Abstract
Canine mammary epitheliosis (ME) is a poorly studied dysplasia that may have premalignant potential. In this study, the clinicopathological relevance of ME was prospectively studied in 90 female dogs with mammary tumors (MTs) that underwent radical mastectomy. ME distribution, extent, and coexistence with benign and malignant MTs were evaluated for each case (505 mammary glands). ME was macroscopically undetectable and was present in 47/90 (52%) cases, frequently bilateral. In dogs with malignant MTs and ME, diffuse ME throughout the mammary chain was present in 10/39 (26%) cases. A histological ME-carcinoma transition was evident in certain histotypes. By immunohistochemistry (AE1/AE3, cytokeratin 14 [CK-14], CK-8/18, vimentin, calponin, p63, Ki-67, estrogen receptor, progesterone receptor, and human epidermal growth factor receptor 2), ME was a slow-growing, triple-negative process with a strong predominance of basal-like nonmyoepithelial cells. ME was associated with older dogs (P = .016), malignant tumors (P = .044), worse clinical stages (P = .013), lymph node metastasis (LNM, P = .021), higher histological grade tumors (P = .035), and shorter overall survival (OS) in univariate analysis (P = .012). Interestingly, ME was distantly located to the malignant tumor in most cases (P = .007). In multivariate analyses, LNM (P = .005), histological grade (P = .006), and tumor size (P = .006) were independent predictors of OS. For the pathologist, the observation of ME should be clearly stated in the MT biopsy report to alert the surgeon/oncologist. Given the differences between canine ME and its human histopathological counterpart (atypical ductal hyperplasia), “epitheliosis” should remain the preferred term for the dog.
Keywords
Mammary gland (MG) tumors are the most common neoplasms in sexually intact female dogs, representing 50–70% of all tumors.26,30,41,62,67 Mammary neoplasms in dogs are 3 times more frequent than their human counterparts, and many epidemiological, biological, and clinical features are shared between both species. Therefore, canine mammary cancer has been proposed to be a good spontaneous model to study human breast cancer, 45 especially some types of carcinomas such as inflammatory mammary carcinoma.18,51,53
The basic functional and histopathological unit of the mammary tissue in both humans and dogs is the terminal ductal-lobular unit (TDLU). TDLUs are formed by 2 layers of cells attached to the basement membrane. The innermost layer is composed of luminal epithelial cells, and the outermost layer is composed of basal/myoepithelial cells.31,66 Luminal cells express low-molecular-weight cytokeratin (LMW-CK), and basal/myoepithelial cells express high-molecular-weight cytokeratin (HMW-CK) such as cytokeratin 14 (CK-14). In addition, luminal epithelial cells typically express the estrogen receptor (ER) and progesterone receptor (PR). Myoepithelial cells express other markers such as smooth muscle actin, calponin, p63, cluster of differentiation 10, and vimentin.9,50,72 One of the main histological differences between human and canine MTs is that, in the latter, the proliferation and epithelial-mesenchymal transition of myoepithelial cells are much more frequent, complicating their immunolabeling as they lose their original immunophenotype.25,50,72
The classification of molecular subtypes in human breast cancer helps determine the prognosis and allows the selection of targeted therapies depending on the molecular markers expressed in each individual tumor, which can be detected by immunohistochemistry (IHC). 54 The classical human breast cancer molecular subtypes are: luminal A, luminal B, human epidermal growth factor receptor 2 (HER-2)-enriched breast cancer, and triple-negative breast cancer (TNBC; ER-, PR-, HER-2-), which can be further subdivided into luminal androgen receptor, mesenchymal, and 2 basal-like subtypes (immunosuppressed and immune-activated, both positive for HMW-CK). TNBC is the molecular subtype with the poorest clinical outcome, and no specific adjuvant therapy has been established.23,31,35,48,54
Although after surgery, chemotherapy and/or radiotherapy are routinely used as adjuvant therapy for human breast cancer, 31 great advances in the treatment of human breast cancer have been made over the past several decades, targeting individual mammary neoplasms at the molecular level. Few studies have been published on canine mammary cancer chemotherapy, and its effectiveness in advanced metastatic canine mammary cancer is not considered satisfactory.5,60,70 Targeted therapies for female dogs with mammary cancer are under development.20,28,29,52,61
Despite efforts to standardize the methods for the detection of molecular markers in canine mammary cancer, the results obtained by different researchers are still variable.12,13,19,50,71 Therefore, surgery is the main and often only therapy for dogs with MTs, with the exception of the specific clinicopathological entity inflammatory mammary cancer17,51,67 and advanced metastatic disease,14,40 for which surgery is generally not recommended. Nevertheless, tumor removal is not always successful. After surgical excision, the presentation of new tumors in nonoperated canine MGs is frequent during follow-up, although the data vary among studies according to the type of tumor and surgery. For example, the rate is high (70%) in dogs with mammary cancer resected by nodulectomy 59 and regional mastectomy (58%). 68 In a recent study of 159 female dogs that underwent resection of all visible MTs with complete margins, local recurrences or new tumors occurred in 31.5% and 58.6% of cases with or without ovariohysterectomy at the time of surgery, respectively. 67
Besides neoplasms, human and canine MGs can present with dysplasia. Canine mammary dysplasias are currently classified as: duct ectasia, lobular hyperplasia, epitheliosis, papillomatosis, fibroadenomatous change, and gynecomastia. 72 Among them, mammary epitheliosis (ME) stands out as a potential premalignant lesion, although it has been poorly studied and its clinical and pathological characteristics are mostly unknown.4,42,44,72 Histologically, ME is characterized by the intraductal proliferation of epithelial cells that grow toward the lumen of mammary glandular ducts in small solid aggregates formed by cords and tubules. The cells are monomorphic, small, and cuboidal with hyperchromatic nuclei and scant eosinophilic cytoplasm. Nuclear and cellular pleomorphism is mild.25,26,30,72 The immunophenotype is unknown. ME, along with other mammary dysplasias, can coexist with benign/malignant neoplasms in the same MG.4,21 The same intraductal structures show an increase in pleomorphism, mitoses, and occasionally, necrotic foci, named in situ carcinoma25,30 or epitheliosis with atypia/severe atypia. 72
In women,3,6,10,32,37,38 intraductal proliferative lesions are well-recognized premalignant changes that significantly increase the risk of developing invasive carcinomas. These lesions are classically divided into 3 different categories: usual ductal hyperplasia (UDH), atypical ductal hyperplasia (ADH), and ductal carcinoma in situ (DCIS), with risk levels ranging from 1.5-fold, 4- to 5-fold, and 8- to 10-fold, respectively. According to its nuclear features, DCIS is further divided into low-, intermediate-, and high-grade DCIS.3,6,10,32,37,38
Genetic studies have shown that ADH and all forms of DCIS represent intraepithelial neoplasms (clonal populations) that lead to different defined types of invasive carcinomas. 36 While most cases of UDH are unlikely to represent a direct precursor lesion, a slight proportion contains clonal populations of cells that may give rise to ADH. 27 UDH is characterized by haphazardly oriented benign epithelial cells that grow into the lumen of the duct. The cells have indistinct borders with variably sized nuclei and show mosaic pattern labeling for both LMW-CK (luminal type) and HMW-CK (basal type) with heterogeneous ER expression. Cells in ADH are evenly spaced and monotonous with scant cytoplasm containing rounded small hyperchromatic nuclei. They are positive for ER and negative for HMW-CK. 37 ADH is thought to be a very early neoplastic step in the pathway to low-grade DCIS and ER-positive low-grade invasive ductal carcinomas. 39 Low-grade DCIS shares architectural characteristics with ADH, and their differentiation remains problematic, while the diagnosis of intermediate- and high-grade DCIS is more straightforward. 37 DCIS has variable immunoprofiles that can be classified into the same molecular subtypes as invasive carcinomas (luminal A, luminal B, HER-2-enriched, and triple-negative). 69 The intraductal lesions are strictly noninvasive; therefore, a layer of intact myoepithelium can be detected by IHC (eg, p63, HMW-CK, calponin). 33
Considering the histological characteristics indicated in the several histological classifications of canine MTs and dysplasias,25,30,72 the histological morphology of human ADH might correspond with canine dysplasia named “epitheliosis,” while human DCIS is similar to in situ carcinoma25,30 or “atypical epitheliosis.” 72 Immunolabeling of myoepithelial cells in dogs and their identification as a continuous layer to determine an in situ state can be misleading, since myoepithelial proliferation in canine MTs is much more common than their human counterpart (ie, complex tumors), and new peripheral layers of interstitial myoepithelial cells can be generated.50,72 The authors of the latest international canine MT classification 72 preferred the term “atypical epitheliosis” instead of carcinoma in situ, to avoid the confusion created by the need to detect a layer of immunolabeled myoepithelial cells.
In veterinary medicine, despite being poorly studied, mammary dysplasia may be a relatively common undetectable lesion in female dogs. In one of the few published studies on canine mammary dysplasia, 44 dysplastic lesions and DCIS were histologically found in the MG of 52% of female dogs that underwent necropsy due to a nonmammary-related disease. It is hypothesized 72 that the probable malignant tumor subtypes derived from ME are comedocarcinoma, tubular carcinoma, and tubulopapillary carcinoma. Nevertheless, the biological behavior of these carcinomas derived from ME is unknown, but might be different from those carcinomas not derived from epitheliosis. 72
Despite its presumptive malignant potential and the prognostic importance of ME in female dogs, no previous studies have evaluated its relevance and extension in female dogs with MTs or the expression of molecular markers. Thus, the goal of this study was to determine the immunophenotype, frequency, and distribution of ME in the mammary chain of female dogs with MTs and to assess whether its presence is related to epidemiological, clinical, and pathological characteristics, to determine the pathological and clinical relevance of this dysplastic mammary change in female dogs.
Materials and Methods
Animals and Clinical Procedures
This prospective study included female dogs with at least 1 MT that underwent radical mastectomy (unilateral or bilateral) at the Veterinary Teaching Hospital of the Complutense University of Madrid (VTHC, Madrid, Spain) from November 2016 to December 2020. At presentation, the following epidemiological data were obtained: age, breed, weight, reproductive/hormonal background (spayed status and time of spay, pregnancies, pseudolactations, hormonal treatments), history of previous MTs, and growth rate of the mammary nodules. Complete general and mammary clinical examination, including abdominal ultrasound and radiographic evaluation of the thoracic cavity, was performed to rule out the existence of distant metastases. The clinical stage was established for each case following the modified tumor-node-metastasis system, 56 and only cases without distant metastases (stages I–IV) were included in the study. Animals underwent total mastectomy (unilateral or bilateral) according to the following criteria of the Unit of Mammary Oncology (VTHC): unilateral mastectomy in dogs weighing ≤10 kg with 1 nodule larger than 3 cm or larger than 5 cm for dogs weighing >10 kg, or multiple nodules throughout the mammary chain regardless of nodule size; if both mammary chains were affected, 2-stage bilateral total mastectomy was performed.
In dogs with at least 1 malignant tumor recruited until June 2019, postsurgical follow-up visits were recommended every 3 months during the first year, and every 6 months during the second year. Clinical examination, thoracic radiographs, and abdominal ultrasound were performed at every visit to detect recurrence and/or metastases. Phone consultation with the dog owners was done at the end of the second year (end of the follow-up period) if the follow-up visits were discontinued by the will of the owners. Follow-up data were used to calculate the disease-free interval (DFI) and overall survival (OS), defined as the time from surgery to the development of recurrences/metastases and cancer-related death, respectively. All procedures were performed in accordance with the ethical guidelines of the VTHC and with owner consent.
Macroscopic Evaluation and Sampling of Mammary Chains
After surgical resection, entire mammary chains including the regional lymph nodes (superficial inguinal lymph node removed en bloc during total mastectomy and/or axillary lymph node) were fixed for 24–48 hours in 10% buffered formalin. Once fixed, mammary chains were thoroughly examined and palpated. Serial 1-cm sectioning was made from the cranial to the caudal end, and macroscopic alterations (ie, thickening, cysts, presence of nodules) present in each individual MG were described and annotated together with macroscopic description of the tumors, location, and size (T1 <3 cm, T2 3–5 cm, T3 >5 cm). Systematic sampling of each MG (even if they did not contain macroscopic changes) and tumors was performed. The lymph nodes were serially sectioned every 0.2 cm and sampled according to international guidelines. 50 All samples were processed and embedded in paraffin.
Histopathological Study Including Evaluation of ME
Histopathological diagnoses 72 of all tumors and histological grading (low, intermediate, high) 49 of the malignant neoplasms were established by hematoxylin and eosin-stained 3-µm-thick deparaffined sections. The presence of ME was evaluated in each MG. The number and location of affected MGs, ME distribution, and coexistence with mammary neoplasms were evaluated for each case. The coexistence of ME with benign or malignant mammary neoplasms was annotated. In dogs with malignant MTs, the location of ME in the same, contiguous, or distant (separated by more than 1 MG) MG was evaluated. If ME was found in the same, contiguous, and distant MG of the malignant tumor, diffuse involvement of the mammary chain was considered.
Immunophenotyping of ME
To establish the immunophenotype of ME, 30 selected mammary samples with multiple ME affecting more than 5 ducts were immunolabeled for wide-spectrum CKs (AE1/AE3), CK-14 (HMW-CK), CK-8/18 (LMW-CK), vimentin, calponin, p63, Ki-67, ER, PR, and HER-2. After deparaffination and antigen retrieval in a Lab Vision PT Module (Thermo Fisher Scientific, Waltham, Massachusetts) by immersion in 1-mM EDTA at 95°C for 20 minutes, the sections were cooled and immunolabeled using a polymer-based method and peroxidase detection system (UltraVision Quanto MAD-021881QK; Master Diagnóstica, Granada). The primary antibodies, positive and negative controls, and immunolabeling conditions are depicted in Table 1. Pan CK AE1/AE3, CK-14, CK-8/18, vimentin, calponin, p63, and the proliferation index (Ki-67) were expressed as the percentage of positive neoplastic cells in ME. When the sample had more than 1 ME, the mean percentage of positive cells in the ME was calculated. HER-2 was considered positive when >30% of cells had moderate-to-strong, complete, homogeneous membrane labeling. 1 Finally, the samples were considered ER- and PR-positive when at least 10% of the ME cells were immunolabeled. 50
Primary antibodies and protocols used for canine mammary epitheliosis immunophenotyping.
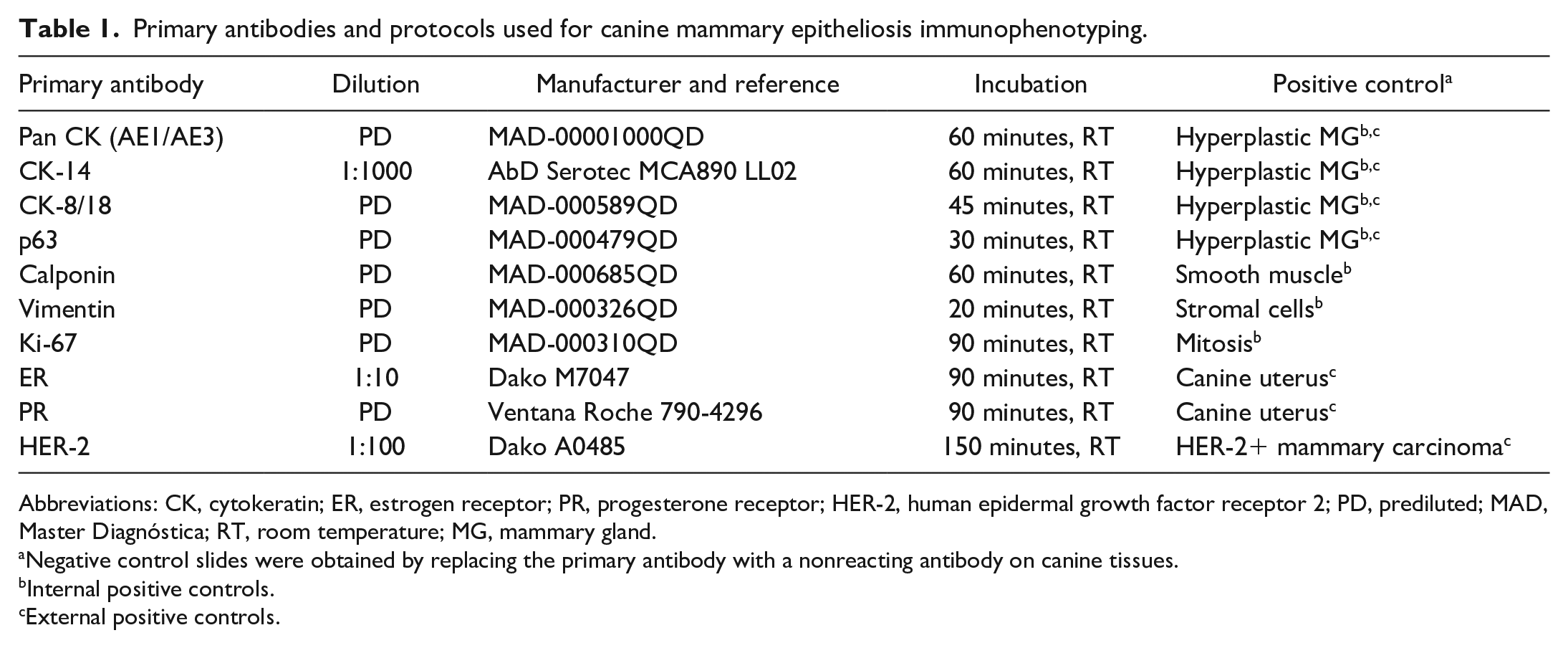
Abbreviations: CK, cytokeratin; ER, estrogen receptor; PR, progesterone receptor; HER-2, human epidermal growth factor receptor 2; PD, prediluted; MAD, Master Diagnóstica; RT, room temperature; MG, mammary gland.
Negative control slides were obtained by replacing the primary antibody with a nonreacting antibody on canine tissues.
Internal positive controls.
External positive controls.
Statistical Analyses
For the statistical study of the variables associated with ME, 2 databases were used: (1) Database of cases and (2) Database of mammary glands. In animals with more than 1 malignant tumor, the most malignant one (based on histopathological and clinical criteria, ie, histotype, histological grade of malignancy, and clinical stage) was selected for statistical purposes for database A. 49 The epidemiological, clinical, and pathological variables included in each database are shown in Table S1. The Fisher’s exact test and chi-square test were used to compare binary categorical variables and categorical variables with more than 2 categories, respectively. Pearson’s correlation coefficient was used to evaluate the correlation between continuous variables. Two-tailed P-values less than .05 were considered statistically significant. For survival analyses (database A), only animals with at least 1 malignant tumor were included. Those cases that were lost to follow-up, died for a reason not related with MTs, or were alive at the end of the study were censored for survival analyses. The Kaplan-Meier method with the log-rank test was used to estimate OS in dogs with malignant MTs and to compare among groups (comparison between cases that had ME in at least 1 MG versus cases that had no ME). The multivariate Cox proportional hazards model (Cox regression) was used to determine the influence of multiple variables (age; small/large breed; spay status; clinical staging; presence of ME; and size, grade, and diagnosis of the most malignant tumor) on the OS. Covariates were selected according to the Wald forward method that enters variables into the system in several steps according to their significance (P < .05). Variables that were significant at one step were considered constants for evaluation of the combined effect on OS. Models were evaluated with respect to their discrimination based on Harrell’s C-statistic. Statistical analyses were carried out using the STATA statistical package (Version 13.1) for Harrell’s C-statistic and IBM SPSS Statistics program (Version v.25; Armonk, New York) for the remaining analyses. All data analyzed in this study are available upon request to the authors.
Results
Clinical and Surgical Features
A total of 90 female dogs between ages 3 and 14 (mean, 10.20 ± 2.16 years) were prospectively recruited for the study (71 unilateral total mastectomy, 19 bilateral total mastectomy). In cases of bilateral total mastectomy, the time between the 2 surgeries varied between 21 and 107 days (mean, 54.83 ± 24.28 days). Of 90 cases, 25 were mixed breed dogs (28%), followed by 10 Yorkshire Terriers (11%), 9 German Shepherds (10%), and 6 French Bulldogs (7%). The remaining 25 breeds consisted of less than 5 individuals each. When dogs were classified as small (<10 kg) or large (≥10 kg), most of the population was small in size (56/90, 62%), whereas large dogs (34/90) accounted for 38%. Most of the animals (76/90, 84%) were intact, with the exception of 14 spayed females (16%) that had been castrated after 5 years of age (mean age at castration, 7.31 ± 2.08; mean age at presentation, 10.57 ± 2.59). The remaining animals were neutered at surgery. In 36/90 cases (40%), the dogs had previous pseudopregnancy, 10/90 (11%) had been pregnant, and 9/90 (10%) had received some hormonal treatment. At presentation, 55/90 (61%) of the dogs were in clinical stage I (T1, without regional or distant metastasis), 10/90 (11%) in stage II (T2, without metastasis to regional or distant lymph nodes), 12/90 (13%) in stage III (T3, without lymph node metastasis [LNM] or distant metastasis), and 13/90 (14%) of the cases had regional LNMs and were classified as clinical stage IV.
Macroscopic Findings
After the inclusion period, 109 complete mammary chains were collected for macroscopic examination and histopathology, and a total of 505 neoplastic and nonneoplastic MGs were examined. Small female dogs (34/90; 38%) commonly had 4 instead of 5 pairs of MGs (P = .004). In the majority of cases (82/90; 91%), at least 1 MG of the mammary chain was grossly thickened (mild, moderate, or severe). Of 505, 328 (65%) neoplastic and nonneoplastic MGs had diffuse thickening of different severity levels as follows: mild in most of the MGs (156/505; 30.9%), moderate in 103/505 (20.4%), and severe in 69/505 (13.7%). Macroscopic duct dilation (macroscopic ectasia) was present in 32/90 (36%) cases and in 77/505 (15.2%) of MGs evaluated. At gross examination, 216 of 505 MGs (42.8%) had nodules. The tumors were mostly smaller than 3 cm in diameter (T1, 185/216; 85.6%), followed by tumors larger than 5 cm (T3, 16/216; 7.4%), and 15/216 (6.9%) tumors were between 3 and 5 cm in diameter (T2).
Histopathology
MG tumors
Malignant tumors were present in 67/90 (74.4%) of the cases. In total, 248 neoplasms were found: 129/248 (52%) were malignant and 119/248 (48%) were benign MTs. All tumor margins were clean. The majority of malignant tumors were low grade (grade I, 72/129; 55.8%), while those of intermediate grade of malignancy (grade II) and those of high grade (grade III) accounted for 36/129 (27.9%) and 21/129 (16.3%), respectively. LNMs were histologically confirmed in 12/90 (13%) of the cases.
Mammary epitheliosis
In total, ME (Fig. 1) was present in 47/90 (52%) of the cases in the study. ME was observed in 55 of the 109 (50.5%) mammary chains evaluated; it was detected in 1 or 2 MGs within the same mammary chain in 41/55 (75%) and in 3 or more MGs in 14/55 (25%) of the cases. ME was bilateral in 8 of the 19 (42%) animals that underwent bilateral mastectomy. Thirty-nine dogs had at least 1 malignant tumor and ME. Regarding the location of ME and malignant tumors, ME was located only in the same MG as the malignant tumor in 2/39 (5%) of the cases, only in the contiguous MGs in 2/39 (5%), and only in a distant MG in 9/39 (23%) of cases. Diffuse ME (coexistence of ME in the MG affected by the malignant tumor and in contiguous and distant MGs) was present in 10/39 (26%) of cases. ME was present in 103/505 (20.4%) of the MGs. Duct ectasia (174/505; 34.5%) and atypical hyperplasia (83/505; 16.4%) were other mammary dysplasias found. The coexistence of ME with MTs by histotype is depicted in Table 2. In some malignant neoplasms with ME (n = 6), a histological ME-carcinoma transition was evident (Fig. 2) in certain histological tumor types: tubular carcinoma (n = 2), ductal carcinoma (n = 1), intraductal papillary carcinoma (n = 1), tubulopapillary carcinoma (n = 1), and comedocarcinoma (n = 1).
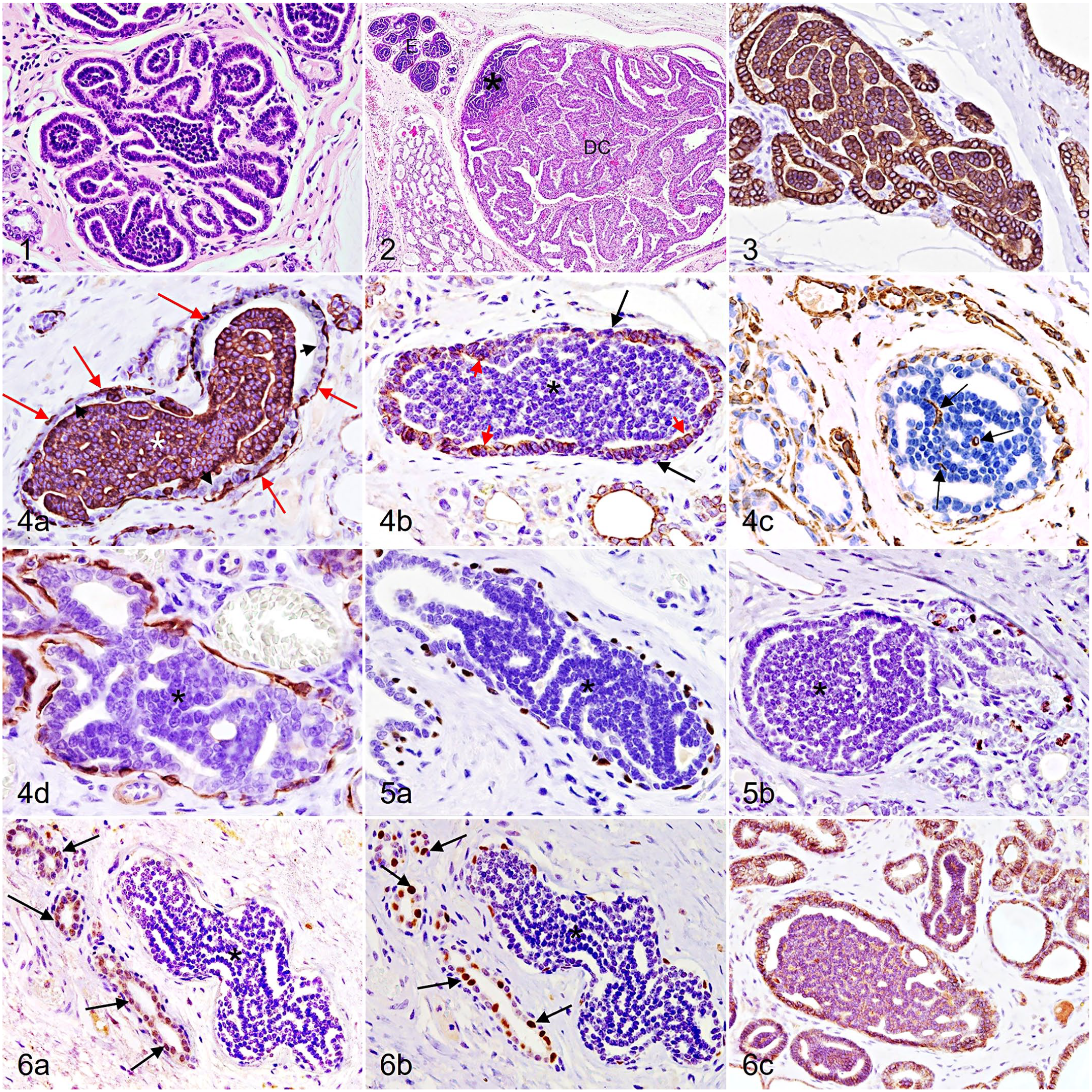
Mammary gland, dog.
Coexistence of mammary epitheliosis and mammary tumors by histotype in the same mammary gland, in dogs with mammary tumors.
ME Immunophenotyping
In the 30 samples evaluated, ME had a similar pattern of immunolabeling (Figs. 3–6), showing a mixed population with a strong predominance of basal-like phenotype cells. ME cells were consistently positive (100%) for wide-spectrum CKs (AE1/AE3). HMW-CK-14 was generally expressed by ME cells (mean, 84.48 ± 20.78%), whereas LMW-CK-8/18 immunolabeling was lower and variable among cases (mean, 3.03 ± 3.25%). Of note, when multiple mammary ducts were affected by ME in a single sample, a similar percentage of HMW-CK- and LMW-CK-positive cells was observed. Calponin- and p63-positive cells were only found in the basal layer of ducts that contained epitheliosis but not in the epitheliosis itself. Low numbers of stellate to fusiform vimentin-positive cells were found between cords of ME epithelial cells (mean, 1.03 ± 1.05%). The proliferation index of ME was very low (<1%) in all cases, except in 1 case which was 13%. Invariably, all ME cells in all samples were triple-negative (ER-, PR-, and HER-2-). The ducts containing ME had typical immunolabeling in the periphery, consisting of luminal cells (positive for LMW-CK-8/18, ER, and PR) and a surrounding layer of basal/myoepithelial cells (positive for HMW-CK-14, p63, and calponin).
Associations of ME and Clinicopathological Data
Database of cases
The advanced age of female dogs was significantly correlated with a greater number of MGs affected by ME (P = .016). ME was associated with several indicators of malignancy as follows. The presence of ME in any MG was more frequently found in dogs with at least 1 malignant tumor (P = .044). In dogs with malignant tumors, the presence of ME in any MG, the number of glands affected by ME, and a greater extent of ME through the mammary chain (diffuse ME) were significantly associated with regional LNM (P = .021, P = .001, P =.012, respectively) and a worse clinical stage (P = .013, P = .001, and P = .015, respectively). Diffuse ME was also associated with the grade of malignant tumor (grade III; P = .035). Interestingly, ME was frequently located in MGs other than the malignant tumor itself (P = .007).
Database of mammary glands
An increased number of MGs affected by ME and a greater extension in the mammary chain were associated with a higher frequency of microscopic ductal ectasia (P = .043) and lobular hyperplasia with atypia (P = .022). Mammary gross thickening was associated with the presence of hyperplasia with atypia (P = .042). The statistical association of ME with clinicopathological variables is depicted in Table 3.
Statistical associations of mammary epitheliosis and clinicopathological features in dogs with mammary tumors.
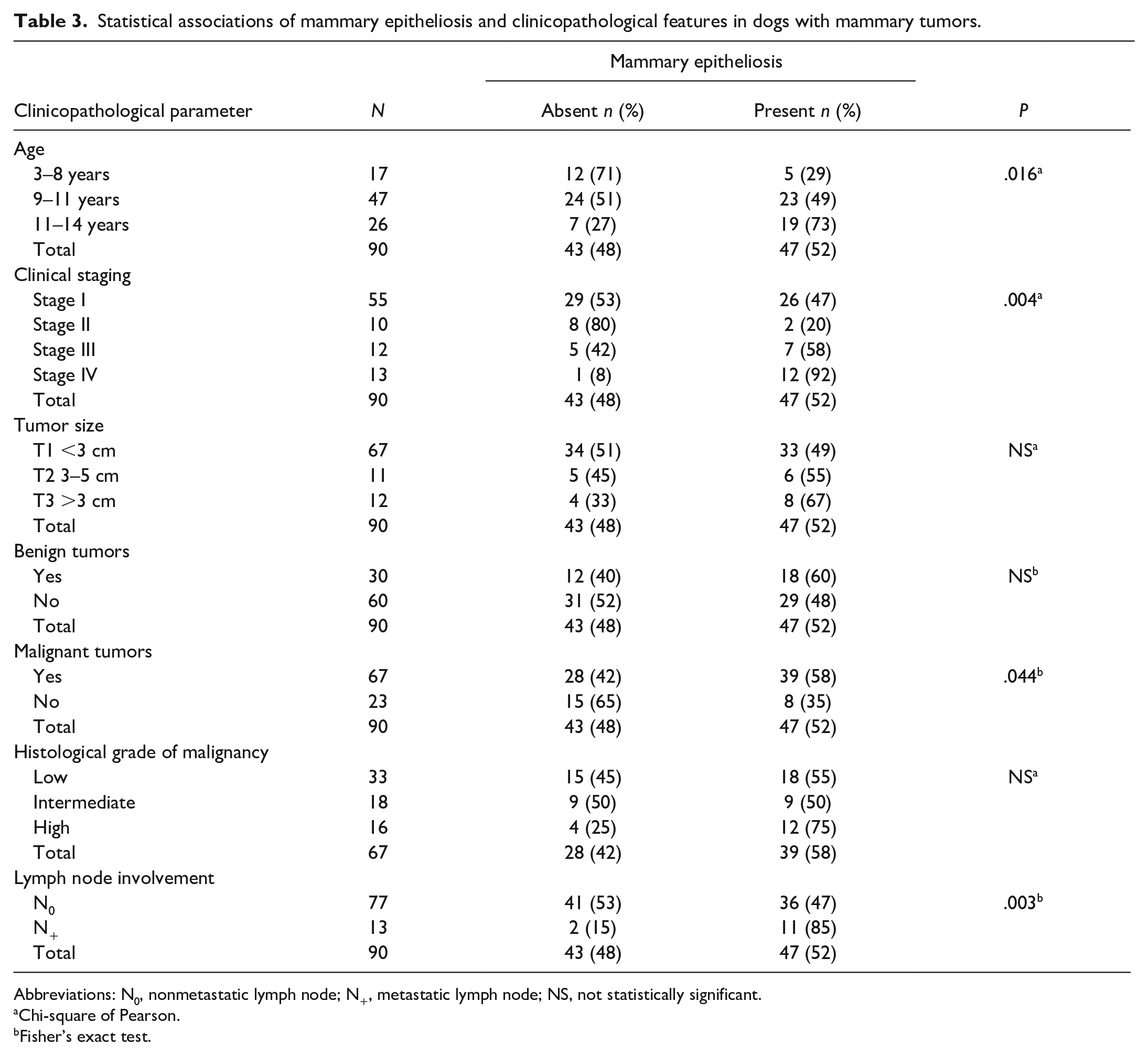
Abbreviations: N0, nonmetastatic lymph node; N+, metastatic lymph node; NS, not statistically significant.
Chi-square of Pearson.
Fisher’s exact test.
Survival Analyses
Information regarding follow-up variables was obtained in 60 of 67 cases with malignant tumors. Only 8/60 (13%) of the animals died because of the tumor within the first year (7 with ME, 1 without ME), and 16/60 (27%) died (14 with ME, 2 without ME) by the end of the second year (end of the follow-up). In 31 cases where all follow-up visits were complete, the DFI was calculated. Recurrences were found in 5/31 (16%) of the dogs (4 with ME, 1 without ME) and distant metastasis in 9/31 (29%) of the dogs (7 with ME, 2 without ME). Although the statistical level of significance was not reached (P = .206), DFI was longer in dogs without ME (mean, 611.31 ± 119.46 days) compared with cases with ME (mean, 451.56 ± 174.47 days). In total, 60 dogs with at least 1 malignant tumor were included in the univariate analysis of OS. The OS (Fig. 7) was significantly lower (mean, 554.87 ± 39.64 days) in cases that had ME in at least 1 MG compared with cases without ME (mean, 693.64 ± 26.05 days; P = .012). ME was not selected as an independent prognostic variable. The Cox proportional hazards regression model showed that only LNM (clinical stage IV; hazard ratio [HR], 5.64; 95% confidence interval [CI], 1.67–19; P = .005), histological grade of malignancy (HR, 3.30; 95% CI, 1.41–7.72; P = .006), and size of the malignant tumor (HR, 1.27; 95% CI, 1.07–1.52; P = .006) were independent predictors of OS with a Harrell’s C-statistic of 0.88.
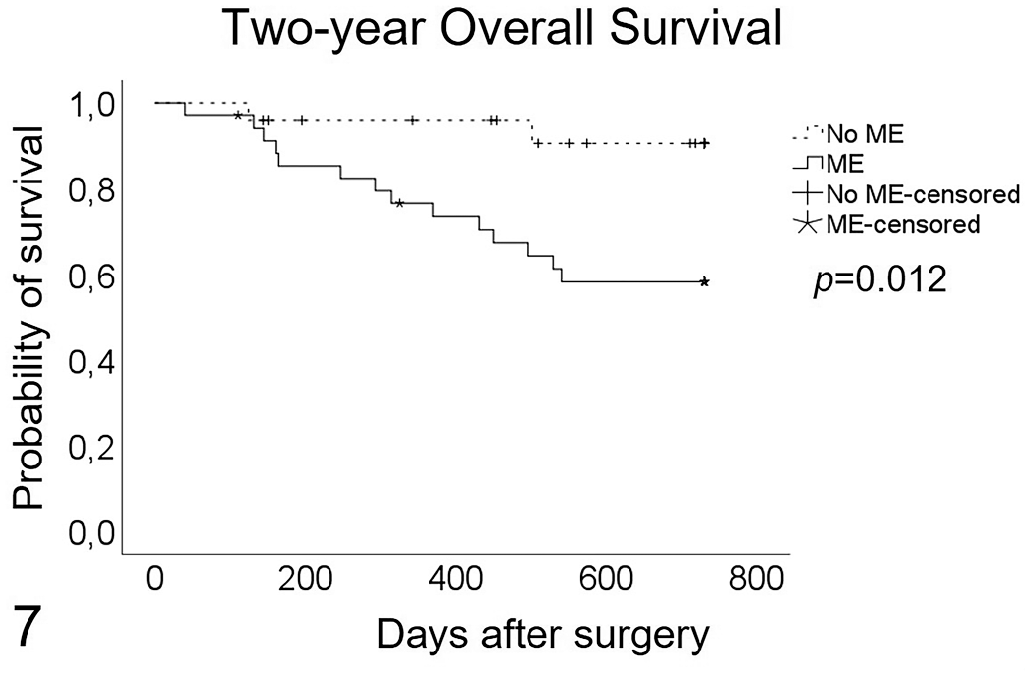
Kaplan-Meier survival curve. Overall survival of dogs with malignant mammary tumors with and without mammary epitheliosis (ME; ME versus no ME) in at least 1 mammary gland. The difference is significant (log-rank test, P = .012). Cases that died by other causes or were alive at the end of the study were censored.
Discussion
To the best of our knowledge, this is the first study to describe the pattern of distribution, extension, and immunophenotype of ME in female dogs with MTs, demonstrating the association of ME with malignancy and a poor prognosis.
The animals included in the study were predominantly small-sized dogs with an average age of 10.2 years and a ratio of malignant to benign MTs (52% versus 48%) consistent with previously reported studies.11,57,63,64,67 After the evaluation of 505 MGs, the vast majority of nodules were <3 cm (T1 size), similar to other reports.11,15 However, this small size was followed in frequency by nodules >5 cm (T3), which can be explained by the selection criteria of the animals included in the study that fulfilled the criteria for total mastectomy: large tumors (eg, T3) or multiple small nodules distributed throughout the mammary chain. The fact that all animals included in the study underwent radical unilateral or bilateral mastectomy is unique compared with existing literature on canine MTs and provides important histological information on nonneoplastic MGs that are not systematically analyzed by a pathologist. Despite this advantage, it is necessary to consider that our results may differ from those of other studies on canine MTs, in which the animals included had any type of mammary surgery.
One of the singularities of this study was the macroscopic evaluation of entire mammary chains, which provided information on macroscopic changes present in MGs affected and nonaffected by neoplastic processes. The few macroscopic studies of mammary chains11,15,24,46,47,49,58,64,65 have only depicted tumor size as a predictor of malignancy. In this study, a thorough evaluation and description of neoplastic and nonneoplastic features (eg, thickening, duct ectasia) revealed that thickening of MGs was a common finding not associated with any clinical or histopathological variable, with the exception of atypical lobular hyperplasia. ME was not associated with any macroscopic changes.
According to our results, ME should be considered a frequent lesion with no macroscopic changes. ME was present in 52% of the cases, in accordance with the general findings on intraductal proliferations reported by other authors4,21,44 evaluating female dogs with no clinical mammary disease at necropsy 44 or mammary biopsies.4,21 According to human histopathological criteria, these authors classified intraductal proliferations as UDH, ADH, and DCIS of low, intermediate, or high grade. Furthermore, morphological characteristics among the different authors differed, as features consistent with ME were named ADH by one group 21 and DCIS by others.4,44 According to the description of canine MG tumors, dysplasia histological classifications, and the authors’ observations, the histological characteristics of canine ME coincide with those of human ADH.
ME was found within ducts that consistently showed a surrounding single layer of myoepithelial cells (positive for p63 and calponin). Although in canine mammary tumors with myoepithelial proliferation, the presence of a surrounding p63-/calponin-positive layer does not exclude invasiveness; in simple carcinomas its absence indicates stromal infiltration in canine 50 and human 37 MTs. In this study, the noninfiltrative nature of all ME analyzed was confirmed by IHC.
There have been no previous reports on ME immunophenotyping. While UDH in human breast pathology is composed of a mixed population of luminal and basal cells, ADH and low- to intermediate-grade DCIS are formed by a homogeneous population of luminal epithelial cells, are negative for HMW-CKs, and positive for ER and PR.32,37 High-grade DCIS is less likely to be ER-positive, and about 10% of cases are HMW-CK-positive (eg, CK-14) and are therefore classified as basal-like subtype.2,37 In contrast to the histological similarities between canine ME and human ADH, the IHC results of this study showed that cells in ME were a mixed population with a strong predominance of basal-like immunophenotype, as the vast majority of cells were CK-14+, CK-8/18−, and negative for myoepithelial immunomarkers (p63 and calponin). A minor proportion of cells had luminal differentiation (CK-8/18+). The presence of a few stellate to elongate vimentin-positive cells located between the rows of cuboidal ME epithelial cells in all ME could be interpreted as incipient stroma. The invariable negativity for ER, PR, and HER-2 receptors observed in all ME conflicts with the reported ER- and PR-positive expression in human intraductal dysplasia (a mix of positive and negative ER/PR cells in UDH and an entirely positive population in ADH and low- to intermediate-grade DCIS).2,37 Previous studies on intraductal lesions in dogs4,8,21,42–44 showed negative-to-low ER expression. In human breast pathology, ER-negative ADH and low-grade DCIS are considered discordant cases. 2
These differences in canine ME versus ADH or low-grade DCIS immunophenotype indicate that caution should be taken when considering canine ME as a model for ADH or low-grade DCIS, at least from the hormonal point of view and when the human terminology for intraductal proliferations is used. Taking into account these results and even though they are histologically similar, we believe that canine ME and human ADH or low-grade DCIS represent different entities and therefore the term “mammary epitheliosis” should be still used for this canine dysplasia. However, when severe atypical features in ME (eg, epitheliosis with atypia/severe atypia) are present, carcinoma in situ should be suspected. The distinction between this condition and an invasive state can be established by the lack of a surrounding myoepithelial layer,50,72 as occurs in humans. 37 Our findings indicate that most ME cells have a mammary triple-negative nonmyoepithelial basal cell origin (basal-like molecular subtype, ER-, PR-, and HER-2-negative) which suggests a poor prognosis, 48 and despite the low Ki-67 growth index, ME may have a malignant nature and not only premalignant as noted to date in the veterinary literature.25,72 In human breast cancer, most invasive ductal basal-like tumors, positive for HMW-CKs 5 and 14, originate from basal/suprabasal multipotent progenitor cells and can gradually differentiate into glandular (luminal) cells and coexpress LMW-CK-8/18. 34 Similarly, canine ME might originate from these multipotent progenitor cells and could potentially follow a progression pathway into luminal neoplastic cells, although its triple-negative nature would tend to a more malignant progression.
The possible evolution of canine ME into some malignant histotypes was proposed in the most recent international classification of canine MTs. 72 In human medicine, the use of mammography, routine incisional biopsies, and subsequent surgery has led to the establishment of a temporal sequence, the relative risk of generating carcinomas from each intraductal proliferative dysplastic lesion, as well as the risk of generating specific types of carcinomas. 22 In veterinary medicine, a similar longitudinal prospective study with an adequate number of cases, detected by mammography without mammary malignancies and incisional biopsies, is not feasible today. In this study, the histological evidence of progression of ME to mammary carcinomas in a variety of simple (eg, tubular carcinoma, tubulopapillary carcinoma, comedocarcinoma) and ductal-associated (eg, ductal carcinoma, intraductal papillary carcinoma) histotypes was reported for the first time. However, no statistical association was found between the presence of ME and any specific histological type.
Besides being of great interest to the veterinary pathology community, our results regarding ME are of epidemiological, surgical, and clinical relevance in diverse aspects. ME is a slow-growing, age-related lesion that needs to be considered a priori if the case is elderly, as ME is more widespread in the mammary chain. There was no statistical correlation between the presence of ME and the reproductive status (spayed/not spayed). It is well known that early ovariohysterectomy (before the first or second estrous cycle) in female dogs has a protective effect against the development of MTs. 59 The population included in the study had a predominance of not-spayed animals or animals spayed after the age of 5 years, so the protective effect of early castration on the development of ME cannot be properly evaluated in this cohort of animals.
Regarding the surgical importance of ME, in more than one quarter of the cases, ME was found not only in the same gland as a malignant tumor but also in adjacent and distant glands. Dogs with malignant tumors more frequently had ME located in a different MG (P = .007). This is an indicator of the risk of performing nodulectomy or regional mastectomy as a surgical approach, although indicated by other clinical parameters, due to the probability of leaving behind unresected ME located in other MGs. The development of new tumors is very frequent after nodulectomies (70%) and regional mastectomies (58%).59,68 In light of our results, because ME is highly present in nontumor-affected MGs, ME might be the origin of these reported new tumors in unresected MGs. Moreover, ME was seen in both mammary chains in 42% of cases that underwent total bilateral mastectomy, showing that the risk of presenting ME is bilateral as in human dysplasia (eg, ADH). 16 The finding of ME in dogs with resected MTs in one chain indicates the need for follow-up and precise mammary examination of the contralateral mammary chain.
Previous studies on the ultrasonographic and radiographic characteristics of surgical mammary biopsies have reported subtle microcalcifications associated with intraductal dysplasias, with no reference to epitheliosis. 42 Ideally, future studies carried out on the ultrasonography characteristics of ME, demonstrating preoperative detectable changes, and ultrasonography of the mammary chain in dogs with MTs will lead to a better approach for determining the type of surgery to perform.
ME was statistically associated with worse clinical stages, the presence of malignant tumors, LNM, a higher histological grade, and shorter OS in univariate analysis. However, multivariate Cox proportional hazards regression analysis revealed that the decrease in survival was more due to an aggressive neoplastic disease rather than to the presence of ME. Clinical stage IV (LNM) was the strongest independent predictor of cancer-related death, followed by histological grade of malignancy and size of the malignant tumor, in accordance with a previous study by our group 49 and other studies7,45,55,63 including animals with any kind of surgery.
In summary, our results showed that ME is a common slow-growing mammary lesion that mainly affects older dogs, is related to malignant neoplasms, and can be distributed throughout the mammary chain, even in glands distant from those affected by tumors, showing no macroscopic alteration. ME is associated with malignancy indicators, is able to progress to carcinomas, and is possibly linked to the appearance of new mammary neoplasms in unresected MGs. For the pathologist, since the detection of ME in a malignant MT indicates a high probability of the existence of ME in other MGs (P = .007), and is associated with malignancy and a poorer prognosis, the observation of ME in a biopsy of a malignant MT should be clearly stated in the biopsy report. In the case of nodulectomies and regional mastectomies, the report should warn the surgeon/oncologist who could consider the possibility of extending the surgery (radical mastectomy) in a preventive way or establishing closer follow-up. The results obtained from this study are of pathological and clinical importance in the veterinary field, and reinforce the need for further studies on the role of canine mammary dysplasia as a spontaneous model for their human counterpart.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858221092013 – Supplemental material for Epitheliosis is a histopathological finding associated with malignancy and poor prognosis in dogs with mammary tumors
Supplemental material, sj-pdf-1-vet-10.1177_03009858221092013 for Epitheliosis is a histopathological finding associated with malignancy and poor prognosis in dogs with mammary tumors by Guillermo Valdivia, Ángela Alonso-Diez, Daniel Alonso-Miguel, María Suárez, Paloma García, Gustavo Ortiz-Díez, Maria Dolores Pérez-Alenza and Laura Peña in Veterinary Pathology
Footnotes
Supplemental material for this article is available online.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Spanish Ministry of Science, Innovation and Technology, project PGC2018-094516-B-I00; a PhD grant funded through the Mexican Council for Science and Technology (CONACYT; 515916 to G.V.); the ECVP specialization residency in Veterinary pathology grant from Complutense University (69/2018 to A.A.D.); and a PhD contract at Complutense University (7026349846-Y0SC001170 to D.A.M.).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
