Abstract
A 3-year-old Texas Longhorn steer had a long history of progressive swelling of the soft tissues of the jaw and neck. At necropsy, multifocal to coalescing dermal and subcutaneous pyogranulomas were surrounded by fibrous tissue. Microscopically, the pyogranulomas contained aggregates of gram-negative coccobacilli surrounded by Splendore-Hoeppli material and were separated by bands of fibrovascular tissue (botryomycosis). Phylogenetic analysis of multilocus sequence-typing data revealed that the bacteria recovered in pure culture from swabs of submandibular tissue were most closely related to Bibersteinia [Pasteurella] trehalosi. The bacterial colonies were immunohistochemically reactive with a rabbit polyclonal anti-Pasteurella class C acid phosphatase antibody. Botryomycosis is a pyogranulomatous inflammation caused by a variety of nonbranching, nonfilamentous bacteria that elicit the formation of Splendore-Hoeppli material. This case of botryomycosis is unique for its association with Bibersteinia trehalosi.
Keywords
Botryomycosis, also known as bacterial pseudomycetoma, bacterial pseudomycosis, bacterial granuloma, or granulomatosis, is a chronic, pyogranulomatous inflammatory disease that mimics the clinical and microscopic lesions of mycetoma caused by infection with fungi or Actinomyces species. 16 Microscopically, botryomycosis is characterized by multifocal pyogranulomatous inflammation with colonies of gram-positive or gram-negative bacteria rimmed by Splendore-Hoeppli material (asteroid bodies) and separated by bands of fibrous tissue. 14 The skin and lungs are the most common sites for botryomycosis, although it has also been reported in the pericardium, spermatic cord, skeletal muscle, mammary gland, peritoneum, lymph nodes, and nasopharynx. 11,13,20,23 Staphylococcus aureus is most commonly recovered, but other reported bacteria include Pseudomonas aeruginosa, Proteus vulgaris, Escherichia coli, Streptococcus, Actinobacillus, Pasteurella multocida, and Arcanobacterium pyogenes. 6,9,14,17,19,24
Bibersteinia trehalosi causes septicemia in lambs and pneumonia in cattle, domestic sheep, and bighorn sheep in North America 8,18,22,26 but, to our knowledge, has not been reported as a cause of botryomycosis. The purpose of this report is to describe a case of subcutaneous botryomycosis caused by B. trehalosi in a Texas Longhorn steer.
Case Presentation
A 3-year-old Texas Longhorn steer with a history of weight loss and progressive (over several weeks) submandibular swelling was presented for postmortem examination. The steer was euthanatized because of poor prognosis. At necropsy, the soft tissue at the caudoventral aspect of the mandible and the cranioventral aspect of the neck was expanded bilaterally by communicating abscesses or pyogranulomas up to 10 cm in diameter with abundant thick, opaque, yellow-white exudate. Thick bands of fibrous tissue surrounded and separated the pyogranulomas. No cutaneous ulcers, fistulous tracks, or bony involvement of the mandibles was observed. Submandibular and retropharyngeal lymph nodes were enlarged up to 10× normal size. Approximately 5% to 10% of the cranioventral aspects of both lungs were consolidated and dark red, but neither pulmonary abscesses nor granulomas were observed.
Histological Findings
Multifocal to coalescing pyogranulomas with intervening fibrosis expanded the subcutis (Fig. 1). The granulomas had central accumulation of degenerated neutrophils with a peripheral layer of epithelioid macrophages and scattered lymphocytes and plasma cells. Colonies of gram-negative coccobacilli were surrounded by intensely eosinophilic, radiating, club-shaped material (Splendore-Hoeppli phenomenon or asteroid bodies; Figs. 2,3). The inflammation extended multifocally into adjacent salivary glands. The consolidated areas of the lungs were affected by acute suppurative bronchopneumonia with hyperplasia of bronchus-associated lymphoid tissue. Regional lymph nodes had follicular hyperplasia with prominent germinal centers; medullary sinuses were filled with numerous macrophages and fewer neutrophils.
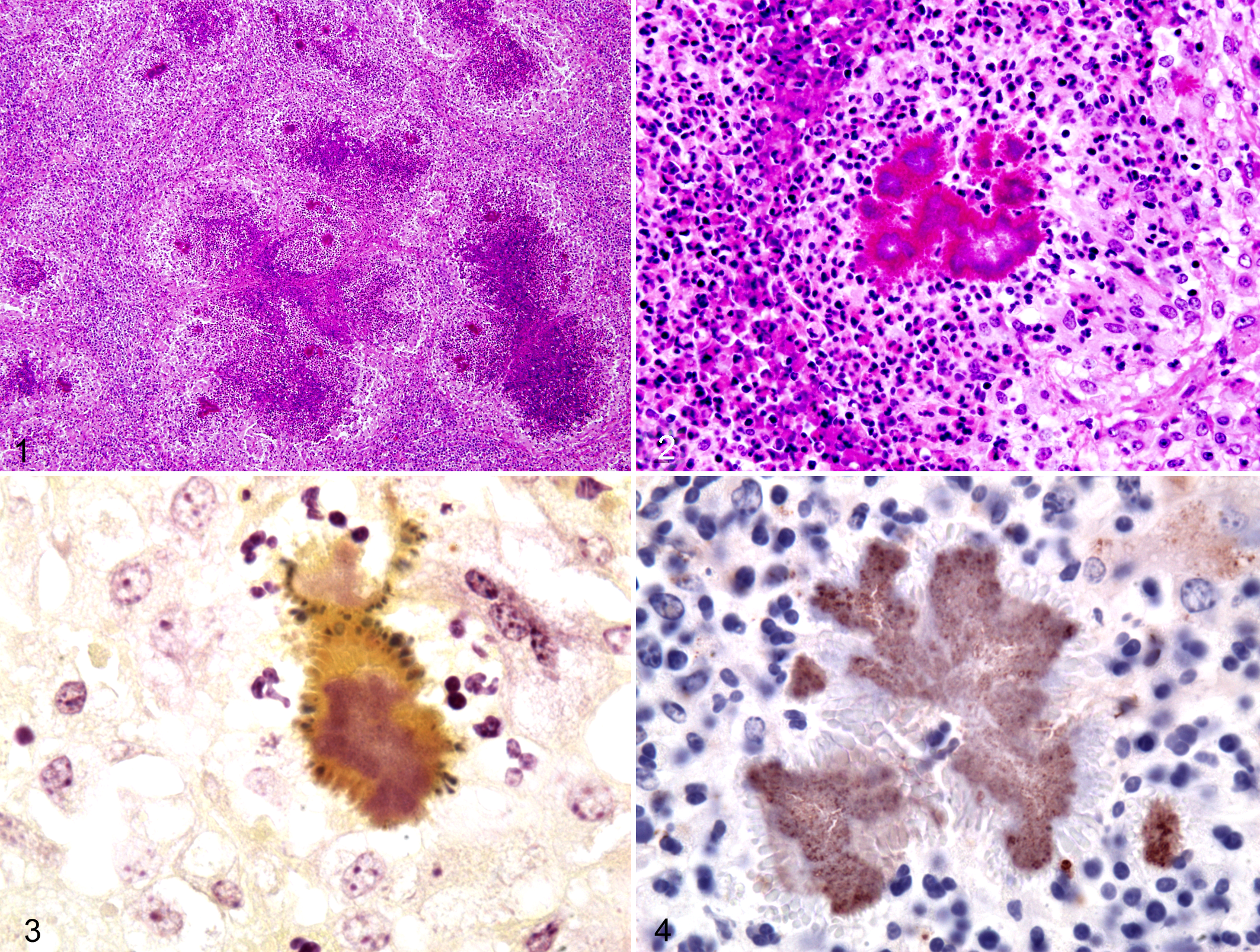
Bacteriological Findings
The bacteria, recovered in pure culture from swabs of affected subcutis, were presumptively identified as Mannheimia haemolytica using the Sensititre AP 80 (Trek Diagnostic Systems, Cleveland, OH). Because M. haemolytica is not a commensal organism of ruminant skin and had not been reported previously as a cause of botryomycosis, additional tests were performed. The bacterial colonies were strongly positive by immunohistochemistry using a rabbit polyclonal Pasteurella class C acid phosphatase antibody (Fig. 4), which was consistent with the isolation of Pasteurella species from bacterial swabs. 21
To determine the nucleotide sequences of selected housekeeping genes, multilocus sequence typing (MLST) was used. A genomic DNA template was used in standard polymerase chain reaction (PCR) amplifications (Phusion high fidelity DNA polymerase, Fermentas, Glen Burnie, Maryland) to retrieve regions of the 16S rRNA, rpoB, recN, rpoA, and tdhF genes. The resulting amplicons were purified through spin columns (Qiagen, Valencia, California) and sequenced with BigDye chemistry using gene-specific oligonucleotide primers (IDT, Coralville, Iowa) at the University of Missouri DNA Core Facility.
When compared with GenBank databases using BLASTn, 1 the nucleotide sequences had 99% homology with orthologous genes from B. trehalosi, with less homology to Mannheimia species (M. haemolytica, 88%; M. granulomatis, 87%; M. ruminalis, 87%). For each gene, Haemophilus parasuis had the most homology outside the B. trehalosi taxon. In summary, the comparative sequence analyses indicated that the recovered bacterial strain was B. trehalosi. The sequences were deposited in GenBank as JF957191 (rpoB), JF957192 (rpoA), JF957193 (recN), JF957194 (tdhF) and JF957195 (rrn).
Discussion
The [Pasteurella] haemolytica complex was formerly divided into biovars A and T. Biovar A organisms are now assigned to the genus Mannheimia 2 ; biovar T was reclassified as B. trehalosi. 4,22 B. trehalosi produces leukotoxin, which is considered an essential virulence factor. 7 It is a cause of septicemia in sheep in the United Kingdom and the United States. 8 In the United States, B. trehalosi can cause a fibrinous bronchopneumonia with high mortality in bighorn sheep. 18,26 In contrast, B. trehalosi infection is usually subclinical in cattle, although it has been associated with bronchopneumonia. A fatal case of necrotizing hepatitis was reported in a cow in the United Kingdom. 25 B. trehalosi has also been isolated from granulomas and joints of cattle and the brain of a Roe deer. 4,25 It is considered normal flora in the tonsils of bison. 10
The recovery of B. trehalosi was unexpected in this case, because Bibersteinia species have not been associated with botryomycosis, although other Pasteurella species have been isolated from botryomycosis in a dog and from jaw abscesses in a cow. 3,19,24 In humans, P. multocida has been isolated from hepatic pyogenic granulomas, pulmonary pyoepithelioid granulomas, and abdominal granulomas. 5,12,15
At necropsy, the differential diagnosis included mandibular osteomyelitis caused by Actinomyces bovis and actinobacillosis caused by Actinobacillus lignieresii. Concurrent infection with Arcanobacterium pyogenes was considered because of the pyogenic nature of the granulomas. Lymphoma was not considered in the differential diagnosis because of the apparent involvement of soft tissue beyond the lymph node and the presence of abundant purulent exudate. The absence of bony lesions in the mandible and the presence of gram-negative, rather than gram-positive, bacteria were inconsistent with actinomycosis and with infection by A. pyogenes or Nocardia spp. Gram-negative bacteria commonly associated with botryomycosis, such as Actinobacillus spp, Pseudomonas spp, Proteus spp, and E. coli, were considered as possible causes. However, B. trehalosi was recovered in pure culture from samples of subcutaneous lesional tissue and identified by molecular methods. In addition, the intralesional bacteria were immunoreactive with Pasteurella class C acid phosphatase antibody. This case of subcutaneous botryomycosis is unique in the recovery of B. trehalosi, which, to our knowledge, is not a known cause of botryomycosis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
