Abstract
Meningiomas are the most common intracranial tumors in dogs. A variety of inflammatory cells have been shown to invade these tumors in people, but little is known about interactions between the immune system and naturally occurring brain tumors in dogs. The purpose of this study was to investigate the presence of a variety of immune cell subsets within canine intracranial meningiomas. Twenty-three formalin-fixed, paraffin-embedded tumor samples were evaluated using immunohistochemistry with antibodies specific for CD3, CD79a, CD18, CD11d (αD), CD45RA, forkhead box P3, and Toll-like receptors 4 and 9. Immune cell infiltration was evident in all samples, with a predominance of CD3+ T cells. Large numbers of CD18+ microglia and macrophages were noted surrounding and infiltrating the tumors, and a subset of these cells within the tumor appeared to be CD11d+. Scattered macrophages at the tumor–brain interface were TLR4+ and TLR9+. Rare CD79a+ B cells were noted in only a small subset of tumors. Lesser numbers of lymphocytes that were CD11d+, CD45RA+, or FoxP3+ were noted in a number of the meningiomas. Although the function of these cells is not yet clear, work in other species suggests that evaluation of this immune cell infiltrate may provide important prognostic information and may be useful in the design of novel therapies.
Meningiomas are the most common brain tumor in dogs and cats, and they also frequently occur in humans. 37,54,66 Histology typically reflects a benign neoplasm in all species, although atypical and anaplastic variants are well recognized. 36,47 In addition, invasion of adjacent brain parenchyma has been reported with all tumor grades and appears to occur more frequently in dogs than in other species. 46 This invasive nature complicates therapy in canine patients. Surgical excision of low-grade meningiomas usually results in long-term control or cure in cats and humans. 23,24,61 Despite some recent promising results with advanced surgical techniques including long-term survival in some cases, 27,35 many canine meningiomas have poorly defined margins and an unclear plane of dissection, making complete surgical removal challenging. 19,43 Therefore, adjunctive radiotherapy is often recommended to prolong survival, which results in additional adverse effects and a prolonged course of therapy, and it usually dramatically increases the expense of treatment. 4 As a result of these shortcomings, novel therapies are required to improve outcome. One such novel intervention is immunotherapy, which involves manipulation of the immune system in order to generate an anti-tumor response. Effective immunotherapy requires detailed knowledge of the different populations of immune cells present within tumors as well as an understanding of their function.
The relationship between the immune system and brain tumors in humans has been extensively evaluated. The majority of studies have examined glial cell tumors, which typically show a robust infiltrate of macrophages and T cells with a surrounding proliferation of parenchymal microglia. 34,42,50,52 Large mononuclear cells expressing macrophage markers are also found in the majority of meningiomas from human patients. 3,8,51 Tumors with a higher grade tend to have larger numbers of infiltrating macrophages. 8,50 Large numbers of lymphocytes have also been documented in human meningiomas, and the majority of these cells appear to be CD8+ T cells, with sparse populations of CD4+ cells and B cells. 8,51
The role of these infiltrating immune cells is not clear, and both anti-tumor and pro-tumor effects have been suggested. Several early studies documented a better prognosis in patients with glial cell tumors and larger lymphocytic infiltrates, 9,20,45 although other studies failed to show such a benefit. 10 The heterogeneous nature of the infiltrating lymphocyte population may, in part, account for this discrepancy but was not examined in these reports. Whereas CD8+ cytotoxic T cells might have anti-tumor effects, other lymphocyte populations, such as regulatory T cells (Tregs), are important in contributing to peripheral tolerance and the escape of tumors from an immune response. Regulatory T cells are classically defined as being CD4+CD25+ and express the forkhead box P3 (FoxP3) transcription factor, which contributes to their development. 32 Populations of Tregs have been shown to contribute to immune escape in a variety of cancers, 74 and recent studies have also shown these cells in human brain tumors. 30,33
Toll-like receptors (TLRs) on the surface of immune cells recognize a variety of microbial molecular patterns, and they initiate immune and inflammatory responses through several signal transduction pathways. For example, TLR4 recognizes lipopolysaccharide (LPS), and TLR9 recognizes unmethylated cytosine-guanine dinucleotide (CpG) motifs characteristic of bacteria. Agonists of both TLR4 and TLR9 have shown promise in immunotherapeutic trials in experimental models. 40
There is very little information concerning interactions between the immune system and naturally occurring brain tumors in companion animals. The purpose of this study was to examine macrophage, microglial, and lymphocyte populations within and surrounding canine intracranial meningiomas using immunohistochemistry.
Materials and Methods
Sample Preparation and Medical Record Review
Tissue samples from 23 canine intracranial meningiomas were obtained from the pathology archives at North Carolina State University. Samples had been collected from patients intraoperatively as part of a therapeutic resection or at necropsy. All tissues were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 5 μm, and mounted on glass slides. Sections of lymph nodes from clinically normal dogs were used as positive control tissue. The histologic variant of meningioma was obtained from the pathology report generated at the time of surgery or necropsy, and standard veterinary references were used as an aid in classification of these tumors. 36,62 Details regarding therapeutic interventions including radiation therapy, chemotherapy, and glucocorticoid administration were obtained from the medical records.
Immunohistochemistry
Slides were deparaffinized in xylene, and the tissue was rehydrated using graded alcohols. Antigen retrieval was performed by placing tumor slides in a pH 9.0 solution (Target retrieval solution, pH 9, Dako, Carpinteria, CA) within a heated pressure chamber (Pascal, Dako) for fifteen minutes at 123°C and 20 psi, except for CD18, which required incubation in a proteinase K solution (Dako). Endogenous peroxidase and nonspecific staining were blocked using 0.5% hydrogen peroxide in methanol and 2.5–3% normal serum from the appropriate species, respectively. Immunohistochemical staining was performed using primary antibodies directed against a variety of immune cells and markers. Sections were incubated for sixty minutes at room temperature (RT) or overnight at 4°C (Table 1). All tumors were incubated with primary antibodies against CD3, CD79a, CD45RA, CD18, CD11d, and FoxP3, and a subset of tumors was also stained for TLR4 and TLR9 (cases 1–3 and 5–14). Biotinylated secondary antibodies (1:300, Vector Laboratories, Burlingame, CA) were applied for thirty minutes at RT followed by an avidin-horseradish peroxidase reagent (1:500, Vector Laboratories) for thirty minutes. For some primary antibodies, a nonbiotin, peroxidase-based detection system (ImmPRESS anti-mouse Ig polymer detection kit, Vector Laboratories) was used in place of the secondary antibody and avidin-horseradish peroxidase reagent. Substrates used for detection included 3,3′-diaminobenzidine (DAB, Bioexpress, Kaysville, UT) and NovaRed (Vector Laboratories). Tissues were counterstained using hematoxylin (Hematoxylin QS, Vector Laboratories), dehydrated, cleared, and mounted using Permount (Fisher Chemicals, Fair Lawn, NJ). Negative controls were generated by substitution of a nonspecific, species-matched immunoglobulin or by omission of the primary antibody.
Primary Antibodies Used for Immunohistochemistry
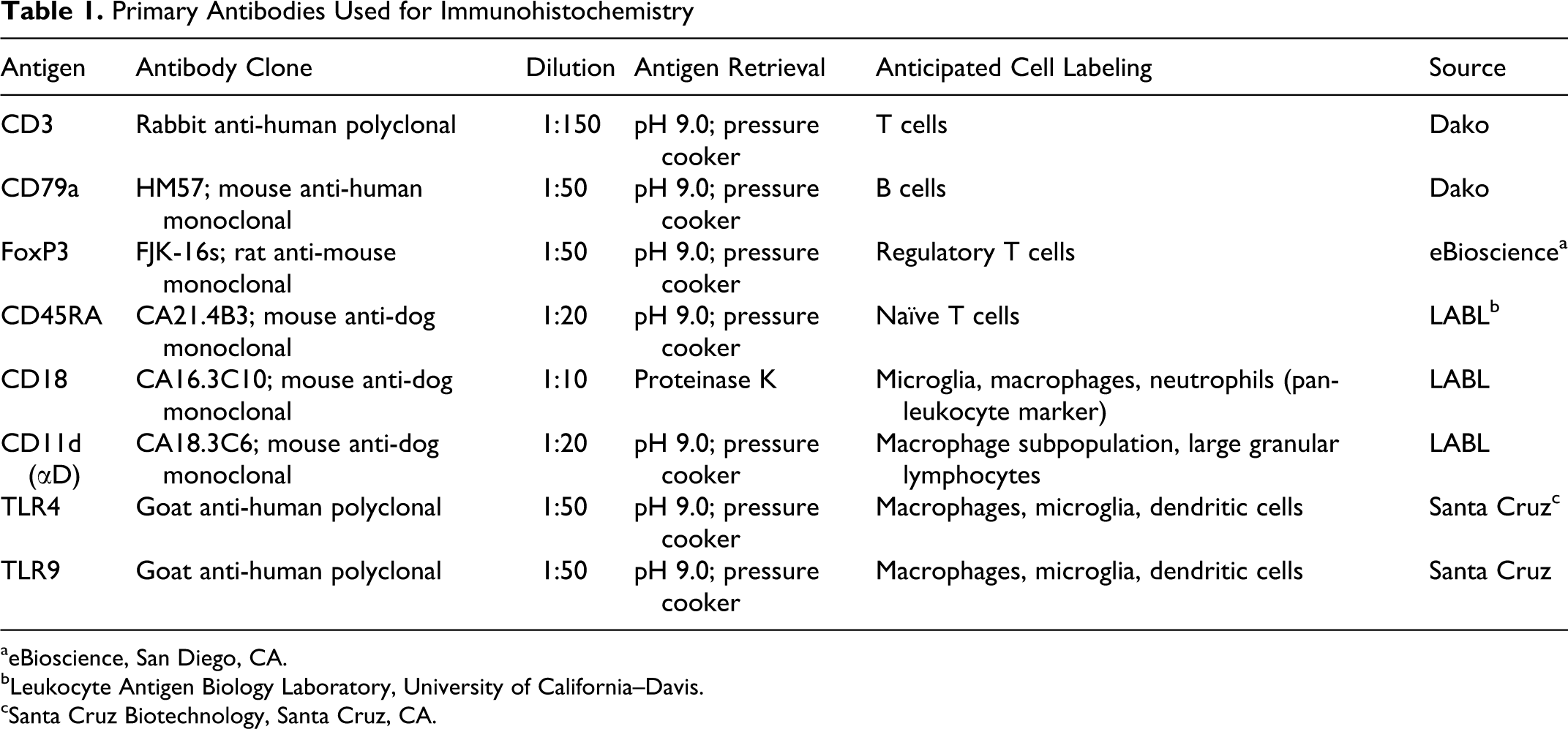
aeBioscience, San Diego, CA.
bLeukocyte Antigen Biology Laboratory, University of California–Davis.
cSanta Cruz Biotechnology, Santa Cruz, CA.
Analysis
Original H&E sections were reviewed to confirm the diagnosis, and immunostained sections were analyzed qualitatively by two of the authors (LB Borst and CLM) using light microscopy. In addition, cell numbers for CD3, CD79, CD45RA, CD11d, and FoxP3 were quantified by counting positively labeled cells per high-power (400X) field (hpf). Sections were scanned on low power to identify areas with the highest concentration of labeled cells. A minimum of 10 hpf were counted and a mean value generated for each antibody. A D’Agostino and Pearson test determined that the cell counts did not follow a Gaussian distribution, and therefore, nonparametric tests were used to evaluate differences in cell populations. To compare populations of immune cells within meningiomas, positively labeled cells for each antibody were evaluated with a Kruskal-Wallis one-way analysis of variance followed by Dunn multiple comparison test. Cell counts associated with different meningioma subtypes (meningothelial vs transitional), intracranial locations (frontal-olfactory vs cerebellum), and therapeutic intervention (glucocorticoid administration or not) were compared using the Mann-Whitney test. An association between patient age and immune cell infiltration was investigated using a Spearman correlation calculation.
We also attempted to account for potential effects on immune cell populations related to the size of the tumor or regions of necrosis. The volume of the tumors was estimated by measuring the tumor dimensions in 3 planes (medial-lateral, ventral-dorsal, and rostral-caudal) from magnetic resonance (MRI) or computed tomographic (CT) images (18 dogs) or from estimates provided in necropsy reports when images were not available (3 dogs). In 2 dogs with surgical biopsies, images were not available for review, and these dogs were excluded from the size analysis. Regions of necrosis were identified using H&E sections, and the proportion of the section that was necrotic (expressed as a percentage of area) was estimated. Associations between tumor volume or percentage tumor necrosis and immune cell infiltration (cell counts) were investigated using a Spearman correlation calculation. All statistical calculations were performed using a commercially available software program (Prism, Graphpad Software, La Jolla, CA). P < .05 was considered significant.
Results
Samples
Twenty-three tumor samples were included in the analysis, including 10 biopsy samples and 13 collected at necropsy. Dogs ranged in age from 6 to 17 years (mean = 10.5 years), and there were 11 males and 12 females (see Table 2). Breeds represented included the Golden Retriever (n = 5), Boxer (n = 4), Labrador Retriever (n = 3), Beagle (n = 2), and one patient each from the Dachshund, Schnauzer, West Highland White Terrier, Old English Sheepdog, Bassett Hound, Chesapeake Bay Retriever, Shetland Sheepdog, German Shepherd, and mixed breeds. Meningioma variants included meningothelial (n = 8), transitional (n = 13), psammomatous (n = 1), and anaplastic (n = 1) patterns. Fifteen of the 23 tumors (65%) had some degree of necrosis, although this was usually a very small portion of the tumor, and only 3 of the meningiomas had greater than 5% of their area that was necrotic. Intracranial locations are detailed in Table 2, and they included frontal lobe (2), frontal-parietal lobes (1), frontal-olfactory region (9), rostral skull base (1), parasellar (1), tentorial (1), cerebellum (5), and cerebellar-pontine angle (1). In 2 dogs, forebrain was the only recorded location. The duration of clinical signs varied from 1 day to 2 years, but it was between 1 and 3 months for most of the dogs.
Tumor Type and Immune Cell Numbers
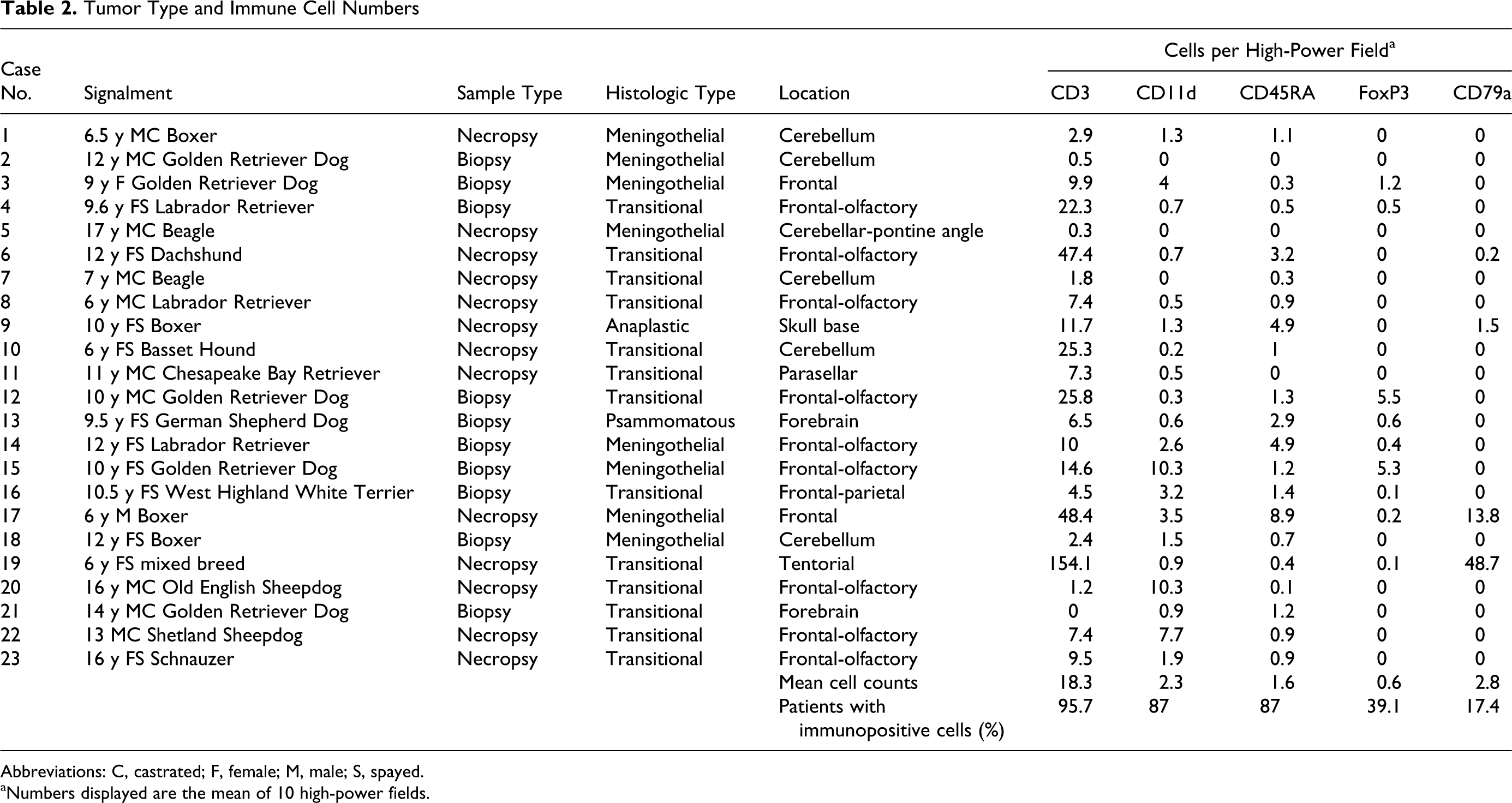
Abbreviations: C, castrated; F, female; M, male; S, spayed.
aNumbers displayed are the mean of 10 high-power fields.
Immunohistochemistry
The results of the immunohistochemical evaluation are presented in Table 2. The antibodies for CD3, CD79a, and CD45RA appeared to stain primarily the cell membrane of select lymphocytes, although the scant amount of cytoplasm present in these cells made determination of cytoplasmic staining difficult. CD3+ lymphocytes were detected in most tumors (22/23, 96%), and several patterns of expression were noted. In tumors in which adjacent brain parenchyma was present, CD3+ cells were prominent in perivascular areas surrounding vessels at the brain–tumor interface (Fig. 2), although these cells were often less numerous around vasculature within the tumor. CD3+ cells were also often randomly interspersed throughout the tumor, either singly or in small clusters (Fig. 1). There were variable numbers of labeled cells, both between tumors and within different areas of the same tumor (Table 2). CD79a+ B cells were noted in only 4 tumors (17%) and were confined to perivascular cuffs at the tumor–brain interface (Fig. 3). Such cuffs always had large numbers of CD3+ T cells present. In addition, populations of cells with a lymphoid morphology that were CD3 and CD79a negative were frequently noted admixed with CD3+ lymphocytes. The smooth muscle layer of larger blood vessels was consistently stained by the CD79a antibody, as has been previously reported. 53 CD45RA+ cells were found in the majority of tumors (20/23, 87%), although in relatively small numbers. Distribution of these cells was similar to CD3+ cells, with positive cells found in a perivascular distribution at the tumor–brain interface and smaller numbers interspersed throughout the tumor. FoxP3 immunoreactivity resulted in positive staining of the nucleus of labeled cells (Fig. 4). Labeled cells were found in small numbers in 9 tumors (39%) and were diffusely distributed throughout the tumor tissue.
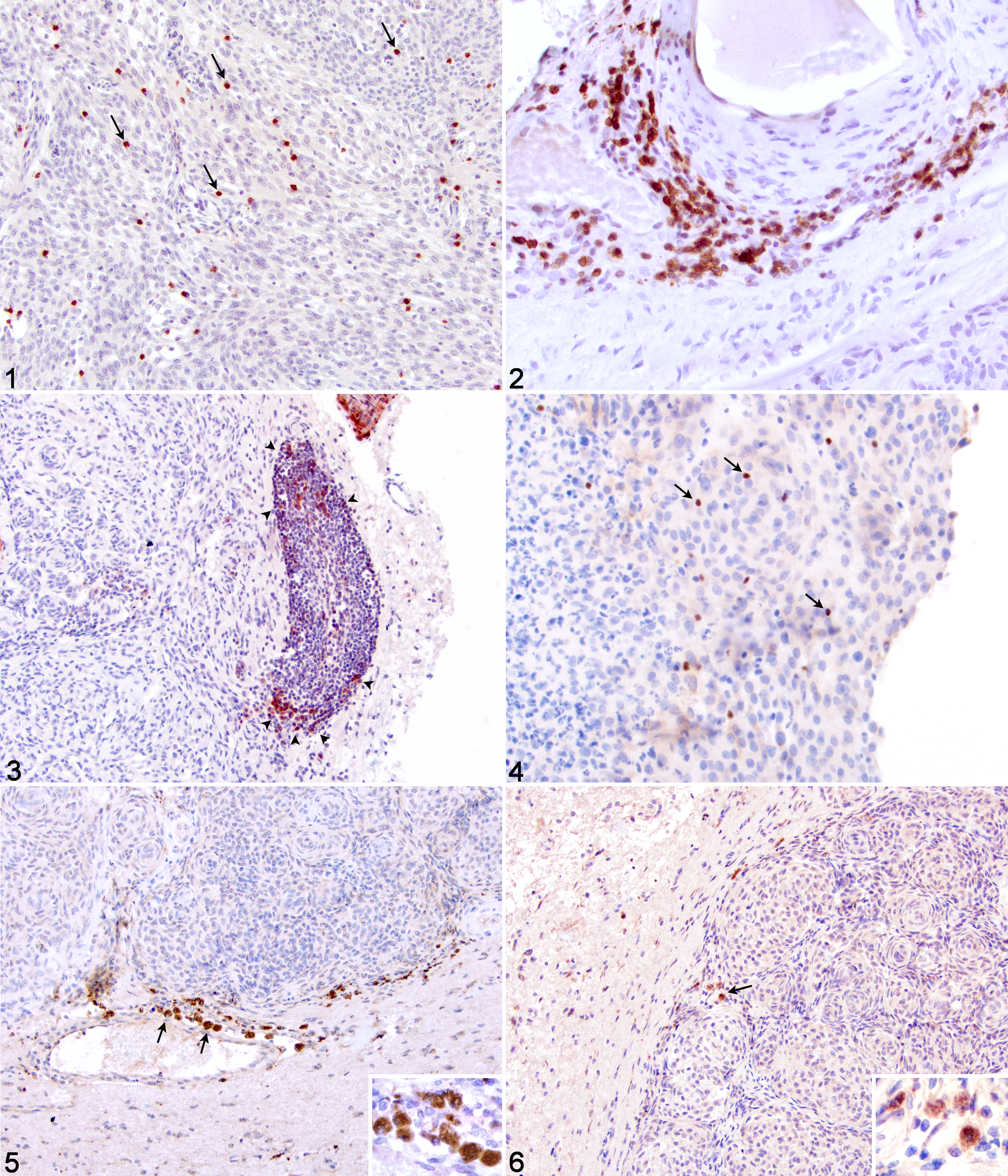
Intracranial meningioma; dog No. 4. CD3+ lymphocytes within tumor (arrows). Immunohistochemistry, rabbit polyclonal antibody to CD3 and hematoxylin counterstain.
CD18 immunostaining resulted in positively labeled cells in the vast majority of meningiomas (22/23, 96%). Large numbers of CD18+ cells with morphologies consistent with ramified and activated microglia were noted within normal brain parenchyma surrounding the tumors and invading into the neoplastic tissue itself (Fig. 7–Fig. 9). Within the meningiomas, positive cells with a macrophage morphology were also noted in some areas (often associated with necrosis; Fig. 10), and CD18+ neutrophils were occasionally seen. Counting of CD18+ cells was not possible because of the dense nature of the cellular infiltrate and the ramified morphology of the cells. Most CD11d+ (αD) cells had similar ramified, dendritic, or rounded morphologies suggestive of microglia and macrophages (Fig. 11 and Fig. 12). These cells also had a variable distribution between different tumors as well as within individual tumors. Interestingly, CD11d+ cells were found almost exclusively within the neoplastic tissue, and adjacent brain parenchyma was typically devoid of labeled cells. Most tumors had positively labeled cells (20/23, 87%), although these cells were found in relatively smaller numbers compared with CD18. Small numbers of cells with a lymphocyte morphology were CD11d+. The pattern of labeling with the CD18 and CD11d antibodies primarily involved the cell membrane, although some cells did appear to have cytoplasmic expression. Immunostaining with the TLR4 and TLR9 antibodies produced similar results, labeling small numbers of cells with a macrophage morphology located at the interface of the meningioma and the normal brain parenchyma, sometimes in a perivascular distribution (Figs. 5 and 6). Cytoplasmic labeling appeared to predominate with these TLR antibodies.
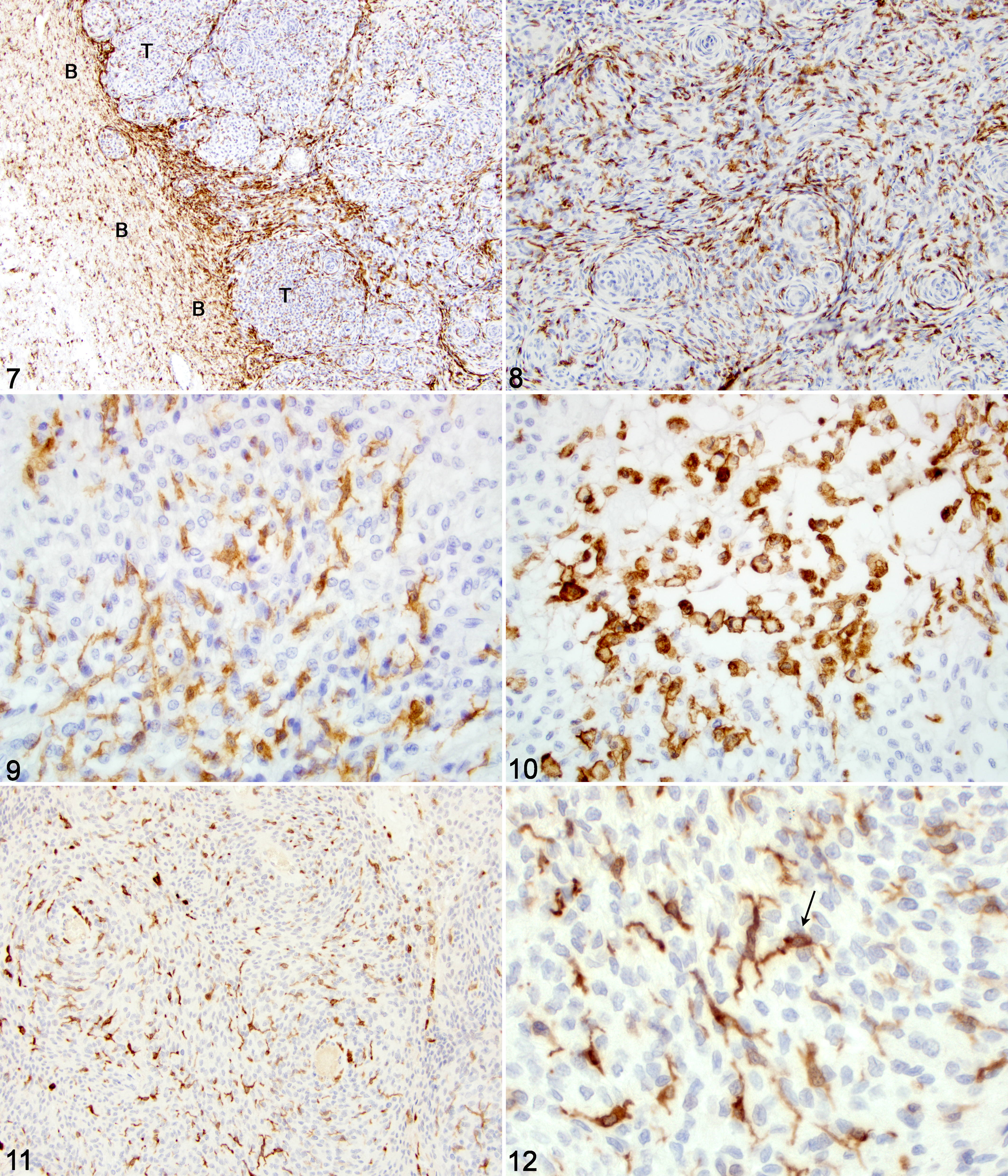
Intracranial meningioma; dog No. 8. CD18+ cells at tumor (T)-brain (B) junction and invading into neoplastic tissue. Immunohistochemistry, mouse monoclonal antibody to CD18 and hematoxylin counterstain.
Considering all tumors in the study, there were significantly more CD3+ cells than other lymphocyte subsets, and more CD45RA+ cells than CD79a+ cells (P < .001 for Kruskal Wallis one-way analysis of variance and P <.05 for comparisons between groups). For meningioma subtype, only meningothelial and transitional tumors were compared, as the psammomatous and anaplastic variants accounted for only one tumor each. No differences in cell counts were detected between these histologic subtypes. CD3 cell counts were found to correlate negatively with age (Spearman r = -0.504, P = .014), although such a correlation was not found for any of the other cell counts. For intracranial location, the frontal, frontal-parietal, and frontal-olfactory groups were combined and compared with the cerebellar group (which also included the cerebellar-pontine angle tumor). Tumors in the frontal region had significantly higher numbers of CD11d+ (P = .015), CD45RA+ (P = .039), and FoxP3+ (P = .029) cells than cerebellar tumors, and CD3+ cell numbers approached significance (P = .063).
Regarding therapeutic intervention, no dogs received radiation therapy, but one dog received lomustine (CCNU; dog No. 1) and one dog received hydroxyurea (dog No. 18). Because of varying doses and different glucocorticoid formulations, administration of glucocorticoids prior to obtaining the histopathological sample was considered only as a yes or no phenomenon. For 4 dogs, it was not possible to definitively discern this information from the medical record. Fifteen dogs received glucocorticoids prior to biopsy or necropsy, whereas 4 did not. Comparison of cell counts between dogs that received glucocorticoids and those that did not did not reveal any statistically significant differences (data not shown). In addition, we found no correlation between immune cell counts and tumor volume or degree of necrosis (data not shown).
Discussion
The meningiomas evaluated in this study showed robust infiltration of CD18+ and CD11d+ cells with microglial and occasional macrophage morphologies. There was concurrent infiltration of large numbers of CD3+ T cells and smaller numbers of CD45RA+ and FoxP3+ cells. CD79a+ B cells were found in only a few tumors, and they were confined to perivascular regions at the periphery of the tumor. Small numbers of TLR4+ and TLR9+ cells with a macrophage morphology were present at tumor–brain boundaries.
Although antibodies against CD18 label the β2 integrin subunit present on most leukocytes, cells of myeloid origin tend to stain more robustly, including microglia. 1,56 There are several populations of CD18+ cells present within the brain, including parenchymal microglia, which are dispersed throughout the neuropil, as well as perivascular cells (also known as perivascular macrophages or perivascular microglia), which as their name implies, are present within the perivascular space surrounding central nervous system (CNS) vasculature. In an undisturbed, normal brain, parenchymal microglia have a finely branched, ramified morphology. After a variety of pathological insults, these cells may acquire a more activated phenotype, characterized by cellular hypertrophy, with thickening and shortening of the branching processes. 58 In some situations (eg, necrosis), these cells may acquire a large, rounded, overtly phagocytic morphology indistinguishable from a macrophage. 58 In addition to phagocytosis, microglia perform a variety of functions, including the secretion of cytokines and growth factors, cytotoxicity, and the presentation of antigen to T cells. 2 The parenchymal microglia represent a population of cells that colonize the brain during embryogenesis with little subsequent contribution from bone marrow sources, but with the potential for local self-renewal. 26 In contrast, perivascular cells normally display a rounded macrophage morphology and undergo continuous replenishment from bone marrow sources. 31 Their perivascular location is well suited to sample CNS antigen, and these cells constitutively express MHCII on their surface. 31 Evidence suggests that these cells are the primary antigen-presenting cells of the CNS. 31,71 Several techniques are available to differentiate parenchymal microglia from perivascular cells in normal brain tissue, including immunohistochemistry for markers such as MHC II and Iba1 and quantification of the relative expression of CD45 with flow cytometry. 6,22,31,56 However, activated microglia can alter the expression of these molecules as well as their morphology, making differentiation from perivascular cells or blood-derived macrophages difficult or impossible with these techniques. 18
Microglia and macrophage infiltration has been documented in brain tumors in both experimental animal models and human patients, 5,25,72 including meningiomas. 3,8,51 However, the role of these cells is unclear and continues to be debated. 5,59 These cells might be acting as antigen-presenting cells. Infiltration of large numbers of T cells in most tumors suggests that tumor antigen is being processed and presented to lymphocytes in peripheral lymphatic organs, and restimulation of these invading T cells by activated microglia and macrophages within and around the tumor might elicit an anti-tumor response. Conversely, tumor-associated microglia have been shown to secrete immunosuppressive cytokines (eg, IL-10, TGF-β) in other species, potentially contributing to tumor invasion and downregulation of an anti-tumor response. 16,70 Increased numbers of macrophages infiltrating meningiomas have been associated with increasing grades of malignancy in human patients. 28 The dense microglial/macrophage response seen at the margins of the tumors in our study resembles the patterns seen in higher grade invasive meningiomas in humans. 28 Traditional guidelines for classification of canine meningiomas have 2 established grades, benign and anaplastic. 36 More recently, however, some authors have suggested the use of an intermediate atypical designation, which would bring the veterinary classification more in line with established human grading schemes. 60 Only one tumor in this study was graded as anaplastic, and atypical grades were not assigned in this study. In addition, adjacent brain parenchyma was often not available for study in these tissue blocks because of sampling limitations, which limited the assessment of invasive activity. However, further study of the relationship between immune cell infiltration, tumor invasion, and meningioma grade in canine patients appears to be warranted.
CD11d is the αD portion of the αDβ2 integrin. Antibodies against this protein label a subpopulation of canine leukocytes including macrophages within the spleen and lymph nodes as well as large granular lymphocytes within the peripheral blood. 17 This integrin appears to be important in binding to extracellular matrix and has a role in leukocyte migration and retention in tissues. 73 Binding to VCAM-1, a vascular adhesion molecule present within the CNS, has been described, which may facilitate entry of peripheral inflammatory cells into the brain or spinal cord. In support of this hypothesis, CD11d expression has been documented in macrophages and neutrophils infiltrating the spinal cord after experimental injury in the rat. 39 However, to our knowledge, CD11d expression on microglia has not been documented, and the role of this molecule in immune cell infiltration into brain tumors has received little attention. Studies of canine microglia analyzed ex vivo have failed to show expression of CD11d in normal dog CNS as well as in dogs infected with distemper virus, 56,57 and the importance of this integrin to tumor–immune system interactions remains unknown.
Similarly, the role of invading lymphocytes remains speculative at this time, as these cells represent a number of potential subsets. CD3+ T cells accounted for the majority of lymphocytes invading meningiomas, and small numbers of B cells were seen in only 17% of the tumors. These findings are very similar to what has been observed in human meningiomas and glial cell tumors, 51,55 and they are distinct from the rare entity known as inflammatory pseudotumor. Inflammatory pseudotumors have been documented in the human CNS, 29 and a recent case report describes a dog with meningeal involvement over the thoracolumbar junction. 38 However, this condition lacks the classic histological features of meningioma and consists of a robust infiltration of plasma cells, which were not noted in our cases. We sought to further classify the invading T cell infiltrate; traditional classification systems have divided T cells into CD4+ “helper” cells and CD8+ “cytotoxic” cells. Whereas the former subtype includes Th1 and Th2 cells, which aid in cell-mediated and humoral immunity, respectively, CD8+ lymphocytes have potential effector function against virally infected or transformed cells. Previous studies of human brain tumors have shown that the majority of invading T cells are CD8+ and may reflect the generation of an anti-tumor response, whereas CD4+ cells are present in smaller numbers. 51,69 However, currently available antibodies against canine CD4 and CD8 are not effective in formalin-fixed tissue and therefore could not be used in this study. We did identify an additional population of lymphocytes that were neither CD3+ nor CD79a+, and the identity of these cells is unclear. It is possible that these may represent natural killer cells, as has been described in human patients. 67,68 The lack of definitive markers for this cell type in canine tissue make confirmation of this hypothesis difficult at this time, although the expression of CD11d noted on small numbers of lymphocytes might support this contention.
CD45RA is an alternatively spliced isoform of CD45, and it has traditionally been considered to be a marker of naïve T cells. 48 Although T cells usually require activation in order to enter and surveil the CNS, CD45RA+ cells have been documented in normal canine cerebrospinal fluid. 65 It is unclear whether these cells enter the CNS in this state or regain CD45RA expression after CNS entry. Most meningiomas in this study contained small numbers of CD45RA+ cells, a finding that has also been noted in experimental rodent tumors. 49 The discrepancy between CD3+ and CD45RA+ cells in this study suggests that most T cells invading canine meningiomas are in an activated state.
Another T cell subset that has recently generated considerable interest are the Tregs. These are cells that suppress the immune response through a variety of mechanisms, including alteration of antigen presentation by dendritic cells, direct contact and lysis of effector T cells, and the secretion of cytokines such as IL-10 and TGF-β. 74 The FoxP3 transcription factor controls development of these cells and serves as a useful marker in several species, including the dog. 7 Regulatory T cells have recently been shown to play an important role in the progression of a variety of different neoplasms, 74 including gliomas. 30,33 Small numbers of FoxP3+ cells were documented in a subset of the canine meningiomas in this study, suggesting that Tregs may be playing a role in tumor escape from the immune system in these patients.
The negative correlation between age and CD3+ cell numbers, although relatively weak, is intriguing and may suggest a depressed immune effector response to tumors with advancing age. Several lymphocyte subsets were found in larger numbers within meningiomas affecting the forebrain compared with those in the cerebellar region (CD45RA+ and FoxP3+ cells showed significant differences, and CD3+ cells approached significance), and CD11d + cells had a similar distribution. The significance of this difference in cell numbers in different brain areas is unknown at this time, but it may indicate regional differences in the immune response to CNS tumors. Alternatively, these differences related to age and tumor location might be statistical artifacts related to the relatively small number of tumors analyzed in each group.
Detectable expression of TLR4 and TLR9 was confined to small numbers of macrophages at the tumor–brain interface. Although microglia have been documented to express these receptors in previous studies in other species, 44 expression of these molecules on the surface of microglia in these patients was either absent or below the limit of detection by this immunohistochemical method. Expression of TLR9 has been documented in microglia and macrophages present within human glioblastoma samples. 41
The original hypothesis that the immune system could detect and eliminate tumor cells was developed by Burnet and Thomas in the 1950s. 11,64 They postulated that tumor cells arising in an organism may possess new antigenic variants that will be recognized by the immune system, leading to destruction of the tumor, often without any “clinical hint of its existence.” However, tumors form in immunocompetent individuals despite tumor immunosurveillance. Newer work provides evidence that as tumors develop and grow, they are “sculpted” by the immune system, which selects for the growth of nonimmunogenic tumor variants. Thus, the term “immunoediting” has been proposed to reflect this complex tumor–immune system dynamic. 21
Immunotherapy seeks to alter the balance of this tumor-immune system network, and the desired result is immune rejection of the tumor. There are a multitude of potential strategies to accomplish this goal, including nonspecific immunotherapy. This strategy involves the administration of either cytokines or microbial products that interact with cell surface receptors (such as TLRs) in order to effect an anti-tumor response. Nonspecific immunotherapy involving TLR agonists has shown utility in animal models of glioma, 14,15,40 as well as a modest benefit thus far in limited human clinical trials. 12,13 The presence of TLR4- and TLR9-positive cells in a peritumoral location may provide a rationale for future immunotherapeutic intervention with TLR agonists in canine patients.
Correlation of immune cell infiltration with outcome has been attempted in a number of studies examining human patients with brain tumors. Increased infiltration of lymphocytes into gliomas has been associated with an improved prognosis in a number of reports, 9,20,45 although other studies have failed to confirm this finding. 10 Some of this discrepancy likely relates to failure to adequately identify the varying subpopulations of lymphocytes present within such tumors. More recent studies suggest that populations of CD8+ cells may correlate with an anti-tumor response and improved outcome, whereas CD4+ cells and Tregs may contribute to an immunosuppressive environment and tumor progression. 63 Determining outcome in canine patients with brain tumors is difficult for a number of reasons, including the lack of an antemortem histologic diagnosis in most patients, nonuniform therapeutic protocols, variable owner tolerance for clinical signs and therapeutic side effects, and difficulties inherent in trying to interpret tumor histology at necropsy in light of previous treatments and progression of disease. As a result, correlation with outcome was not possible in this study, although future studies aimed at investigating a possible relationship between the antemortem immune response against the tumor and prognosis should be considered.
In conclusion, this is the first study focused on characterization of the immune response in naturally occurring canine brain tumors. Intracranial meningiomas in these dogs had substantial numbers of associated immune cells. Large numbers of CD18+ microglia and macrophages were noted surrounding and infiltrating the tumors, and a subset of these cells appeared to be CD11d+. Scattered macrophages at the tumor–brain interface were TLR4+ and TLR9+. The lymphocyte infiltrate consisted mainly of CD3+ T cells, and sparse numbers of CD79a+ B cells were noted in only a small subset of tumors. Smaller numbers of lymphocytes that were CD11d+, CD45RA+, or FoxP3+ were noted in a variable number of the meningiomas. Elucidation of the function of these cellular subsets and their effect on tumor behavior and ultimate prognosis awaits further study. These findings begin to reveal the complex pathophysiology of the immune response and its relationship to the invasive growth of these tumors, and further work may provide important prognostic information, as well as a basis for future immunotherapeutic trials.
Footnotes
Acknowledgement
The authors acknowledge Stan Dunston for assistance with preparation of the histological sections.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported in part by the Merck-Merial Veterinary Scholars Program, the American College of Veterinary Internal Medicine Foundation and by the Department of Clinical Sciences, North Carolina State University College of Veterinary Medicine.
