiated cases. Immunohistochemical markers commonly used to identify neoplastic endothelial cells include factor VIII–related antigen, CD31, and CD34. In this study of vascular neoplasms in cats, CD34 was the marker with the highest labeling intensity (2.95) for neoplastic endothelial cells and had a moderate background (1.22). FVIII-ra had the highest background (1.71) with a moderate labeling intensity (2.46). CD31 had the lowest background (0.21) and also had a moderate labeling intensity (2.1). A labeling index was calculated by subtracting the background scores from the labeling intensity scores for each endothelial marker. The endothelial cell labeling index for CD34 was significantly higher than that of FVIII-ra and not different from that of CD31. A large difference between specific labeling intensity and nonspecific background labeling is characteristic of an ideal immunohistochemical marker, 22 so based on this feature, CD34 may be particularly useful when there is significant background staining with factor VIII-ra in the diagnosis of vascular neoplasms in cats.
CD31 is expressed in nearly all human vascular neoplasms 31 and was expressed in all cat vascular neoplasms in this study. Well-differentiated neoplasms, such as hemangiomas, might be expected to label equally or more intensely with a specific marker than less well-differentiated neoplasms. However, the average labeling intensity score for CD31 in the hemangiomas (1.5) was less than that in hemangiosarcomas (2.27). This unexpected difference may be due in part to the low number of hemangiomas examined (11 vs 50 hemangiosarcomas) and may not be representative. For FVIII-ra, the labeling intensity score was slightly lower in hemangiosarcoma (2.43) than in hemangioma (2.62). In human angiosarcomas, inconsistent FVIII-ra antibody labeling has been reported. 16 Alternatively, overexpression of some genes has been reported in some canine vascular neoplasms. 26
Despite the sensitive labeling of neoplastic endothelial cells with antibodies to CD34, many nonvascular (epithelial, mesenchymal, leukocytic, and melanocytic) neoplasms also had positive labeling for CD34. CD34 labeling has been well-characterized in human endothelial cell neoplasms 31 and in nonvascular neoplasms, such as human precursor lymphoid and myeloid neoplasms, 29,31 human and canine gastrointestinal stromal neoplasms, 8,9,17,19,32 and canine and feline meningeal neoplasms. 23 CD34 labeling has also been sporadically described in human fibroblastic, myofibroblastic, lipomatous, smooth muscle, perivascular wall, melanocytic, follicular, and peripheral nerve sheath neoplasms. 1,3,4,13,17,27,28 In our study, CD34 labeling was observed in most intestinal adenocarcinomas (5/7) and parathyroid neoplasms (2/2) and sporadically in apocrine gland neoplasms (2/6), squamous cell carcinomas (2/7), follicular neoplasms (1/5), and mammary carcinomas (1/14). In the sole thymoma, CD34 labeling was restricted to the neoplastic thymic epithelial cells (confirmed by immunohistochemistry for pancytokeratin). CD34-positive labeling has not been reported in human thymomas.
About one-third of the leukocytic neoplasms in this study were CD34-positive. Lymphoma derived from precursor lymphocytes may be CD34-positive, 29,31 so the 2 (of 14) CD34-positive lymphomas in this study may have been derived from precursor lymphocytes. Two of 5 mast cell neoplasms had CD34 labeling. CD34 labeling has been described in mature murine mast cells, 7 although to our knowledge, CD34 labeling of mast cell neoplasms in domestic species has yet to be evaluated. All histiocytic (2/2) and plasmacytic (2/2) neoplasms evaluated were positive for CD34.
Ninety-one percent of the mesenchymal neoplasms expressed CD34. Nonvascular spindle cell neoplasms may be considered in the differential diagnosis for otherwise ambiguous or poorly differentiated vascular neoplasms. The widespread labeling of nonvascular spindle cell neoplasms with CD34 antibodies in our study suggests that CD34 is not a distinguishing immunomarker for hemangiosarcoma, in particular when vascular formation is not apparent and other spindle cell neoplasms are considered in the differential diagnosis.
Both the melanomas in this study were CD34-positive. CD34-positive melanomas have been occasionally reported in humans, 2 although CD34 immunoreactivity was not observed in a recent evaluation of the immunohistochemical phenotype of canine oral melanoma. 25
The difficulty in evaluating the immunoreactivity of the nonvascular neoplasms is in interpreting the specificity of the CD34 immunolabeling. Polyclonal antibodies are considered to be more sensitive markers because of recognition of multiple epitopes. However, with this increased sensitivity comes inherent epitope cross-reactivity. In addition, formalin fixation–associated cross-linking of proteins modifies antigenic properties and can affect antigen availability and immunoreactivity, 20 so a component of nonspecific CD34 labeling in the examined nonvascular neoplasms cannot be excluded. Many of the CD34-positive neoplasms in this study have not, to our knowledge, been reported to be CD34-positive in human counterparts, making it difficult to interpret positive immunolabeling as true CD34 protein expression in the neoplastic cells. Evaluation for CD34 mRNA expression may better characterize the specificity of CD34 immunoreactivity in these nonvascular neoplasms. Finally, neoplastic cells are well-known for their aberrant and varied expression of nonnative proteins, and aberrant CD34 protein expression within these nonvascular tumor cells is possible.
In a similar study of CD34, CD31, and FVIII-ra expression in human vascular and nonvascular neoplasms, 16 the authors concluded that a combination of CD34 and CD31 antibodies provided the best sensitivity and specificity in the diagnosis of human vascular neoplasms. Our study results are similar and suggest that although CD34 is a sensitive label for vascular neoplasms of cats, low specificity may hinder the sole use of this marker in the diagnosis of feline vascular neoplasms, in particular when nonvascular spindle cell sarcomas are included in the differential diagnosis. In summary, the broad expression of CD34 in neoplasms of cats does not warrant its use as a marker of vascular neoplasms in this species.
Footnotes
Acknowledgements
We thank Dee Dusold and the Animal Disease Diagnostic Laboratory histology laboratory staff for technical assistance. Preliminary results from this study were presented at the 2010 annual meeting of the American College of Veterinary Pathologists.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
