Abstract
Clinical and pathologic features of neuronal ceroid-lipofuscinosis in a 4-month-old ferret are reported. Clinical signs including neurological symptoms appeared at 3 months of age and progressed rapidly. By magnetic resonance imaging, severe cerebral atrophy was recognized. Histopathologically, there was severe neuronal loss and diffuse astrogliosis with macrophage accumulations; lesions were found predominantly in the cerebral cortex. Intracytoplasmic pigments were observed in surviving neurons and macrophages throughout the brain. The pigments were intensely positive for periodic acid–Schiff, Luxol fast blue, and Sudan black B and exhibited a green autofluorescence. Electron microscopic examination revealed the accumulation of electron-dense granular material within lysosomes of neurons and macrophages. Immunohistochemically, a large number of saposin-positive granules accumulated in the neuronal cells, astrocytes, and macrophages of the lesions, but significant immunoreactivity for subunit c of mitochondrial adenosine triphosphate synthase was not observed. Based on these findings, the animal was diagnosed as affected by neuronal ceroid-lipofuscinosis.
Neuronal ceroid-lipofuscinosis (NCL) is the most commonly diagnosed lysosomal storage disease in both human and domestic animals.10,29 In general, NCL is clinically characterized by progressive, usually nonspecific neurological signs. Lysosomal accumulations of ceroid-lipofuscin in neurons, microglia, and macrophages are the most characteristic microscopic findings. The accumulated ceroid-lipofuscins are autofluorescent and are positive for periodic acid–Schiff (PAS), Luxol fast blue (LFB), Sudan black B, and the Ziehl-Neelsen acid fast stain. 29
Recent reports have revealed biochemical and genetic features of some of the diseases, and approximately 160 NCL-causing mutations have been identified in 9 human genes (CLN1, CLN2, CLN3, CLN5, CLN6, CLN7, CLN8, CLN10, and CLCN7). 10 Subunit c of mitochondrial adenosine triphosphate synthase (SCMAS) is the main storage material in the diseases caused by the mutations in CLN2, CLN3, CLN5, CLN6, CLN7, CLN8, and CLCN7, whereas the accumulation of saposins (A and D) is recognized in some subtypes of NCL (those affected by mutations in CLN1 and CLN10). 10 Clinical and pathological features of the disease have been reported in several animal species, such as dogs,1,2,13,14,18,19,22,23,26,28 cats,16,21 sheep,512,24,25 cattle,6,8,11 goats, 4 horses,9,31 and nonhuman primates. 29 Even in the veterinary field, mutations of causative genes have been identified: CLN5, 5 CLN6,24,25 and CL10 30 in sheep; CLN5 in cattle 8 ; and CLN2 (TPP1), 1 CLN5, 18 CLN8, 13 and CLN10 (CTSD) 2 in dogs.
Because ferrets (Mustela putorius furo) are popular companion animals, detailed knowledge of their diseases is needed in veterinary medicine. Neurological disorders including canine distemper virus (CDV) infection 27 and influenza infections combined with metabolic failure (such as Reye syndrome–like disease) have been reported in ferrets.3,20 However, only limited information concerning lysosomal storage diseases in ferrets including NCL 17 is available. This article describes the clinical and pathological features of a spontaneously occurring NCL in a ferret.
History
A spayed female ferret was imported and kept with other young ferrets. There was no information concerning the pedigree of the ferret. At 3 months of age, the ferret showed ataxia and was isolated from the others because it was suspected to have an infectious disease. The clinical signs progressed gradually with additional signs including weight loss, dysphagia, swimming-like movements, tremors, diarrhea, and dysstasia appearing by 4 months of age. No aggressive behavior was recognized during the clinical course. Blood biochemistry and radiographic examinations revealed no abnormalities, except for a mild increase in the activities of liver enzymes (aspartate aminotransferase 301 units per liter, alanine aminotransferase 277 units per liter, and alkalinephosphatase 469 units per liter
Pathologic Findings
Severe lesions were found in the cerebrum; these lesions were characterized by marked cortical atrophy, neuronal loss, and diffuse astrogliosis (Fig. 1). Surviving neurons were shrunken, and these cells and macrophages sometimes contained pale pink intracytoplasmic pigments (Fig. 2). The pigments were positive for PAS (Fig. 3a), Sudan black B (Fig. 3b), and LFB (Fig. 3c) but negative for Ziehl-Neelsen. Those pigments exhibited a green autofluorescence under fluorescent microscopy (Fig. 4d). Electron microscopic examinations revealed the accumulation of electron-dense granular materials, approximately 100 to 500 nm in diameter, within the lysosomes of neurons and microglia (Figs. 4a and 4b). These characteristics of the pigments were consistent with those of ceroid-lipofuscin. Proliferating astrocytes exhibited morphological features of gemistocytic astrocytes with abundant eosinophilic cytoplasm and intense immunoreactivity to GFAP (Fig. 5a). A large number of Iba-1– and HLA-DR–positive microglia and macrophages were also diffusely present throughout the cerebral cortex (Fig. 5b). These phagocytic cells were filled with ceroid-lipofuscin. The immunostaining for NF revealed a marked decrease in axons and neurons in the cerebral cortex. In the hippocampus, thalamus, and brainstem, neurons were not diminished but contained variably intense intracytoplasmic accumulations of ceroid-lipofuscin. Astrocytes and microglia were moderately abundant in these regions; however, lesions were very mild compared with those in the cerebral cortex. In the cerebellum, there was a moderate decrease in numbers of Purkinje cells and granule cells with a mild to moderate diffuse accumulation of microglia and macrophages. There were ceroid-lipofuscin accumulations in Purkinje cells and phagocytes. Astrocytosis in the cerebellum was less prominent than that seen in the cerebrum. No CDV antigens were detected in the central nervous system (CNS) tissues.
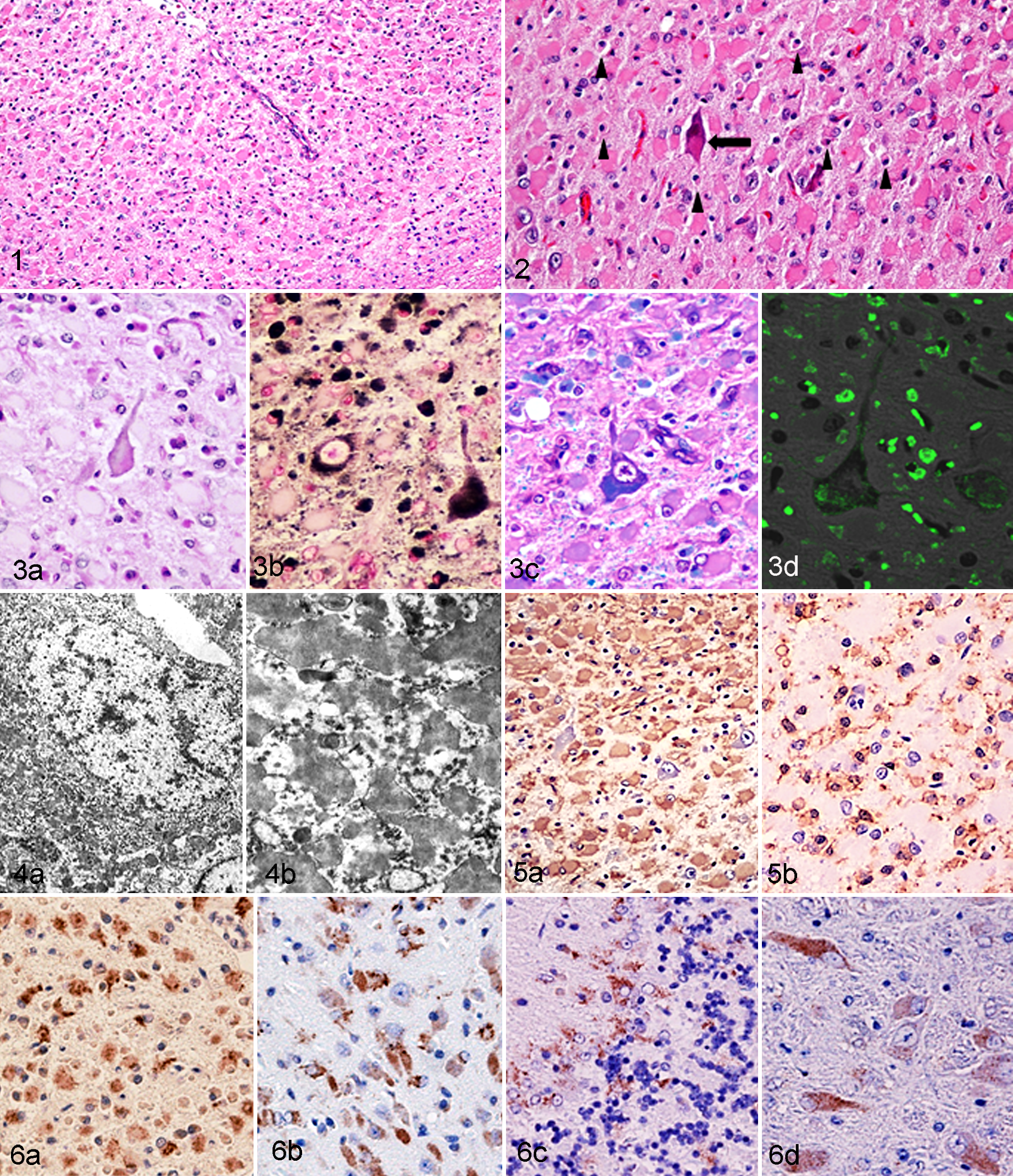
Ceroid-lipofuscin–laden macrophages were also observed in other visceral organs. These cells accumulated around the blood vessels and bronchi in the lungs and were multifocally present within hepatic sinusoids. Similar cells were observed in splenic and lymph node sinuses. There were no significant lesions in the heart, gastrointestinal tract, pancreas, kidneys, or urinary bladder.
After immunostaining for SCMAS, no significant immune-positive material was seen in the brain, lungs, or liver of the ferret, although in the control tissues (brain of NCL dog), many SCMAS-positive granules were found in macrophages and neurons that contained ceroid-lipofuscin. Saposin immunostaining revealed numerous immune-positive granules in the cytoplasm of neurons, astrocytes, microglia, and macrophages in the brain of the ferret. The number of saposin-positive granules increased with the degree of severity of the lesions. In the cerebral cortex, numerous saposin-positive granules were distributed mainly in microglia, macrophages, and astrocytes (Fig. 6a), and in the areas with mild lesions those granules were found also in surviving neurons (Fig. 6b). In the cerebellum, fine saposin-positive granules were mostly distributed in the cytoplasm of Purkinje cells, basket cells, and macrophages in the Purkinje cell layer (Fig. 6c). In the other brain regions, such as medulla oblongata, a small number of fine saposin-positive granules were found within the cytoplasm of neurons (Fig. 6d). In the brain of the NCL dog, saposin-positive granules were also found in the surviving neurons, but the number of affected cells was much less than those seen in the ferret brain, especially in the cerebrum. In the lungs and liver, there was no significant immunoreactivity for SCMAS or saposin. In the brains of both ferret and dog examined, cathepsin D immunostaining revealed intensely staining intracytoplasmic granules within the surviving neuron, whereas the immunopositive materials were reduced in the severely affected regions of the brain attributable to neuronal loss.
Discussion
Based on the clinical and pathological features in the present study, the ferret was diagnosed as affected by NCL. Clinical features of this ferret, such as onset at an early age and progressive neurological signs, are consistent with those of some types NCL reported in humans and other animals.10,29 Antemortem MRI examination revealed severe cerebral atrophy with a marked ventricular dilation. The atrophic cerebrum was characterized by ceroid-lipofuscin deposition as well as very severe astrogliosis and neuronal loss. In dogs affected by NCL, brain atrophy with ventricular dilation has been observed by MRI examinations, 14 although these features also occur in aged dogs infected with CDV, a condition commonly termed “old dog encephalitis.” 7 However, early clinical onset, histopathological features, and absence of CDV antigens in the CNS rule out the possibility of the viral infection in the present case.
In the described ferret, the most prominent pathologic features are astrocytosis and severe neuronal loss in the cerebrum. The infiltration of microglia and macrophages was most severe in the cerebral cortex compared with other CNS regions. Such lesions might result from a severe inflammatory reaction rather than a direct effect of the neuronal ceroid-lipofuscin, because accumulation of the pigment was commonly observed throughout the CNS. In humans, different distribution patterns of lesions and severity of neuronal degeneration and glial activation among NCL subtypes have been recognized. 10 Although the subtypes have not been as well elucidated in animals affected by NCL, severe cerebral atrophy was reported in NCL of South Hampshire sheep affected by the mutation of the ovine NCL6 gene.24,25 Oswald et al 25 suggested that prominent activation of astrocytes and microglia precedes neuronal degeneration and that in this ovine NCL, the glial reaction is more important for neuronal loss than ceroid-lipofuscin accumulation. A similar hypothesis might explain the brain lesions seen in the affected ferret.
The results of immunohistochemical studies for SCMAS and saposin may indicate that the major storage materials in the brain of the ferret are saposins, as seen in humans with mutations in CLN1 or CLN10 10 or the NCL affecting Miniature Schnauzers. 26 Because the SCMAS antibody recognizes an epitope that is well preserved among mammals, 15 the lack of significant immunoreactivity for SCMAS in the brain of the ferret case is not likely due to species differences. Numerous SCMAS-immunopositive granules were found in the brain of the Border Collie dog affected by NCL caused by the mutation of canine homologue to CLN5, 18 whereas the number of saposin-immunopositive granules was much less than that seen in the ferret brain. In both humans and animals affected by some types of NCL, saposins A and D have been shown to be the major materials stored.10,26,30 Because the saposin antibody used in this study could not precisely distinguish between saposin A, B, C, and D, further studies using more specific antibodies available for paraffin sections will be needed to confirm the specific saposins stored in the affected ferret. A deficiency of cathepsin D encoded by a mutated CLN10 gene has been known to cause NCL in both humans and animals.2.10,30 In this study, cathepsin D immunostaining revealed intensely staining intracytoplasmic granules in surviving neurons throughout the brains of both the affected canine control tissues and the affected ferret. These findings may be due to the normal distribution of this protease in the ferret. Ceroid-lipofuscin–laden macrophages were observed also in other visceral organs, including the lung, liver, spleen, and lymph nodes, whereas those were negative for both saposins and SCMAS. These findings may indicate that these pigment-laden macrophages accumulate because of different pathological mechanisms.
Further studies, especially those focused on the type of NCL caused by the mutation of CLN1 gene, will be necessary to identify complete pathogenesis of this disease in ferrets. Although the biochemical and genetic characteristics remain unclear, the present study has more clearly demonstrated the unique clinical and pathological features of NCL in the ferret.
Footnotes
Acknowledgement
We extend special thanks to Dr Junji Ezaki, Department of Biochemistry, School of Medicine, Juntendo University, for kindly providing rabbit antiserum against SCMAS and scientific information concerning biochemical features of human NCL.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
