Abstract
Mucopolysaccharidosis (MPS) types I and VII are inborn errors of metabolism caused by mutation of enzymes involved in glycosaminoglycan catabolism, which leads to intralysosomal accumulation of glycosaminoglycans. In children, severe forms of MPS I and VII are characterized by somatic and neurologic manifestations, including a poorly understood hearing loss. The purpose of this study is to describe the age-related histopathologic changes of the ear in spontaneous canine models of MPS I and VII. Pathologic changes in the ear were assessed in MPS I and VII dogs ranging from 1.6 to 9.3 months of age. Paraffin-embedded sections of the whole ear and Epon-embedded semithin sections of the cochlea were examined. The following lesions were blindly scored in the middle and inner ear: inflammation, cells vacuolization, thickening of osseous and membranous structures, perivascular vacuolated macrophages infiltration, and bone resorption. All dogs had lysosomal storage within cells of tympanic membrane, ossicles, tympanic bone and mucosa, cochlear bone, spiral ligament, limbus, and stria vascularis. The MPS I dogs mainly had progressive cochlear lesions. The MPS VII dogs had severe and early middle ear lesions, including chronic otitis media and bone resorption. The MPS I dog only partially recapitulates the pathology seen in humans; specifically, the dog model lacks inflammatory middle ear disease. In contrast, the MPS VII dog has severe inflammatory middle ear disease similar to that reported in the human. In conclusion, the canine MPS VII model appears to be a good model to study MPS VII–related deafness.
Keywords
Mucopolysaccharidoses (MPSs) are hereditary diseases of humans and domestic animals. They constitute a collection of heterogeneous conditions all characterized by a defect in glycosaminoglycan (GAG) catabolisms. Eleven types of MPS are reported in the human, with a global impact of 1.75 to 4.5 in 100,000 births.
1,9,10,16
Six types of MPS are reported in domestic animals,
28
each of which is defined by the deficiency of a ubiquitous lysosomal enzyme required for the catabolism of specific GAGs. MPS I is the first- or second-most prevalent of all the MPSs in people, depending on the geographic area.
1,9
–11,15,16
It is an autosomal recessive disease caused by deficient activity of the enzyme α-
For MPS I and VII, genetically engineered models (MPS I mouse model) and spontaneous models (MPS I and VII dog and cat models, MPS VII mouse model) are available. Plott hound dogs with an autosomal recessive α-
Hearing and otic pathology have been investigated in murine models of MPS; mostly MPS VII and, rarely, MPS I. 2,18,21 However, there is no published electrophysiologic (ABR) or histopathologic study of hearing in canine MPS models. This work is the first to provide histopathologic data relevant to hearing pathology in spontaneous large animal models of MPS I and VII.
The purposes of this study are to (1) thoroughly describe the otic histopathology in canine MPS I and VII, (2) compare otic lesions observed in dogs to those reported in human patients and discuss the relevance of dogs as models for otic pathology, and (3) give insights in the pathogenesis of MPS-related deafness.
Materials and Methods
Animals
Ten male dogs were included in this study: 5 MPS I–/– mixed-breed Beagle dogs, 1.6 to 9.3 months of age, homozygous for the mutant α-
The 5 MPS I–/– animals were 1.6, 7.2, 9.3, 9.3, and 9.3 months old at the time of their euthanasia; the 5 MPS VII–/– animals were 1.9, 3.5, 3.8, 5.4, and 5.6 months old. In all animals, histopathologic lesions in the ears were assessed using paraffin-embedded sections. Epon-embedded samples of the right inner ear were examined in 3 MPS I–/– dogs (all 9.3 months old) and 2 MPS VII–/– dogs (1.9 and 3.8 months old). The control group comprised 3 wild-type male Beagle dogs that were bred and kept under the same conditions as the diseased animals and euthanized at 2.0, 2.7, and 7.1 months old.
Euthanasia and Sampling
Animals were deeply anesthetized, euthanized with an intravenous overdose of barbiturics, and then rapidly injected with an intracarotid perfusion of 4% paraformaldehyde. The left temporal bones, encasing middle and inner ears, were collected for paraffin embedding in all animals. The right temporal bone of 3 MPS I–/– dogs (9.3 months), 2 MPS VII–/– dogs (1.9 and 3.8 months), and a wild-type dog (2.7 months) were collected and used for Epon embedding of the cochlea. The remaining right temporal bones were embedded in paraffin.
Macroscopic Examination
External ear canals, ear drums, and middle ear cavities were grossly investigated in all dogs.
Paraffin embedding
Temporal bones were postfixed in 4% paraformaldehyde at 4°C for at least 2 days with the bulla having been opened to facilitate fixation before decalcification. Ears were decalcified for 2 weeks by immersion in a solution of 5% formic acid/5% formalin in distillated water changed daily and kept at 4°C. A long and gentle decalcification was necessary to preserve fragile structures, such as the organ of Corti and associated hair cells. Once decalcified, samples were cut to section and assess, on the same slide, a longitudinal midmodiolar section of the cochlea and the entire middle ear cavity with the first ossicle (malleus) and the tympanic membrane (Figs. 1, 2 ). Sections were cut at a thickness of 4 μm and stained with hematoxylin and eosin (HE), periodic acid–Schiff, and a silver impregnation associated with Luxol fast blue.
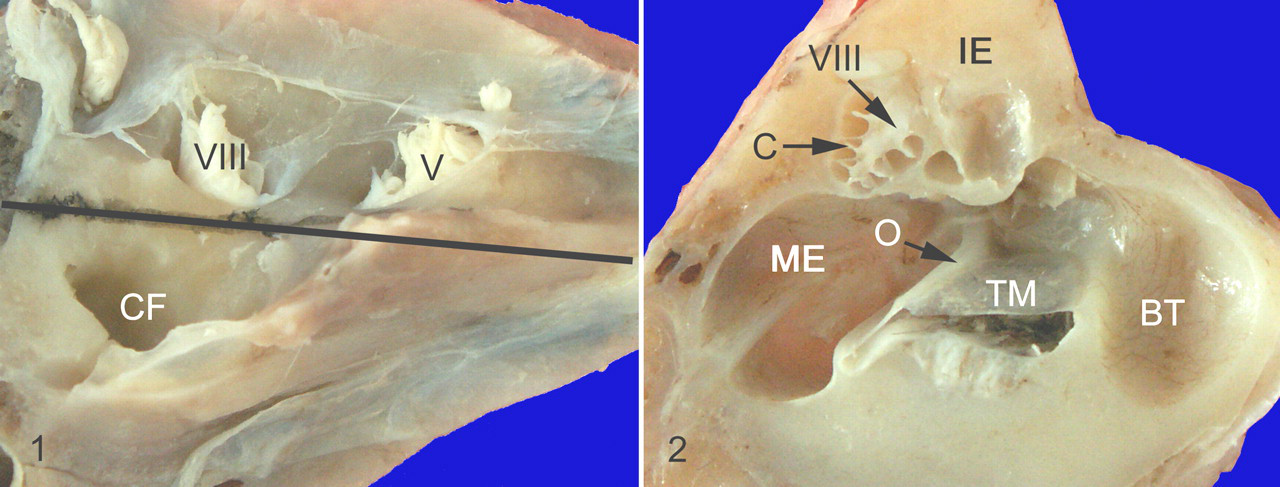
Epon embedding
The right temporal bones of 3 MPS I, 2 MPS VII, and 1 wild-type dog were postfixed in 4% glutaraldehyde for 48 hours at 4°C. The cochleae were isolated and decalcified for 3 weeks by immersion in a solution of 7% EDTA in phosphate buffered saline prepared fresh, changed daily, and kept at 4°C. Samples were postfixed in 4% glutaraldehyde, then in buffered 2% osmium tetroxide, dehydrated in an ascending ethanol series, and embedded in Epon. Semithin sections were cut at a thickness of 0.5 μm and stained with methyl azure blue.
Semiquantitative Scoring of Ear Lesions
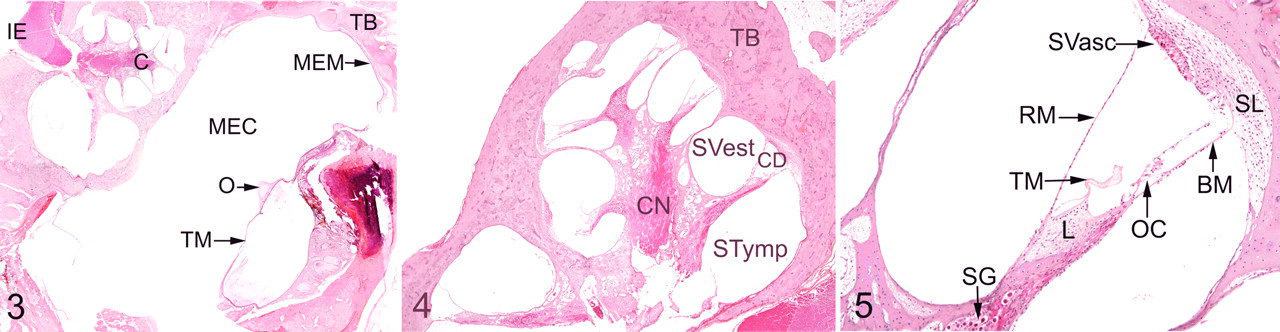
The following structures of the middle and inner ear were thoroughly observed: temporal bone, tympanic membrane, tympanic cavity, middle ear mucosa, malleus, all parts of the cochlea (Figs. 3–5). They were compared with control animals that did not display ear lesion at any age. Thorough descriptive analysis of ear lesions led to the identification of 6 major criteria used to quantify middle and inner ear lesions: (1) lymphocytic and plasma cell infiltration, (2) epithelial or mesenchymal cell vacuolization, (3) thickening of osseous structures, (4) thickening of membranous structures, (5) perivascular infiltration with vacuolated macrophages, and (6) bone resorption. In all dogs, middle and inner lesions were blindly scored on hematoxylin–eosin–safran sections according to the previously described major criteria (Table 1 ). Some criteria were not quantifiable and were therefore scored 0 (to indicate an absence) or P (to indicate the presence of the corresponding lesion). The other lesion criteria were quantified according to a scale ranging from 0 to 2 or 4, depending on severity (Table 1). The composite score had a possible range from 0 to 18 for middle ear and 0 to 19 for inner ear.
Scoring Grid of Middle and Inner Ear Lesions in Mucopolysaccharidosis Dogs
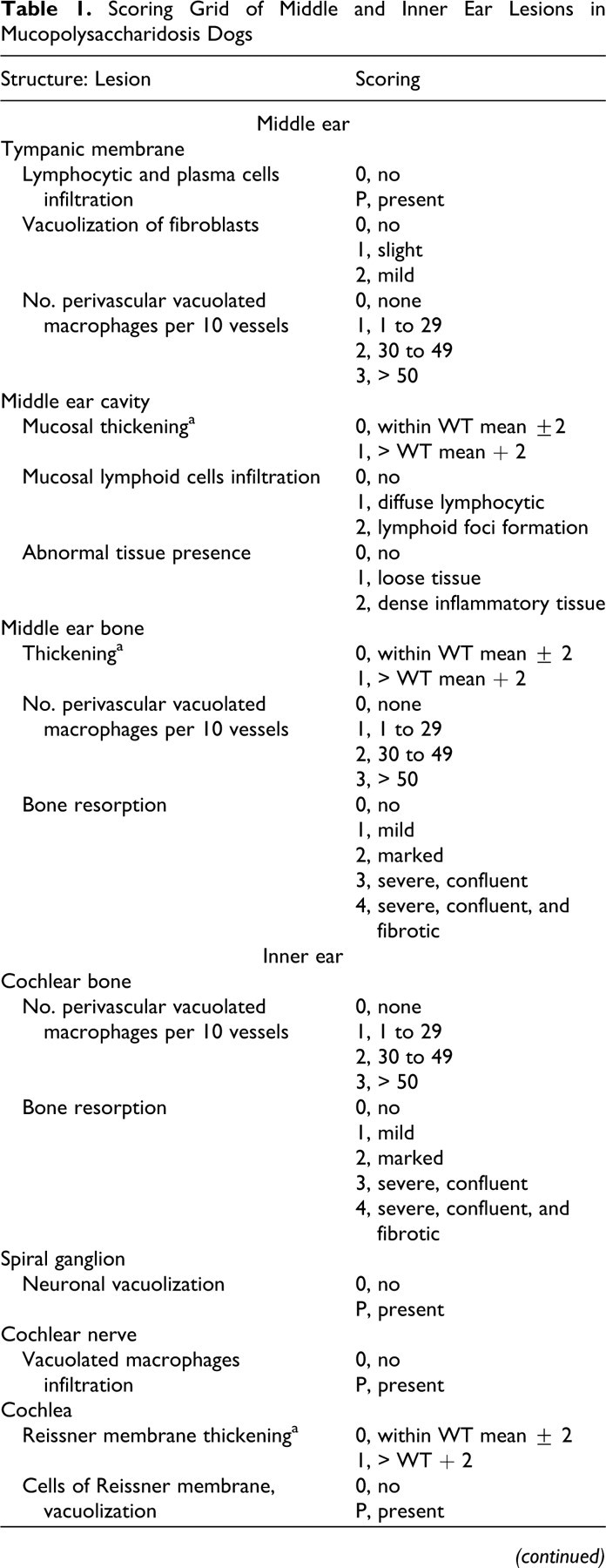
a Thicknesses were compared with mean values in controls. Normative range was control mean value, plus or minus 2 standard deviations. Basilar membrane thickness was always measured at the level of the middle turn of the cochlea. WT, wild type.
Results
Control animals had no ear lesion at any age. Because the middle and inner ear lesions differed between MPS I and VII dogs, we present the 2 ear compartments separately.
Middle Ear Lesions: Predominant Finding in MPS VII Dogs
There was no gross lesion in MPS I dogs. Gross examination revealed a marked stenosis of the external ear canal and middle ear cavity in all MPS VII dogs. The temporal bone lining the middle ear cavity was markedly thickened.
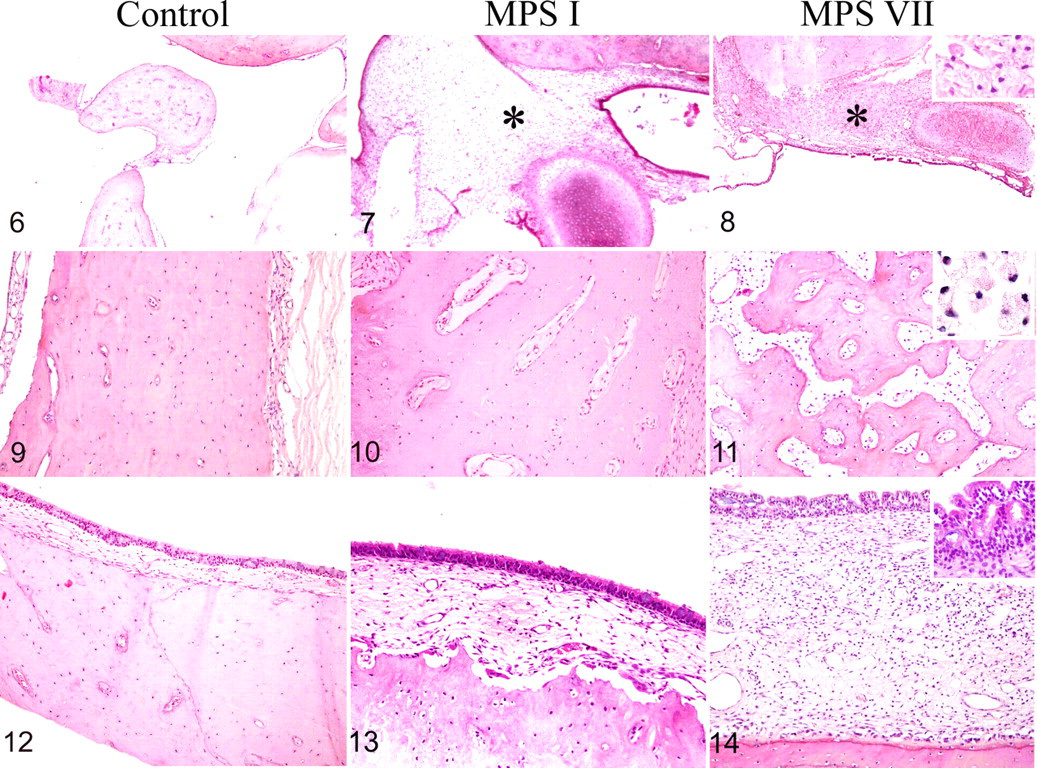
The tympanic membrane was moderately thickened in MPS I dogs compared with controls. This thickening was mostly due to hypertrophy of lamina propria, infiltration of perivascular spaces with numerous vacuolated macrophages, and vacuolization-associated fibroblast hypertrophy. Thickening was more pronounced in MPS VII dogs regardless of age and was chiefly due to lymphocytic and plasma cell infiltration and hyperkeratosis of the external epithelial lining.
Osteocytes and chondrocytes in the ossicles of MPS I and VII dogs were markedly enlarged and vacuolated. Independent of age, ossicles were remodeled by perivascular lacunae filled with osteoclasts and vacuolated macrophages in both models. In MPS VII dogs, perivascular lacunae led to a moth-eaten appearance at low magnification.
The middle ear cavity was partly obstructed by tissue encasing the ossicles in the 3 oldest MPS I dogs and in all MPS VII dogs from the age of 1.6 months (Figs. 6–8). In MPS I dogs, this tissue contained little collagen and abundant vacuolated macrophages (Fig. 7); in MPS VII dogs, it was dense and inflammatory with numerous lymphocytes (Fig. 8). In MPS VII only, the obstruction of middle ear cavity was exacerbated by a severe thickening of middle ear mucosa from the age of 1.9 months on and by temporal bone changes from the age of 3.8 months on.
The overall thickness of the bone lining the middle ear cavity was increased in MPS VII dogs. In MPS I dogs, the temporal bone of the bulla tympanica had a moderate enlargement of perivascular spaces when compared with the controls (Figs. 9, 10 ). Lacunae were centered on capillaries and filled with osteoclasts and a connective tissue containing numerous vacuolated macrophages. In MPS VII dogs, the lacunae were confluent with a subsequent trabecular aspect of the bone (Fig. 11 ). These lacunae resulted from increased bone resorption rather than a lack of formation, given that they contained osteoclasts and macrophages. The overall thickening of the temporal bone and its abnormal trabecular aspect indicate the coexistence of increased bone deposition and perivascular bone resorption. In the oldest MPS VII dogs, the bone resorption spaces were filled with a mature collagenous tissue.
When compared with that of the controls, the middle ear mucosa was moderately thickened in MPS I dogs and severely so in MPS VII dogs (Figs. 12–14). In MPS I dogs, the thickening was due to a hypertrophy of the lamina propria, infiltration of the perivascular spaces with numerous vacuolated macrophages, and fibroblast hypertrophy and vacuolization (Fig. 13). In MPS VII dogs, additional findings included epithelial hyperplasia and diffuse lymphocytic infiltration related to chronic otitis media (Fig. 13), which worsened with age.
The cytoplasmic vacuoles observed in all cell types did not stain positive by periodic acid–Schiff or Luxol fast blue, probably because of the processing technique and the long decalcification that likely led to dissolution of storage material.
To conclude, MPS I dogs had only slight middle ear lesions at all ages, characterized by perivascular infiltration of vacuolated macrophages and an absence of middle ear inflammatory lesions. The MPS VII dogs had severe restrictive middle ear lesions as early as 1.9 months of age, characterized by severe bone thickening and remodeling, vacuolated macrophages infiltration, and marked chronic otitis media.
Inner Ear Lesions: Numerous and Prominent in MPS I Dogs
When compared with that of the controls, the overall cochlear micro-organization was unchanged in all dogs. The cochlear bone showed lesions similar to those described in the rest of the temporal bone.
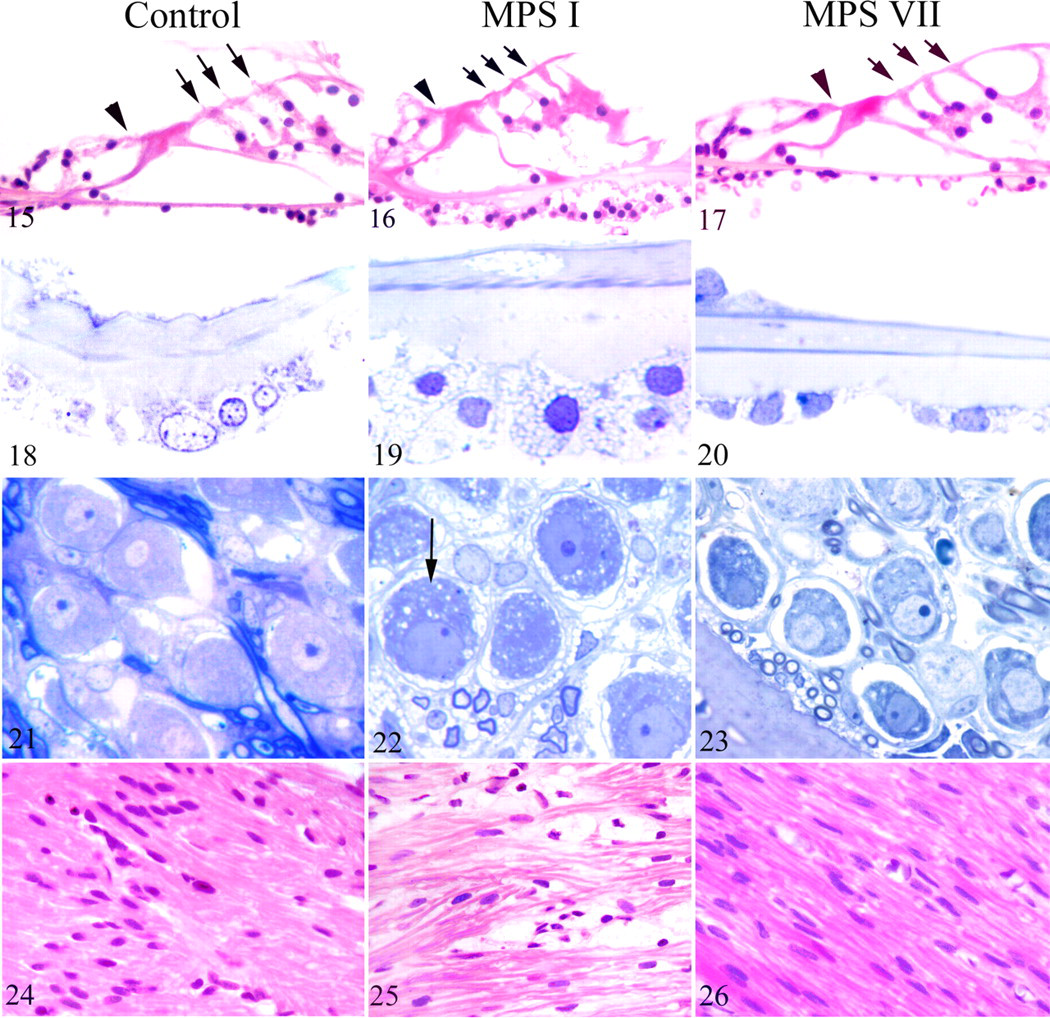
The organ of Corti did not display degenerative change or hair cell loss in any dog (Figs. 15–17). The basilar membrane was thickened in the 3 oldest MPS I dogs (9.3 months) when compared with the controls (Figs. 18, 19 ). Thickening was chiefly due to marked vacuolization of the mesenchymal cells (Fig. 19). Basilar membrane cell vacuolization was slight in dogs with MPS VII and only visible on the semithin sections (Fig. 20 ).
In the spiral ganglions (Figs. 21–23), vacuoles were observed in neurons and satellite cells of the 3 oldest MPS I dogs (Fig. 22). Vacuolization was more subtle in dogs with MPS VII and observed on only the semithin sections (Fig. 23).
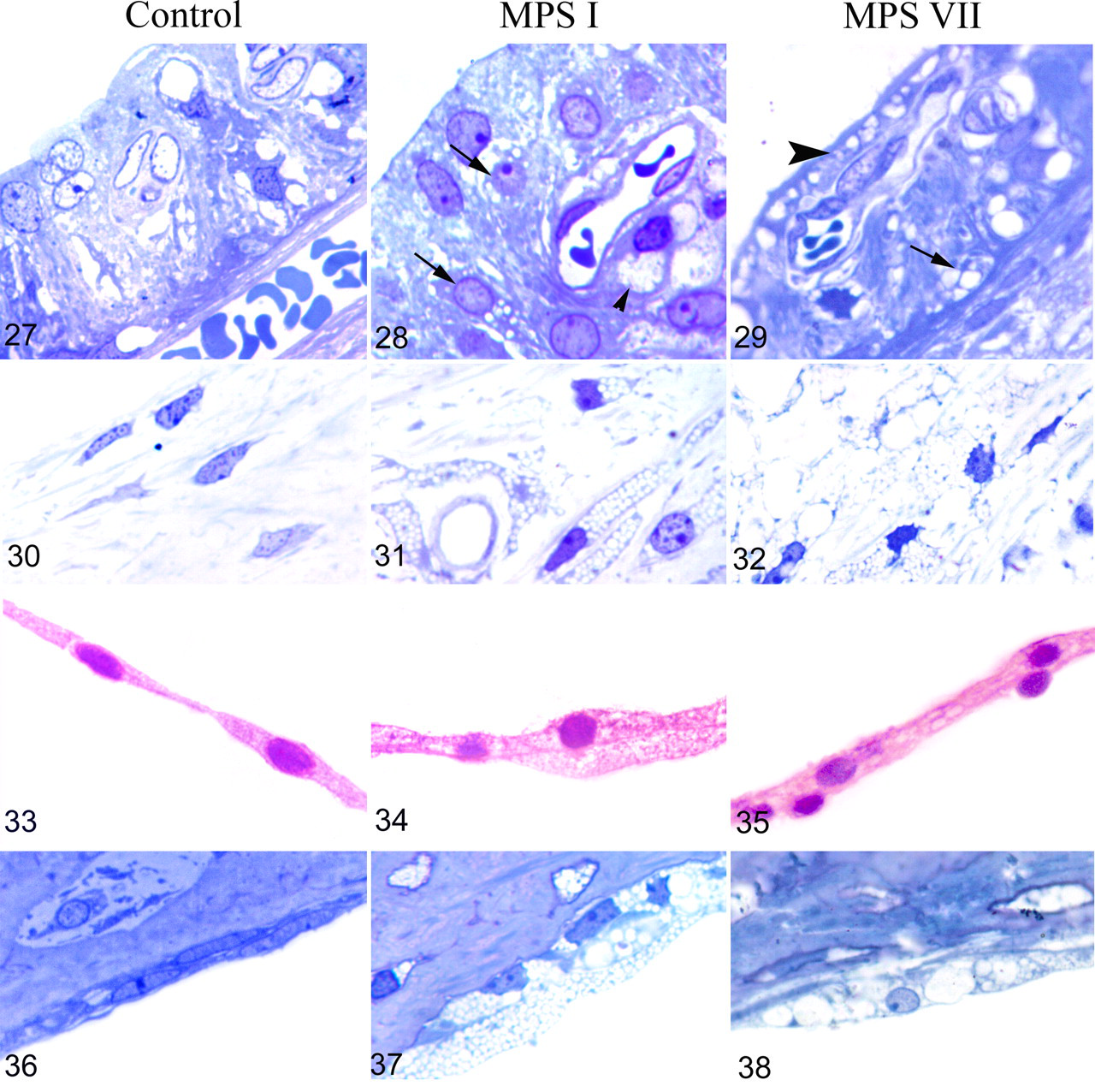
The cochlear nerve (Figs. 24–26) was infiltrated by vacuolated swollen cells (presumably macrophages), leading to a disruption of nerve fibers in MPS I dogs (Fig. 25), seen as early as 1.6 months. No cochlear nerve lesion was observed in MPS VII dogs at any age (Fig. 26). In the stria vascularis (Figs. 27–29), vacuolization was observed in perivascular macrophages and epithelial intermediate cells in MPS I dogs (Fig. 28). Marginal and basal epithelial cells were vacuolated in MPS VII dogs (Fig. 29). Fibroblasts of the spiral ligament (Figs. 30–32) and limbus were markedly vacuolated in both MPS I (Fig. 31) and MPS VII (Fig. 32) dogs with an age-related worsening. Reissner membrane was increasingly thickened from the age of 7.2 months in MPS I dogs and almost normal in MPS VII dogs when compared with the controls (Figs. 33–35). Cells lining the scala tympani (Figs. 36–38) were markedly hypertrophied and vacuolated in MPS I (Fig. 37) and MPS VII (Fig. 38) dogs.
To conclude, MPS I dogs had numerous inner ear lesions, which worsened with aging. MPS VII dogs, euthanized younger, had similar but slighter lesions apart from cochlear nerve infiltration, which was never present in MPS VII dogs. The organ of Corti was normal in all dogs, and there was no otitis interna.
Semiquantitative Scoring of Ear Lesions
Semiquantitative scoring confirms that middle ear lesions predominated in MPS VII dogs. In MPS I dogs, the inner ear was the most involved structure. The middle ear scores ranged from 5 and 8 in the MPS I dogs and between 8 and 15 in the MPS VII dogs (Table 2 ). The middle ear mean score was 6.6 ± 1.1 in the MPS I dogs and 12.4 ± 2.9 in the MPS VII dogs. The relatively higher middle ear scores in MPS VII dogs are chiefly explained by the marked otitis media and severe tympanic bone lesions observed (Table 2). Those lesions were present in the youngest animal (1.9 months) and worsened slightly with age. Interestingly, although most MPS I dogs were older than MPS VII dogs at the time of their euthanasia, they showed only slight middle ear lesions. Hence, middle ear disease is a prominent feature in only canine MPS VII.
Comparative Semiquantitative Scoring of Ear Lesions in Mucopolysaccharidosis I and VII Dogs, by Age (Mos) a
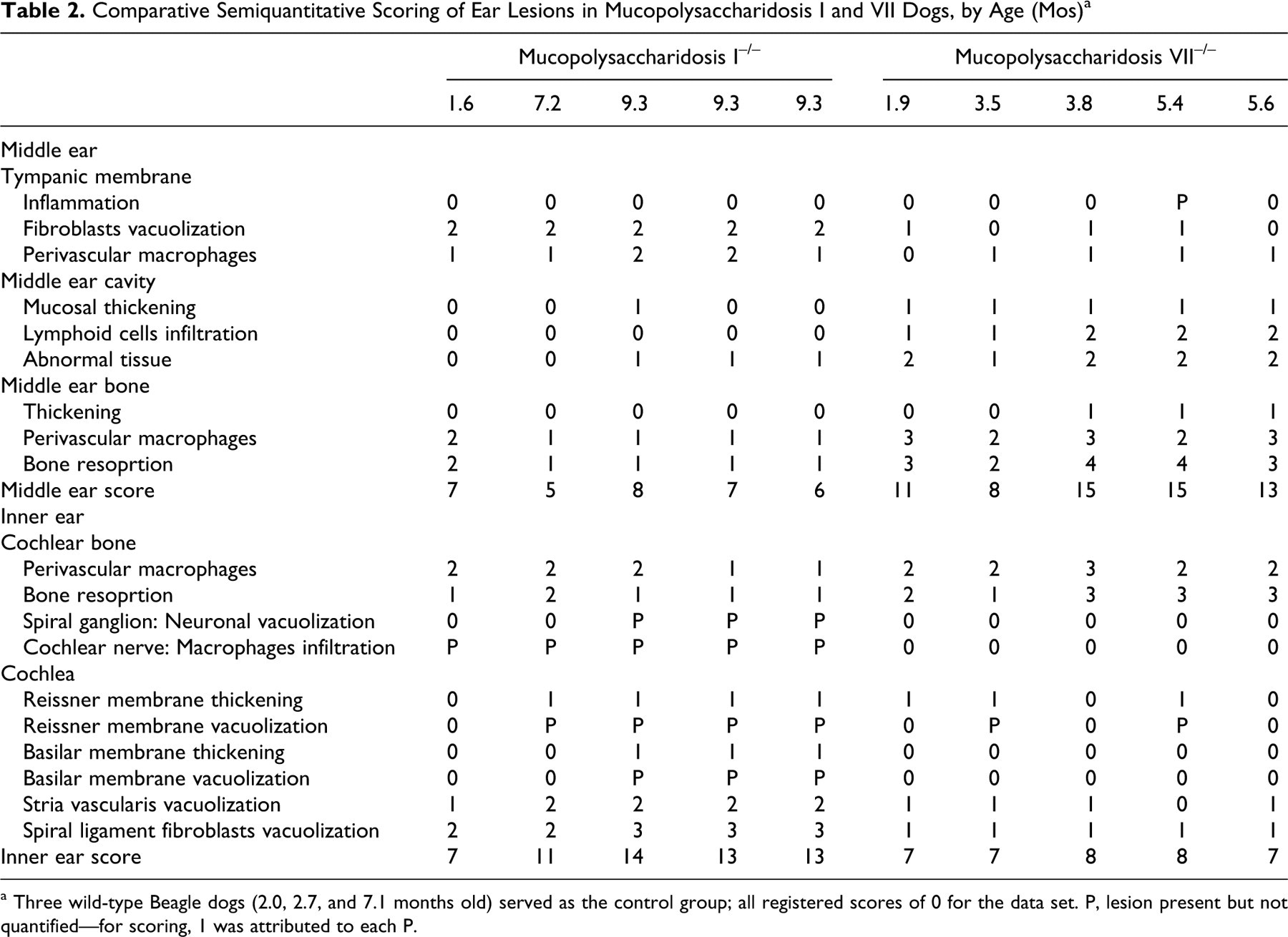
a Three wild-type Beagle dogs (2.0, 2.7, and 7.1 months old) served as the control group; all registered scores of 0 for the data set. P, lesion present but not quantified—for scoring, 1 was attributed to each P.
The higher inner ear scores in older MPS I dogs were due to the numerous cochlear lesions, which were absent or milder in MPS VII: cochlear nerve disruption by vacuolated cells, spiral ganglion neuronal vacuolization, basilar membrane thickening, and spiral ligament fibroblasts vacuolization. Cochlear lesions were characterized by an age-related worsening in MPS I dogs: The youngest animal had a score of 7 and the oldest had a score of 14 (Table 2). MPS VII dogs had a stable inner ear score (7 or 8), but their severe phenotype did not allow us to collect ear samples after the age of 5.6 months.
Discussion
This report is the first to describe ear histopathology in large animal models of MPS I and MPS VII. The MPS I dogs have mild middle ear lesions, which remain stable from the age of 1.6 to 9.3 months, and cochlear lesions, which increase in number with aging. The MPS VII dogs mainly have severe and early middle ear lesion from the age of 1.9 months on.
Age-related evolution of the disease probably explains the difference observed in inner ear lesions between canine models of MPS I and VII. In MPS I dogs, inner ear lesion scores vary from 7 in the youngest animal (1.6 months) to 14 in the oldest (9.3 months). Hence, the most prominent inner ear lesion are observed in only the oldest dogs (9.3 months), which reveals an age-related worsening of inner ear lesions. Because of humane and ethical reasons, no MPS VII dog was maintained after the age of 5.6 months. With aging, dogs with MPS VII might have developed cochlear lesions similar to those of dogs with MPS I. Their short life expectancy may explain the scarcity of inner ear lesions in this model.
In MPS VII dogs, otitis media is probably secondary to poor drainage owing to middle ear cavity and external ear canal stenosis (both absent in MPS I dogs). Inflammatory lesions then worsen middle ear obstruction and lead to a self-perpetuating cycle. Middle ear stenosis is related to a more severe temporal bone deposition and thickening of middle ear mucosa in dogs with MPS VII, when compared with MPS I. The severe middle ear lesions in canine MPS VII correspond to those reported in murine models of MPS VII and I. 2,21 The literature contains no description of otic pathology in MPS VII human patients. Middle ear lesions observed in MPS VII dogs are similar to those described in Hurler disease patients (ie, MPS I, severe phenotype): thickened middle ear mucosa, obstruction by mesenchyme, middle ear effusion owing to otitis media, and lytic bone lesions. 6,7,22 The MPS I dogs, on the contrary, have mild middle ear lesions and do not have the otitis media reported in their human counterparts. Hence, the MPS I dog is not an appropriate model to study middle ear lesions and conductive hearing loss described in Hurler syndrome. However, the MPS VII dog has marked middle ear lesions and otitis media and is thus a good model to study middle ear lesions and their impact on conductive hearing loss in Sly syndrome (MPS VII).
Two hypotheses could explain the absence of middle ear tissue hypertrophy and subsequent otitis media in canine MPS I. The first is the nature of the stored material. MPS VII is characterized by the accumulation of heparan sulfate (HS), dermatan sulfate (DS), and chondroitin sulfate (CS), whereas only HS and DS are accumulated in MPS I. HS and CS present at the cell surface and in the context of proteoglycans, are necessary for the activation of FGF receptors and the stimulation of fibroblasts growth. 12,13,19 Overstimulation of FGF receptors on fibroblast mediated by HS and CS in MPS VII might be involved in the hypertrophy of lamina propria in middle ear mucosa. This hypothesis cannot, however, explain the difference observed between dogs with MPS I and patients with Hurler syndrome, both accumulating the same GAGs. Canine MPS I is characterized by a mild phenotype (resembling Hurler-Scheie syndrome). Dogs with MPS I store less GAGs in liver and spleen when compared with patients with Hurler syndrome. 23 Therefore, as compared with patients with Hurler syndrome, dogs with MPS I may have less GAG accumulation in their middle ear and, subsequently, milder lesions.
MPS I dogs have inner ear lesions similar to those reported in murine models: vacuolization of fibroblasts, basilar membrane and Reissner membrane cells, spiral ganglion cells, and auditory nerve cells. 2,18,21 Those lesions have not been reported in Hurler syndrome (MPS I-H) patients. 6,7,22 This discrepancy could be explained by an age-related evolution of the disease observed in animal models compared with the very short life expectancy of MPS I-H patients. Otic histopathologic changes of MPS I-H have been described in young children between 2.5 and 6.0 years old. 6,7,22 In our study, the most prominent inner ear lesions were observed in older dogs only. Similarly, the age-related study of otic pathology in MPS I mice reported an absence of lesions in newborn mice and a progressive worsening of inner ear lesions from the age of 2 months (young adults) to 1 year (old adults). 21 Our results suggest that mature MPS I dogs—which present well-developed inner ear lesions and almost no middle ear lesions—may be a good model to study, by electrophysiology, the sensorineural component of hearing loss in Hurler syndrome. It is indeed difficult to perform functional assessment of hearing with ABR when a marked conductive hearing loss is present. Our preliminary ABR data in MPS I dogs show a predominantly sensorineural and retrocochlear hearing loss (J.H., American College of Veterinary Pathologists/American Society for Veterinary Clinical Pathology meeting 2008, abstract No. 155). The mild involvement of middle ear disease and the progressive worsening of inner ear lesions in MPS I dogs make them a good model to characterize, by electrophysiology, the neurogenic component of deafness.
Otic lesions with sensorineural deafness assessed by ABR are also reported in caprine and bovine β-mannosidosis. 20,21 Middle and inner ear cells (including supporting and hair cells of the organ of Corti) are markedly vacuolated in both models. Otic lesions are closed but more severe in caprine or bovine β-mannosidosis when compared with canine MPS.
There was no degenerative change and no hair cell loss in the organ of Corti in either canine model. However, preliminary ABR testing in our dogs demonstrated endocochlear dysfunction. The lesions observed in the stria vascularis and spiral ligament could explain this sensorineural hearing loss. Epithelial cells of the stria vascularis and fibroblasts of the spiral ligament and limbus are vacuolated in MPS I and VII dogs. Stria vascularis epithelial cells and inner ear fibroblasts are responsible for the maintaining of cochlear ion homeostasis. An imbalance in ionic composition of endolymph could explain a sensorineural hearing loss in the absence of degeneration of the organ of Corti. 21
The temporal bone lytic lesions are one of the most striking findings in MPS VII dogs. The large and confluent lacunae observed in temporal bone and ossicles are also reported in Hurler disease patients 6,7 and MPS VII mice. 2 They are similar to the bone resorption defects described in human otosclerosis, 8 a complex disease attributed to persistent measles virus infection as well as genetic factors. 29 Otosclerosis is morphologically characterized by proliferation of spongiotic bone with lacunae centered on capillaries and filled with osteoblasts, osteoclasts, and macrophages. 8 In MPS VII dogs, temporal bone lesions are similar and characterized by increased bone deposition (thickening), with the presence of confluent perivascular osteolytic spaces filled with osteoclasts and numerous vacuolated macrophages. Pathogenesis of human otosclerosis is unclear. Given that abnormal oversulfated GAGs are reported to be increased in otosclerotic bone matrix, 20 the accumulation of unprocessed HS, DS, and CS in MPS VII may lead to abnormal bone remodeling. Osteolytic lesions are present but milder in dogs with MPS I. The GAGs stored in MPS I and VII differ in their forms, in particular at the nonreducing terminal which might impact any pathologic effect they exert. Additionally, endoglycosidase activity may differ in these 2 diseases and so render different fragment lengths, which could further affect pathology. Storage of CS and the presence of differently processed GAGs might explain the severity of temporal bone lesions in dogs with MPS VII. As discussed above, dogs with MPS I may have less marked lesions because of their relatively mild phenotype. Interestingly, otosclerosis is associated with sensorineural hearing impairment owing to tumor necrosis factor α (TNF-α) release in the perilymph. 29 To our knowledge, there is no information concerning TNF-α concentration in cochlear lymph in MPS models. Investigations are necessary to determine if a release of TNF-α from osteolytic temporal bone in MPS is implicated in sensorineural hearing loss.
In conclusion, this work provides a thorough evaluation of ear pathology in the canine models of MPS I and VII, and it gives insights into the histopathologic correlates of conductive and sensorineural hearing loss in these diseases. Both models are complementary in the study of MPS-related hearing loss. The MPS VII dog is a good model to study middle ear lesions and the conductive component in Sly syndrome–related deafness, whereas the MPS I dog has a predominant inner ear disease that will permit precise investigation of the histopathologic and electrophysiologic components of the sensorineural deafness in Hurler syndrome. The availability of complete ear function assessments in large animal models will enable the evaluation of specific therapies for MPS-related hearing loss.
Footnotes
Acknowledgements
We thank Dr N. Matthew Ellinwood, who assisted in the revision of English-language usage in the manuscript. We thank the staff of the Gene and Cellular Therapy Center of Boisbonne (Nantes, France) for animal care and breeding. This work was supported by the Association Française contre les Myopathies.
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
This work was supported by a grant from the AFM (Association Française contre les Myopathies).
