Abstract
Six cases of fatal myocarditis associated with encephalomyocarditis virus occurred over a 14-month period in a group of outdoor-housed juvenile rhesus macaques. All animals were younger than 3 years of age and died or were euthanized following acute onset of dyspnea or pulmonary effusion (3 of 6) or were found dead without premonitory signs (3 of 6). Gross findings included pulmonary congestion (6 of 6), variable degrees of pleural effusion (4 of 6), multifocal pale tan foci throughout the myocardium (3 of 6), hepatomegaly and hepatic congestion (3 of 6), and pericardial effusion (1 of 6). Histologically, affected myocardium was infiltrated multifocally by lymphoplasmacytic and histiocytic inflammation admixed with necrotic and degenerate myofibers and infrequent mineralization (6 of 6). Pulmonary edema was present in all animals. Encephalomyocarditis virus was confirmed in 6 of 6 hearts by immunohistochemistry, and virus was isolated from one case by polymerase chain reaction. Sequencing of virus isolated from 1 affected animal indicated infection with a novel encephalomyocarditis virus. Encephalomyocarditis virus should be considered as a differential etiology in outbreaks of myocarditis and pulmonary edema in juvenile primates.
Keywords
Encephalomyocarditis virus (EMCV) is a nonenveloped RNA virus within the Picornaviridae family and Cardiovirus genus that causes sporadic, frequently fatal outbreaks of disease in swine, rodents, various zoo animals, cattle, horses, and nonhuman primates. Based on tissue tropism in mice, EMCV strains are divided into neurotropic and myocardiotropic variants. 2 Myocardiotropic variants can be further stratified into nondiabetogenic and diabetogenic on the basis of their ability to infect pancreatic β cells 3 and induce type 1 diabetes in mice. 4,15 EMCV infection has been documented in humans; however, in contrast to that of many domestic animal species, clinical disease appears to be uncommon, manifesting as nonspecific febrile illness. 13 Rodents are thought to act as silent carriers of the virus, and infrequent outbreaks among animals may occur following consumption of infected rodents or feed contaminated with rodent excreta. 1 Severely affected primates typically are found dead with few antecedent signs. Pathologic findings predominantly consist of necrotizing nonsuppurative myocarditis, pulmonary congestion, and pericardial effusion. 11 Encephalitis and fetal loss may also manifest in some species, particularly baboons. 8,11,12 Milder infections can occur; however, the spectrum and incidence of subclinical disease is poorly understood. Definitive diagnosis is based on immunohistochemistry, electron microscopy, and/or virus isolation, and effective control during outbreaks requires strict rodent control and vaccination of susceptible hosts, where possible. Here we report clinical, gross, histopathologic, and molecular features of an EMCV outbreak in a group of outdoor-housed juvenile rhesus macaques.
Materials and Methods
Animals
Six juvenile rhesus macaques (Macaca mulatta; 4 male, 2 female) ranging in age from 1 to 3 years (mean, 1.9 ± 0.9 years) either died or were euthanized with symptoms of cardiopulmonary disease at the Caribbean Primate Research Center (Sabana Seca, Puerto Rico), which houses approximately 2000 rhesus monkeys in outdoor corrals divided between specific pathogen–free and conventional colonies. The affected animals were housed in corrals throughout the center, and the cases occurred in a random pattern throughout the facility. Necropsy was performed at the center, and selected formalin-fixed (10% neutral buffered formalin) and frozen tissues were submitted for gross and histopathologic evaluation and viral isolation. Fixed tissue specimens were embedded in paraffin, sectioned at 5-μm intervals, and either stained with hematoxylin and eosin for histopathologic examination or subjected to immunohistochemical staining.
Immunohistochemistry
Immunohistochemical localization of EMCV antigen was performed on heart tissue and liver from all affected cases and 2 unaffected animals (negative controls) using the monoclonal antibody 3E5 (kindly provided by the Istituto Zooprofilattico Sperimentale della Lombardia e dell’Emilia Romagna, Brescia, Italy) in a dilution of 1:3000. Pretreatment of the slides for antigen retrieval was done by heating for 1 hour in the LabVision PT Module (Thermo Scientific, Fremont, CA) in citrate buffer (pH, 6.0). The reaction was performed in a Thermo Autostainer 360-2D System (Thermo Scientific) using the Ultravision LP Detection System (Thermo Fisher Scientific, Fremont, CA) and DAB Plus (Thermo Fisher Scientific) as chromogen.
Cell Culture and Viral Isolation
Heart tissue from case No. 5 was minced and transferred to confluent Vero cells (African green monkey kidney cells) in a 25-cm 2 flask with 8 ml of 10% fetal calf serum, minimal essential medium, and 10 μg/ml of gentamicin. When more than 90% of the infected cells showed cytopathic changes, the cultures were clarified by centrifugation for 10 minutes at 2200 rpm. The cell lysates and pellets were saved at –80°C for total RNA isolation.
RNA Extraction, Reverse Transcription Polymerase Chain Reaction, and Sequencing
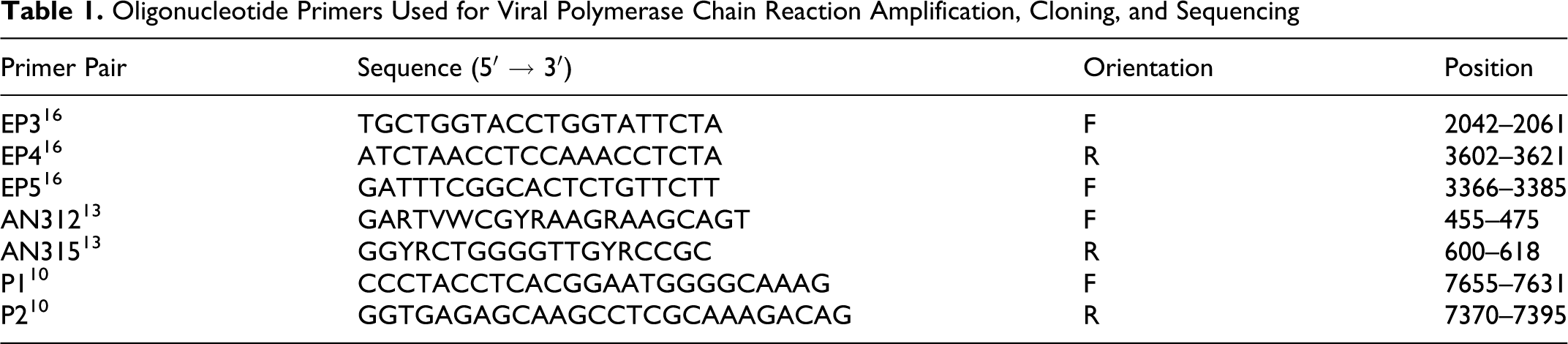
RNA was extracted from the virus isolates by using QIAamp Viral RNA Mini Kit (Qiagen, Valencia, CA). The first-strand cDNA was synthesized following the protocol of QuantiTect Reverse Transcription Kit (Qiagen). Oligonucleotide primers were used to amplify 5′-NTR, 3D, and the corresponding regions of BJC3 and HB1 strains (Table 1). 10,13,16 The cDNA samples were initially amplified with EP3F (5′-TGCTGGTACCTGGTATTCTA-3′) and EP4R (5′-ATCTAACCTCCAAACCTCTA-3′). Amplification was carried out in 25 μl of 1X GoTag Green Master Mix (Promega, San Diego, CA) with 25 pmol of each primer and 5 μl of cDNA. The cycling condition consisted of 2 minutes of denaturation at 94°C, followed by the first 10 cycles of 45 seconds at 94°C, 45 seconds at 53°C, and 100 seconds at 72°C; then, 25 cycles of 45 seconds at 95°C, 45 seconds at 55°C, 100 seconds at 72°C, and a final 10-minute extension step at 72°C. An aliquot (2 to 5%) of these amplification products (EP3F–EP4R) was then used as a template in a subsequent nested polymerase chain reaction (PCR) with the primer EP5F (5′-GATTTCGGCACTCTGTTCTT-3′) and EP4R. The products of the secondary nested PCR amplification were electrophoresed in 1.5% gel and visualized by irradiation with ultraviolet in the presence of ethidium bromide. The nested PCR products (256 base pairs) were cloned into PCR 2.1-TOPO vector (Invitrogen, Carlsbad, CA) and sequenced (Retrogen, Inc., San Diego, CA).
Oligonucleotide Primers Used for Viral Polymerase Chain Reaction Amplification, Cloning, and Sequencing
Electron Microscopy
Vero cells were cultured with affected heart tissue from case No. 5, shown to be positive for cardiovirus by PCR. The cell culture samples were fixed in 3% glutaraldehyde, postfixed in 1.0% osmium tetroxide, dehydrated through graded ethanols and propylene oxide, and embedded in eponate 12 epoxy resin (Ted Pella Inc., Redding, CA). Thin sections were prepared on a Leica Ultracut R microtome and stained with uranyl acetate and Sato lead stain. The grids were examined with a JEOL 1010 Electron Microscope (JEOL Ltd., Tokyo, Japan).
Results
Clinical Signs and Gross Findings
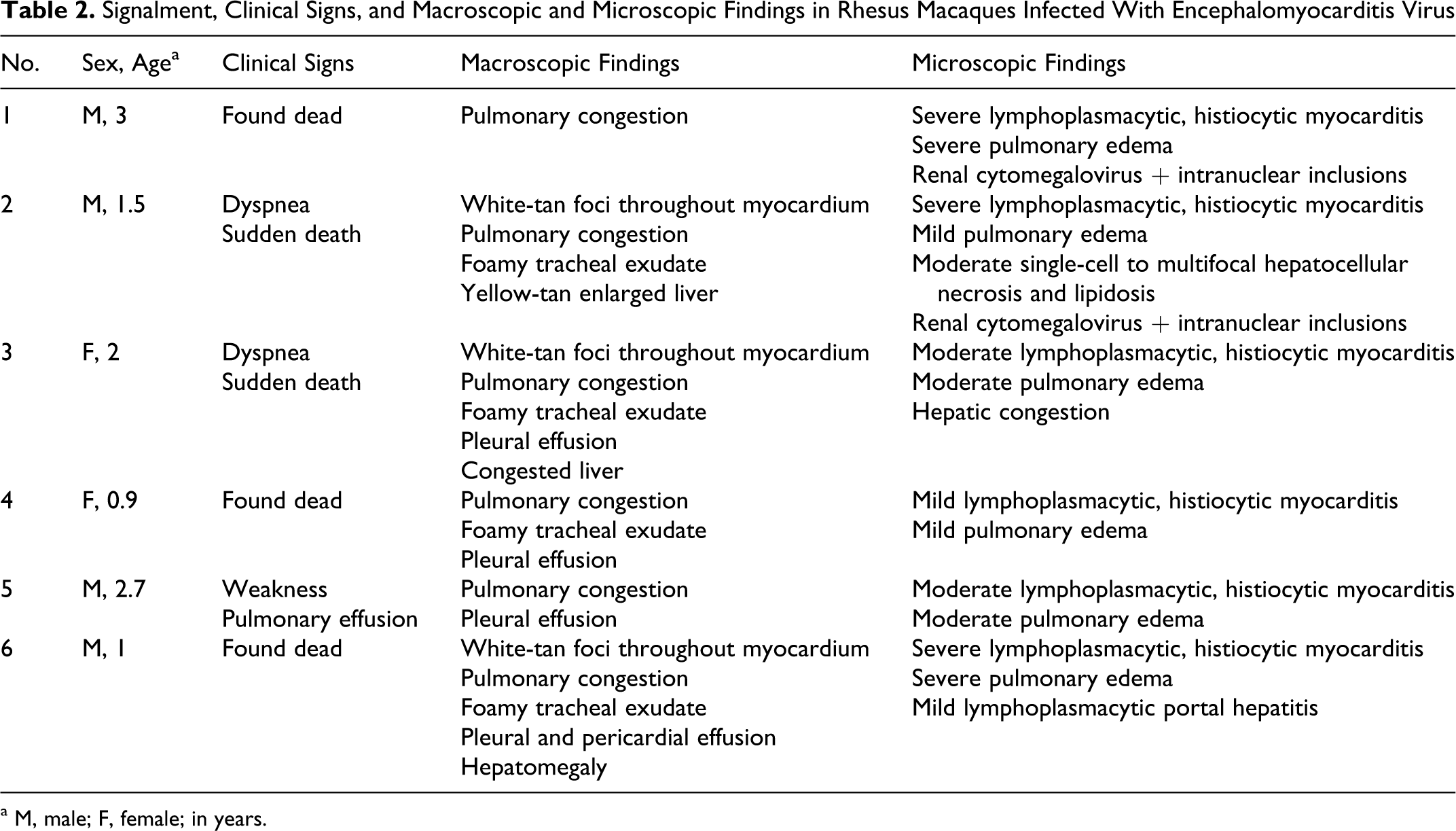
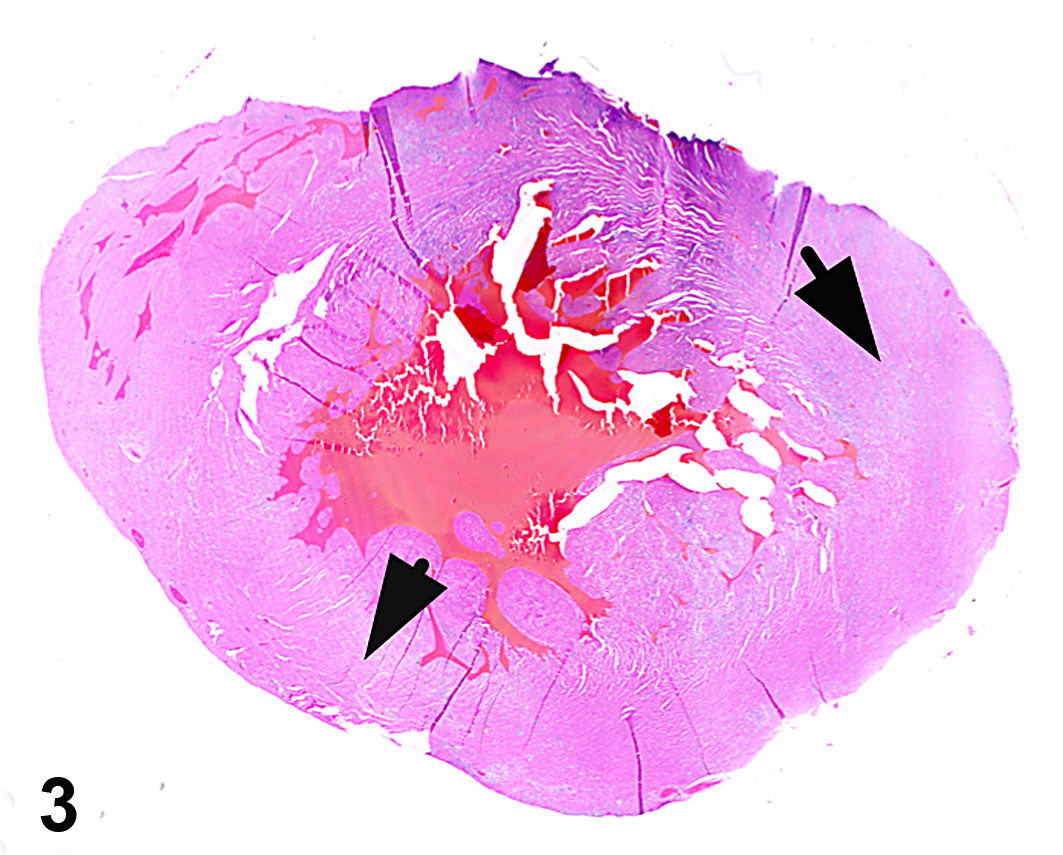
Over a 14-month period, between October 2008 to December 2009, 6 juvenile rhesus macaques (4 male, 2 female) ranging in age from 1 to 3 years (mean, 1.9 ± 0.9 years) either died or were euthanized with symptoms of cardiopulmonary compromise, including dyspnea and pulmonary effusion (Table 2). Three of the animals were found dead, without premonitory signs. When present, gross lesions were confined to the cardiopulmonary systems and liver. Diffuse, marked pulmonary congestion was present in all cases, and 4 of 6 animals had foamy exudate throughout the lower and upper airways. Pleural effusion was a common finding (4 of 6 animals) and was identified radiographically in 1 case (case No. 5) prior to death. In half the cases (3 of 6), gross lesions were present in the myocardium (Fig. 1) and consisted of multifocal, random, variably sized pale white-tan foci affecting all regions of the heart. Some lesions were transmural (Fig. 2) and, in case No. 6, accompanied by pericardial effusion. Liver lesions (3 of 6) included hepatomegaly, lipidosis, and hepatic congestion.
Signalment, Clinical Signs, and Macroscopic and Microscopic Findings in Rhesus Macaques Infected With Encephalomyocarditis Virus
a M, male; F, female; in years.
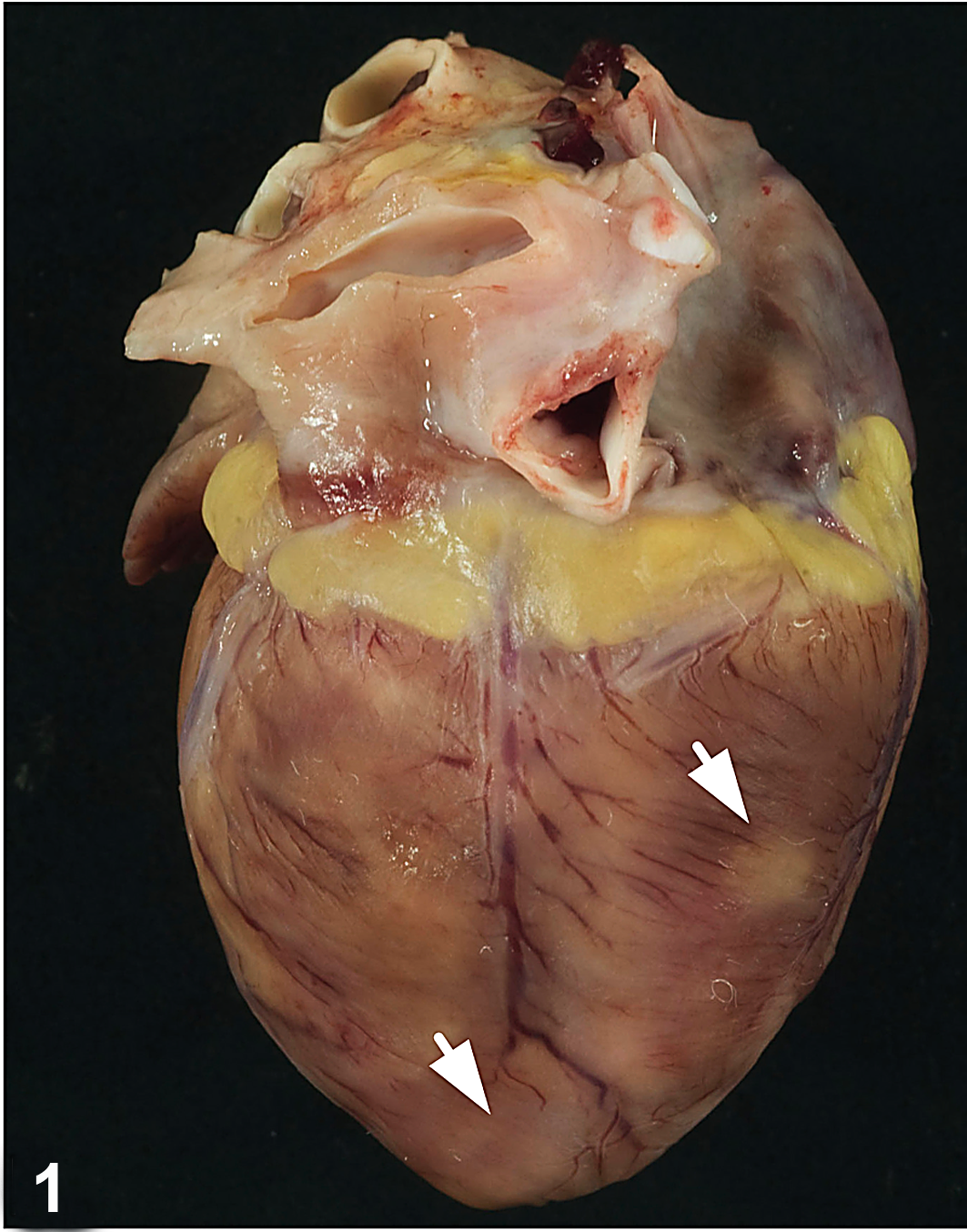
Heart; macaque, case No. 3. Multifocal white-tan foci (arrows) are present on the surface of the heart.
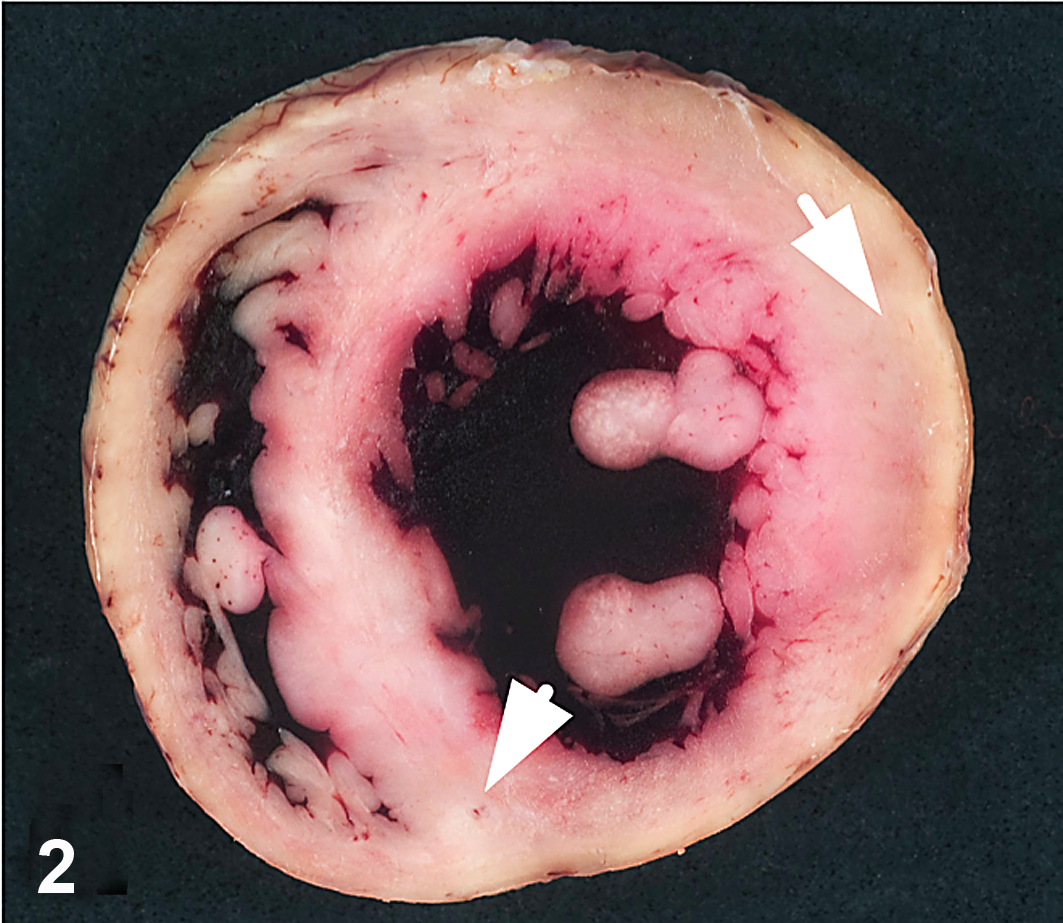
Heart; macaque, case No. 3. White-tan foci are random and multifocally transmural (arrows).
Histologic Findings and Differential Diagnosis
Histologic lesions were consistently observed in the myocardium (6 of 6) and characterized by variably severe lymphoplasmacytic and histiocytic inflammation admixed with necrotic and degenerate cardiomyofibers and infrequent foci of mineralization (Figs. 3,4). Coalescent regions of pulmonary edema were also observed in all cases. Histologic lesions were present in the livers in 3 cases and included single-cell and multifocal hepatocellular necrosis accompanied by mild lipidosis (case No. 2), marked sinusoidal congestion (case No. 3), and mild periportal lymphoplasmacytic infiltrates confined within the limiting plate (case No. 6). An unusual finding in the kidneys in 2 animals (case Nos. 1 and 2) was rare, enlarged cells that contained basophilic intranuclear inclusion bodies surrounded by marginated chromatin. Affected cells were not associated with inflammatory reaction. These inclusions were positive for cytomegalovirus (CMV) by immunohistochemistry (data not shown).
Heart; macaque, case No. 3. White-tan foci correspond to regions of lymphoplasmacytic and histiocytic inflammation (arrows). HE.
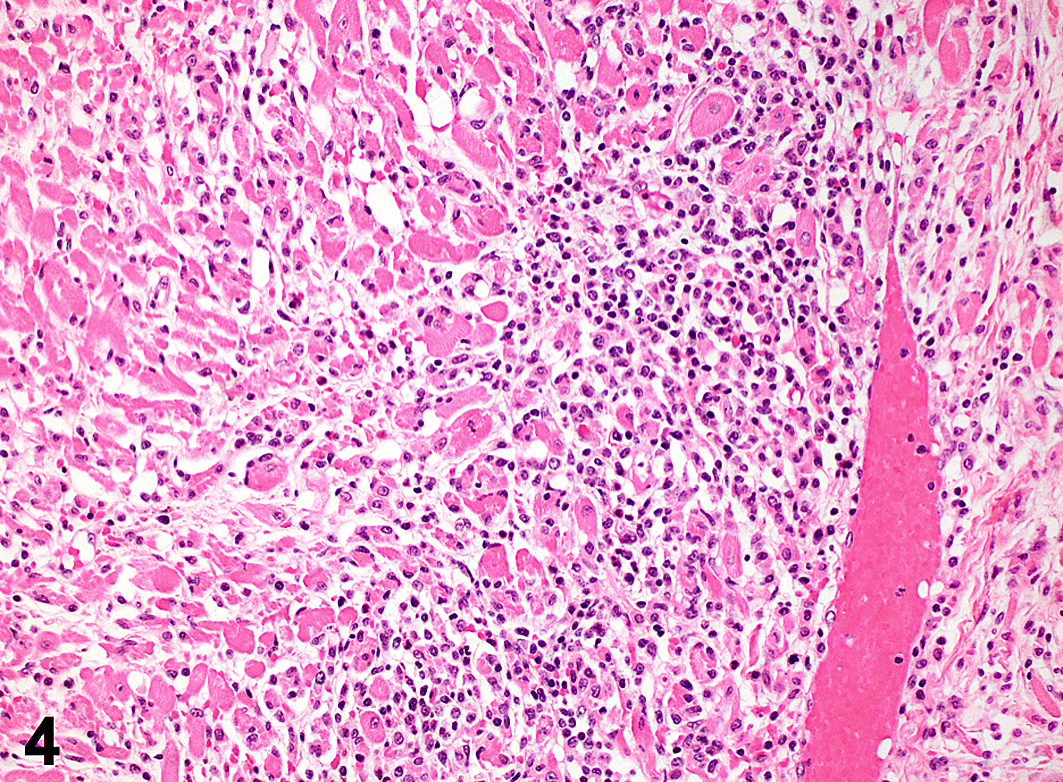
Heart; macaque, case No. 3. Affected myocardium is multifocally necrotic and degenerate and heavily infiltrated with mononuclear inflammation. HE.
Based on history, clinical signs, and gross and histopathologic findings, the major differential diagnoses that were considered for this outbreak included Trypanosoma cruzi, Toxoplasma gondii, Coxsackie virus, EMCV, or other cardiovirus. Serial sections from affected hearts were negative for protozoa, and evaluation for viral etiology was pursued.
Immunohistochemistry and PCR
Infection with EMCV was strongly suspected on the basis of history, location, clinical signs, and gross and histopathologic lesions, and further evaluation was conducted on affected heart tissue by immunohistochemistry. In all cases, antigenic staining was present within the cytoplasm of cardiomyocytes (Fig. 5) but not in heart samples from 2 unaffected animals examined in parallel as negative controls (data not shown). Rare inflammatory cells presumed to be macrophages based on cell morphology were also positive in some heart sections from affected animals (data not shown); however, it is not known whether this reflects direct viral infection or phagocytosis of infected cellular debris. Immunohistochemistry was also performed on sections of liver from each case, but staining was not observed in any section (data not shown). EMCV infection was confirmed in Vero cells cocultured with heart tissue from case No. 5 by PCR using primers against the 5′NTR, VP1, and 3D regions of the virus (Table 1). Resultant bands were sequenced and determined to be from a cardiovirus closely related to porcine isolates of EMCV (Table 3). Based on criteria recommended by the International Committee on Taxonomy of Viruses, 6 classification of the virus as EMCV was further supported by predicted amino acid sequences from the 3D and VP1 regions, which indicated 91% to 95% and 89% to 96% identity, respectively, with those regions from previously characterized EMCV isolates (data not shown). Sequence data from EMCV strains recovered from outbreaks among nonhuman primates are limited; however, VP1 and 3D regions shared 79% and 82% homology, respectively, with an orangutan-isolated strain (Table 3), indicating infection with a novel virus strain not previously reported.
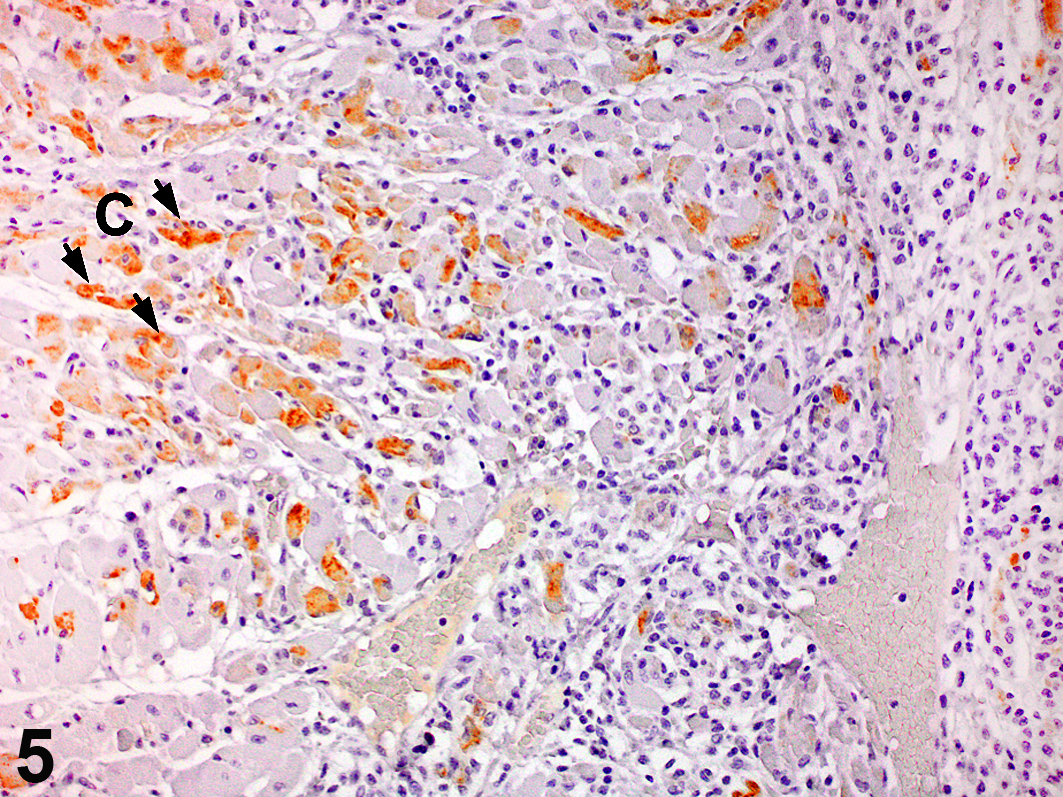
Heart; macaque, case No. 3. Cardiomyocytes (C; arrows) are immunoreactive for encephalomyocarditis virus. Ultravision LP Detection System and DAB Plus.
Sequence Similarity of Virus Isolated From Vero Cells Cultured With Heart Tissue From Case No. 5 to Known Encephalomyocarditis Virus Strains
Electron Microscopy
To attempt virus isolation, Vero cells were cultured with affected heart tissue from case No. 5, shown to be positive for cardiovirus by PCR. Vero cell cultures exhibited marked cytopathic change (data not shown). Transmission electron microscopy was conducted on affected cell cultures, and significant ultrastructural findings included large masses of cytoplasmic membrane material that displaced the nuclei of degenerating cells (Fig. 6). Additionally, many cells contained accumulations of electron-dense granular particles measuring 20 to 30 nm in diameter, which were occasionally membrane bound (Fig. 7). These particles were interpreted to represent degenerative material or possibly accumulations of ribosomes, as has been reported for Picornaviridae. 5 Distinct viral particles in paracrystalline array were not observed. Other ultrastructural findings were consistent with cellular degeneration and necrosis.
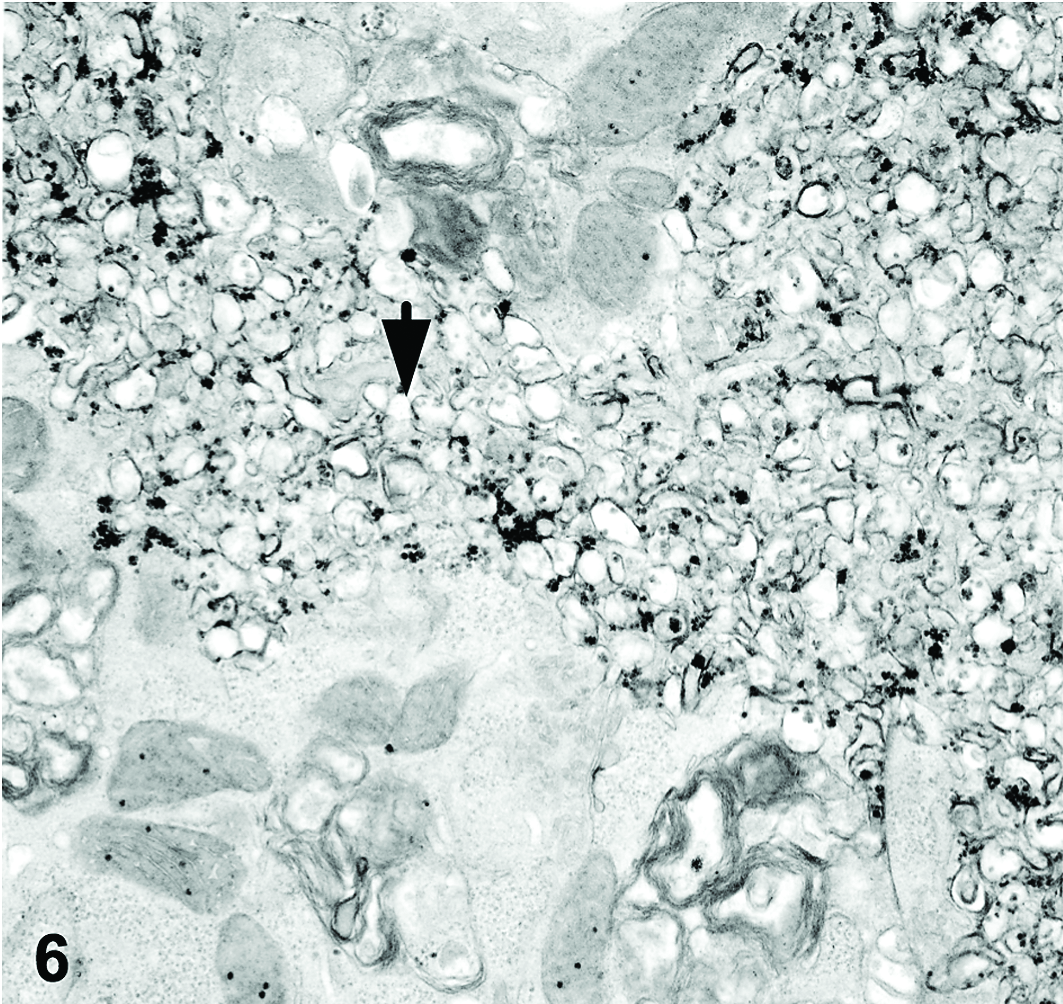
Vero cells. Large masses of cytoplasmic membrane material accumulate within degenerate cells (arrow). Electron microscopy.
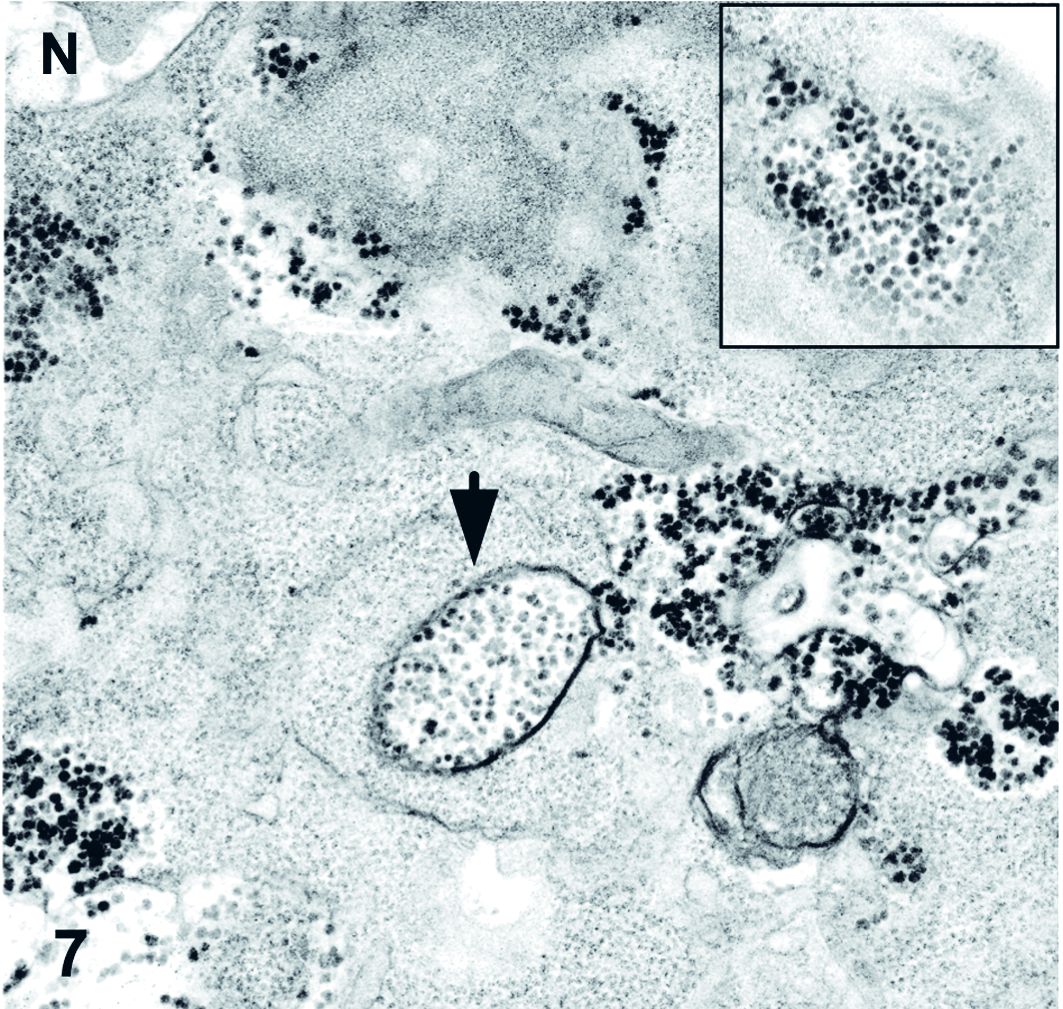
Vero cells. Many cells contain accumulations of electron-dense particles, which are in some cases membrane bound (arrow). Inset: Higher magnification of granular material. N: cell nucleus. Electron microscopy.
Discussion
Here we describe the clinical, gross, histopathologic, and molecular findings associated with an outbreak of myocarditis among a group of juvenile macaques. All affected animals were less than 3 years of age, and the most frequent gross findings included pulmonary congestion and foam-filled airways. In half the cases, there were white-tan foci visible throughout the heart, corresponding to lymphoplasmacytic and histiocytic myocarditis. Pulmonary edema was another consistent finding in all the animals and likely contributed to the cause of death in all cases. Clinicopathologic findings were predominantly localized to the cardiopulmonary system in these animals; however, lesions were also found in liver in some cases, possibly secondary to perfusion abnormalities from cardiac dysfunction. Immunohistochemistry for EMCV antigen in liver was negative, suggesting that hepatic lesions did not result from direct viral infection. 7 History, clinical signs, and gross and microscopic findings were consistent with findings reported in previous outbreaks among nonhuman primates. 1,8,11,14 In addition to cardiopulmonary lesions, encephalitis has been associated with EMCV infection in nonhuman primates; 11,12 however, brain and spinal cord were not available for evaluation in the present outbreak, and thus incidence of neurologic lesions is unknown with this strain.
EMCV infection in heart was confirmed in affected cases by combinations of immunohistochemistry, PCR, and direct viral sequencing. EMCV sequence data are most abundant from porcine outbreaks, whereas data from strains causing disease among nonhuman primates are very limited. Sequencing of portions of the viral isolate from case No. 5 indicates a novel EMCV that shares some homology with EMCV recovered from an orangutan; however, the relationship of this strain to those in previous outbreaks among primates is unknown. Epizootics of EMCV within farms, zoological collections, and primate centers are frequently fatal for a variety of domestic and wild animal species, and the lack of understanding of host susceptibility factors and viral determinants of virulence hinders efforts to prevent outbreaks and limit mortality when they occur. In the future, increasing availability of molecular tools will likely make it possible to better characterize and compare emergent viral strains causing disease among animals and humans.
Ultrastructural examination of cell cultures cocultured with heart from 1 affected animal was also used to evaluate viral infection. Electron microscopy failed to reveal viral particles; however, secondary changes suggestive of EMCV were present. These included distinctive masses of membrane observed in the cytoplasm, as well as 30-nm granular deposits interpreted as degenerative material or possibly ribosomes. These changes have been reported previously in picornavirus-infected cells, 5 although their cause and significance is poorly understood.
The source of the virus in this outbreak is unknown, but initiation of a strict rodent control program coincided with a markedly diminished incidence of disease, suggesting that, consistent with previous reports, 1 exposure to rodents was a key factor in this outbreak. Interestingly, the affected macaques were each housed in nonabutting corrals around the facility, indicating that these animals either had exceptional exposure to the virus or were uniquely susceptible to infection and disease. Precedent for genetic susceptibility to EMCV is found in mice infected with diabetogenic myocardiotropic variants, in which certain strain backgrounds are at increased risk of developing fulminant type 1 diabetes. 9 It is possible that genetic factors may predispose to differential outcomes following infection with neurotropic and myocardiotropic variants in natural outbreaks as well. Moreover, in a 14-month period, 6 cases of fulminant myocarditis associated with EMCV were observed, yet it is likely that more animals were infected than those showing clinical signs, and the incidence of subclinical infection in the colony is currently being studied. An interesting finding in 2 cases was the presence of intranuclear inclusion bodies confirmed as CMV in the kidney. These inclusions were not associated with host tissue reaction in either case, and it is presumed that they resulted from newly acquired infections in these young animals. It is not known whether coinfection with CMV affects susceptibility to EMCV, but it is possible that these animals may have had underlying immune dysfunction that contributed to fatal EMCV disease.
In conclusion, EMCV should be considered in outbreaks of sudden death, myocarditis, and pulmonary edema among primates, and the present report details the molecular signature of a viral strain associated with fatal infection among a group juvenile macaques.
Footnotes
Acknowledgements
We thank Norval King for his expert opinion interpreting electron microscopy images, as well as Basel Assaf, Karen Boisvert, Nicole Evangelous, Heather Knight, Shiva Shanmukhappa, and Nilsa Silva for sample processing, staining, and interpretation, and Kristen Toohey for photographic images of gross specimens.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This research was funded by National Institutes of Health grant Nos. RR00168, RR007000, and RR003640.
