Abstract
Introduction: Pentoxifylline (PTX) reduces the levels of pro-inflammatory cytokines; however, its effects on immune system is not well understood. The aim of this study was to investigate the effect of PTX on T cells under inflammatory conditions in co-culture with THP-1-derived macrophages.
Methods: Toll-like receptor 4 (TLR4) and macrophage migration inhibitory factor (MIF) levels were measured after addition of PTX to lipopolysaccharide (LPS)-stimulated differentiated THP-1 cells. T cell viability and MIF levels were measured after PTX was added to prostaglandin E2 (PGE2)-stimulated Jurkat T-cell leukemia line. Co-culture was conducted to determine the effect of LPS-stimulated differentiated THP-1 cells that are affected by PTX on Jurkat cells. To prevent the direct effects of LPS and PTX on Jurkat cells, LPS and PTX were washed from THP-1 cells before co-culture. T cell viability and interleukin-2 (IL-2) levels were determined in Jurkat cells. Results: Increase in the MIF concentration and TLR4 expression level in differentiated THP-1 cells stimulated with LPS were reversed after PTX addition. However, PTX did not improve T cell viability in PGE2–stimulated Jurkat cells. Co-culturing Jurkat cell and LPS-stimulated differentiated THP-1 cells resulted in a decreased viability of T cells. The addition of PTX restored T cell viability to normal control levels and IL-2 expression level in Jurkat cells. Conclusion: LPS-stimulated THP-1-derived macrophages reduced the T cell viability under inflammation. However, PTX restored T cells viability and IL-2 back to normal levels. Therefore, the immunomodulatory action of PTX may be mediated by macrophage-T cell interactions.
Introduction
In the event of shock caused by major trauma, hemorrhage, or infection, many patients die despite the initial proper treatment.1–3 This is caused by an excessive hyper-inflammatory response and the occurrence of immunological paralysis resulting in the disturbance of homeostasis, leading to multiple organ dysfunction syndrome, sepsis, and other complications. In general, immune system has sufficient biological defenses against injury, infection, or unwanted biological invasions and preserves homeostasis in inflammatory syndromes. However, severe hyper-inflammatory conditions, caused by various triggers, can lead to many complications including, systemic inflammatory response syndrome, immune-paralysis, and compensatory anti-inflammatory response syndrome.4,5 In the case of various injuries, monocytes and neutrophils react in the early stages of the injury. Monocytes in the blood vessels migrate to the damaged tissue and differentiate into local macrophages. Antigens are captured by macrophages in peripheral tissues and processed to form major histocompatibility complex-peptide complexes.6,7 Because of antigen deposition and inflammation, macrophages begin to mature and express molecules that bind to and stimulate T cells in lymphoid tissues. After T blasts leave the blood at the original site of antigen deposition, molecular recognition occurs in the inflamed blood vessels. Macrophage play a key role in the recognition of bacterial components during infection. Toll-like receptor (TLR) activation promotes the secretion of inflammatory cytokines and chemokines, and T-cells play important roles in cell-mediated immunity.8,9 However, there are few studies that show the stimulation effect and the hyper-inflammation control of macrophages on T cells.
Pentoxifylline (PTX), a methylxanthine derivative and nonspecific phosphodiesterase inhibitor, is commonly used as a vasodilator and anticoagulant, which decreases the resistance of vessels, blood viscosity, and platelet aggregation, and augments blood flow in ischemic tissue. 10 A crucial effect of PTX is its suppression of inflammatory response through blockage of extracellular signal-regulated kinase phosphorylation, especially the downregulation of tumor necrosis factor-α (TNF-α). 11 Although studies on the mechanism of the PTX have been conducted, the effect of PTX on the immune system remains unclear. The aim of this study was to investigate the effects of PTX on lipopolysaccharide (LPS)-induced TLR4 expression in differentiated THP-1-macrophage-like cells and to examine the effects of PTX on Jurkat leukemia T-cells in co-culture with hyperinflammatory differentiated THP-1 cells.
Materials and methods
Cell culture, cell stimulation and co-culture
A human acute monocytic leukemic cell line (THP-1) (ATCC TIB-202, Manassas, VA, USA) and a lymphoblast cell line (Jurkat cells clone E6-1) (ATCC TIB-152, Manassas, VA, USA) were maintained in RPMI-1640 (Invitrogen, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum, 2 mM glutamine, 10 mM HEPES, and 100 U/mL penicillin/streptomycin at 37°C in 5% CO2 incubator. The cells were cultured at a concentration of 5 × 105 cells/mL. Cell viability was >99% as determined by trypan blue dye exclusion. THP-1 cells were incubated with Phorbol-12-myristate-13-acetate (PMA) for 3 days to induce THP-1 cell differentiation with macrophage properties. The cells were added to 24-well plates at a density of 1 × 104 cells/mL, divided into control, LPS (1 μg/mL) (Sigma-Aldrich Co., St. Louis, MO, USA), and PTX (0.1 mM, 1 mM, 2 mM) (Sigma-Aldrich Co., St. Louis, MO, USA) groups, and incubated for 8 h. The macrophage migration inhibitory factor (MIF) concentration was determined using Enzyme-linked immunosorbent assay (ELISA). Toll-like receptor 4 protein levels were detected using flow cytometry technique. Toll-like receptor 4 mRNA expression was detected using a real-time polymerase chain reaction (RT-PCR). Jurkat cells (2 × 106 cells/mL) were incubated into 96-well plates, divided into control, PGE2 (1 μg/mL) (Sigma-Aldrich Co., St. Louis, MO, USA), and PTX (0.1 mM, 1 mM, 2 mM) (Sigma-Aldrich Co., St. Louis, MO, USA) groups, and incubated for 12 h. T cell viability was measured by the MTT assay, and MIF concentration was determined using specific ELISA. For co-culture experiments, differentiated THP-1 cells were plated at a density of 1 × 104 cells/mL × 0.5 mL in 24-well plates and stimulated with LPS (1 μg/mL) in the presence or absence of PTX (2 mM). After 8 h incubation, the cells were washed twice in phosphate buffered saline (PBS) and resuspended in fresh medium. While differentiated THP-1 cells were transferred to the lower layer of the transwell plate, Jurkat cells were placed on the upper layer at a concentration of 1 × 104 cells/mL × 0.5 mL and 2 × 106 cells/mL × 0.5 mL respectively, and co-culture was maintained for 12 h. T cell viability in Jurkat cells was measured using MTT assay, and the expression of interleukin-2 (IL-2) was analyzed employing RT-PCR and western blotting. This study was approved the Institutional Review Board of Korea University Guro Hospital (approval number: 2018GR0022).
Enzyme-linked immunosorbent assay for MIF
The MIF concentration in the culture supernatants was measured using ELISA (LotP174268, R&D Systems). Optical density at 450 nm was measured by an automated microplate reader (Bio-Rad Laboratories Inc., Hercules, CA, USA). A standard curve was generated by plotting the optical density versus the log of MIF concentration. Experiments were performed in triplicate in three consecutive wells of 96-well plates. Each experiment was repeated at least 12 times.
Flow cytometry analysis for TLR4
The expression of cell surface TLR4 was determined using flow cytometry. Macrophages were harvested by centrifugation. Cells were divided into control, LPS-stimulated, and LPS-stimulated cells treated with 0.1 mM, 1 mM, and 2 mM of PTX. The assay was conducted according to the manufacturer’s protocol (eBioscience, San Diego, CA, USA) for the measurement of cell surface TLR4 and analyzed by flow cytometry using a Cytomics FC 500 (Beckman Coulter, Brea, CA, USA) and CXP software (Beckman Coulter).
Real-time polymerase chain reaction for TLR4, IL-2 expression
For the measurement of TLR4 and IL-2 mRNA expression, total RNA was extracted using the RNeasy Mini Kit (Qiagen, Hilden, Germany), which was then reverse transcribed using a high-capacity complementary DNA (cDNA) reverse transcription kit (Applied Biosystems, Foster City, CA, USA). Real-time qPCR was performed on a 7500 Real-Time PCR system (Applied Biosystems, Foster City, CA, USA) using TaqMan® Gene Expression Master Mix (Applied Biosystems, Foster City). Each reaction contained cDNA that was amplified with Inventoried TaqMan Gene Expression Assay products (TLR4 assay ID: Hs00152939_ml; IL-2 assay ID: Hs00174114_ml; and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) assay ID: Hs02758991_gl). The threshold cycle (Ct) values were normalized to those of GAPDH, and the amount of target mRNA relative to the control was calculated using the 2-△△Ct method. Relative mRNA expression was expressed as fold-change relative to the average of GAPDH expression.
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide viability assay
MTT assay is widely used to assess the viability and metabolic state of cells. The MTT colorimetric monocyte-mediated cytotoxicity assay is based on the ability of living cells to reduce MTT into formazan by mitochondrial succinate dehydrogenase in viable cells. After treatment under different incubation conditions, Jurkat cells were placed in 96-well flat-bottom tissue culture plates at a final concentration of 2 × 106 cells/mL. After 12 h of incubation at 37°C, the probability of survival of Jurkat cells was measured using the MTT viability assay (ATCC, Manassas, VA, USA).
Western blot analysis for IL-2 expression
The cells were centrifuged for 10 min and washed twice in cold PBS. Cell pellets were suspended in 10 μL of pro-prepTM protein extraction buffer. The samples were kept on ice for 10 min and centrifugated at 4 degrees C for 15 min at 3000 × g. The total protein concentration was determined using the Bradford method (Sigma Co. Louis, MO, USA). IL-2 protein expression was quantified using western blot analysis. Protein samples (20 μg) were fractionated in 15% sodium dodecyl sulfate-polyacrylamide gel (Bio-Rad Laboratories Inc.) and transferred onto a nitrocellulose membrane. Membranes were blocked for 1 h in 5% skim milk (Bio-Rad Co.) and then incubated with the primary antibody anti-human IL-2 (1:500; R&D System). After washing, the membranes were incubated with a 1:1000 dilution of horseradish peroxidase-labeled anti-rabbit antibody (R&D System) as the secondary antibody. Proteins were detected using an ECL chemiluminescence kit (Cyanagen).
Statistical analysis
Paired t-tests and post hoc tests were used for statistical analysis using the SPSS software package (version 13.0, IBM-SPSS Inc., Chicago, Illinois, USA). A p-value < .05 was considered statistically significant.
Results
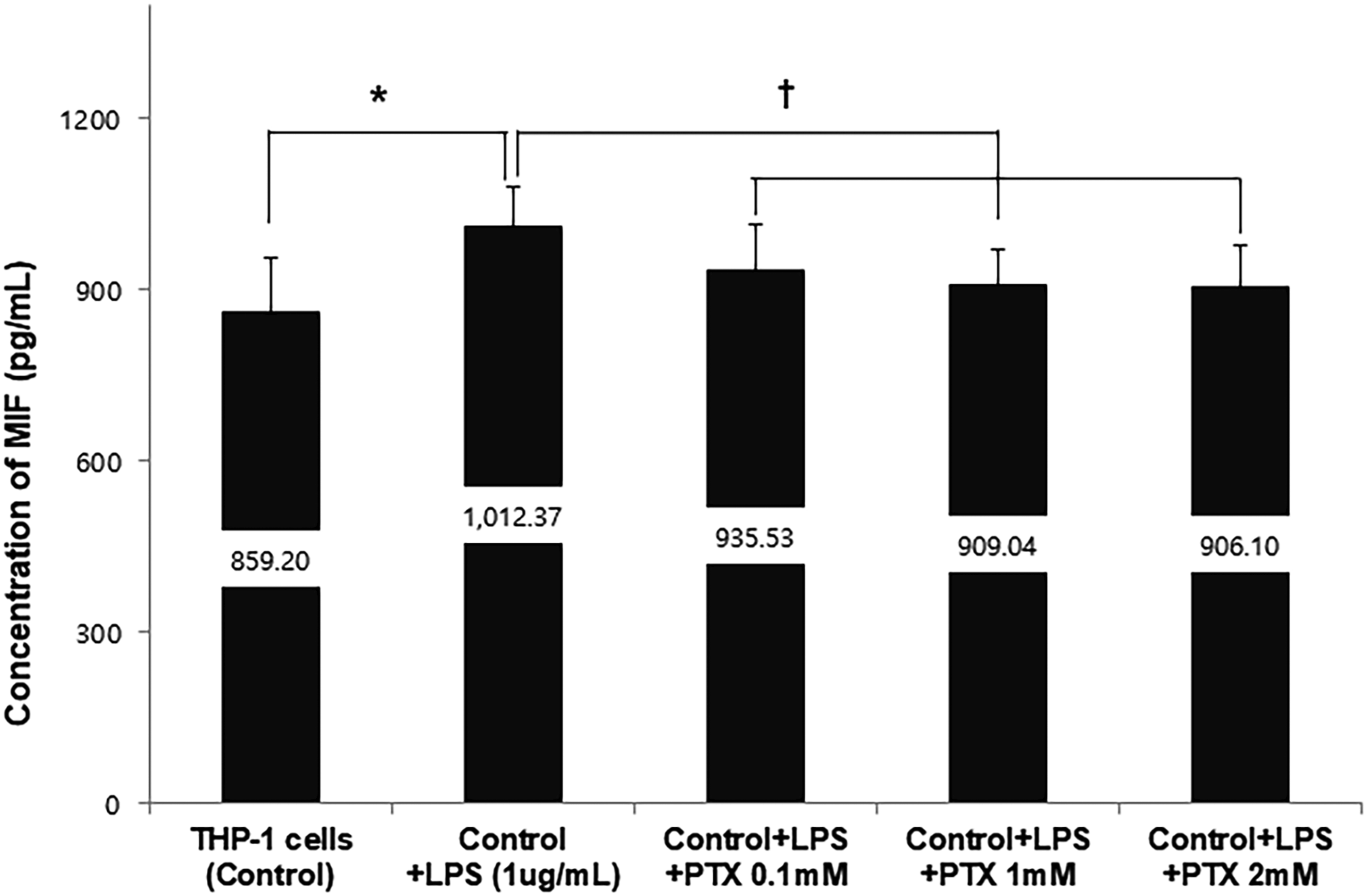
The effect of PTX on LPS-induced TLR4 and MIF in differentiated THP-1 cells
Toll-like receptor 4 mRNA expression increased 6 and 8 h after LPS stimulation in differentiated THP-1 cells. The mRNA expression was significantly higher after 8 h of LPS stimulation than after 2, 4, and 6 h. Moreover, TLR4 mRNA expression was significantly higher after 6 h than after 2 h (Figure 1). The proportion of TLR4 positive cells increased in LPS-stimulated differentiated THP-1 cells compared to that in the control group. After 8 h of incubation with added PTX, the proportion of TLR4 positive cells in LPS-stimulated differentiated THP-1 cells further decreased as the PTX concentration increased as shown by flow cytometry analyses (Figure 2). Lipopolysaccharide increased the concentration of MIF in differentiated THP-1 cells; PTX reduced the concentration of MIF in differentiated THP-1 cells stimulated with LPS, as determined by ELISA (Figure 3). The effect of LPS on TLR4 mRNA expression over time in differentiated THP-1 cells. TLR4 expression in LPS-stimulated differentiated THP-1 cells increased at 6 h and 8 h as determined by RT-PCR. The bar graph shows the TLR4 mRNA expression ratio (TLR4/GAPDH ratio) from repeated measurements (n = 12). Data are presented as mean ± SD. *p = .005, †p < .001, ‡p = .01. TLR4; toll like receptor 4, LPS; lipopolysaccharide, PTX; pentoxifylline. The effect of PTX on TLR4 surface expression in LPS-stimulated differentiated THP-1 cells. (A). The expression of cell surface TLR4 was determined using flow cytometry analysis. Cells were divided into control, LPS stimulated cells, and LPS stimulated cell with PTX (0.1 mM, 1 mM, and 2 mM). After staining, the cells were analyzed by flow cytometry using Cytomics FC 500 (Beckman Coulter, USA) and CXP software (Beckman Coulter, USA). (B). TLR4 expression was 0% in the control, 49.3 ± 12.2 in the LPS stimulated cells, 44.3 ± 11.8 at 0.1 mM PTX, 35.9 ± 11.3 at 1 mM PTX, 27.75 ± 11.6 at 2 mM PTX. Data are presented as mean ± SD. *p = .01, †p = .045, ‡p =.026, #p = .034. TLR4; toll like receptor 4, LPS; lipopolysaccharide, PTX; pentoxifylline. The effect of PTX on MIF concentration in LPS-stimulated differentiated THP-1 cells. LPS increased the concentration of MIF in differentiated THP-1 cells; however, PTX restored the increased concentration of MIF in differentiated THP-1 cells stimulated with LPS, as revealed by ELISA. The bar graph shows the concentration of MIF from repeated measurements (n = 12). Data are presented as mean ± SD. *p = .015, †p < .05. LPS; lipopolysaccharide, MIF; macrophage migration inhibiting factor, PTX; pentoxifylline.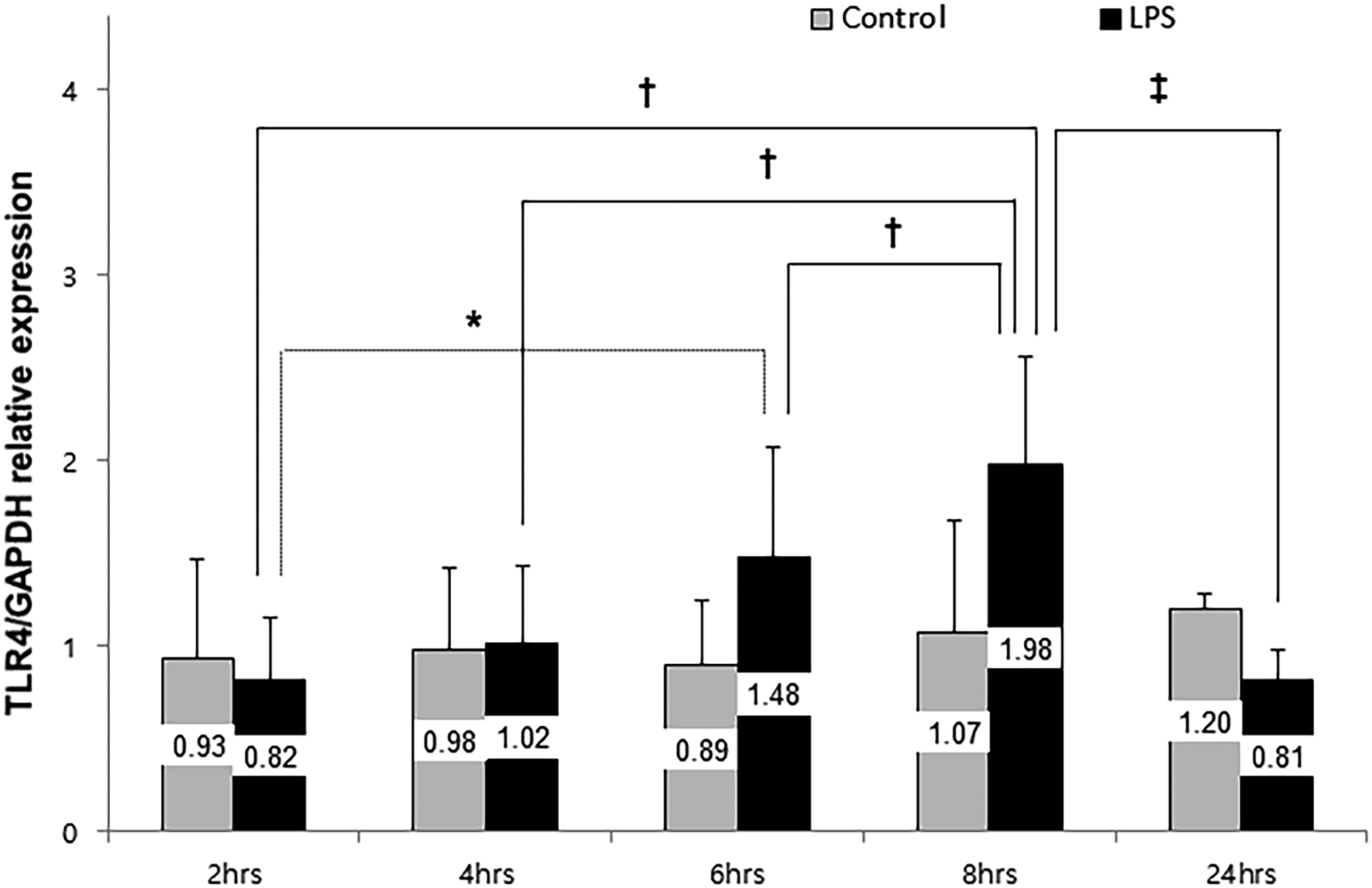

The effect of PTX on PGE2-induced T cell viability and MIF in Jurkat cells
PGE2 decreased T cell viability in Jurkat cells and PTX did not restore the cells’ viability estimated using MTT assay (Figure 4). PGE2 also decreased the concentration of MIF in Jurkat cells; however, PTX did not restore normal levels of MIF, as determined by ELISA (Figure 5). The effect of PTX on cell viability in PGE2-stimulated Jurkat cells. PGE2 decreased T cell viability in Jurkat cells. PTX did not restore normal T cell viability measured using MTT assay. The bar graph shows the cell viability from repeated measurements (n = 12). Data are presented as mean ± SD. *p = .027. PGE2; Prostaglandin E2, PTX; pentoxifylline. The effect of PTX on MIF concentration in PGE2-stimulated Jurkat cells. PGE2 decreased the concentration of MIF in Jurkat cells and addition of PTX did not restore normal PGE2-induced concentration of MIF determined using the ELISA method. The bar graph shows the concentration of MIF from repeated measurements (n = 12). Data are presented as mean ± SD. *p = .045, †p < .05. PGE2; prostaglandin E2, MIF; macrophage migration inhibiting factor, PTX; pentoxifylline.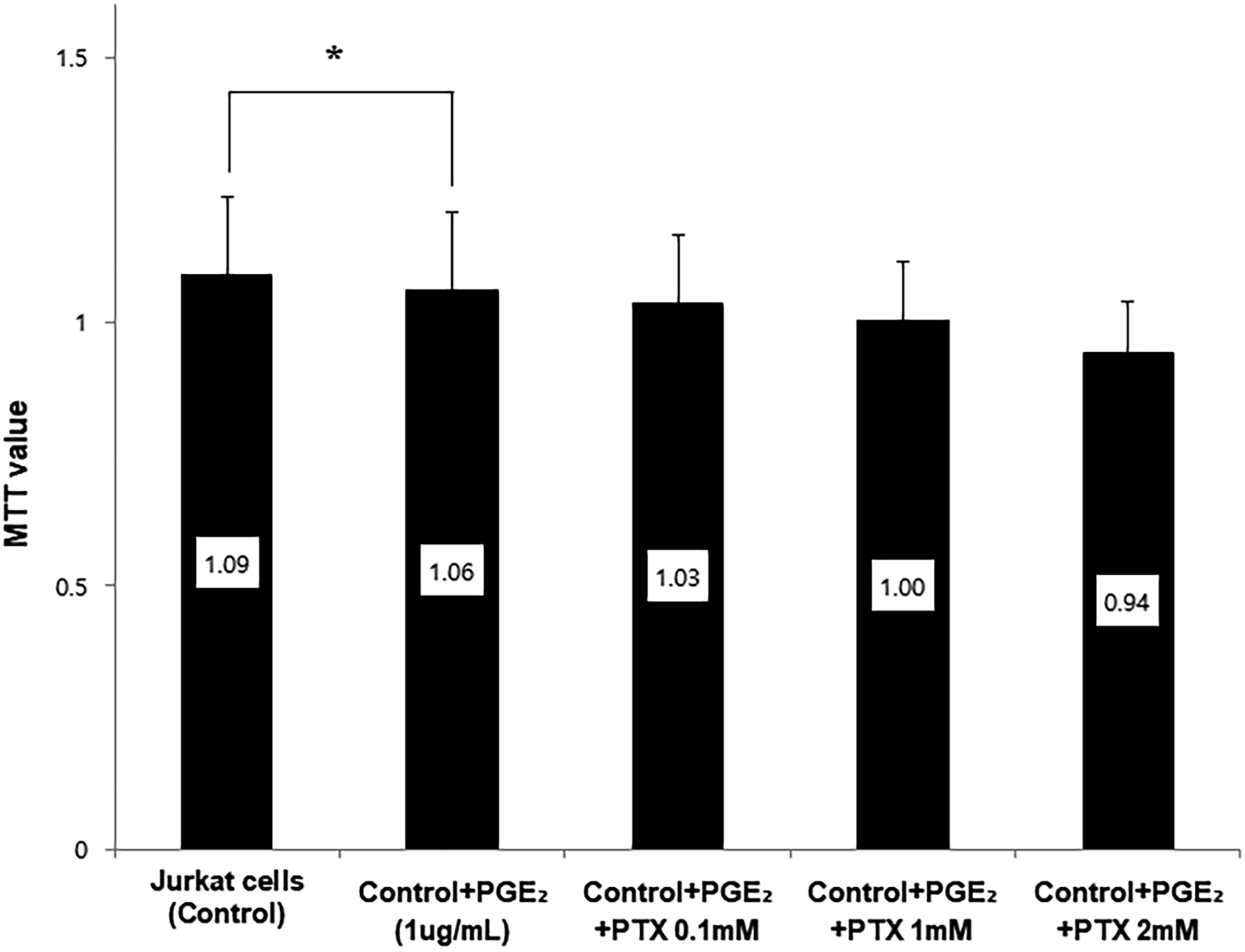

The effect of PTX on Jurkat cells co-cultured with hyper-inflammatory differentiated THP-1 cells
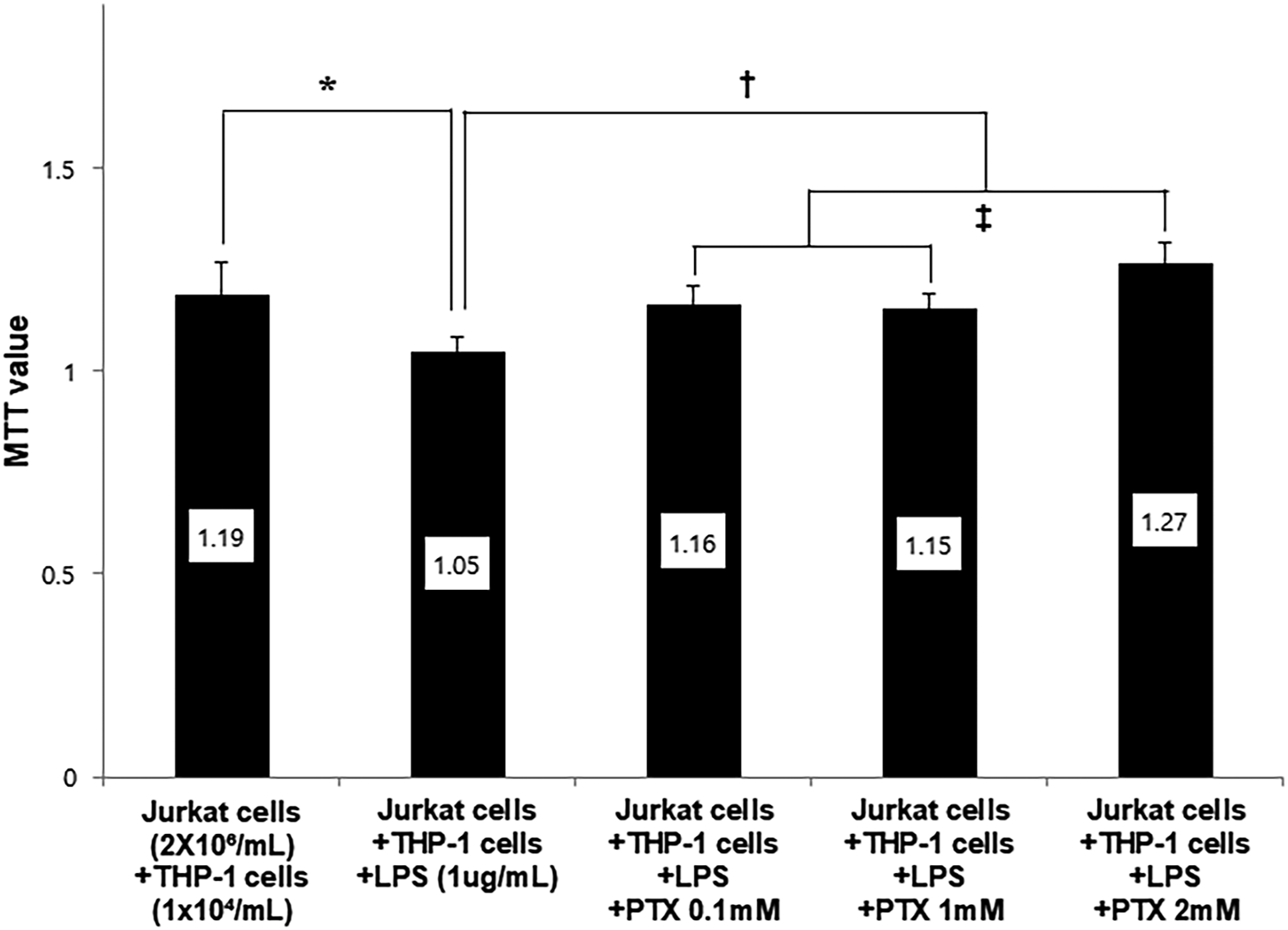
To determine the effect of differentiated THP-1 cells on Jurkat cells, the cells were co-cultured with Jurkat counterparts at various concentrations. Jurkat cell viability was not affected. Accordingly, the ratio of differentiated THP-1 cells to Jurkat cells was 1 × 104 cells/mL to 2 × 106 cells/mL (Figure 6). The percentage of T cell viability was reduced in Jurkat cells co-cultured with LPS-stimulated differentiated THP-1 cells compared to non-stimulated cells. The addition PTX restored the T cell viability to normal levels and increased the most in the presence of 2 mM PTX (p < .05) (Figure 7). Similarly, IL-2 mRNA expression was reduced in co-cultured Jurkat cells with LPS-stimulated cells. Addition of PTX to the same co-culture setup significantly restored normal levels of IL-2 mRNA (p < .05) (Figure 8). IL-2 expression level in western blot experiment was similar to that of PCR findings (p < .05) (Figure 9). The changes in cell viability of Jurkat cells according to THP-1 cells concentration changes. To determine the appropriate ratio between differentiated THP-1 cells and Jurkat cells for the co-cultue experiments, various co-culture concentrations of THP-1 cells were employed. The mean MTT values (Jurkat cell density; 2 × 106 cells/mL) were .87 ± .08 in the control group, .70 ± .13; .74 ± .14; .73 ± .20; .68 ± .12 in Jurkat cells co-cultured with differentiated THP-1 cells (density; 1 × 104 cells/mL; 5 × 104 cells/mL; 1 × 105 cells/mL; 5 × 105 cells/mL). After conducting various combinations of co-culture experiments, the optimal ratio determined was 1 × 104 cells/mL differentiated THP-1 cells to 2 × 106 cells/mL Jurkat cells. The bar graph shows the concentration of MTT value from repeated measurements (n = 12). Data are presented as mean ± SD. *p < .05. The effect of PTX on co-cultured Jurkat cell viability in LPS-stimulated differentiated THP-1 cells. The mean MTT value in Jurkat cells under non-stimulated differentiated THP-1 cells was 1.19 ± .08 in the control group, 1.05 ± .03 in Jurkat cells under the differentiated THP-1 cells stimulated with LPS, the mean MTT value in Jurkat cells was 1.16 ± .05, 1.15± .04, 1.27 ± .05 under the differentiated THP-1 cells stimulated with LPS and 0.1 mM, 1 mM, 2 mM PTX. Therefore, T cell viability was reduced in Jurkat cells co-cultured in LPS-stimulated differentiated THP-1 cells than in non-stimulated cells. Addition of PTX restored T cell viability of Jurkat cells to normal levels and increased most in the presence of 2 mM concentration PTX. Data are presented as mean ± SD. *p = .006,†p < .05, ‡p = .005. LPS; lipopolysaccharide, PTX; pentoxifylline. The effect of PTX on IL-2 mRNA expression in co-cultured conditions. IL-2 mRNA expression was measured using RT-PCR. The IL-2 mRNA expression decreased in Jurkat cells co-cultured with differentiated THP-1 cells stimulated by LPS, and was restored to normal levels when PTX was added to the experimental setup. The bar graph shows the IL-2 mRNA expression from repeated measurements. Data are presented as mean ± SD *p = .006, †p = .017. IL-2; Interleukin 2, LPS; lipopolysaccharide, PTX; pentoxifylline. The effect of PTX on IL-2/Actin expression in co-cultured conditions. In the western blot experiments, the mean of IL-2 expression ratio (IL-2/Actin ratios) in Jurkat cells co-cultured with differentiated THP-1 cells were .97 ± .52 in the control group, .88 ± .31 in Jurkat cells co-cultured in the differentiated THP-1 cells stimulated with LPS, 1.10 ± 0.42 in Jurkat cells co-cultured in the differentiated THP-1 cells stimulated with LPS and 2 mM PTX. The bar graph shows the IL-2 expression levels from repeated measurements. Data are presented as mean ± SD *p = .026, †p = .010. IL-2; Interleukin 2, LPS; lipopolysaccharide, PTX; pentoxifylline.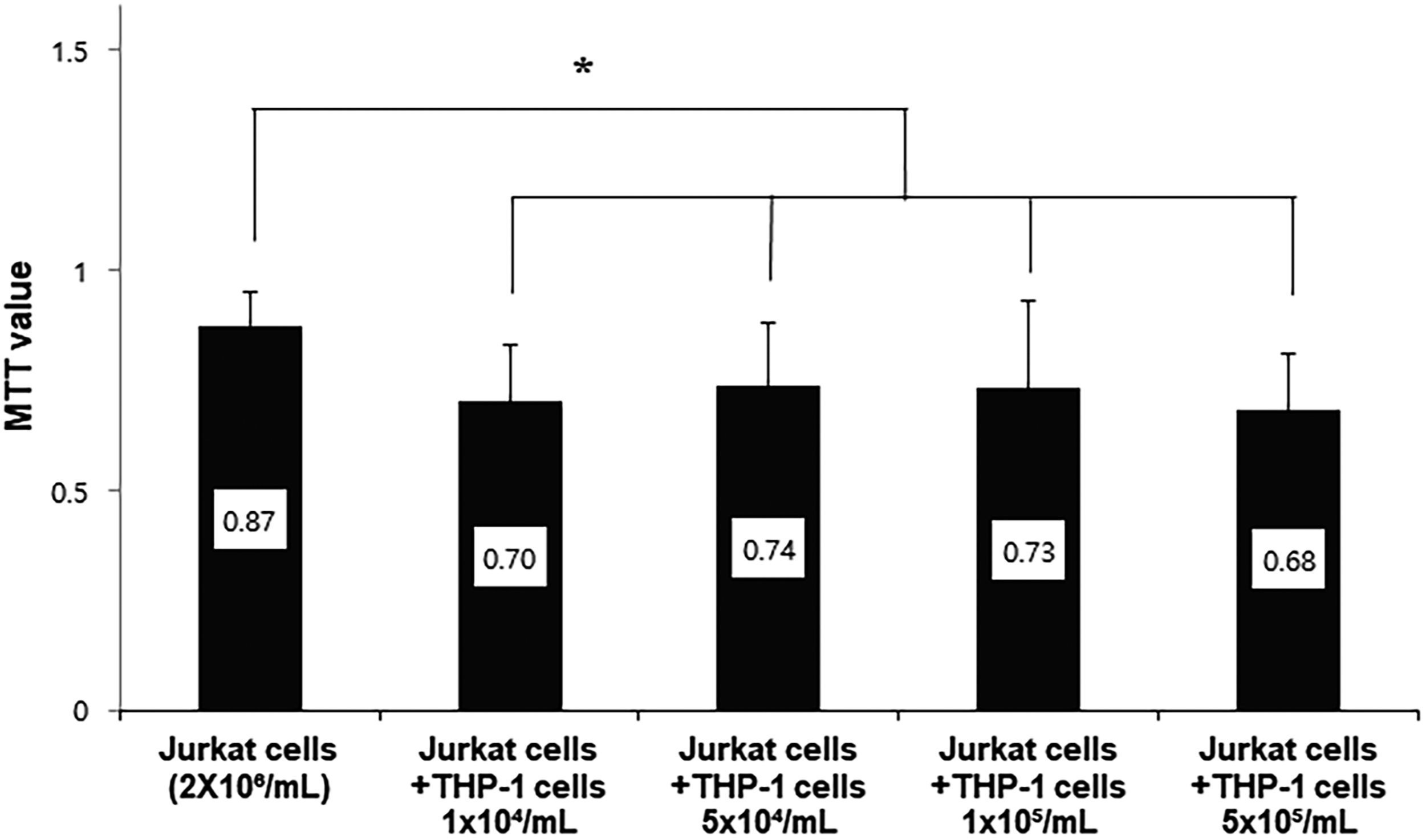


Discussion
Immune reactions caused by infection and trauma can cause severe complications such as sepsis and multiple organ failure despite proper treatment, which can have a significant impact on the patient’s prognosis. These immune responses are caused by macrophages and T lymphocytes, which play an important role in innate immune responses, and cytokines and chemokines caused by the interaction of these cells.1,3,12,13 A human acute monocytic leukemic cell line (THP-1 cell) is differentiated by PMA into macrophages and a lymphoblast cell line (Jurkat cell) was used as a model to study the crucial role of T lymphocytes.4,5 Therefore, this study was conducted using THP-1 cells and Jurkat cells. This study is meaningful not only to confirm the action of PTX in macrophages and T lymphocytes but also to investigate the effects of PTX on T lymphocytes with hyper-inflammatory macrophages using co-culture. To the best of our knowledge, this is the first study to confirm the immunomodulatory effect of PTX through co-culture.
Toll-like receptor 4 is expressed on the surface of monocytes and macrophages by LPS and activates NF-κB and ERK/MAPK pathways to secrete cytokines like TNF-α, IL-1β, IL-6, IL-10, etc.3–5,14 Migration inhibitory factor is secreted into the bloodstream during the process in which TLR recognizes bacteria or immune complexes to produce NF-κB, which in turn activates CD44/CD74 on the cell surface, thereby upregulating the expression of TLR on the cell surface and activation of NF-kB in the cell.7,8 Also, MIF inhibits p53-mediated apoptosis, promotes cell proliferation, and overrides the anti-inflammatory effects of glucocorticoids. 15 Therefore, MIF plays an important role in sepsis by controlling inflammatory reactions, including the activation of various cytokines in macrophages, neutrophils, and T lymphocytes.12,13 As is known, in this study, when LPS was added to THP-1 cells, the expression of TLR4 increased and the MIF level also increased. Pentoxifylline downregulates the production of various inflammatory cytokines (TNF and IL-6) and anti-inflammatory cytokines (IL-10) produced during infection, bleeding, and shock. Also, PTX is known to inhibit the activation of NF-kB, thus reducing TLR expression and TLR induced pro-inflammatory cytokine.16–19 In this study, similar to other studies, PTX decreased TLR4 expression in LPS-stimulated THP-1 cells. Pentoxifylline also reduced the MIF level in this condition. Although the effect of PTX on MIF is not well known yet, it is thought that reducing MIF level is associated with the mechanism of PTX that reduce NF-kB, but further study is needed. The relationship between MIF and T cell proliferation was also established. When T-cell proliferation was reduced by PGE2 which inhibits T lymphocyte activity, the MIF concentration was decreased. This indicated weakened immune function along with the reduction in T cell proliferation, signifying immune paralysis.20,21 Cyclic adenosine monophosphate (cAMP) is a potent regulatory factor that inhibits the activation of T lymphocytes, which inhibits the expression of T-cell receptor (TCR) needed for the formation and proliferation of cytokines. Pentoxifylline is known to inhibit the activation of T lymphocytes by inhibiting the decomposition of this cAMP.22,23 In this study, PTX did not decrease the proliferation of T lymphocytes in PGE2-stimulated T lymphocyte, but decreased MIF level.
The two cell types were co-cultured to determine the effects of macrophages on T lymphocytes. In THP-1 cells stimulated by LPS, the T lymphocyte counts decreased compared to the non-stimulated cells group, indicating that macrophages in hyper-inflammatory conditions could induce a decrease in T lymphocyte numbers. As with this study, several studies have shown that macrophages activated by the host defense mechanism by inflammation inhibit the proliferation of T lymphocytes. 9 On the other hands, MTT value of co-cultured T lymphocytes with THP-1 cells pretreated with PTX and LPS showed PTX restored the viability of T lymphocyte to the baseline level. Immature T lymphocytes are differentiated into CD4+ T lymphocytes and CD8+ T lymphocytes mainly by IL-2.24,25 Also, secreted IL-2 promotes differentiation of T-lymph cells by upregulating the activation of immature CD4+ T lymphocytes.12,13,22 Therefore, it was considered that the change in T-lymphocytes in co-culture would be related to IL-2, and the IL-2 level was measured. Similar to the viability of the T lymphocytes measured by MTT, the IL-2 level was reduced in the co-culture of the T lymphocytes and LPS stimulated THP-1 cells. Also, when co-cultured the T lymphocytes with THP-1 cells pretreated with PTX and LPS, The IL-2 level was restored to the baseline level like the MTT value in the same situation. Since IL-2 is known to increase immunity by inducing T cell proliferation, 26 the supplement of PTX to LPS-stimulated macrophages could be used to increase T cell proliferation and hence, improving immunity.
This study had several limitations. First, studies on the intercellular effects of macrophages and T lymphocytes, excluding neutrophils, cannot represent the effects of PTX during injuries, infection, and hemorrhage. However, it is meaningful to use macrophages that react in the early stages of damage, which will be a cornerstone for future studies. Second, in the co-culture of LPS-stimulated macrophages and T lymphocytes, PTX restored T cell proliferation to normal levels. However, since immune status cannot be determined just by the viability of T lymphocytes, further studies are needed.
Third, in our study, 0.1 mM, 1 mM, and 2 mM PTX were used. If the high concentration of PTX is used in vivo, it is expected to exhibit various biological effects, so further clinical research will be needed. Fourth, Generally, the number of lymphocytes in normal healthy people is approximately 7–20 times that of monocytes, but in the co-culture setting, the Jurkat cells were set at 2 × 106/mL and THP-1 cells were set at 1 × 104/mL Since our experiment showed similar MTT values of Jurkat cells in THP-1 cells at 1 × 105/mL and 5 × 105/mL, it is thought that it did not significantly affect the results of the experiment, but further research is needed. Finally, in vivo, since several cells interact in addition to macrophage and T-lymphocyte, further research is needed to evaluate the effect of PTX.
Conclusions
The occurrence of hyperinflammatory and immune paralysis conditions can lead to death due to sepsis and multiple organ failure. After infection, excessive immunological paralysis may be managed by altering the communication between macrophage and T cell. The intercellular effects, as shown in our study, and other unknown effects coordinating hyper-inflammatory conditions may reduce immune paralysis. Therefore, PTX may help maintain immunity by maintaining the number of T lymphocytes as a relief from hyperinflammation, however, further studies are needed to confirm these findings.
Footnotes
Acknowledgements
We are grateful to our colleague Qiuyu C, Bang IY, and Korean shock society members for participation in the study and helpful discussion. We appreciate the efforts of Song DJ for reviewing statistical analyses.
Authors’ contributions
SJP, YDC, and SHC conceived this idea. SHC, JYK, and WJK designed the experiments and interpreted the data. SJP, HJC, KHK, and SHC performed experiments. SJP and SHC wrote the manuscript. All authors have read and approved the final manuscript.
Declaration of conflicting interests
The authors (s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (2018R1A2B6004283) and partially supported by a Korea University Grant.
Ethics approval
Ethical approval for this study was obtained from Korea University Guro hospital institutional review board (approval number/2018GR0022).
Data sharing
The data and materials used in this study are available from the corresponding author upon reasonable request.
