Abstract
Cerebellar abiotrophy, a premature degeneration of cerebellar neurons, has been described in most domestic animals. Affected animals typically present with progressive neurologic signs after a variable period of postnatal normalcy. This report describes the clinical, histopathologic, and immunohistochemical (IHC) findings of cerebellar abiotrophy in an alpaca. The alpaca developed intention tremors, hypermetria, and a wide-based stance at 1.5 years of age. Histologic lesions, confined to the cerebellar vermis, included marked absence of Purkinje cells, decreased granule cells, narrowing of the molecular layer, and thinning of white matter tracts consistent with abiotrophy. Increased cell processes in the molecular layer immunolabeled for glial fibrillary acidic protein, whereas immunoreactivity for neurofilament was reduced in the molecular layer and cerebellar folia white matter. To the investigators' knowledge, this is the first report of cerebellar abiotrophy in a camelid and the first documentation of IHC findings associated with this condition.
Cerebellar abiotrophy denotes premature degeneration of fully formed cerebellar neurons due to an intrinsic metabolic defect, often suspected to be inherited as an autosomal recessive condition.1, 11 This differs from hypoplasia, in which the cerebellum fails to form completely during development. Clinically, animals with cerebellar abiotrophy are born with normal neurologic function followed by delayed onset of clinical signs ranging from a few weeks to several years of age, corresponding to the onset of neuronal degeneration. The disease is progressive, although in some cases affected animals reach a stage at which clinical signs plateau.5, 11 Typically, Purkinje cells undergo primary degeneration; granule cell loss is considered secondary, as are reactive gliosis and Wallerian-type degeneration.1, 11 Cerebellar abiotrophy has been reported in most domestic species,11 although not in camelids, to our knowledge. The present report describes the clinical, microscopic, and immunohistochemical (IHC) features of a putative case of cerebellar abiotrophy in an alpaca.
A 1.5-year-old female alpaca presented to Purdue University Veterinary Teaching Hospital with a primary complaint of trembling of the head and neck and abnormal gait of 3 months' duration. The animal had been treated for suspected meningeal worm infestation without noticeable improvement. The neurologic examination showed normal behavior and no cranial nerve deficits. Subtle head tremors were present at rest and worsened when the animal reached for food. The alpaca would stand with a wide-based stance and, when walking, showed moderately severe ataxia and hypermetria. A cerebrospinal fluid (CSF) aspiration and a computed tomography (CT) scan were performed; the protein content of the CSF was 20 mg/dl (reference range <66.8 mg/dl) and there was 1 white blood cell/μl and 0 red blood cells.13 Serologic testing for antibodies against West Nile and Equine Herpes virus was negative by virus neutralization, and no viruses were isolated from the CSF. On CT, there was increased area of fluid opacity along the dorsal margin of the rostral cerebellum. This suggested a small cerebellum such as from hypoplasia, atrophy, or degeneration, or less likely a cystic area near the tentorium cerebelli. A tentative diagnosis of cerebellar degeneration (abiotrophy) was made based on the history and clinical findings. Neurologic signs persisted and the alpaca was humanely euthanized and submitted for necropsy examination at 23 months of age.
On gross examination of the brain, the dura mater was lightly adhered to the leptomeninges of the dorsal cerebellum and could be peeled away with gentle digital pressure. Although an overall reduction in cerebellar size was not visibly apparent, the folia of the vermis were mildly narrowed and slightly granular when compared with the smooth, rounded contour of the lateral hemisphere folia (Fig. 1). Approximately one third of the brain, including a portion of cerebellum, was frozen for possible ancillary tests; the remainder of the cerebrum, cerebellum, and brain stem were fixed in 10% neutral-buffered formalin. Selected samples were routinely processed, paraffin-embedded, sectioned at 5 μm, and stained with HE. Additional 5-μm-thick, formalin-fixed, paraffin-embedded sections were stained with luxol fast blue or immunolabeled for glial fibrillary acidic protein (GFAP, rabbit polyclonal antibody, Z0334; Dako Corp., Carpentaria, CA), neurofilament (mouse monoclonal antibody, clone SMI-31, SMI-31R; Covance, Berkley, CA), or synaptophysin (rabbit monoclonal antibody, clone SP-11, RM-9111; LabVision, Fremont, CA) according to previously published methods.8, 12 The unaffected lateral hemisphere provided an internal positive control.
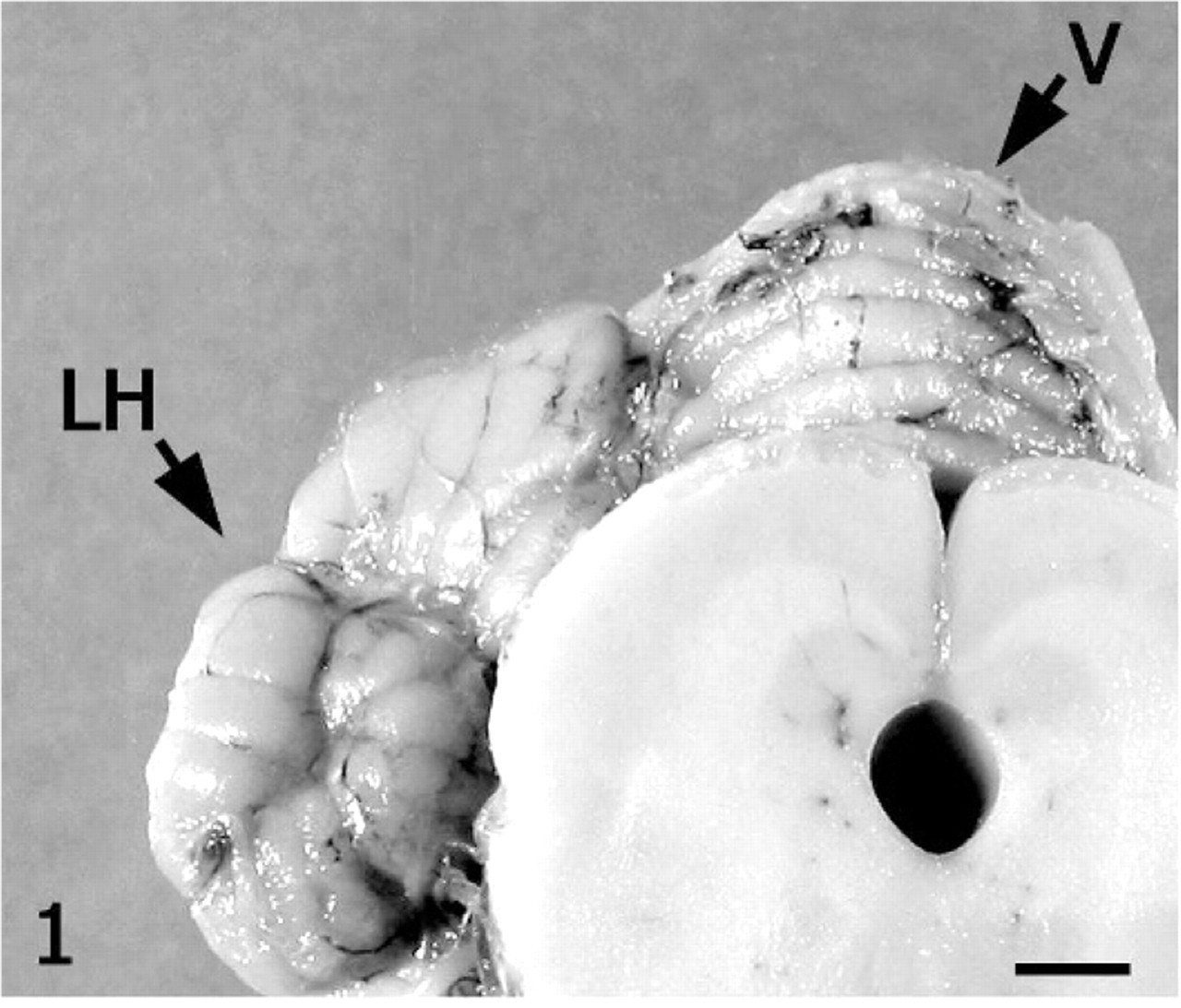
Cerebellum; alpaca. Folia of the vermis (V) are narrow and roughened compared to the right lateral hemisphere (LH). Bar = 3 cm.
On subgross assessment of HE cerebellar sections, the lateral hemisphere had distinct, trilaminar tinctorial features characteristic of normal cerebellar cortex (molecular and granular layers oriented around white matter), whereas folia forming the vermis were pale-stained with a poorly discernable granular layer (Fig. 2). The diameter of individual folia in the vermis was reduced, and sulci were widened. The transition from affected to unaffected folia was abrupt. Microscopically, the cerebellar cortex of the vermis had marked segmental depletion of Purkinje cells and moderate reduction in granule cells. Purkinje cells remaining in affected folia were widely separated and multifocally displayed karyolysis, swelling of the soma, and dispersion of Nissl substance (chromatolysis). The granular layer was hypocellular, with scattered karyorrhectic debris and swollen, round, homogeneously pale eosinophilic structures consistent with ghost cells or spheroids. The molecular layer was thin due to loss of neuronal processes and collapse of neuropil. White matter tracts in affected folia were narrow relative to unaffected cerebellum. Multiple nonstaining vacuoles in the white matter (dilated myelin sheaths) were either empty or contained swollen eosinophilic axons (spheroids) or individual phagocytic cells (digestion chambers) oriented in chains consistent with Wallerian-type degeneration. The examined lateral hemisphere was well-populated with both Purkinje cells and granule cells (Figs. 3, 4).
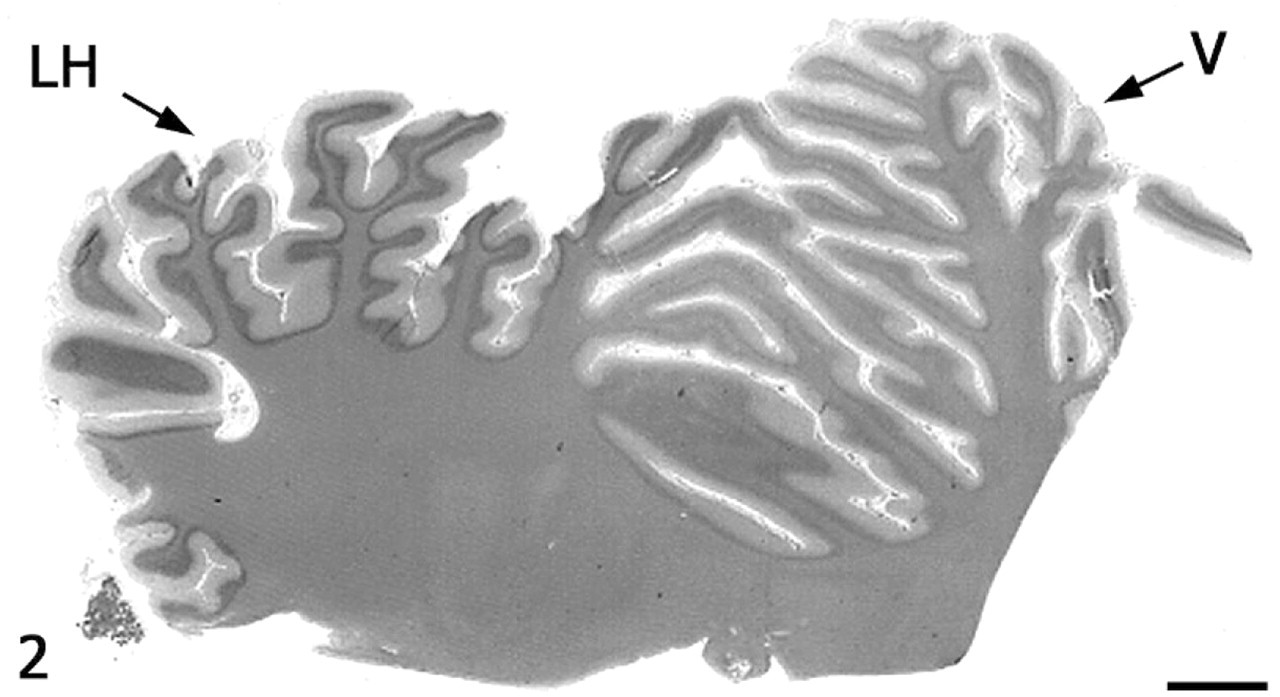
Cerebellum; alpaca. Folia of the vermis (V) lack the distinct tinctorial layering typical of unaffected cerebellar cortex in the lateral hemisphere (LH). HE. Bar = 3 cm.
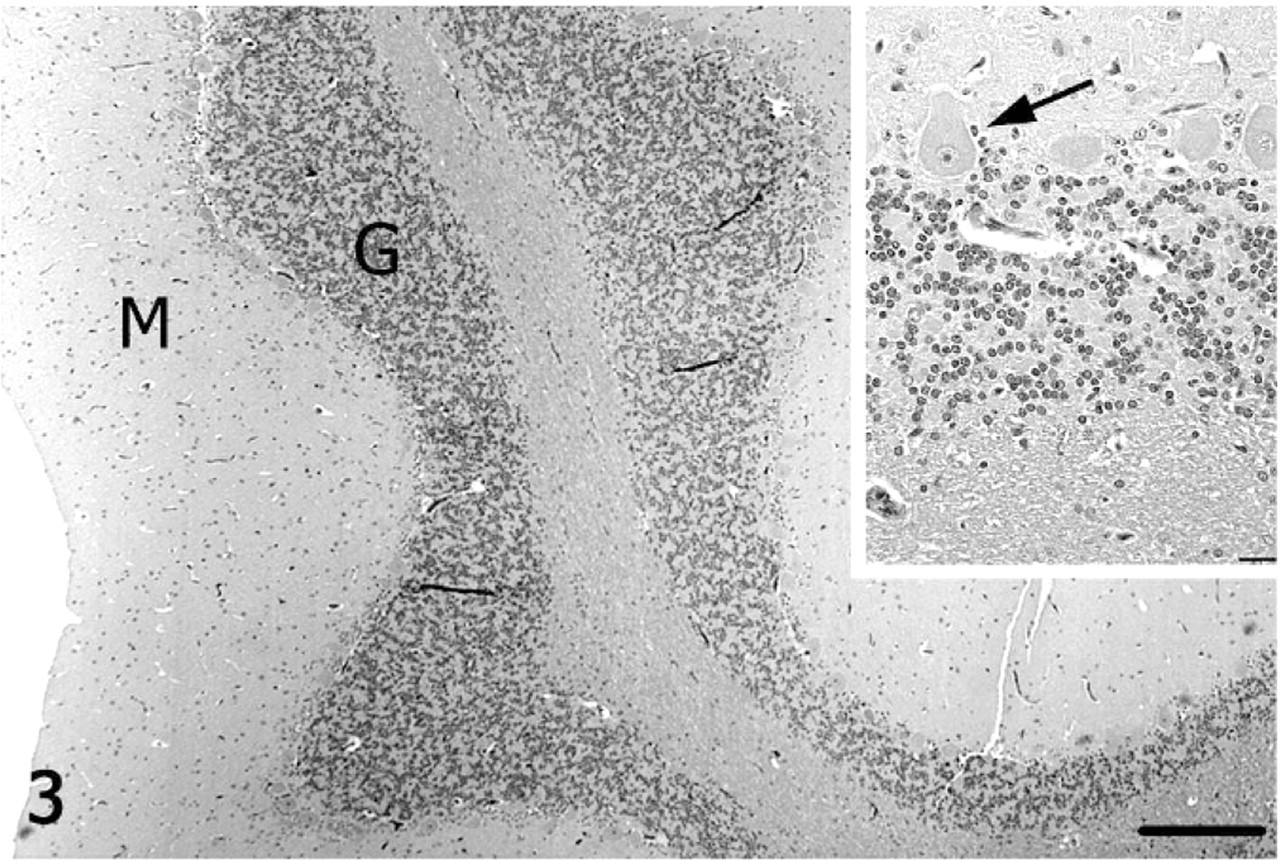
Cerebellum; alpaca. Unaffected cerebellar lateral hemisphere with well-populated granular (G) and Purkinje cell layers, and a well-defined molecular (M) layer. HE. Bar = 250 μm. Inset: Higher magnification of Purkinje cells (arrow). HE. Bar = 25 μm.
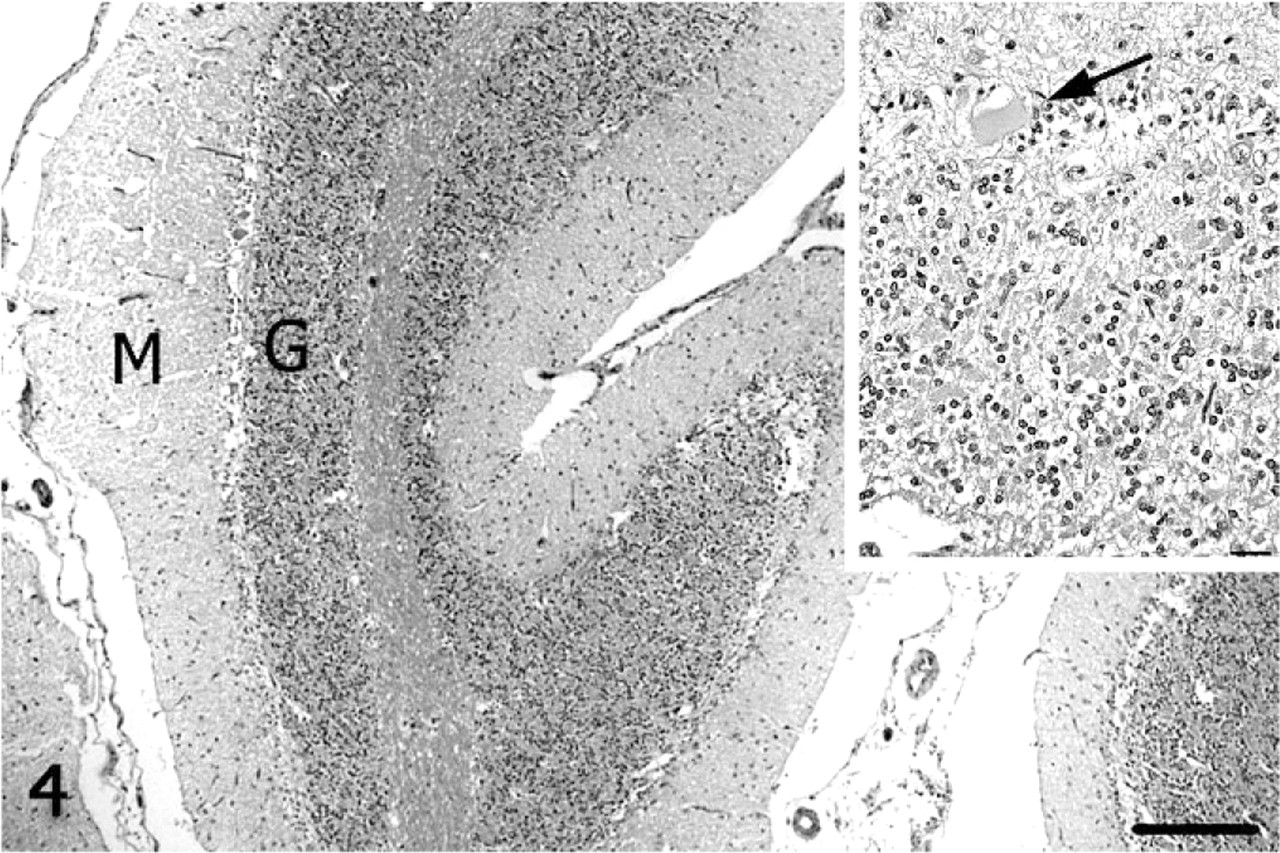
Cerebellum; alpaca. Folia of the vermis lack Purkinje cells and have thin granular (G) and molecular (M) layers. HE. Bar = 250 μm. Inset: Higher magnification of a degenerate Purkinje cell (arrow). HE. Bar = 25 μm.
With luxol fast blue, cerebellar white matter in the vermis had decreased staining amid nonstaining vacuoles compared with the lateral hemisphere, consistent with loss of myelin (Figs. 5, 6). Immunoreactivity for neurofilament was reduced in both the molecular layer and the white matter of the vermis compared with the lateral hemisphere (Figs. 7, 8). Purkinje cell soma in the lateral hemisphere had strong neurofilament immunolabeling, providing stark contrast to the marked, segmental depletion of this layer in the vermis. A marked increase in astrocyte cell processes (astrogliosis) was identified in the molecular layer of the vermis with GFAP immunolabeling. Astrocyte numbers were relatively increased (astrocytosis) in the granular and Purkinje cell layers (Figs. 9, 10). Intensity of synaptophysin immunoreactivity was mildly increased in the granular layer of the vermis compared with the lateral hemisphere, emphasizing the enhanced visualization of neuropil due to the decrease in granule cell bodies (not shown).
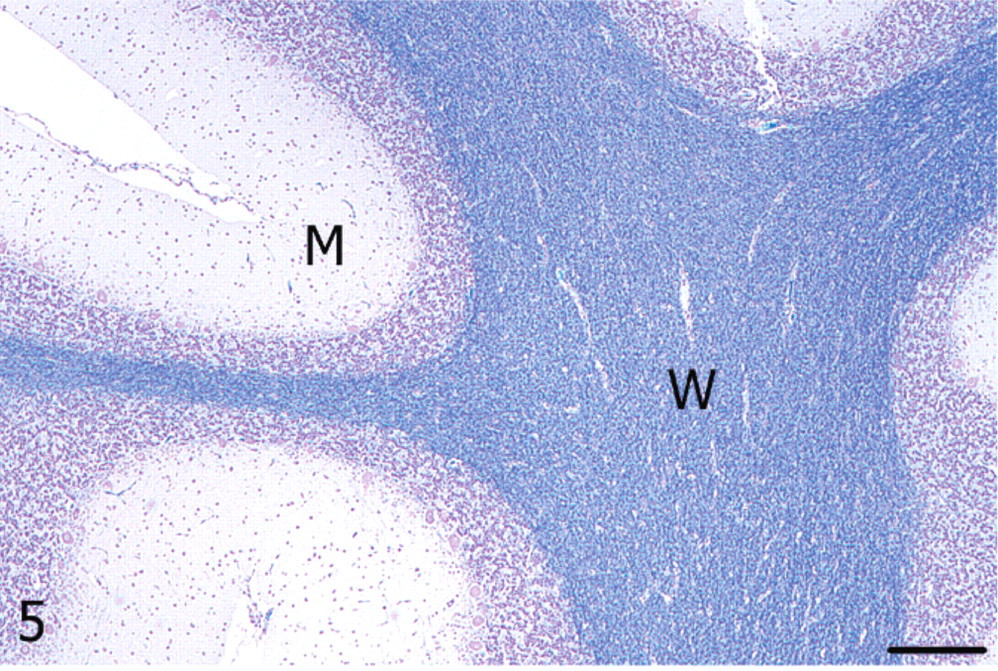
Cerebellum; alpaca. Unaffected cerebellar lateral hemisphere with dense myelin staining in the white matter (W); M = molecular layer. Luxol fast blue. Bar = 250 μm.
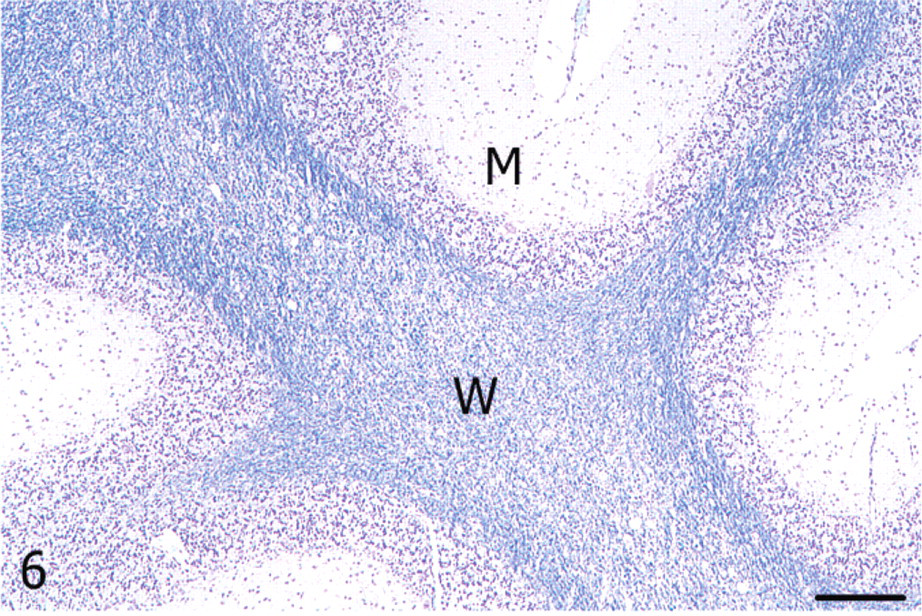
Cerebellum; alpaca. White matter (W) of the vermis showing vacuolation and reduced myelin staining; M = molecular layer. Luxol fast blue. Bar = 250 μm.
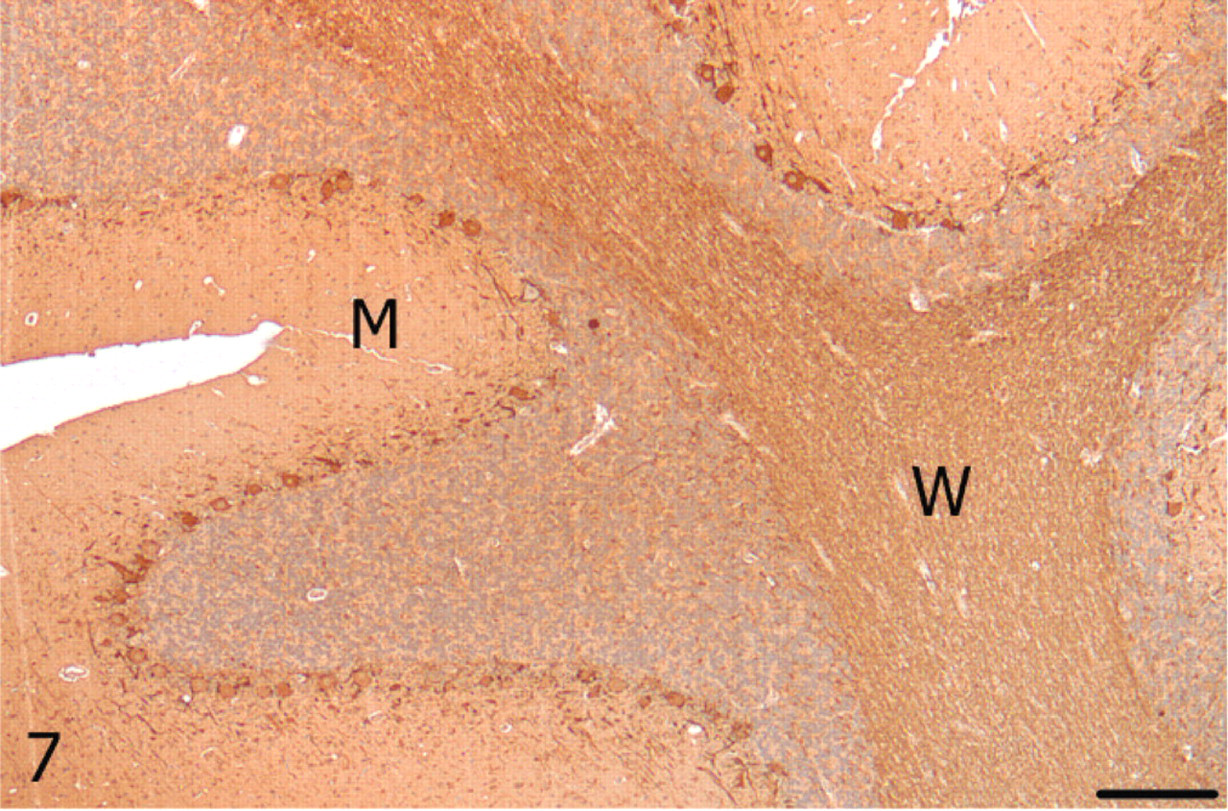
Cerebellum; alpaca. Unaffected cerebellar lateral hemisphere with strong neurofilament immunoreactivity in Purkinje cells and white matter (W), and lighter but diffuse immunolabeling in the molecular (M) layer. Neurofilament, streptavidin-biotin immunoperoxidase. Bar = 250 μm.
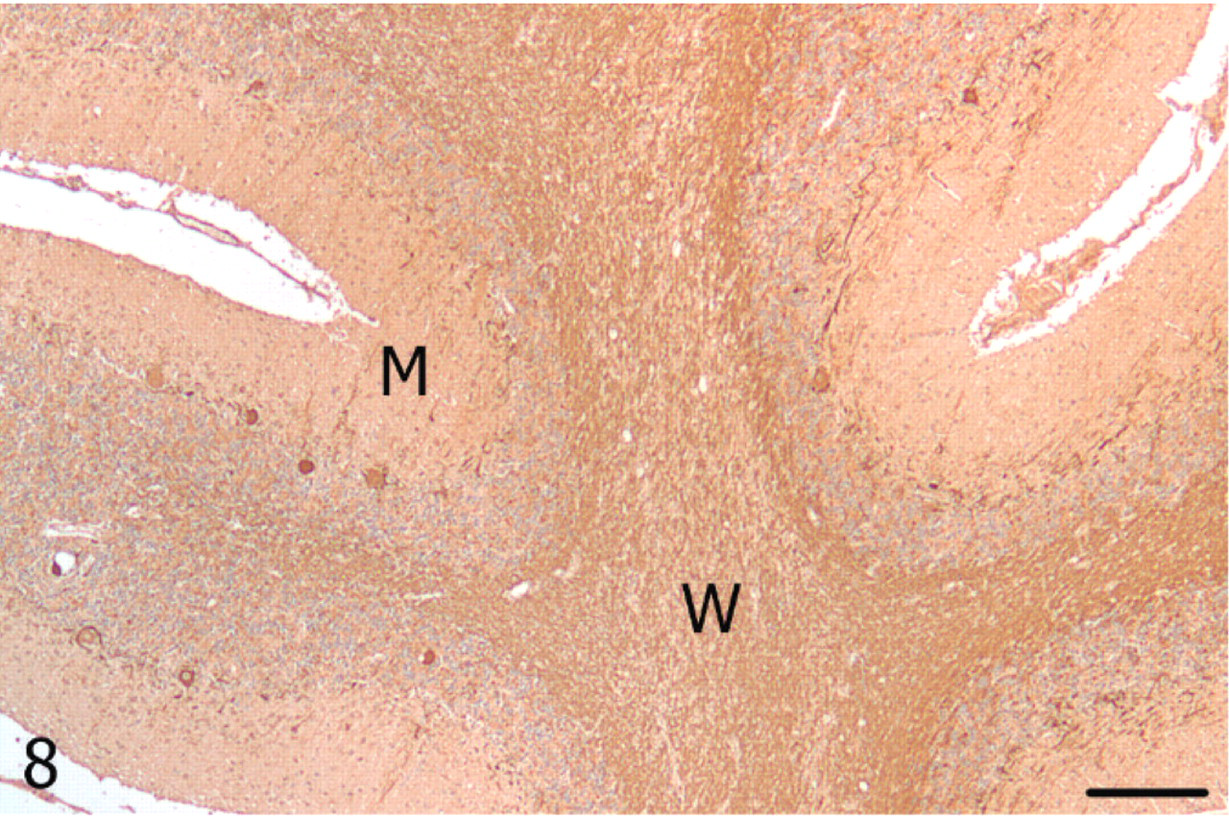
Cerebellum; alpaca. A paucity of Purkinje cells immunolabeling for neurofilament in the vermis, and reduced immunoreactivity in the molecular (M) layer and white matter (W). Neurofilament, streptavidin-biotin immunoperoxidase. Bar = 250 μm.
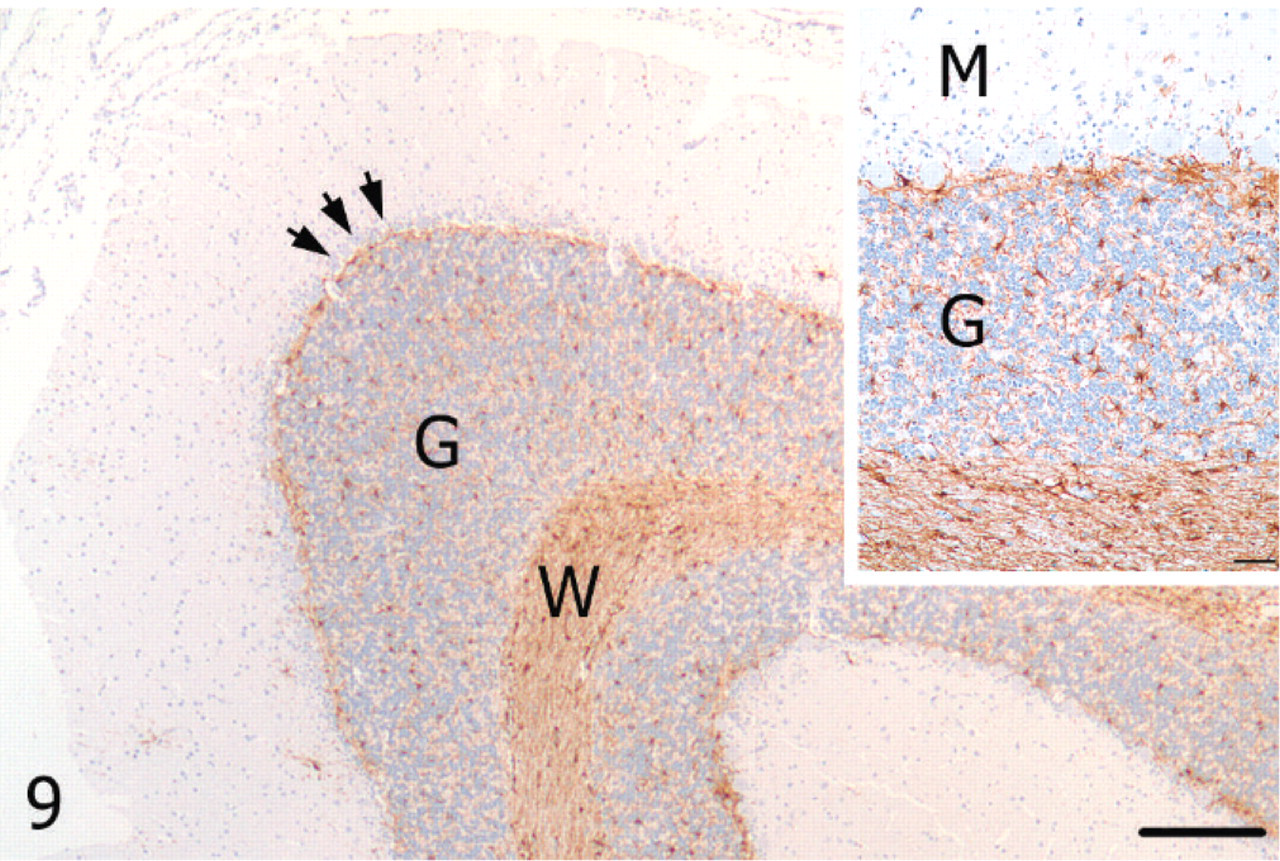
Cerebellum; alpaca. Unaffected cerebellar lateral hemisphere with immunoreactivity for GFAP in astrocytes of the Purkinje cell layer (arrows), granular (G) layer, and white matter (W). GFAP, streptavidin-biotin immunoperoxidase. Bar = 250 μm. Inset: Higher magnification of unaffected cerebellar cortex. M = molecular layer, G = granular layer. Bar = 50 μm.
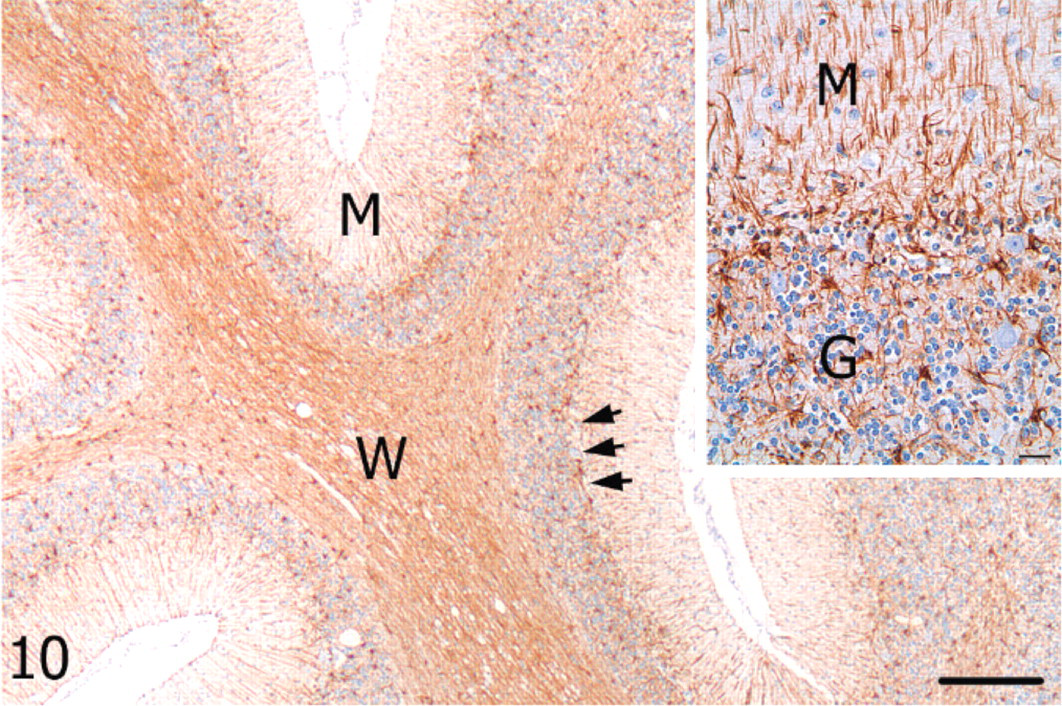
Cerebellum; alpaca. Marked astrogliosis evidenced by GFAP immunoreactivity in cell processes of the vermis molecular (M) layer and moderate astrocytosis of the Purkinje cell (arrows) and granular layers; W = white matter. GFAP, streptavidin-biotin immunoperoxidase. Bar = 250 μm. Inset: Higher magnification of cerebellar cortex from the vermis. M = molecular layer, G = granular layer. Bar = 25 μm.
The clinical history and histopathologic findings in this alpaca support a diagnosis of cerebellar abiotrophy. A second differential diagnosis was cerebellar degeneration, a term that describes a similar clinical syndrome and histologic lesion but implies an extrinsic etiology.3 In the present case, no other alpacas on the farm were affected to suggest a toxic or nutritional cause. In addition, no evidence of infectious disease was detected microscopically or with ancillary diagnostic tests. It is unknown whether normal offspring had previously been produced by the same breeding pair that sired this alpaca to evaluate potential inheritance. Histologic findings in this case were similar to those described in several breeds of sheep3, 5, 10 and calves.4, 6, 11 Interestingly, the vermis was particularly affected in the present alpaca, similar to reports in Wiltshire sheep3 and Holstein Friesian calves.4 Summers et al.11 suggested that abiotrophy may arise in the vermis and progress to other regions of the cerebellum. Also, these investigators noted the impressive transition from normal and abnormal cortex within a single folia, as seen in the present case.
Application of IHC markers in this alpaca provided additional information regarding lesion development. In particular, antibodies against GFAP depicted marked astrogliosis with processes extending into the molecular layer. However, astrocytosis within the Purkinje cell layer (Bergmann astrocytes) or in the granular layer was not as remarkable. Although many reports described gliosis as a secondary change in cerebellar abiotrophy,3–6, 11 visualization of these cells and their processes by GFAP immunolabeling emphasized that the marked cellular response was one of hypertrophy and proliferation of cell processes rather than hyperplasia. The mechanism of astroglial reaction demonstrated in the present case may reflect a specific spectrum of cytokines associated with cellular degeneration compared with the array of cytokines that might be associated with outright neuronal necrosis. Although a comparison to cerebellar hypoplasia was not made in this alpaca, GFAP immunoreactivity may provide differentiation between hypoplasia and early-onset abiotrophy, as gliosis in the former occurs mostly in the white matter.9 Because animals with cerebellar hypoplasia demonstrate neurologic signs at birth compared with the postnatal disease development in abiotrophy, the distinction between these two conditions would only be important when the onset of clinical signs in a young animal is unknown. Although loss of Purkinje cells and thinning of the molecular layer and white matter was apparent with routine stains in this case, neurofilament immunolabeling enhanced these lesions and may prove useful in mild or early cases of abiotrophy.
Neurologic disease in South American camelids can result from several infectious and noninfectious etiologies, including Eastern equine encephalitis virus, West Nile virus, Equine herpesvirus-1 virus, rabies virus, Listeria monocytogenes, Coccidioides immitis, meningeal worm (Parelaphostrongylus tenuis), trauma, and polioencephalomalacia (thiamine deficiency).2, 7, 14 The history, clinical findings, and neuroanatomic localization should help distinguish these conditions from abiotrophy. Based on the findings in the present case, cerebellar abiotrophy should be considered as a differential diagnosis for progressive neurologic disease in alpacas, and potentially other camelids. If a herd experiences multiple affected animals, pedigrees should be assessed to identify possible modes of inheritance. Ideally, molecular analysis to detect the intrinsic defect should be pursued. However, even with individual cases, producers should be informed of the likelihood that the condition is inherited before subsequent matings of the same breeding pair.
Footnotes
Acknowledgements
We thank the histology and immunohistochemistry laboratories of the Purdue University Animal Disease Diagnostic Laboratory for performing histologic and immunohistochemical procedures and Dr. Jacob J. Rohleder for the interpretation of the CT scan.
