Abstract
The misfolded form of cellular prion protein (PrPc) is the main component of the infectious agent of transmissible spongiform encephalopathies and the validated biomarker for these diseases. The expression of PrPc is highest in the central nervous system and has been found in peripheral tissues. Soluble PrPc has been detected in cerebrospinal fluid, urine, serum, milk, and seminal plasma. In this study, attempts were made to characterize prion protein in urine samples from normal and scrapie-infected sheep. Urine samples from scrapie-infected sheep and age-matched healthy sheep were collected and analyzed by Western blot following concentration. A protease K-sensitive protein band with a molecular weight of approximately 27–30 kDa was visualized after immunoblotting with anti-PrP monoclonal antibodies to a C-terminal part of PrPc, but not after immunoblotting with monoclonal antibodies to an N-terminal epitope of PrPc or with secondary antibodies only. The amount of PrPc in the urine of 49 animals (control group:
Keywords
Introduction
Transmissible spongiform encephalopathies (TSEs) are a group of neurodegenerative diseases such as bovine spongiform encephalopathy (BSE) in cattle, scrapie in sheep and goats, chronic wasting disease (CWD) in elk and deer, and Creutzfeldt-Jakob disease (CJD) in humans. TSEs are characterized by amyloid-like deposition of the protease-resistant pathological isoform (PrPSc) of host cellular prion protein (PrPc) primarily in the central nervous system and lymphoid organs. 21,27 The expression of PrPc is highest in neural tissues and also has been found in non-neural tissues. 4,5,7,11,17 PrPc is located on the cellular surface, but soluble PrPc has also been detected in cerebrospinal fluid, 20,28 blood plasma, 29 milk, 8,15 ovine seminal plasma, 6 and human urine. 18 It is unknown whether TSE-associated PrP (PrPSc) circulates in body fluids and is eventually eliminated from the body. Blood has been repeatedly stated to be infectious, 3,12,16,25 and recently, results on CWD transmission by saliva have been reported. 16 Urine also has been found to be infectious in cases of coincident scrapie infection and chronic kidney inflammation in autoimmune mice 22 and in scrapie-infected hamsters, 13 but not in CWD-infected deer. 16
A protease-resistant isoform of the prion protein was reported once in the urine of scrapie-infected hamsters, BSE-infected cattle, and humans with CJD, 24 but later these results were declared as a consequence of cross-reaction of secondary antibodies with either contaminating bacterial proteins 9 or immunoglobulin fragments excreted in urine. 10,14,23 TSE-infectivity in urine of scrapie-infected hamsters 13 and mice with coincident TSE and kidney inflammation 22 reopened an inquiry into the presence of TSE-associated proteins in urine. In this study, attempts were made to characterize prion protein in urine samples from normal and scrapie-infected sheep, to determine the conditions for its optimal detection, and to quantify the amount of PrP in urine of healthy and scrapie-infected animals.
Materials and methods
Animals
Scrapie-infected sheep were sourced from a farm quarantined for scrapie and were identified by a PrPSc immunohistochemistry (IHC) assay of the “third eyelid” or the nictitating membrane (NM)-associated lymphoid tissue. 19 NM-test positive animals used in this study were kept in a designated animal facility at Ottawa Laboratory Fallowfield (Ottawa, ON, Canada). Age-matched control animals were housed in a different “clean” building. All experimental procedures involving animals were carried out according to Canadian Council on Animal Care guidelines.
Sheep urine samples collection and processing
Urine samples (450–600 ml) from scrapie-positive and age-matched control animals were collected in the morning after the intramuscular administration of 100 mg of furosemide.
a
Urine was cleared of debris by centrifugation for 20 min at 4,000 ×
Preparation of brain tissue homogenates
Sheep brain tissue was obtained from normal or scrapie-infected animals with clinical signs confirmed by histopathology and PrPSc IHC. A 20% (w/v) homogenate was prepared in cold homogenization buffer (10 mM Tris-HCl. pH 7.2, 300 mM sucrose) using a FastPrep Cell Disrupter
e
at a speed setting of 6.5 for 45 sec. The homogenate was cleared of cell debris by centrifugation at 1,500 ×
Proteinase K digestion
Concentrated and dialyzed urine samples and brain tissue homogenates of scrapie or control healthy sheep were incubated with 1–50 μg/ml proteinase K c for 60 min at 37°C. The reaction was stopped by the addition of PMSF to a final concentration of 5 mM.
Peptide: N-Glycosidase F treatment of urine and brain PrP
Deglycosylation was performed using a Peptide: N-Glycosidase F (PNGase F) kit f according to the manufacturer's protocol. A volume of 50 μL of 250-fold concentrated and dialyzed sheep urine or 50 μL of 10% (w/v) ovine brain tissue homogenate were denatured by heating for 10 min at 100°C in the presence of 0.5% sodium dodecyl sulfate (SDS) and 1% β-mercaptoethanol, and incubated for 60 min at 37°C in buffer containing 100 mM sodium phosphate, 10 mM Tris-HCl, 1% NP-40, and 1–4 units of PNGase F. Treatment was stopped by the addition of 2X SDS loading sample buffer followed by incubation at 100°C for 5 min.
Sodium dodecyl sulfate polyacrylamide gel electrophoresis and Western blot detection of PrP
For Western blot analysis, samples were subjected to sodium dodecyl sulfate Polyacrylamide gel electrophoresis (SDS-PAGE) using reducing conditions and were subsequently transferred to a polyvinylidene difluoride (PVDF) membrane g by semidry blotting in 10 mM 3-(cyclohexylamino)-1-propanesulfonic acid c (CAPS) buffer plus 10% methanol, pH 11.0. Membranes were blocked with 1% casein in 10 mM Tris-HCl, 150 mM NaCl, 0.1% Triton X-100, pH 7.5 (Tris buffered saline Tween [TBST]) overnight at 4°C, and then incubated for 60 min at ambient temperature with 1 μg/ml of one of the following anti-PrP monoclonal antibodies: F99/97.6.1, h M2188, M2197, M2932. 1 Hybridization with primary antibodies was omitted in control experiments on verification of the specificity of the PrP binding. The membrane was washed 3 times with 20 ml of TBST for 20 min and incubated with peroxidase-conjugated AffmiPure donkey anti-mouse IgG (H+L) (DAM-HRP) i with minimal cross-reaction to bovine, chicken, goat, guinea pig, Syrian hamster, horse, human, rabbit, and sheep serum proteins at a 1/5,000 dilution for 60 min at ambient temperature. All dilutions of detection antibodies were in TBST with 1% casein. After a final washing of the membranes with TBST, prion protein bands were visualized by enhanced chemiluminescence j using the ChemiDoc System g or Bioflex MSI film. k
Mass spectrometry analysis of urine proteins
A urine sample from a scrapie-infected animal was processed as described above and loaded into 2 lanes on an SDS-PAGE gel. One line was analyzed by Western blotting with an anti-PrP monoclonal antibody M2188. A segment in the unprocessed parallel lane was excised according to the position of the immunoreactive band on the Western blot and analyzed by liquid chromatography-tandem mass spectroscopy (LC-MS/MS) at the Eastern Quebec Proteomics Core Facility (Quebec City, Canada). The peptide masses and sequences obtained were matched to proteins using the Mascot Search program (http://www.matrixscience.com).
Quantitative analysis of PrP in urine samples
The amount of PrP in urine samples was estimated by comparison with a known amount of sheep recombinant PrP25–233 ARQ (alanine-136 arginine-154 glutamine-171).
1
Urine concentrates (equivalent to approximately 2.5 ml of nonconcentrated urine) were tested in parallel with 0.03–4.0 ng of sheep recombinant PrP25–233. Western blotting was performed as described above. The amount of PrP was quantified by densitometry according to the intensity of PrP bands with known protein concentration using a ChemiDoc System
g
and a Quantity One 1-D Analysis Software, version 4.4.
g
Skewness of data was assessed using the Kolmogorov-Smirnov test. The nonparametric Mann-Whitney
Results
Urine samples from scrapie-infected and control sheep were collected, concentrated 250-fold, and analyzed by Western blot with anti-PrP monoclonal antibodies (mAbs). A diffuse protein band (in some preparations a double band) with MW 27–30 kDa was visualized in some urine samples after immunoblotting with anti-PrP mAb M2188 (binds with epitope SRPLIHFG; Fig. 1A). Sheep PrP peptides YYRENMYRY (amino acid positions 129–138) and VVEQMCITQYQRE (amino acid positions 189–202) were identified by LC-MS/MS in an anti-PrP antibody immunoreactive segment of the SDS-PAGE gel. To examine if nonspecific binding of the secondary antibodies to urinary immunoglobulin fragments or contaminating bacterial proteins occurred during the immunoblotting procedure as previously reported, 9,10,23 control immunoblotting experiments were performed using secondary antibodies only (omitting the hybridization step with primary antibodies). No immunoreactive bands were detected with secondary anti-mouse antibodies (Fig. IB) or with non-PrP antibodies (anti-Salmonella mAb M105, data not shown), confirming that PrPc was detected in sheep urine. Concentrated urine samples were also probed with various anti-PrP monoclonal antibodies (Fig. 2A). Thus, PrPc in urine was detected by the monoclonal antibody M2197 (binds with epitope HVAGAAAAGAVVG; Fig. 2B), F99/97.6.1 (binds with epitope QYQRES; Fig. 2B), but not after immunoblotting with mAb M2932 (binds with epitope YPPQGGGGW; Fig. 2B). Digestion of concentrated urine samples with proteinase K eliminated the immunostaining of PrPc on Western blots (Fig. 2A), indicating that PrPc excreted in urine was in a proteinase K-sensitive form.
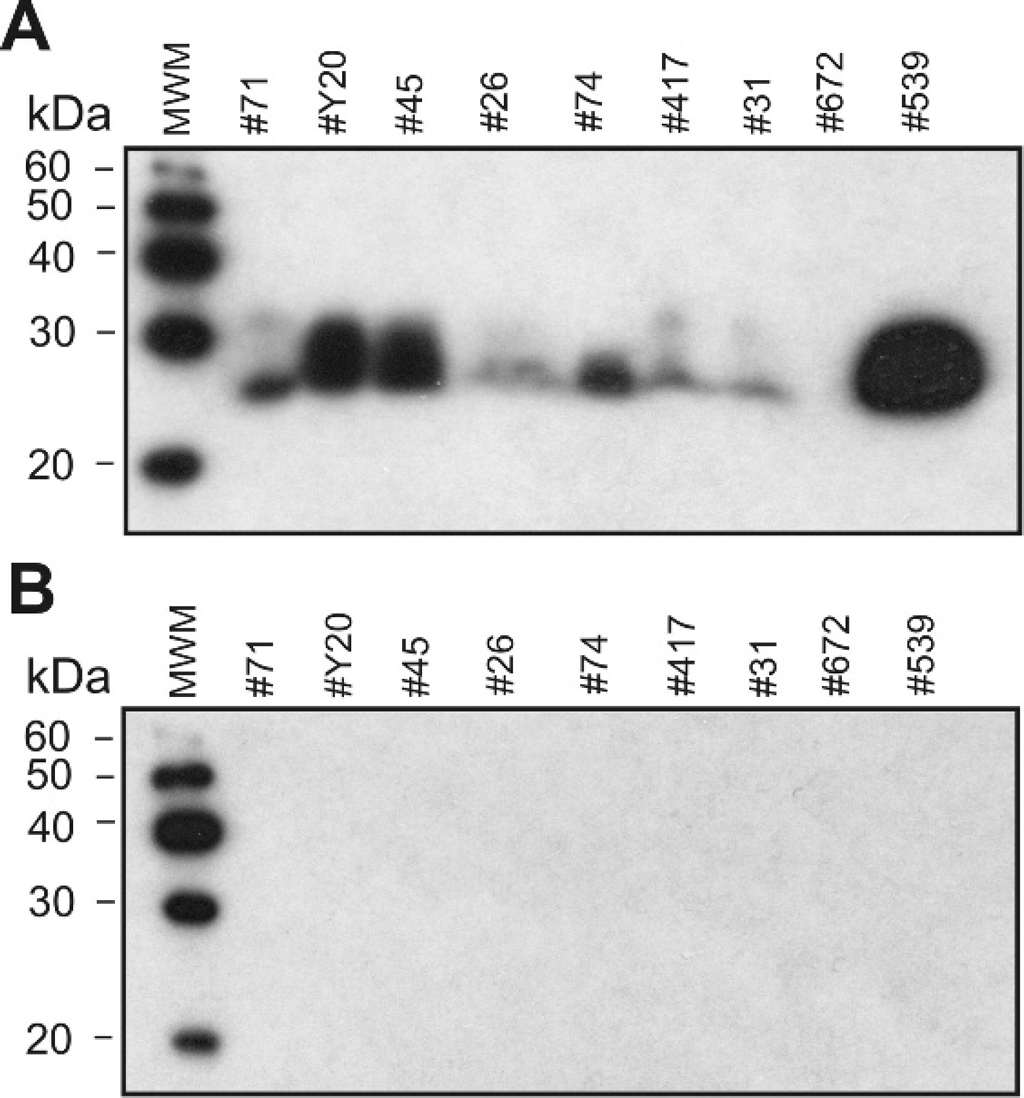
Western blot analysis of concentrated urine specimens from sheep naturally infected with scrapie.
Treatment of concentrated urine samples with PNGase F (an enzyme that removes oligosaccharides from N-linked glycoproteins) resulted in the shift of the molecular weight from 27–30 kDa to 17–18 kDa (Fig. 2C). In contrast, the deglycosylated PrPc from brain tissue had a molecular weight of approximately 25 kDa (Fig. 2C). After partial cleavage with PNGase F, 3 isoforms of urine PrPc were identified: 28–30 kDa, 23–24 kDa, and 17–18 kDa (data not shown). These observations suggested that the urinary PrPc was excreted in a diglycosylated-truncated form. A PK titration (1–50 μg/ml) was performed to compare the protease sensitivity of PrPc from urine and brain tissue homogenate samples. PrPc in urine from a scrapie-infected sheep was more PK sensitive than PrPc in brain tissue homogenate from a scrapie negative sheep (Fig. 3).
It was essential to analyze whether the excretion of PrPc in urine was associated with the scrapie status of the sheep. The amount of PrPc in the urine of 49 animals (control group:
Discussion
The sheep urine samples were concentrated approximately 250-fold, providing an opportunity to estimate a concentration of PrP in the urine of scrapie-infected and control animals by Western blot. Proteinase K-sensitive PrP (i.e., PrPc) was identified by a range of anti-PrP monoclonal antibodies that bind to distinct epitopes located at the C-terminus of PrP. Sheep PrP peptides were identified by LC-MS/MS in an anti-PrP antibody immunoreactive segment of the SDS-PAGE gel. No proteins were detected by Western blotting when monoclonal antibodies with other than prion protein specificity were used or when secondary antibodies were applied alone. Therefore, the detection of PrPc in sheep urine was not a result of the cross-reaction with bacterial proteins or immunoglobulins excreted in urine as was previously reported for the false-positive detection of protease K-resistant PrP in urine samples. 9,10,23,24 Interestingly, the urinary prion protein did not react with anti-PrP mAb M2932, which binds to an N-terminal epitope of PrP. After deglycosylation with PNGase F, urinary PrPc had a molecular weight of 17–18 kDa, which was lower than the molecular weight of deglycosylated PrPc from brain tissue (approx. 25 kDa). This combined data suggested that PrPc was excreted in urine in an N-terminally truncated form. The exact origin of the urinary PrPc is yet to be determined. The blood-borne PrPc may filter though the kidney as the molecular weight threshold for protein excretion is ∼40 kDa. In addition, detectable expression of PrPc in kidneys has been reported 17 ; it is feasible that PrPc can be shed from the cell surface and contribute to the overall PrP concentration in urine. The N-terminally truncated PrPc was previously detected in the urine of healthy human individuals 18 and in the ram reproductive fluid along with the C-terminally truncated and full-length PrPc forms 6 ; only full-length and C-terminally truncated forms of PrPc were reported in ovine milk, 15 suggesting tissue-specific variations in proteolytic cleavage/shedding of cellular PrP.
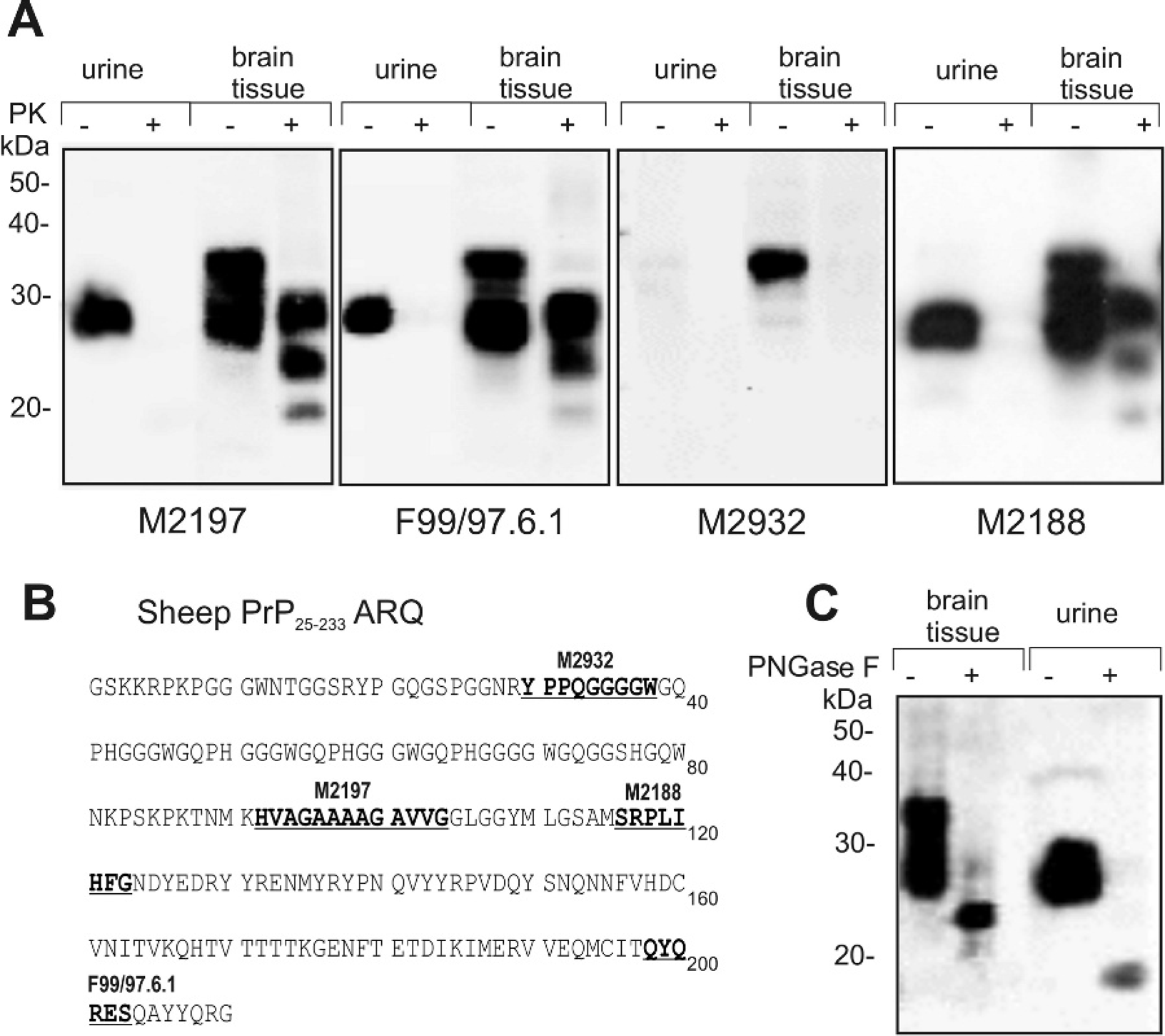
Detection of PrP in urine and brain tissue homogenate samples by anti-PrP monoclonal antibodies (mAbs).
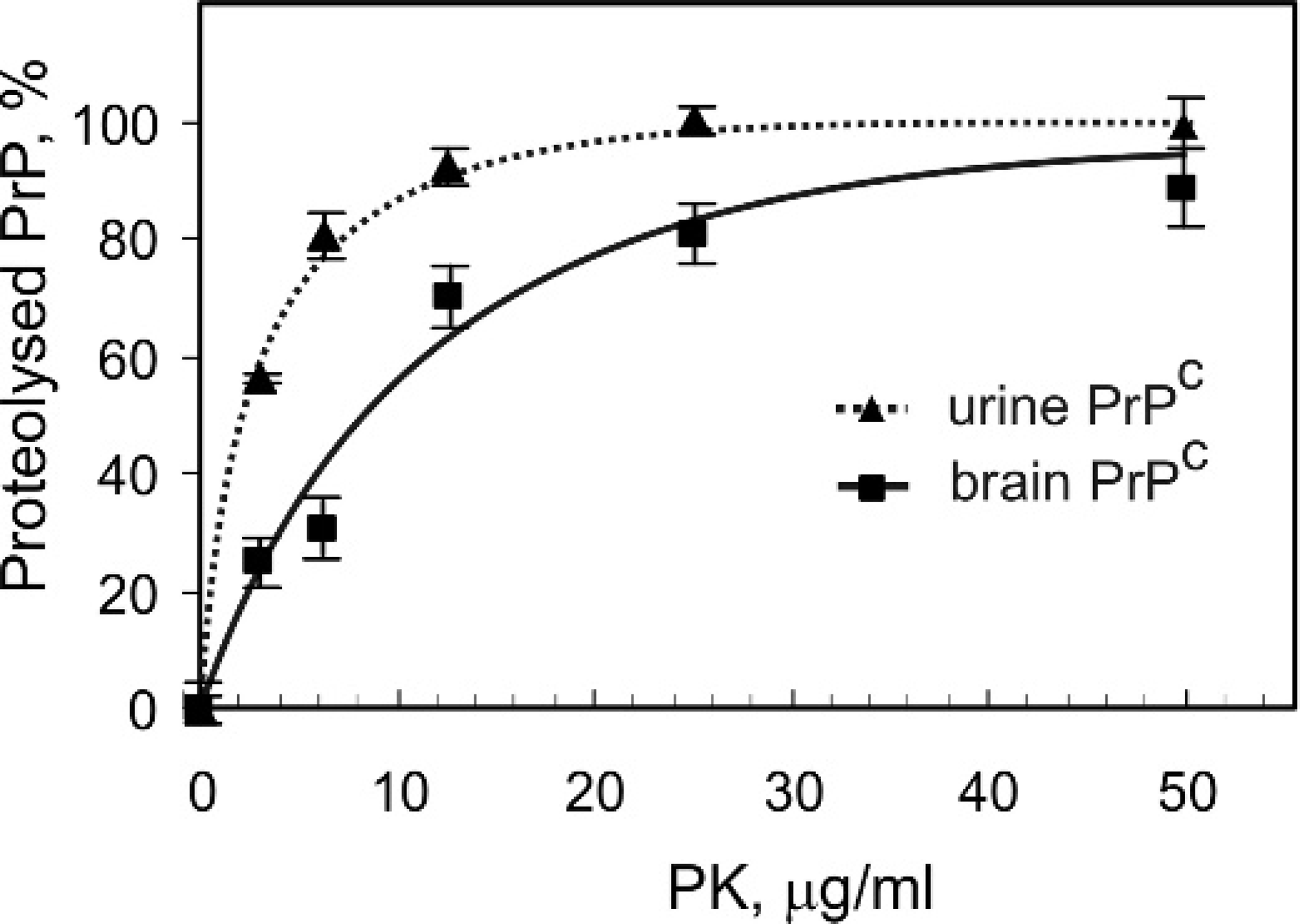
Proteinase K titration and digestion of urine and brain tissue-derived PrPc. Total protein concentration in samples was adjusted to 3–4 mg/ml. PrPc was detected by Western blot using monoclonal antibody M2197 as a primary antibody. Fraction of the proteolysed PrPc is presented as a function of PK concentrations. Results are expressed as the means ± SD.
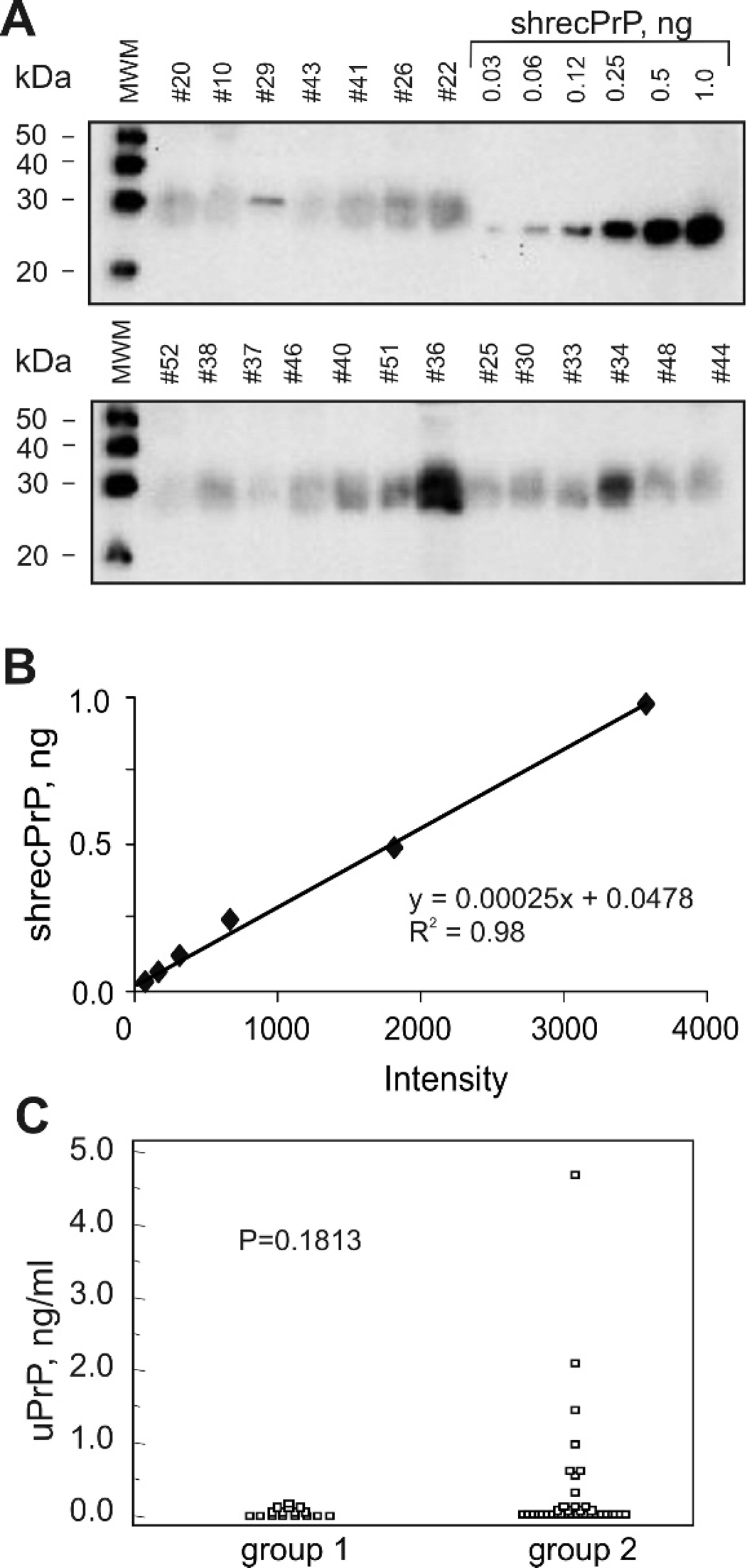
Assay of PrPc concentration in sheep urine.
Considerable variation in the amount of excreted urinary N-terminally truncated PrPc was observed among sheep from a naturally scrapie-infected flock. Thus, the majority of NM-test positive sheep had urinary PrPc concentration in the same range as control animals: the median value for both groups was 0.04 ng/ml. Seven of 33 NM-test positive animals had an elevated level of PrP in urine: 0.6–4.7 ng/ml. The reason and consequences of the observed variation in urinary PrP excretion in scrapie-infected animals is unclear. No correlation was found between the urinary PrPc concentration, the total protein concentration in urine samples, and severity of scrapie-associated clinical signs. The authors were unable to detect proteinase K-resistant PrP in scrapie-infected sheep urine samples, possibly as a result of levels being below the limit of detection of this method or the lack of proteinase K-resistant PrP excreted in urine. Attempts of other researchers to identify protease K-resistant PrP in urine samples of TSE-affected animals failed, 9,10,23,24 but urine has been reported to be infective in the case of coincident scrapie infection and chronic kidney inflammation in autoimmune mice 22 and scrapie-infected hamsters. 13 A recent study 26 reported immunohistochemical detection of the disease-associated N-terminally truncated form of PrP in kidneys of 48% of experimentally scrapie-infected sheep cases and of 25% of the natural scrapie cases. BSE infectivity has been detected by mouse bioassay in the kidney of ARQ/ARQ Romney sheep orally infected with BSE. 2 Based on this data, the possibility of TSE infectivity in urine of scrapie-infected sheep has to be carefully investigated. The present study provides experimental evidence of the elevated level of N-terminally truncated PrPc in urine of 21% of naturally scrapie-infected sheep. Although these results do not provide the basis of a useful diagnostic test for scrapie, it remains to be established by bioassay whether the elevated level of urinary PrPc is accompanied by scrapie infectivity in urine and potentially contributes to the horizontal transmission of scrapie.
Acknowledgements
The authors are grateful to Brian Cathcart, Pierre Gauthier, and Dean McIntyre for help with sheep urine samples collection; Hongsheng Huang for help with brain tissue homogenates preparation and prion protein quantification; and TSE unit staff for processing biopsies and immunostaining. This work was supported through a collaborative research agreement between Diachemix LLC. Milwaukee, Wisconsin, and the Canadian Food Inspection Agency (RPS#NBR 0421). Copyright by the Crown in right of Canada.
Footnotes
a.
Furosemide Injection Sabex Standard 5% solution, Sabex. Inc., Quebec, Canada.
b.
Millipore, Billerica, MA.
c.
Sigma-Aldrich Co., Oakville, Ontario, Canada.
d.
Pierce, Rockford, IL.
e.
Qbiogene, Carlsbad, CA.
f.
New England BioLabs, Inc., Pickering, Ontario, Canada.
g.
Bio-Rad, Mississauga, Ontario, Canada.
h.
VMRD, Inc., Pullman, WA.
i.
Jackson Immunoresearch Laboratories, Inc., West Grove, PA.
j.
Amersham, Piscataway, NJ.
k.
Clonex Co., Markham, Ontario, Canada.
l.
Alicon AG, Schlieren, Switzerland.
m.
MedCalc Software, Mariakerke, Belgium.
