Abstract
A 3-month-old suckled beef calf from the west coast of Scotland showed neurologic clinical signs for 1 week and was euthanized after failing to respond to treatment. Blood and tissue samples, including the brain, were submitted for diagnosis. Histologic examination of the brain showed neuronal chromatolysis and necrosis in the hind brain and loss of Purkinje cells in the cerebellum, accompanied by mild nonsuppurative encephalitis in the hind brain with a striking lack of inflammation in the cerebellar layers. Other microscopic lesions present were mild nonsuppurative meningitis with perivascular cuffs, diffuse hypergliosis, and occasional foci of neuronophagia. Polymerase chain reaction amplification of viral nucleic acids and specific immunohistochemical labeling allowed the identification of louping ill virus, and serology showed high titers of immunoglobulin M, indicating a recent infection.
Keywords
A 3-month-old female Limousin cross suckled calf showed neurologic clinical signs, including vertical nystagmus, hyperesthesia, and hyperactivity to external stimuli and occasional episodes of opisthotonos for 5 to 7 days. This animal had suffered an episode of diarrhea at 3 weeks of age but no other illness. Sudden deaths due to hypomagnesemia had occurred in 2 two-month-old calves from this herd 4 months previously.
The farm comprised two holding areas, one of which (holding A) is located in an area of hill ground considered to have high numbers of sheep ticks (Ixodes ricinus). The affected calf was a twin and was born at the other holding (holding B), where exposure to ticks is regarded as being less frequent owing to low land localization, but it was moved to holding A at 5 days of age to be adopted by a cow whose calf had died. The animals at holding A were treated with acaricidal spot-on preparations because of the presence of sheep ticks, but this calf was not treated as such, because she was born in the less-exposed holding (holding B). The affected farm, on the island of Islay off the west coast of Scotland, has sheep grazing with the cattle, along with wild deer that occasionally graze close to the farm animals.
At the onset of the clinical signs, the calf was treated empirically with oxytetracycline, corticosteroids, and a multivitamin supplement. No improvement was seen 4 days after treatment, and for humane reasons, the calf was euthanized by an overdose of intravenous barbiturate. Necropsy was performed by the local veterinary practice. No gross abnormalities or ticks were observed, and a neurologic condition involving the brain was suspected. Serum, vitreous humor, bone, and kidney samples were taken for biochemical and serological analysis and bacterial culture. The brain was removed and fixed in 10% formalin for histologic examination, along with samples of internal organs (lung, liver, spleen, kidney, heart, thymus, and small intestine).
Differential Diagnoses
Central nervous clinical signs in young calves can be caused by a number of conditions, depending on geographic location and management of the affected herd. In Scotland, the most common neurologic diseases are bacterial meningitis, thrombotic meningoencephalitis, focal symmetrical encephalomalacia caused by the epsilon toxin of Clostridium perfringens type D, and polioencephalomalacia due to induced thiamine deficiency or lead poisoning. Other possible causes affecting calves less frequently were considered, including listeriosis, louping ill, malignant catarrhal fever, and infection with bovine herpesvirus type 5. Although clinical signs were not suggestive of hypomagnesemia, it was included in the differential diagnosis because previous cases had occurred in this herd.
Microscopic and Immunohistochemical Findings
The major microscopic lesion in the brain of this calf was neuronal necrosis. Mild inflammatory changes were also present, as denoted by lymphoplasmacytic perivascular cuffs, diffuse hypergliosis, and glial foci (Fig. 1). Necrotic neurons were present in several nuclei throughout the brain, most frequently in the hind brain. The majority of neurons within the affected nucleus were frequently damaged, although in a small number of nuclei, only one or a small number of neurons were affected. Neuronal necrosis was most evident in the putamen and the red and vestibular nuclei but was also present in the Purkinje cell layer of the cerebellum, where these cells either appeared shrunken and eosinophilic with pyknotic nuclei or were absent from the Purkinje cell layer (Fig. 2). In brain nuclei, affected neurons were swollen with pale eosinophilic cytoplasm and dissolution of the Nissl substance (neuronal chromatolysis). In the majority of these neurons, no nuclear material was visible, but in some of them, the nucleus was displaced to the periphery. Neuronophagia, as denoted by the remnants of neurons surrounded or obscured by inflammatory cells, was not common but, when present, always appeared in those nuclei showing several necrotic neurons (Fig. 3). Focal gliosis was present in relation to these nuclei but absent in the cerebellum. Swollen axonal sheaths causing white matter vacuolation were present in the reticular formation at the pons and medulla oblongata. These contained swollen and rounded axons (spheroids) and single or occasionally multiple macrophages (digestion chambers), or they appeared as empty vacuoles (Fig. 3). Thin perivascular cuffs formed by one or two layers of lymphocytes, plasma cells, and macrophages were present in all the areas studied. In the meninges, inflammatory infiltrate was scarce and appeared mainly adjacent to blood vessels in the in the depths of the sulci of the parietal and frontal cortex. This infiltrate was formed by lymphocytes, plasma cells, and macrophages. There were no significant lesions in the samples examined from the other organs.
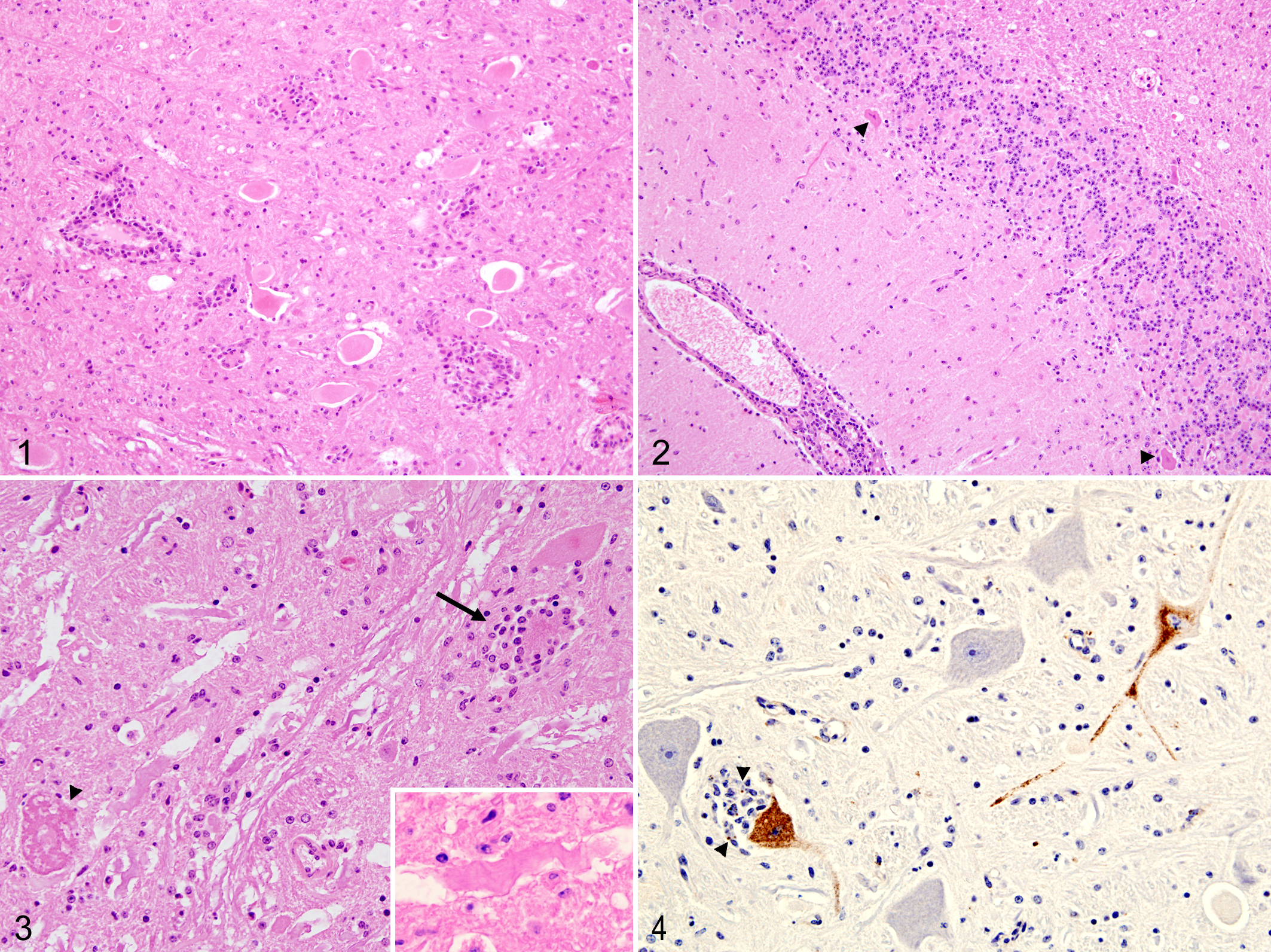
Immunohistochemical labeling of paraffin-embedded sections from areas of severe neuronal lesions (internal capsule, midbrain, pons, and medulla oblongata) was performed using a pool of monoclonal antibodies against the E and NS1 proteins of louping ill virus, as previously described. 2 Although numerous neurons showed degeneration and occasional neuronophagia, only a few were positively labeled within midbrain, pons, and medulla oblongata, with no positive labeling in the internal capsule. Positive labeling appeared as a fine brownish granular deposit in the cytoplasm of neurones. Interestingly, it was observed in both microscopically affected and nonaffected neurons but always within nuclei showing several degenerate neurons. Positive signal was also seen in the cytoplasm of phagocytes within neuronophagic nodules, presumably as a consequence of phagocytosed viral antigen associated with neuronal debris (Fig. 4).
Laboratory Findings
Analysis of calcium and magnesium levels in the vitreous humor, calcium:magnesium ratio in bone, and lead level in the kidney showed them all to be within the normal reference ranges.
Postmortem serum antibody level to louping ill virus was measured by hemagglutination inhibition test. 12 The predominance of immunoglobulin M (IgM) was estimated by heat inactivation of IgM in serum at 64.5°C for 30 minutes; this method has been shown to remove IgM from the sample, as confirmed by immunoelectrophoresis, 11 thus allowing estimation of IgM predominance by comparison of total hemagglutination inhibition (HAI) activity in non-heat-treated serum with the HAI activity in heat-treated serum. In this case, the non-heat-treated HAI titer was 1/640, in comparison with the heat-treated titer, 1/20, a 5-fold difference consistent with IgM predominance and supportive of recent infection. To further confirm louping ill infection, total RNA was extracted from formalin-fixed paraffin-embedded sections of brain by a commercial kit (RecoverAll, Ambion, Austin, TX), and louping ill viral RNA was detected by real-time reverse transcription polymerase chain reaction 9 in all three regions examined (midbrain, pons, and medulla oblongata).
Diagnosis
The histopathologic diagnosis was severe neuronal necrosis, neuronophagia, and axonal degeneration with subacute, moderate, diffuse nonsuppurative encephalitis and meningitis. These lesions are consistent with a neuronotropic viral infection, such as louping ill virus. Positive immunohistochemistry labeling within neurons and high levels of anti–louping ill virus IgM established that a virus belonging to the Flavivirus genus was the likely causative agent of the lesions—probably, louping ill virus. Final confirmation and definitive etiologic diagnosis was supported by polymerase chain reaction amplification of viral RNA at the site of neurologic lesions.
Discussion
Louping ill is a disease primarily affecting sheep in the United Kingdom, although it also causes high levels of mortality in red grouse chicks. Other species can be affected, including humans, as well as several wild or domestic animals, such as horses, pigs, and dogs. The causative agent of louping ill is a virus belonging to the mammalian tick-borne virus group of the Flavivirus genus and is closely related to the tick-borne encephalitis virus, an important human pathogen. Louping ill virus is normally transmitted by the sheep tick (Ixodes ricinus); therefore, the geographic distribution of the disease is closely linked to the habitat of this species. Louping ill is primarily restricted to certain areas of sheep tick habitat within the British Isles, where it has been present for more than 200 years. In recent years, a similar disease caused by a closely related flavivirus has been described in Norway, Spain, Turkey, and Bulgaria. 10 There have been recent reports of an increased incidence of louping ill diagnoses in Scotland, 1 which may be associated with alteration in tick populations with regard to both range and abundance. 14
The movement of this animal from an environment with low exposure to ticks (ie, holding B) to one with a high likelihood of tick exposure (ie, holding A), with the absence of preventive acaricidal treatment, could explain why this animal became infected. The cograzing of cattle with sheep and deer would explain the source of sheep ticks in the area, given that their abundance is strongly influenced by the numbers of these ruminant host species present in the area. 7
Experimentally, subcutaneous inoculation of louping ill virus in cattle can cause fatal disease. 13 However, infection does not always result in severe clinical disease. Serologic evidence indicates that in Great Britain, louping ill infection in cattle does naturally occur, although it is uncommon and only a minority of infected animals develop clinical signs. Across the United Kingdom, 150 seropositive cattle were detected between 2005 and 2010 in 353 blood samples submitted from animals suspected of having louping ill infection based on clinical signs (K. Willoughby, personal communication). It remains unclear why this calf developed the disease after the infection, but a lack of colostral antibodies could have played a role, given that the dam was in the low-exposure holding (holding B). It has already been suggested that outbreaks in cattle may occur when susceptible, unexposed stock are moved to tick-infested areas. 17
The distribution and characteristics of the encephalitis found in this case are similar to those described in natural or experimental cases of louping ill in sheep4,15 and experimental louping ill virus infection in cattle. 13 However, the inflammatory component was less severe and the neuronal necrosis more frequent than that described in those reports. This probably reflects a specific stage of the disease rather than differences in pathogenesis with sheep or experimental infections of cattle. The relatively high number of necrotic Purkinje cells in the cerebellum, compared to descriptions in natural cases of ovine louping ill, 5 is of note and suggests that in cattle these cells may be more susceptible to damage caused by the virus than in sheep.
The degeneration and death of the neurons in louping ill virus infection is thought to be a direct effect of viral infection of the cell. 5 The relative lack of neurons positively immunolabeled as a percentage of the number of neurons showing degenerative changes may be due to the lack of sensitivity of the monoclonal antibodies used for immunohistochemistry. Similar results have been described in natural cases of ovine louping ill where the amount of viral antigen was inversely proportional to the amount of inflammatory changes, suggesting that virus was cleared from the nervous tissue by the inflammation as the disease progressed. 16 Another possible explanation is that neuronal death in louping ill may occur via a bystander neurotoxic mechanism, with no actual infection of the neurons. Such mechanisms have been shown to occur through the release of proinflammatory cytokines in Japanese encephalitis, another flaviviral infection, 6 or by dysregulation of astrocyte-mediated glutamate transport, as in Sindbis virus encephalitis. 3
This case describes the findings in a naturally occurring louping ill infection of a calf. It also highlights the importance of taking protective measures, either acaricidal or vaccination, against louping ill infection when moving susceptible animals to regions with high levels of sheep ticks in areas where louping ill is known to be present. This has been acknowledged in outbreaks of louping ill in sheep 10 as well as in llamas introduced to a Scottish island. 8
Footnotes
Acknowledgments
Thanks are due to Janice Gilray and Maddy Maley, Moredun Research Institute, for technical assistance; Dr Mark Dagleish, for constructive criticism of the manuscript; and to the farmer concerned, for his assistance. J.B., K.W., C.U., G.M., and H.C. are funded by the Scottish Government.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
