Abstract
Inclusions of periodic acid-Schiff-positive, amylase resistant material were found within skeletal muscle fibers adjacent to an osteosarcoma in the proximal femur of an 8-year-old intact female Cocker Spaniel dog (dog No. 1) and adjacent to a synovial cell sarcoma of the stifle joint in a 7-year-old spayed female Bouvier des Flandres dog (dog No. 2). Inclusions were pale blue-gray with hematoxylin and eosin stain and formed irregular inclusions, replacing up to approximately 80% of the fiber diameter. Inclusions from dog No. 2 were of non-membrane-bound granular to filamentous material that occasionally formed discrete, elongate electrondense masses. The features of these inclusions were similar to those of materials previously described as complex polysaccharide, polyglucosan bodies, amylopectin, and Lafora bodies. Evidence for a generalized metabolic disorder was not found in these two dogs, suggesting that storage of complex polysaccharide can occur as a relatively nonspecific response to metabolic alterations in skeletal muscle in a variety of conditions.
Keywords
Inclusions of material that is periodic acid–Schiff (PAS) positive and amylase resistant have been described in skeletal muscles of humans and animals with various metabolic disorders.1–9,11,12,14,15 Names given to this material include complex polysaccharide, polyglucosan bodies, Lafora bodies, and amylopectin. Depending on the type of disorder, inclusions may be confined to skeletal muscle,2,6,7,11 involve only skeletal and cardiac muscle,2,11 or occur within cells of numerous organs.2,4,9,11 In the central nervous system, Lafora bodies and related corpora amylacea are incidental findings within neurons of aged humans and dogs.10 Similar age-related inclusions do not occur in other organs. Lafora bodies have also been described in the central nervous system of dogs with neurologic disease10 and in the skeletal muscle fibers and peripheral nerves of dogs with familial myoclonic epilepsy.3 Inclusions of complex polysaccharide occur within skeletal muscle of dogs with muscle-type phosphofructokinase (PFK) deficiency, also known as glycogenosis type IV. Clinical signs of hemolytic disease predominate in dogs with PFK deficiency, and there are minimal signs of neuromuscular dysfunction. The development of skeletal muscle inclusions in these dogs appears to be a function of age.6 Here, we describe two dogs with no apparent metabolic disease in which intramyofiber inclusions of complex polysaccharide were localized to skeletal muscle fibers adjacent to nonmyogenic malignant mesenchymal tumors.
An 8-year-old intact female Cocker Spaniel dog (dog No. 1) was examined for left hind leg lameness. Radiographs revealed a fracture of the proximal femur with bony lysis and proliferation suggestive of neoplasia, and examination of biopsy samples confirmed the presence of osteosarcoma. Myofibers in adjacent skeletal muscle exhibited marked fiber size variation due primarily to fiber atrophy and were often separated by edema and loose connective tissue. Hematoxylin and eosin (HE) staining revealed scattered fibers containing single to multiple inclusions of blue-gray hyaline material involving multiple contiguous fiber segments. This material occurred most often in the subsarcolemmal regions of the muscle fiber. These inclusions were intensely PAS positive and amylase resistant (Fig. 1). The dog had no history of exercise intolerance or other evidence of neuromuscular or hemolytic disease. Blood samples for PFK analysis were not obtained. The dog died due to metastatic osteosarcoma within 3 months of diagnosis.
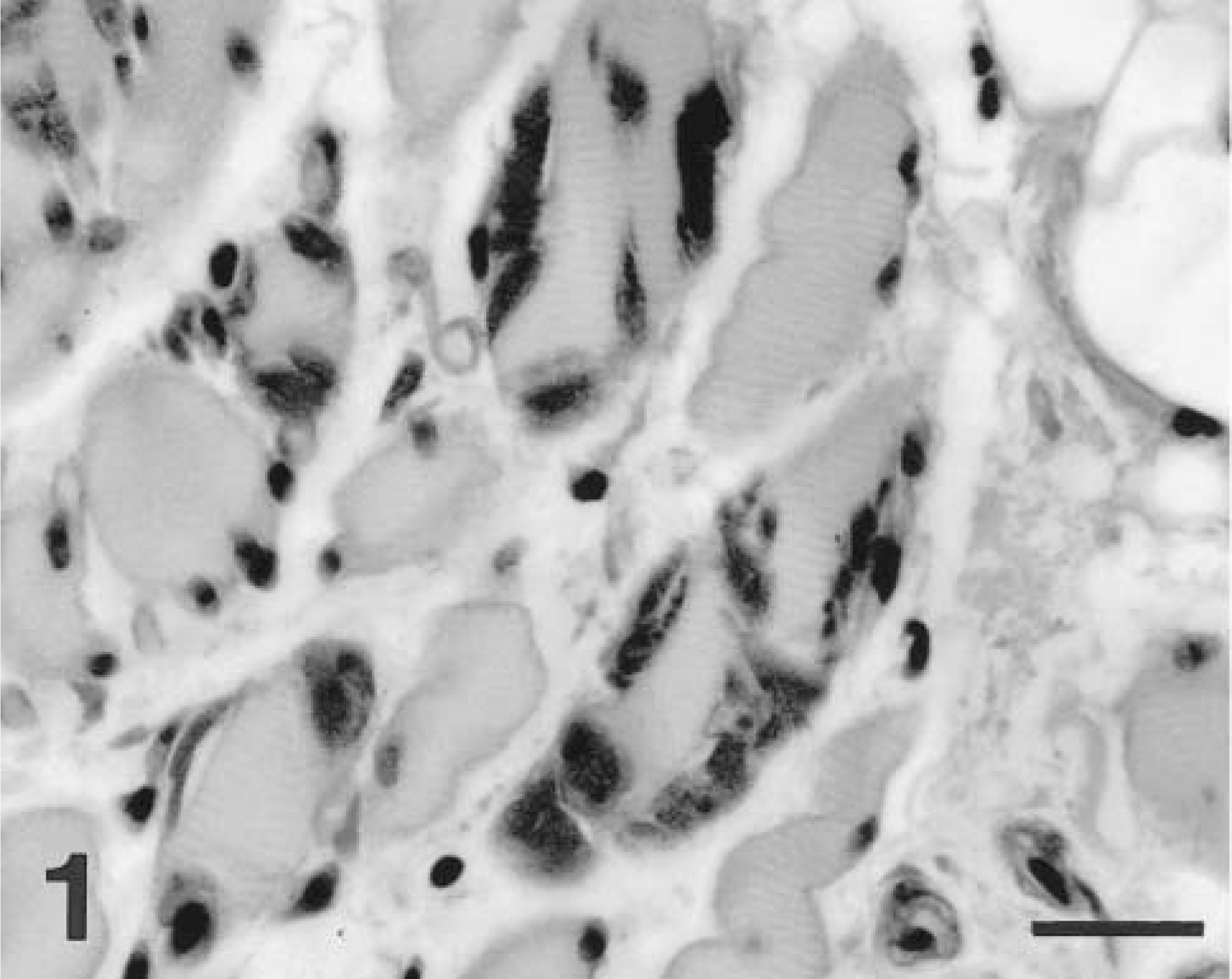
Skeletal muscle; dog No. 1. Myofibers adjacent to the osteosarcoma are atrophic, and many fibers contain one to several subsarcolemmal to intracytoplasmic inclusions of PAS-positive, amylase-resistant material. PAS with amylase digestion. Bar = 27 μm.
A 7-year-old spayed female Bouvier des Flandres dog (dog No. 2) was examined for progressive unilateral hind limb lameness and painful swelling of the stifle joint. The leg was amputated, and a neoplasm involving the joint capsule and synovium and with extension into the adjacent musculature was histopathologically diagnosed as a synovial cell sarcoma. There was marked fiber size variation in adjacent skeletal muscle, with both fiber atrophy and fiber hypertrophy, particularly in regions immediately adjacent to the tumor. There were areas of infiltration of muscle by loose connective tissue and adipose tissue. Many skeletal muscle fibers contained one to several blue-gray hyaline inclusions. This material was most often present in the subsarcolemmal region but in some cases involved almost the entire fiber diameter. Large inclusions involved multiple contiguous fiber segments. Inclusions often appeared to be composed of “packets” of material stacked within the cytoplasm (Fig. 2). This material was intensely PAS positive and amylase resistant. Skeletal muscle in areas more distant from the tumor exhibited mild myopathic changes but did not contain inclusions.
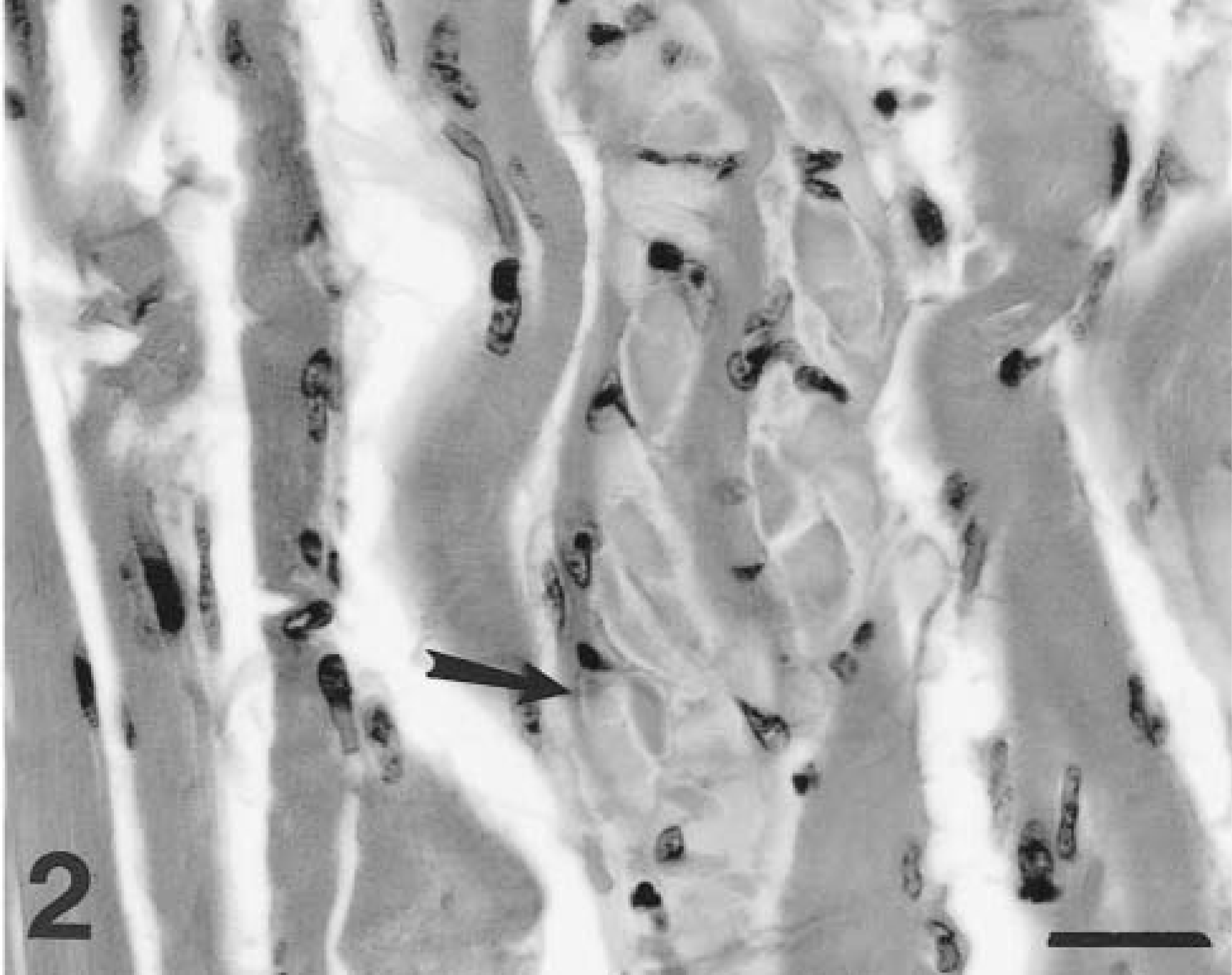
Skeletal muscle; dog No. 2. A myofiber adjacent to the synovial cell sarcoma contains multiple stacked packets of pale hyaline material (arrow). HE. Bar = 27 μm.
Samples of affected muscle were obtained from the paraffin block, rehydrated to water, postfixed in osmium tetroxide, and prepared for routine transmission electron microscopy. Ultrastructural examination revealed intracytoplasmic inclusions composed of non-membrane-bound masses of admixed granular and branching filamentous material. Material was often densely packed centrally, with more loosely arranged material on the periphery. Less commonly, this material formed elongate electron-dense structures surrounded by more loosely arranged granular to filamentous material (Fig. 3). Filamentous material admixed with apparently normal mitochondria was also seen.
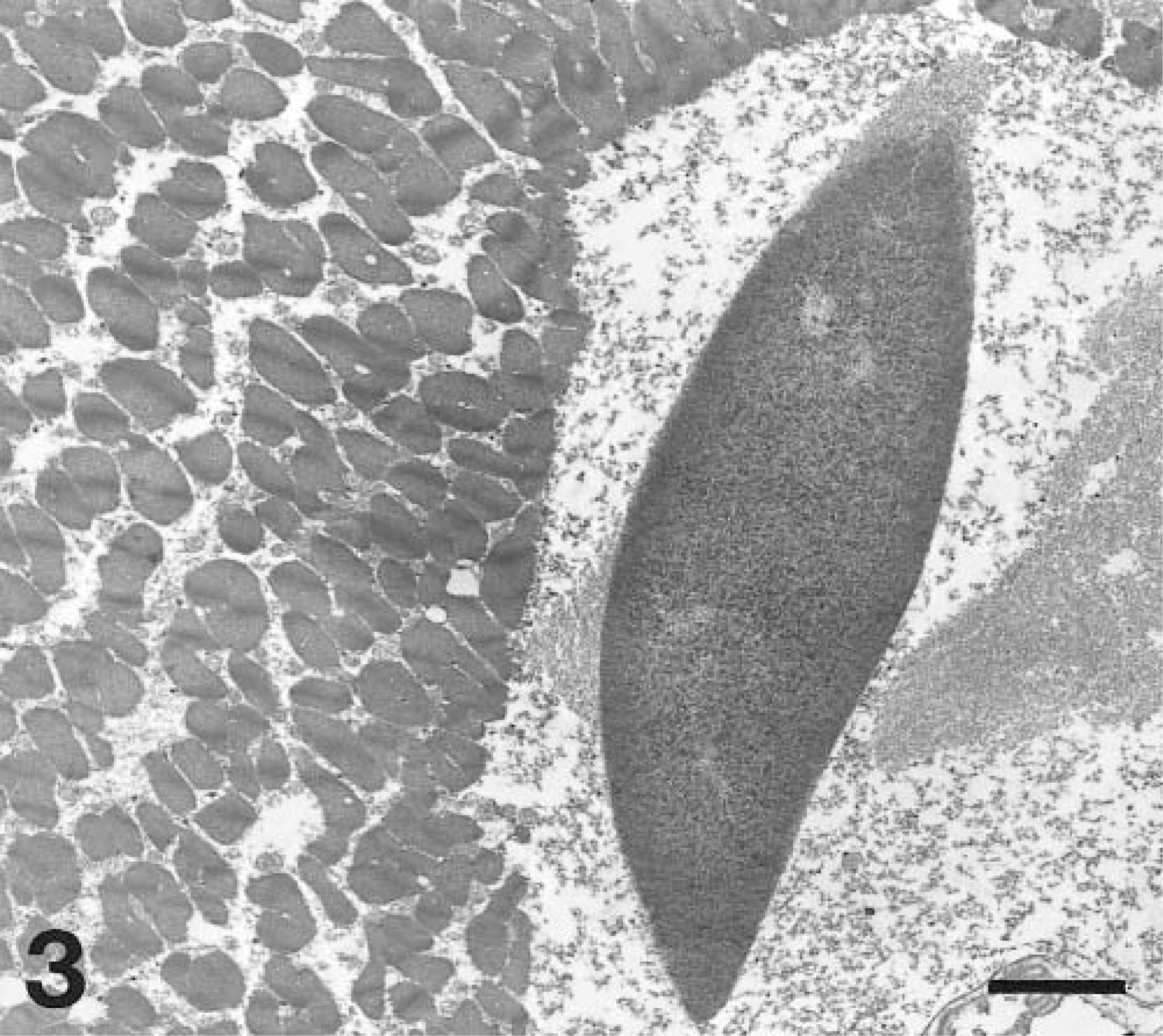
Transmission electron micrograph. Skeletal muscle; dog No. 2. An intramyofiber non-membrane-bound inclusion consists of an elongate central core of electron dense material with tapered ends and peripheral loosely arranged granular and filamentous material. Uranyl acetate and lead citrate. Bar = 0.3 μm.
This dog had no history of signs of neuromuscular or hemolytic disease. Whole blood was obtained from this dog prior to its death due to metastatic disease approximately 3 months after diagnosis. Analysis of DNA (Section of Medical Genetics, University of Pennsylvania) revealed no mutation in the PFK gene, and erythrocyte 2,3-diphosphoglycerate concentrations were normal, excluding a proximal glycolytic enyzmopathy.
Inclusions of PAS-positive, amylase-resistant material occur within skeletal muscle fibers in a variety of human and animal disorders. In humans, inclusions similar to those described here have been associated with generalized metabolic myopathies1,2,8,11 and with myopathy due to hypothyroidism.7 Such inclusions in humans are most commonly associated with carbohydrate metabolic defects.1,2 Similar inclusions have been seen within skeletal muscle of dogs with PFK deficiency6 and in Norwegian forest cats4 and Quarterhorse foals9 with inherited glycogen branching enzyme deficiency. The distinctively crystalline inclusions described in Quarterhorse foals with glycogen branching enzyme deficiency were not seen in these two dogs. Inclusions very similar to those described in these two dogs occur in skeletal muscle fibers of horses with polysaccharide storage myopathy.12,14,15 This equine disorder is thought to reflect abnormal carbohydrate metabolism, although a specific defect has yet to be identified.13 Similarly, in some cases of generalized polysaccharide storage myopathy in humans, defects of glycolysis or glycogenolysis were not found.8,11
Testing for hypothyroidism was not performed in either dog, but there was no clinical evidence of thyroid dysfunction. Dog No. 1 was an American Cocker Spaniel, and PFK deficiency has been reported in this breed.5 Although blood was not obtained from this dog for genetic and biochemical testing prior to death, there was no clinical or laboratory evidence of hemolytic anemia or myopathy. PFK deficiency was ruled out by antemortem blood testing in dog No. 2. Only a small area of skeletal muscle from dog No. 1 was available for examination; therefore, it was not possible to determine the distribution of skeletal muscle lesions. However, clinical evidence of generalized neuromuscular disease was not apparent. Multiple samples containing skeletal muscle were examined from the amputated limb of dog No. 2, and intramyofiber inclusions were present only within those fibers immediately adjacent to the tumor.
The finding of complex polysaccharide inclusions in skeletal muscle fibers adjacent to malignant tumors in these two dogs in the absence of evidence of a generalized neuromuscular disorder suggests that such inclusions may occur as a relatively nonspecific change associated with altered myofiber metabolism. Similar localized inclusions of complex polysaccharide associated with neoplasia have not been previously reported in any species. Intramyofiber inclusion formation in these two dogs was most likely related to alterations induced by the adjacent malignant tumors, but the specific type(s) of underlying metabolic dysfunction is unknown.
Footnotes
Acknowledgements
We thank C. Downie (Bailey Veterinary Clinic, Roseburg, OR) and D. Baum (Bandon Veterinary Hospital, Bandon, OR) for referral of cases and A. Wenski-Roberts for photographic assistance.
