Abstract
Gastrointestinal lymphomas were identified in 120 cats between 1995 and 2006. Lymphomas were classified according to the World Health Organization (WHO) scheme. Cats with mucosal T-cell lymphoma (n = 84) predominated and had a median survival of 29 months. Mucosal T-cell lymphoma matched WHO enteropathy-associated T-cell lymphoma (EATCL) type II. Epitheliotropic T-cell infiltrates were present in 62% of cats and occurred as clusters or diffuse infiltrates of small to intermediate-sized T cells in villous and/or crypt epithelium. Similar lymphocytes infiltrated the lamina propria in distinctive patterns. Cats with transmural T-cell lymphoma (n = 19) had a median survival of 1.5 months. Transmural T-cell lymphoma matched WHO EATCL type I. Epitheliotropic T-cell infiltrates were present in 58% of cats. Large lymphocytes (n = 11), mostly with cytoplasmic granules (LGL–granzyme B+) (n = 9) predominated. Transmural extension across the muscularis propria characterized the lesion. Both mucosal and transmural T-cell lymphomas were largely confined to the small intestine, and molecular clonality analysis revealed clonal or oligoclonal rearrangements of T-cell receptor-γ in 90% of cats. Cats with B-cell lymphoma (n = 19) had a median survival of 3.5 months. B-cell lymphomas occurred as transmural lesions in stomach, jejunum, and ileo–cecal–colic junction. The majority were diffuse, large B-cell lymphomas of centroblastic type. In conclusion, T-cell lymphomas characterized by distinctive mucosal architecture, CD3 expression, and clonal expansion predominated in the feline gastrointestinal tract.
Keywords
Lymphoma is the most common hemopoietic neoplasm in the cat, and the incidence is reported to be the highest for any species.5,9,21,27 Since the decline (in the United States) in the incidence of feline leukemia virus (FeLV) infection, which is highly lymphomagenic, the incidence of feline lymphoma has actually increased. This increase has mainly been in gastrointestinal lymphomas.16,27 Gastrointestinal lymphomas, especially mucosal T-cell lymphomas of the small intestine, have been underreported prior to the advent of T-cell receptor gene rearrangement analysis.18,35 Hence, the increased incidence of feline gastrointestinal lymphoma in recent years is likely to be of far greater magnitude.
The gastrointestinal tract harbors the largest population of lymphoid and accessory immune cells in the body. Not surprisingly, it is often a target for lymphoma. The diffuse, mucosal-associated lymphoid tissue (MALT) of the small intestine, which consists of lamina proprial compartments (LPC) and intraepithelial compartments (IEC), is populated largely by CD3+ T cells in normal cats.12,23 Less than 10% of villous lamina proprial lymphocytes (LPLs) are B cells, and even fewer are found in the IEC. Intraepithelial lymphocytes (IEL) are almost exclusively T cells. The inductive environment for adaptive immune responses is concentrated in the distal small intestine and ileo–ceco–colic junction, where Peyer patches and solitary lymphofollicular structures are most prominent. Antigen-stimulated B cells are induced to class switch to immunoglobulin A and home to the lamina propria adjacent to intestinal crypts, and are most numerous below the crypt–villous junction. Naive T cells in Peyer patches and mesenteric lymph nodes are induced by interactions with migratory antigen-bearing mucosal dendritic cells (DCs) to express cell surface molecules, such as β7 integrins (α4β7 and αEβ7) and chemokine receptors (especially CCR9), that enable them to traffic to the diffuse MALT and enter the LPC and IEC of the small intestine.4,14
Historically, pathologists have relied on the mucosal effacement and transmural invasion by cytologically atypical lymphocytes to confidently diagnose intestinal lymphoma. However, these morphologic criteria are less relevant when examining endoscopic biopsy specimens, which consist of mucosal and scant submucosal tissue with random orientation. Furthermore, lymphoma manifests in the intestinal mucosa long before transmural progression. This concept is supported by serial biopsy or subsequent necropsy findings in the current study and is also discussed in Fondacaro et al. 6 T-cell lymphomas of diffuse MALT of the small intestine are the most predominant gastrointestinal lymphomas, which often coexist with lymphoplasmacytic inflammatory bowel disease (IBD) or occur following a clinical history of IBD. 18
Immunohistochemistry and molecular clonality assays are valuable adjuncts to morphological assessment in diagnosis of lymphoma. Immunophenotypic assessment is most useful in lymphocyte lineage assignment, although it can also highlight distinctive lymphocyte infiltration patterns such as epithelial colonization, which are important in assessing the likelihood of lymphoma.3,18,23
Lymphocyte antigen receptor gene rearrangement analysis has become an important method to establish the presence of clonal lymphocyte expansion in lymphoproliferative disease. In a recent article, we characterized the expressed feline T-cell receptor-γ (TCRG) repertoire. Multiple sequence alignment of TCRG variable (V) and joining (J) segments revealed highly conserved regions, which enabled development of primers for polymerase chain reaction (PCR) amplification of the CDR3 region for detection of the clonality status of T cells in feline intestinal lymphoma. 18 In a companion article, we also characterized the expressed feline immunoglobulin heavy chain (IGH) repertoire and established its usefulness in the diagnosis of B-cell neoplasia. 32
Armed with an understanding of intestinal mucosal trafficking patterns and immunohistochemical and molecular diagnostic tools, we can now reexamine the architectural characteristics of feline gastrointestinal lymphomas and establish the distinguishing morphological features that correlate with clonal expansion of lymphocytes. In this article, we present the characteristic patterns of lymphocytic infiltration of the villous/crypt epithelium and the mucosal lamina propria that raise the index of suspicion of mucosal T-cell lymphoma of the small intestine in morphological terms. Furthermore, we show that mucosal T-cell lymphoma is the dominant lymphoma in the feline gastrointestinal tract.
Materials and Methods
Study Population
A total of 120 cats were included in the study. The majority of cats (n = 98) were presented to the Veterinary Medical Teaching Hospital, UC Davis, for clinical evaluation during the period 1995–2006. Tissues from another cohort of cats (n = 22) were accessed from a local private pathology laboratory (VDx, Davis, California) during the study period. The cases included in the study were limited to those in which a specialist review (P.F.M.) was requested. Tissues were collected by surgical biopsy (n = 47), by endoscopic biopsy (n = 35), and at necropsy (n = 38).
Immunohistochemistry
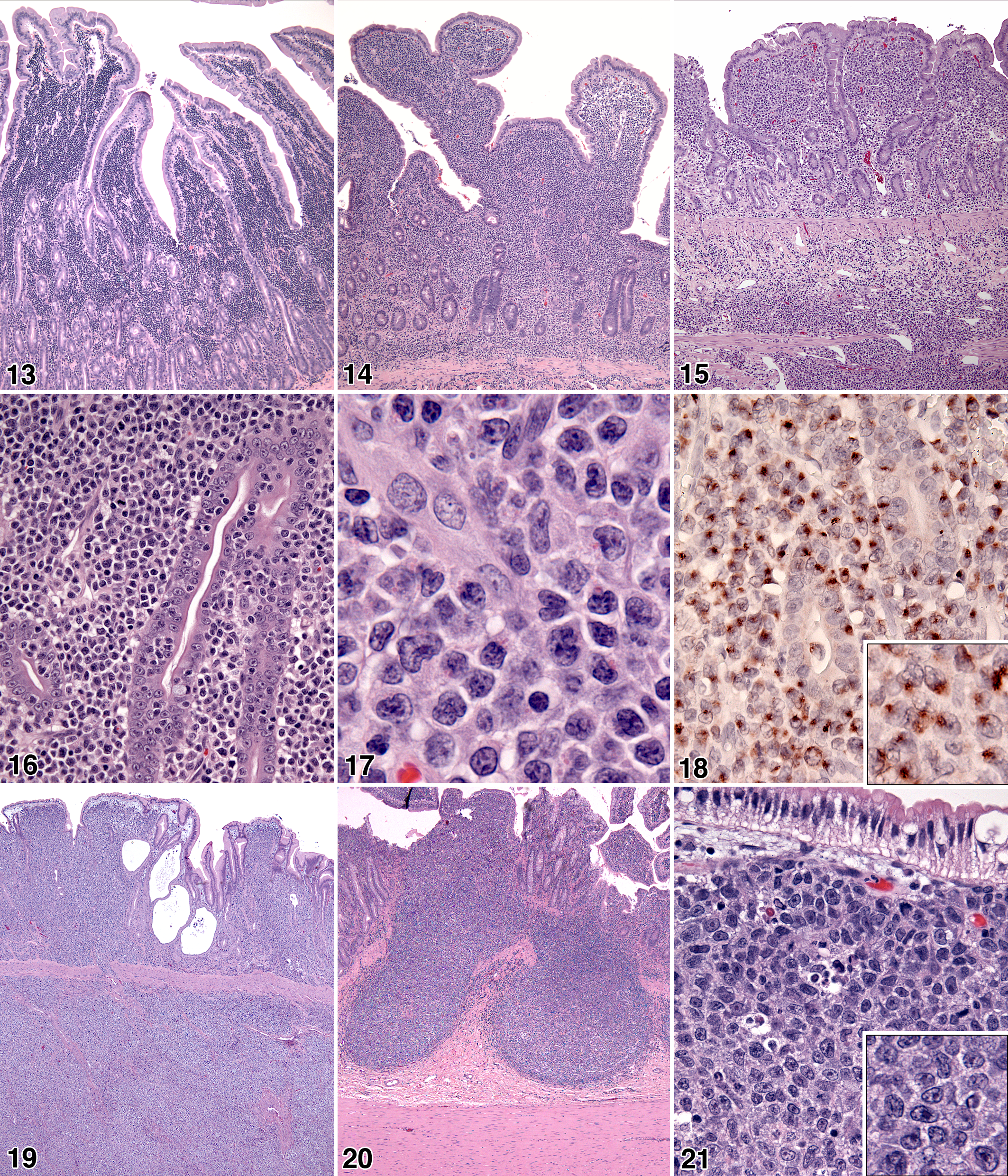
Expression of leukocyte antigens was assessed in formalin-fixed, paraffin-embedded (FFPE) tissue sections by a labeled avidin–biotin immunohistochemical technique and heat-based antigen retrieval. 1 Gastrointestinal tissue from all cats was assessed for the expression of CD3 (clone CD3-12, Serotec Inc, Oxford, UK) and CD79a (clone HM57, Dako Inc, Carpinteria, California). Transmural T-cell lymphomas of large granular lymphocyte type (LGL) (n = 9) were evaluated for expression of granzyme B by immunohistochemistry with a rabbit polyclonal antibody, 26 which is specific for a conserved human granzyme B-peptide sequence (Fig. 1 ) (Spring Bioscience, Pleasanton, California).
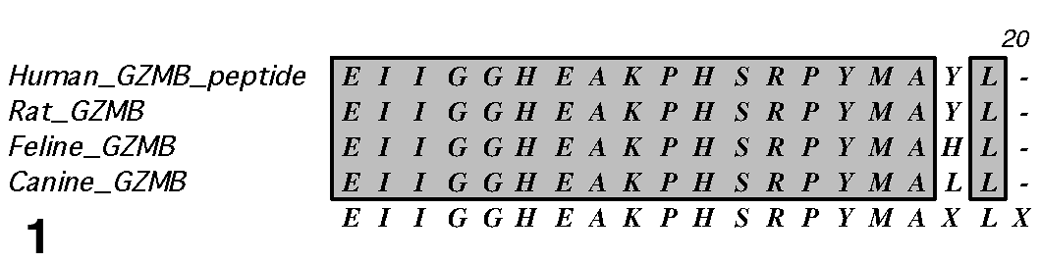
Multiple-species amino acid sequence alignment of the granzyme B-peptide immunogen (human) demonstrates the highly conserved sequence used to generate the polyclonal antibody to granzyme B.
Lymphoma Classification
Lymphomas were classified as B-cell, T-cell, or non-B, non-T cell lymphoma lymphoma by immunohistochemistry to detect expression of CD3 and CD79a. The lymphomas were further classified as mucosal lymphoma (infiltrate confined to the epithelium and lamina propria with minimal extension into the submucosa) or transmural lymphoma (infiltrate extending markedly into the submucosa and muscularis propria). Endoscopic biopsy specimens, although diagnostically adequate, did not include tissue below the muscularis mucosa. 31 Hence, transmural spread was impossible to assess, resulting in a default diagnosis of mucosal lymphoma. The diagnosis of lymphoma was based on the cytomorphology of the infiltrate, the degree of epitheliotropism (if present), and the pattern of lamina proprial infiltration; the specific criteria are fully described and illustrated below. The infiltration patterns were then compared with the World Health Organization (WHO) classification of tumors of the hematopoietic and lymphoid tissues to further classify B- and T-cell neoplasia into clinicopathologic entities.25,28
Statistical Methods
Survival data were obtained for 54 of the 84 cats with mucosal T-cell lymphoma and for 13 of the 19 cats with transmural T-cell lymphoma. Insufficient survival data were obtained for cats with B-cell lymphoma. The effect of cell size and tumor location on survival time was evaluated using the Kaplan-Meier method of survival function estimation and Cox proportional hazards regression. Regression results are reported as hazard ratios (HRs, ie, the ratio of 2 conditional mortality rates) and 95% confidence intervals (95% CIs). Interaction between model covariates was evaluated with a likelihood ratio test. STATA 10.1 (StataCorp LP, College Station, Texas) was used for all calculations.
Lymphocyte Antigen Receptor Gene Rearrangement Analysis
Clonality of lymphocyte antigen receptor genes (TRG and/or IGH) was performed on tissues from 119 of 120 cats as previously described.18,32 Briefly, DNA extraction was performed on 25-μm tissue sections that were cut from FFPE blocks using the DNeasy Extraction Kit (Qiagen, Valencia, California) per the manufacturer’s instructions.
Rearrangement of TCRG was assessed by PCR amplification of the CDR3 region between the V and J segments. Briefly, 100 ng of genomic DNA from each sample was amplified in a 50-μl reaction using a consensus primer derived from the TCRG V segment (5′-AAGAGCGAYGAGGGMGTGT-3′—20 pmol) in conjunction with a consensus primer derived from the TCRG J segments (5′-CTGAGCAGTGTGCCAGSACC-3′—10 pmol) of the feline TCRG cDNA transcripts. Specific amplicon size was expected to occur within a target range of about 80–120 base pairs. Amplification conditions used a 2-step, modified touchdown protocol to increase specificity of the reactions. 11 All PCR reactions were run in duplicate.
Rearrangement of IGH was assessed by amplification of the CDR3 region, which encompasses the junction of V, diversity (D), and J segments. Briefly, 100 ng of genomic DNA from each sample was amplified in a 50-μl reaction using consensus primers derived from the IGH V segment framework 2 (FR2) and framework 3 (FR3) (FR2: 5′-CCAGGCTCCAGGGAAGGG-3′—10 pmol, and FR3: 5′-TCCAGAGACAACGCCAAGAAC-3′—10 pmol) in conjunction with consensus primers derived from IGH J segments (J2: 5′-TGAGGACACTGTGACTATGGTTCC-3′—10 pmol, and JD: 5′-GGACACCGTCACYAKGVYTCC-3′—100 pmol). Amplification conditions used a 2-step, modified touchdown protocol to increase specificity of the reactions. 11 All PCR reactions were run in duplicate.
PCR products were separated by polyacrylamide gel electrophoresis (PAGE) of both native and denatured 15-μl samples of each PCR reaction. Polyacrylamide gels (Criterion Precast gels, Bio-Rad, Hercules, California), which contained both native and denatured PCR products, were run on ice in Tris-borate-EDTA (TBE) buffer at 150 V for 2 hours. Duplicate PCR samples were always run side by side. Gels were then stained for 30 minutes with Gel Star (BioWhitaker Molecular Applications, Rockland, Maine) and visualized with a UV transilluminator (Fisher Scientific, Pittsburgh, Pennsylvania). Samples that produced appropriately and same sized 1 or 2 sharp bands in duplicate were classified as clonal (monoallelic or biallelic rearrangement, respectively). Samples exhibiting 3–5 reproducible bands in duplicate analyses were classified as oligoclonal. Duplicate PCR reactions were used to help distinguish true clonal samples from pseudoclonal samples. Pseudoclonal samples contained 1 or 2 bands of disparate size in duplicate analysis. Samples that produced a broad band, smear, or ladder of bands covering a range of product sizes were classified as a polyclonal.
Results
Signalment
Cats with mucosal lymphoma (n = 86) consisted of 51 males and 35 females; the mean age at the time of diagnosis was 12.6 years (SD 3.4 years; range, 2–20 years). Mucosal T-cell lymphoma predominated (n = 84); non–B- and non–T-cell lymphoma occurred in only 2 cats. Cats with transmural T-cell lymphoma (n = 19) consisted of 7 males and 12 females; the mean age was 13.5 years (SD 2.6 years; range, 7–18 years). Cats with B-cell lymphoma (n = 19) consisted of 11 males and 8 females; the mean age was 12.2 years (SD 4.0 years; range, 2–18 years). There were 120 cats included in the study; 4 cats (cat Nos. 103, 115, 116, and 117) had concurrent B-cell lymphoma and mucosal T-cell lymphoma. Only 3 cats were serologically positive for feline immunodeficiency virus (FIV) (cat Nos. 24, 39, and 120), and none tested positive for FeLV.
Survival
Survival data were available for 54 of the 84 cats with mucosal T-cell lymphoma; 13 of the 19 cats with transmural T-cell lymphoma; and only 6 cats with B-cell lymphoma. Median time to death in the mucosal T-cell lymphoma group (n = 54) was 29 months, in contrast to the transmural T-cell lymphoma group (n = 13), in which it was 1.5 months (Fig. 2 ). The median time to death in the small-cell, T-cell lymphoma group (n = 54) was 28 months, in contrast to the large-cell, T-cell lymphoma group (n = 13), in which it was 1.5 months (Fig. 3 ). The mucosal T-cell lymphoma group largely consisted of small-cell type (n = 50/54), whereas the transmural T-cell lymphoma group was mostly of large-cell type (n = 9/13), with most of these (n = 8/9) being LGL lymphomas. A multivariate model examining the joint effects of cell size and location found a higher rate of death in the large-cell versus small-cell group (HR = 3.7; 95% CI, 1.2–10.9; P = .019) and in the transmural versus mucosal location group (HR = 3.4; 95% CI, 1.3–8.7; P = .010). Limited survival data (n = 6) were available for the B-cell lymphoma group; the median time to death was 3.5 months. Similarly, limited survival data (n = 4) were available for the transmural, small-cell, T-cell lymphoma cats, with a range of survival encompassing 3 days to 28 months.
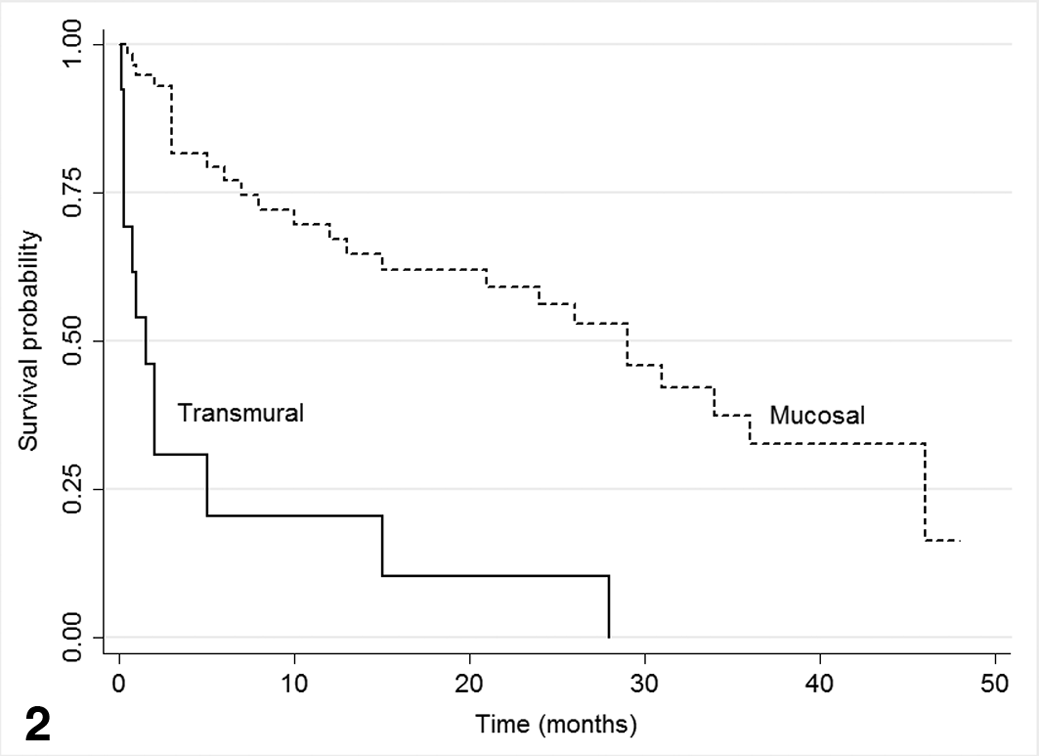
Kaplan-Meier plot of survival comparing cats with mucosal T-cell lymphoma (median survival 29 months) with cats with transmural T-cell lymphoma (median survival 1.5 months).
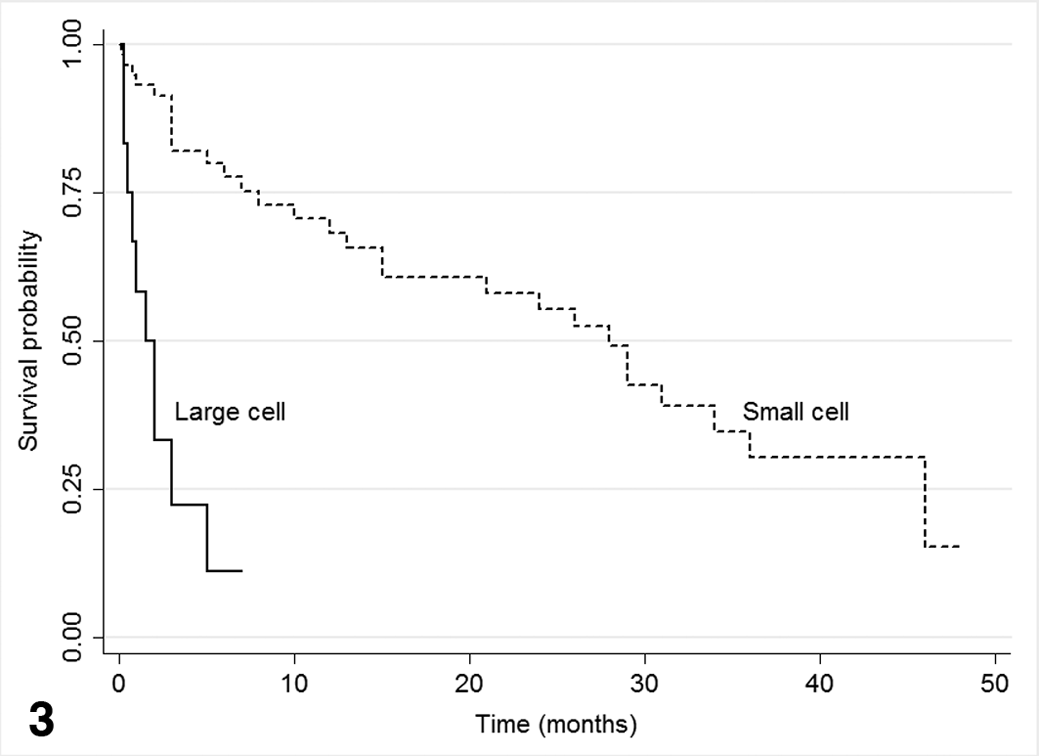
Kaplan-Meier plot of survival comparing cats with T-cell lymphoma, small-cell type (median survival 28 months) with cats with T-cell lymphoma, large-cell type (median survival 1.5 months).
Topographical Features
Feline intestinal lymphoma in our case series was most frequently of T-cell phenotype and occurred predominately in the diffuse MALT of the small intestine as mucosal (n = 84) and transmural lesions (n = 19) (Figs. 4, 5).
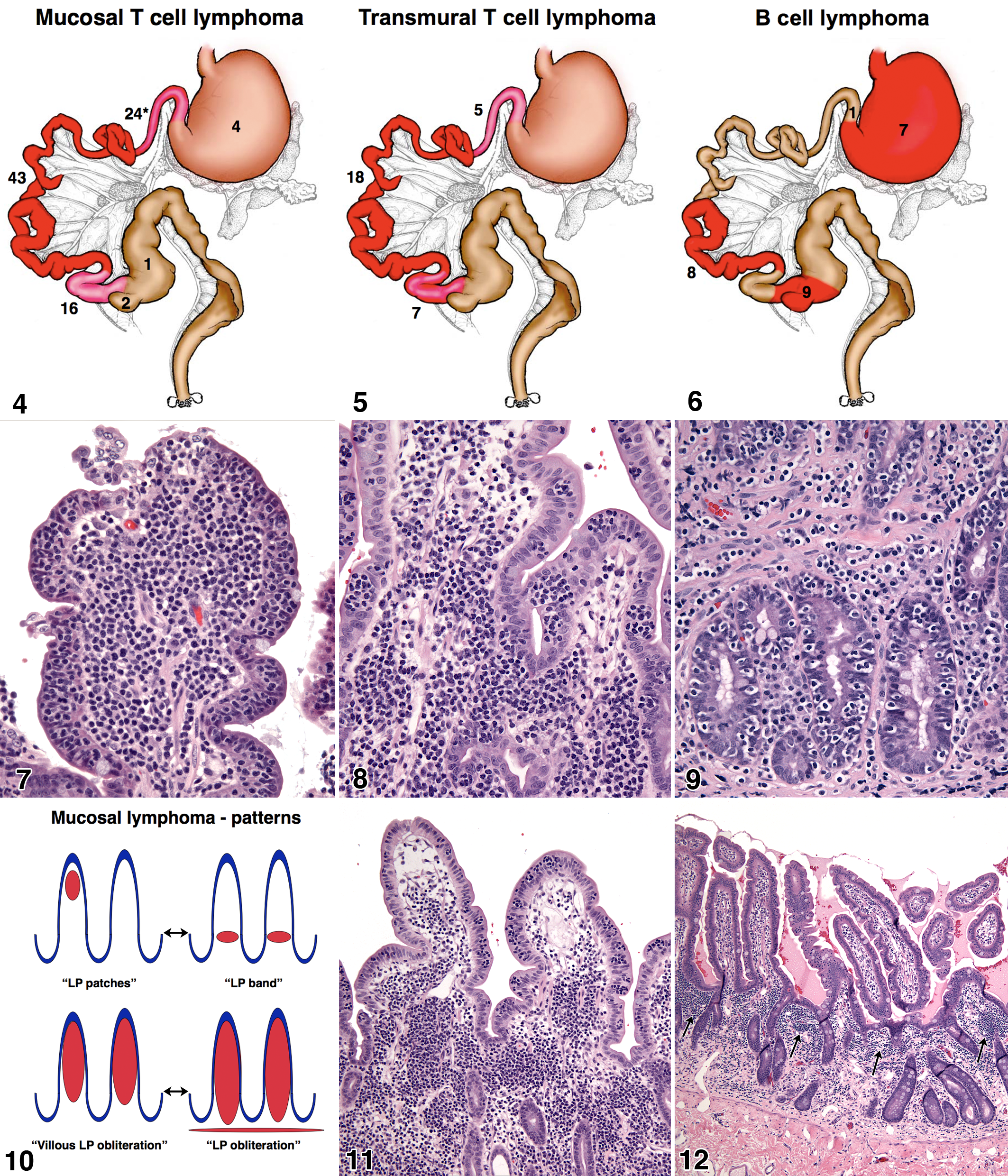
The jejunum was the most common intestinal segment affected in both mucosal and transmural T-cell lymphoma. However, lesions frequently involved more than 1 intestinal segment (Figs. 4, 5). Endoscopic biopsies were excluded from the topographical analysis, because they only sample the duodenum. Extension of mucosal T-cell lymphoma to the stomach and large bowel occurred with markedly lower frequency. Transmural T-cell lymphoma only occurred in the small intestine.
B-cell lymphomas were often multiple and occurred simultaneously in multiple locations in 9 of 19 cats (47%), for example, in the stomach and at the ileo–cecal–colic junction (Fig. 6). We did not encounter B-cell lymphomas that involved the duodenum except in 1 instance (cat No. 114) in which there was direct extension from lymphoma infiltrating the gastric pylorus. B-cell lymphomas occurred in the distal half of the small intestine from midjejunum to the ileum.
Mucosal Architecture in T-Cell Lymphoma
T-cell lymphomas were defined by CD3 expression and characteristic morphological features (see below) and initially arose in the diffuse MALT of the intestine. T-cell lymphomas arose in the IEC and the LPC.
Epitheliotropism was a dominant feature of T-cell lymphoma. Two patterns of T-cell epitheliotropism were encountered: diffuse lymphocytic infiltration of the IEC and discrete clusters of lymphocytes within the IEC (Figs. 7–9). Lymphocyte epitheliotropism occurred most commonly in the villous epithelium and less commonly in the crypt epithelium (Fig. 9). Epitheliotropic lymphocytic infiltrates, which exceeded normal IEL density, 3 occurred in 52 of the 84 cats (62%) with mucosal T-cell lymphoma and in 11 of the 19 cats (58%) with transmural T-cell lymphoma. The recognition of IELs was significantly enhanced in sections stained for CD3 expression.
Mucosal T-cell lymphomas, with and without marked epitheliotropism, involved the lamina propria of the small intestine in a variety of patterns, ranging from discrete, patchy, lymphocytic infiltrates to total effacement of the lamina propria (Fig. 10). In the least severe lesions, discrete regions of increased lymphocyte density occurred within the villous lamina propria; adjacent villi were often uninvolved (Fig. 11). Alternatively, infiltration occurred as a band of increased lymphocyte density that spanned the crypt–villous junction (Fig. 12). In more advanced lesions, dense lymphocytic infiltrates expanded the villous lamina propria (Fig. 13). The most severe lesions were associated with complete lymphocytic infiltration of the villous and crypt lamina propria with the formation of a lymphocytic band beneath the crypt epithelium but above the muscularis mucosae (Fig. 14). In the latter instances, marked mucosal changes, such as villous blunting, villous fusion, and crypt effacement, were evident (Fig. 14).
The lymphoid infiltrates in mucosal T-cell lymphoma were composed of small to intermediate-sized lymphocytes (nuclear diameter <2 red cell diameters) in 80 of 84 cats (Figs. 7, 8). In 2 instances each, the infiltrates consisted of large lymphocytes or large lymphocytes with eosinophilic cytoplasmic granules (LGL) (nuclear diameter >2 red cell diameters). Based on the above array of features, mucosal T-cell lymphoma closely matched the WHO entity enteropathy-associated T-cell lymphoma (EATCL) type II. 25
Transmural T-cell lymphomas of the small intestine occurred both focally and multifocally. Characteristically, the mucosal lamina propria was effaced and the submucosa was heavily infiltrated (Fig. 15). In the least severe transmural lesions, the lymphocytic infiltrate traversed the muscularis propria (tunica muscularis) perivascularly, leading to a trabecular pattern of involvement. In advanced lesions, coalescence of the lymphocytic infiltrate obliterated the muscularis propria and spilled into the serosa and adjacent mesentery. This often resulted in mass formation, intestinal obstruction, and even perforation with peritonitis. Mucosal changes such as villous blunting, villous fusion, and crypt effacement were most severe in transmural lymphomas (Fig. 15). Intestinal segments proximal and distal to transmural foci almost invariably manifested with mucosal lymphoma (Fig. 11).
The lymphoid infiltrates in transmural T-cell lymphoma were composed of large lymphocytes in 11 of 19 cats (Fig. 16, 17); the infiltrate was composed of small to intermediate-sized lymphocytes in the remainder. In 9 of 11 cats, the lymphoid infiltrate was characterized by the presence of eosinophilic cytoplasmic granules (Fig. 17), which are a feature of LGL. All 9 transmural LGL T-cell lymphomas expressed the cytotoxic granule protein, granzyme B (Fig. 18). Based on the above array of features, transmural T-cell lymphomas closely matched the WHO entity EATCL type I. 25
Mucosal Architecture in B-Cell Lymphoma
Gastric and intestinal B-cell lymphomas occurred as transmural lesions in 18 of 19 cats (95%). Neoplastic B cells expressed CD79a in all cases.
In the gastric mucosa, B-cell lymphomas often infiltrated the lamina propria without clear association with preexisting lymphoid follicles (Fig. 19). However, in the small intestine, cecum, and colon, B-cell lymphomas were often clearly associated with mucosal lymphoid follicles (Fig. 20). This association was obscured as foci of proliferation expanded, coalesced, and traversed the muscularis propria to manifest as transmural, diffuse, large B-cell lymphoma in both stomach and intestinal sites (Fig. 19). Incursion of neoplastic B cells into the overlying mucosal epithelium was an infrequent occurrence (cat No. 113).
B-cell lymphomas were classified as the WHO entity diffuse large B-cell lymphoma. 25 Cytologically, B-cell lymphomas most commonly had centroblastic nuclear morphology, in which multiple nucleoli were visible, often adjacent to the nuclear membrane. B-cell lymphomas of immunoblastic type occurred in only 2 of 19 cats (11%); single large central nucleoli predominated in these instances (Fig. 21).
Concurrent Lymphomas
We encountered cats that had more than 1 lymphoma in the gastrointestinal tract. Four cats (cat Nos. 103, 115, 116, and 117) had diffuse large B-cell lymphoma in the stomach and/or the cecum and colon. Additionally, these cats had small T-cell lymphoma of the diffuse MALT of the small intestine, which was confirmed by TCRG gene rearrangement analysis. One of these cats (cat No. 103) also had T-cell lymphoma in the stomach and large intestine adjacent to the foci of B-cell lymphoma (collision tumor). The existence of concurrent T-cell lymphomas originating from distinct T-cell clones was documented in 2 cats (cat Nos. 24 and 38). In each instance, a different T-cell clone (based on PCR product size) was found in the lamina propria of the stomach and the duodenum by analysis of TCRG gene rearrangement in both sites.
Molecular Clonality
Molecular clonality assessment of mucosal T-cell lymphoma revealed clonal rearrangement of TCRG in 66 of 84 cats (78%). Oligoclonal rearrangements were detected in 11 cats (13%). Polyclonal TCRG rearrangements were detected in the remaining 7 cats. Clonality assessment was particularly sensitive in the least severe lesions; only 1 of 21 cats returned a polyclonal result.
Molecular clonality assessment of transmural T-cell lymphoma revealed clonal rearrangement of TCRG in 15 of 19 cats (79%). Oligoclonal rearrangements were detected in 2 cats (11%). Polyclonal TCRG rearrangements were detected in the remaining 2 cats.
Molecular clonality assessment of gastrointestinal B-cell lymphoma revealed clonal rearrangement of IGH in 9 of 18 cats tested (50%). Pseudoclonality was detected in 7 of 18 cats (39%); this occurred when the primers failed to amplify tumor target DNA and, instead, randomly amplified target DNA from scant normal residual B cells. Pseudoclonality was only detectable with duplicate PCR reactions. Polyclonal IGH rearrangements were detected in the remaining 2 cats.
Discussion
We have clearly documented that mucosal T-cell lymphoma of small-cell type, defined by expression of CD3, is the dominant gastrointestinal lymphoma in aged cats (67% of all lymphomas). This lymphoma corresponded closely to human EATCL type II in the WHO classification. 25 Epitheliotropism (villous and/or crypt epithelium) was an important diagnostic feature when present (62%). Distinctive patterns of T-cell infiltration of the intestinal lamina propria were encountered with disease progression and were also diagnostically important. A diagnosis of mucosal T-cell lymphoma was best confirmed by T-cell antigen receptor gene rearrangement analysis; clonal or oligoclonal rearrangements for TCRG were detected in 91% of cats with morphological features of T-cell lymphoma.
Transmural lymphomas (B- or T-cell type) were readily diagnosed by morphological features alone. Immunohistochemistry to detect specific antigen receptor–associated membrane proteins, CD3 and CD79a, was only necessary to determine lineage. Transmural T-cell lymphoma, large-cell type, corresponded closely to human EATCL type I. 25 Together, mucosal and transmural T-cell lymphomas accounted for 83% of all gastrointestinal lymphomas (124 lymphomas). In previous studies, the true incidence of feline gastrointestinal T-cell lymphoma was likely underestimated because of difficulty in distinguishing mucosal T-cell lymphoma from lymphoplasmacytic IBD before widespread availability of molecular clonality assays.7,13,19,20,27,30 Indications that T-cell lymphomas may be more common in the small intestine of cats than previously realized have also been reported.20,34 However, the dominance of T-cell lymphoma in the small intestine of cats was recently confirmed in studies in which T-cell antigen receptor rearrangement analysis was used.18,35 Furthermore, the common occurrence of epitheliotropism in small intestinal T-cell lymphoma of cats has also been described.3,18 In a recent report, the low-grade nature of T-cell lymphoma of small-cell type and its association with prolonged survival (median 18.9 months) were highlighted. 15
T-cell lymphomas (mucosal and transmural) were almost exclusively localized to the small intestine, especially the jejunum. There were only 7 instances (7%) of T-cell lymphoma involving the stomach or large intestine. Trafficking of T cells to the intestinal mucosa is precisely regulated and is dependent in part upon T-cell expression of α4β7 (a mucosal homing integrin). Small intestinal T-cell homing, in particular, is largely dependent upon the additional expression of CCR9. 14 CCL25, the ligand of CCR9, is almost exclusively expressed in the epithelium of the small intestine. Entry of T cells into the epithelium is dependent on CCR9/CCL25 interaction. Epitheliotropism is associated with upregulation of CD103 (αE), which associates with β7 (αEβ7). E-cadherin, which is expressed by enterocytes, is a ligand of CD103 and may contribute to tethering of IELs within the IEC. 14 We have documented the expression of CD103 in IELs of feline small intestine and in epitheliotropic T-cell LGL lymphoma of the small intestine.22,33 Hence, expression of mucosal β7 integrins, especially α4 and CD103, and CCR9 in mucosal T-cell lymphoma would be expected to lead to localization within the lamina propria and IEC of the small intestine preferentially.
Epitheliotropism of T cells is a normal feature in the small intestine of many species including cats, and IELs are an important first line of defense of the intestinal mucosa. 12,23 IELs are also involved in immunoregulatory functions and serve to limit inflammation in an environment shared with many commensal microorganisms.4,10 The density of IELs has often been expressed as a percentage of enterocytes (ie number of IELs per 100 enterocytes). The density of IELs in normal cats as reported covers a wide range. In one study from the United Kingdom, feline IELs ranged from 40 to 80 per 100 villous enterocytes depending on the intestinal segment. 29 This range far exceeded the density of IELs (18 per 100 villous enterocytes) reported in a second study conducted in the United States. 3 A similar study from the United Kingdom conducted in dogs reported an average of 20 IELs per 100 villous enterocytes. 8 The reasons for these marked differences in the 2 feline studies are unknown but likely include the source of the “normal” cats, diet, and environmental factors. The distribution of IELs also differs markedly between villous and crypt epithelium. IEL counts in normal small intestine of cats and dogs were 4- to 10-fold higher in villous epithelium than in crypt epithelium.8,29
The normal IEL distribution pattern was echoed in T-cell lymphoma with prominent epitheliotropism, which occurred in about 60% of T-cell lymphoma cases (mucosal and transmural). Epitheliotropism was more frequent in villous epithelium and less in crypt epithelium. However, prominent crypt epitheliotropism by IELs was distinctly abnormal and readily recognizable, and it increased the likelihood that the disease process was epitheliotropic T-cell lymphoma. Epitheliotropism was manifest as discrete clusters of IELs or as a diffuse infiltrate of the villous and/or crypt epithelium. Clusters of IELs in villous or crypt epithelium were readily recognizable and increased the likelihood of lymphoma. The significance of intraepithelial lymphocyte clusters in establishing a diagnosis of intestinal lymphoma in cats has been previously emphasized.6,21 However, recognition that epitheliotropic lymphocytes were of T-cell lineage relied on studies that used immunohistochemical and immunofluorescence techniques.3,12,18,22,23 Diffuse villous epitheliotropism is more difficult to assess, and interobserver variability is high. However, if the number of IELs exceeds 40 per 100 enterocytes, there is higher likelihood of lymphoma. 3 Importantly, IELs are often underrecognized in HE-stained sections and definitely appear more numerous in CD3 stained sections; this has been previously noticed by others. 3
Epitheliotropism was not a prominent feature in about 40% of T-cell lymphoma cases. However, in both epitheliotropic and nonepitheliotropic T-cell lymphomas, we recognized distinctive patterns of lymphocytic infiltration of the lamina propria by small to intermediate-sized lymphocytes. These patterns were highly correlated with the presence of mucosal T-cell lymphoma and hence were diagnostically important. A characteristic of the least severe T-cell lymphoma (commonly referred to as “emerging”) was patchy infiltration of the lamina propria (and epithelium), such that some villi were infiltrated whereas adjacent villi were spared. In some instances, a band-like lymphocytic infiltration of the lamina propria, which spanned the crypt–villous junction, was observed in early mucosal T-cell lymphoma. These 2 patterns of lamina proprial infiltration have been portrayed as representing early or emerging intestinal lymphoma in cats,6,21 although formal proof of this concept would be difficult to achieve in a clinical setting. Heavy monomorphous lymphoid infiltrates of small to intermediate-sized cells were readily recognized as mucosal T-cell lymphoma when they effaced the villous lamina propria or the entire mucosa. Associated mucosal architectural alterations such as villous blunting and fusion and crypt effacement were only observed in advanced mucosal T-cell lymphoma. It is highly likely that continued expression of mucosal homing molecules by neoplastic T cells would reinforce these mucosal infiltrative patterns and result in an extended sojourn within the mucosal environment without spread to extraintestinal sites.
Analysis of TCRG rearrangement proved to be highly sensitive for detection of mucosal T-cell lymphoma regardless of the severity of the lesion. If clonal and oligoclonal results were combined, the sensitivity was 91%. In transmural T-cell lymphoma, the combined sensitivity (clonal plus oligoclonal results) was 90%. Failure to detect clonality (polyclonal result) can occur if gene segments, which are unaccounted for by the TCRG V and J primers, are used in the rearrangement process. Identification of all TCRG gene segments will only be possible once the feline genome is assembled. The interpretation of TCRG oligoclonality is complex. Oligoclonality is consistent with markedly reduced TCRG repertoire diversity in a chronic inflammatory background or emerging lymphoma with multiple neoplastic clones. 2 The latter is the more likely reason for an oligoclonal result in a lesion that is morphologically consistent with lymphoma. The presence of 2 neoplastic T-cell clones in topologically distinct sites in the gut of a single cat has been reported and also occurred in this series in 2 cats that had concurrent, distinct T-cell lymphomas in different locations. 18 If these clones occurred in the same site, an oligoclonal TCRG rearrangement could be observed in the event of biallelic rearrangement. An alternative explanation for apparent TCRG oligoclonality is the presence of multiple TCRG rearrangements on a single chromosome as occurs in the dog (manuscript in preparation). This is facilitated by the complex genomic structure of the canine TCRG locus, which is organized into 8 V–J–C cassettes, many of which are capable of independent rearrangement. 17 However, until the genomic structure of the feline TCRG locus is known, the likelihood of multiple rearrangements on a single chromosome is unknown.
Cats with mucosal T-cell lymphoma had a median survival of 29 months (n = 54). The vast majority of these cats (n = 50) had small-cell lymphoma (median survival 28 months). The favorable survival of cats with T-cell lymphoma of small-cell type has also been reported by others.6,15 Cats with transmural T-cell lymphoma (n = 13) had a much shorter median survival (1.5 months). The majority of these cats (n = 9) had large-cell lymphoma, especially of LGL type (n = 8). Transmural T-cell lymphoma of small-cell type occurred with low frequency (n = 4) and a wide survival range (3 days to 28 months); meaningful median survival data were not obtained for this group of cats. The poor median survival of cats with transmural LGL lymphoma is reinforced by the findings of our previous work in which the median survival of cats with LGL lymphoma and secondary leukemia was only 19 days. 22 Clearly, the presence of secondary leukemia had a markedly adverse effect on prognosis. Survival time may be influenced by choice of treatment; we did not attempt to correlate treatment regimens with survival time, because these data were not readily available for many of the cats, which were commonly treated by the referring veterinarian (remotely).
Feline gastrointestinal B-cell lymphomas were exclusively classified as diffuse, large B-cell lymphomas. 25 Evidence for progression from an initial mucosal marginal zone lymphoma was lacking. Mucosal marginal zone lymphomas in humans characteristically originate as perifollicular B cell expansions with prominent epitheliotropism; the latter was only observed in a single cat in the absence of perifollicular growth. 25 B-cell lymphomas occurred with higher frequency in the stomach and ileo–ceco–colic junction of cats. The latter sites are enriched for organized lymphoid tissues (mucosal lymphofollicular structures), and evidence of B-cell lymphoma could be found in lymphoid follicles adjacent to transmural lesions. Further indication of postgerminal center origin of gastrointestinal B-cell lymphoma was evident in the results of molecular clonality determination. Analysis of IGH rearrangement in feline gastrointestinal B-cell lymphoma was relatively insensitive (50%). This was likely attributable to somatic hypermutation of IGH V segments that occurs normally in B cells, which pass through follicular germinal centers and are subjected to affinity maturation of the immunoglobulin response to antigen.24,32 However, B-cell lymphomas in the stomach did not clearly originate in mucosal lymphofollicular structures, although it is possible that rapid transmural extension may have obscured these relationships.
Conclusion
We have shown that mucosal T-cell lymphoma of small to intermediate cell type (WHO EATCL type II) is the dominant lymphoma in the gastrointestinal tract of cats and has a relatively long median survival time. Mucosal T-cell lymphoma is characterized by distinctive patterns of infiltration of the villous and/or crypt epithelium and lamina propria. These patterns are readily recognizable in either endoscopic or laparoscopic (full-thickness) biopsy specimens and are highly correlated with clonal (or oligoclonal) TCRG gene rearrangement. However, despite the success in recognition of mucosal T-cell lymphoma in endoscopic biopsy specimens (largely from duodenum), it should be noted that the highest incidence of lymphoma was in the jejunum. Hence, an endoscopic biopsy procedure, despite its convenience, would potentially miss some mucosal T-cell lymphomas. We believe that mucosal T-cell lymphoma has been previously underestimated and that the diagnostic approach to this disease should include morphological assessment of mucosal architecture, enhanced by CD3/CD79a immunohistochemistry, and confirmation by TCRG gene rearrangement analysis in equivocal cases. Transmural T-cell and B-cell lymphomas are readily diagnosed by morphological assessment and immunohistochemistry for CD3/CD79a, usually without the need for molecular clonality analysis. Transmural T-cell lymphoma of large-cell type (WHO EATCL type I) is most often an LGL lymphoma; this is readily demonstrable with immunohistochemistry to detect the cytotoxic granule protein, granzyme B. Transmural T-cell lymphoma of large-cell type has a relatively short median survival time but is the least common T-cell lymphoma in the gastrointestinal tract of cats.
Footnotes
Acknowledgements
We thank Rick Hayes for assistance with artwork and Dr Bill Vernau for critical reading of the manuscript.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
