Abstract
A male cynomolgus macaque at the age of 3 years and 11 months suffered sudden cardiac arrest during a surgical operation. This animal had been clinically asymptomatic for 6 months from the acclimatization period to death. At necropsy, a white mass approximately 5 cm in diameter was found at the base of the heart. Histopathologically, the mass consisted of a granuloma with a number of multinucleated giant cells and multiple necrotic foci. Fungal hyphae characterized by parallel cell walls, distinct septa, and branching were observed in the lesion. The granuloma extended into the thoracic lymph nodes and the subepicardium of the left atrium, compressed the bronchioli, and was separated from the pulmonary parenchyma by a thick fibrous layer. The hyphal morphology and results of polymerase chain reaction assays demonstrated that the pathogen was Aspergillus sp.
A male cynomolgus macaque (Macaca fascicularis) purpose bred in Vietnam was purchased from a breeder at the age of 3 years and 5 months. During quarantine, test results for simian immunodeficiency virus, cercopithecine herpesvirus 1 (B virus), simian retrovirus type D, simian varicella-zoster virus, and Salmonella spp were all negative. In addition, during the 13 months before death, tuberculin tests for Mycobacterium tuberculosis and Mycobacterium bovis were performed 4 times, and all results were negative. During the acclimatization period of 6 months, no clinical signs were observed. The animal was selected for a telemetry study and had open chest surgery under isoflurane anesthesia to implant electrodes on the heart. Sudden cardiac arrest occurred soon after the thoracic cavity was opened. The animal was immediately submitted for necropsy.
A white firm mass with a rough surface approximately 5 cm in diameter was found at the base of heart. The mass adhered to the left atrium and to the pulmonary artery and the left middle and posterior lobes of the lung, but the origin of the mass was unclear. The cut surface of the mass was white and solid (Fig. 1 ). The tracheobronchial lymph nodes adjacent to the mass were enlarged. No significant macroscopic abnormality was observed in the other thoracic or abdominal organs.
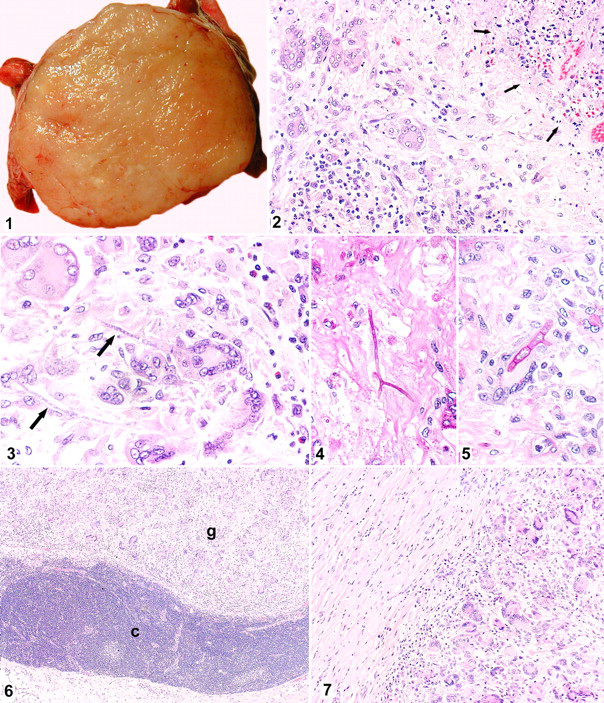
Cut surface of the mass at the base of the heart. The mass is white and firm.
Differential Diagnoses
Differential diagnoses for the thoracic mass included neoplasms such as lymphoma and infectious diseases such as tuberculosis or mycoses.
Microscopic Findings
The entire mass, all the lobes of lung, heart, tracheobronchial lymph nodes, thymus, liver, and spleen were fixed in 10% neutral buffered formalin. The tissues were embedded in paraffin, sectioned, and stained with hematoxylin and eosin (HE). Additionally, sections of the mass and lungs were stained with periodic acid–Schiff (PAS), Gomori methenamine silver (GMS), Ziehl-Neelsen, and Gram (Hucker-Conn method) stains.
The mass consisted of a granulomatous lesion characterized by multinucleated giant cells, epithelioid macrophages, and multiple necrotic foci (Fig. 2), accompanied by infiltration with lymphocytes, plasma cells, macrophages, neutrophils, and eosinophils. A thick fibrous capsule surrounded the mass, and fibrous trabeculae extended into it, suggesting that the granuloma originated in a lymph node. Weakly basophilic cylindric structures were observed in the cytoplasm of the giant cells and/or surrounding stroma and necrotic areas (Fig. 3). These structures had parallel walls that were clearly stained with PAS and GMS stains and thus were determined to be fungal hyphae (Figs. 4, 5). The hyphae were uniformly about 5 μm in diameter, had distinct septa that were strongly PAS positive, and gave off branches at acute or right angles. These characteristics are consistent with Aspergillus spp, 1,4,5 whereas zygomycetes typically stain well with hematoxylin stain and have hyphae with an irregular branching pattern and rare septa.
In the enlarged tracheobronchial lymph nodes, similar granulomatous inflammation as in the mass was observed in the medullary sinuses (Fig. 6). The granulomatous inflammation extended to subepicardial myocardium of the left atrium (Fig. 7). There were no histopathologic changes in the thymus, liver, or spleen.
Ten lung samples representing all the lung lobes were embedded, sectioned, and examined with HE, PAS, and GMS stains. At the site of adhesion of the lung and the mass, the mass compressed bronchioli but was clearly demarcated by a thick fibrous layer and had no continuity with the pulmonary parenchyma. An accumulation of mucus in dilated alveoli in the lobes adherent to the mass and slight focal inflammation in the other lobes were observed, but no fungal organisms or granulomatous inflammation was found in the lung. No bacteria were found in the sections of the mass or the parts of lung adjacent to the mass stained with Ziehl-Neelsen or Gram stains.
Laboratory Findings
To confirm the identity of the organism as Aspergillus spp, polymerase chain reaction (PCR) assay was performed. DNA was extracted from paraffin blocks of the mass, lung, and liver from this animal and from the lung and liver from a healthy control cynomolgus macaque. The duration of formalin fixation of these samples was less than 10 days. Three paraffin sections, 5 μm in thickness for each block, were pooled in a 1.5-ml centrifuge tube and deparaffinized with xylene and ethanol. To disrupt the fungal cell walls, the sections were incubated in lysis buffer containing 300 U/ml lyticase (Sigma, St Louis, MO), 10 mM Tris, 1 mM EDTA, and 20 mM 2-mercaptoethanol, followed by 3 cycles of freezing in liquid nitrogen and thawing. The samples were then incubated in proteinase K overnight, and the DNA was extracted with a commercial kit (QIAamp DNA Mini Kit, Qiagen, Valencia, CA) in accordance with the manufacturer’s instructions. PCR amplification was performed with 0.5 μl (approximately 100 ng) of DNA as the template in a total reaction volume of 25 μl consisting of 12.5 μl of Ex Taq buffer, 2 μl of dNTP mix, 0.125 μl of Ex Taq (all provided by Takara, Shiga, Japan), and 0.5 μM each of the primer pairs. To increase the specificity of the PCR assay, 2 primer pairs were used, both targeting the mitochondrial DNA of Aspergillus spp. Primers P2 (5′-CTTTGGTTGCGGGTTTAGGGATT-3′) and Asp2 (5′-GGGAGTTCAAATCTCCCTTGGG-3′) amplify a 201–base pair fragment, and primers P2 and P1 (5′-GAAAGGTCAGGTGTTCGAGTCAC-3′) amplify a 135–base pair fragment. 9,10 PCR using each primer pair was performed separately. Thirty cycles of amplification were performed after an initial denaturation of DNA at 94°C for 5 minutes. Each of the 30 cycles consisted of a denaturation step of 94°C for 30 seconds, an annealing step of 58°C for 1 minute, and an extension step of 72°C for 1 minute, followed by a final extension at 72°C for 3 minutes. To confirm that the extracted DNA samples did not contain any contaminants that could inhibit the PCR reaction, PCR amplification was performed using primers for β-actin of cynomolgus macaque (sense, 5′-ATCATGTTTGAGACCTTCAACACC-3′; antisense, 5′-TGAGGATCTTCATGAGGTAGTCAG-3′). The method of PCR for β-actin was the same as for Aspergillus spp except for an annealing temperature at 60°C. The PCR products were detected by agarose gel electrophoresis. As shown in Figure 8, PCR assays to detect the DNA of Aspergillus spp were positive in the sample extracted from the mass. The samples from the liver of this case and the liver and lung of the control animal were negative.
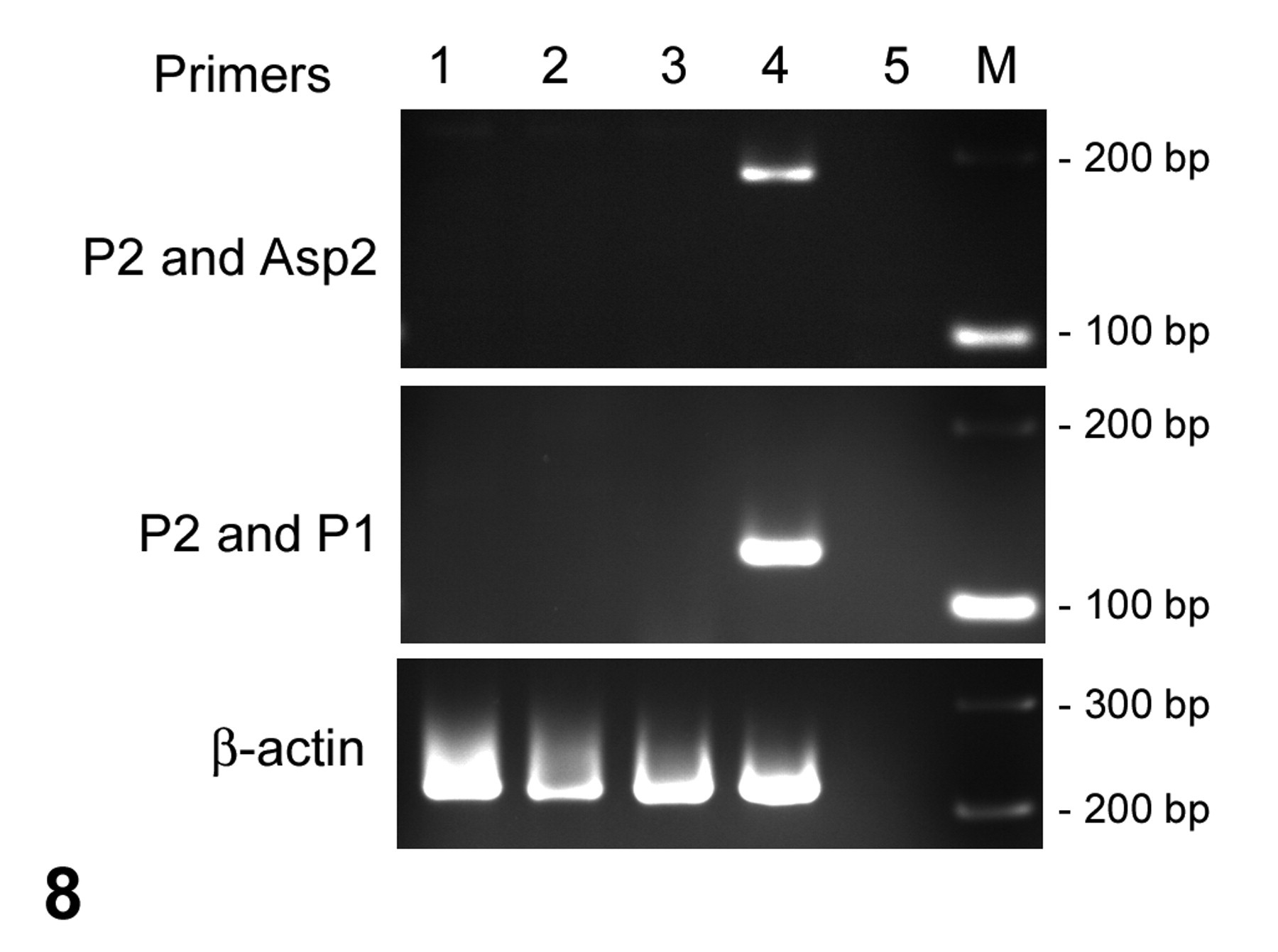
Agarose gel demonstrating the polymerase chain reaction assay. DNA of Aspergillus spp is detected in the samples obtained from the mass at the base of the heart. The polymerase chain reaction for β-actin of cynomolgus macaque is positive in all samples except for the negative control. Lane 1: Liver of the control animal. Lane 2: Lung of the control animal. Lane 3: Liver of this case. Lane 4: Mass at the base of the heart of this case. Lane 5: Negative control amplified without DNA sample. M, band size marker; bp, base pairs.
PCR products of P2 and P1 primers against the DNA extract from the mass were sequenced using a commercial sequencing service (Life Technologies, Carlsbad, CA). According to a BLAST search of the GenBank database (National Center for Biotechnology Information, National Institutes of Health, Washington DC), the sequence was 97% identical to a sequence of Aspergillus flavus (accession No. AAIH02000498) obtained by whole genome shotgun sequencing.
Diagnosis
Fungal granuloma caused by Aspergillus sp.
Discussion
In this case, the fungal morphology strongly suggested that the causative agent was Aspergillus sp. Presence of conidiophores is diagnostic, 4,6,8 but these are typically present only in lesions exposed to the air, such in respiratory tract lumina, and not in solid tissues. In this case, despite extensive histopathologic examination, conidiophores were not detected in the lung or in the granuloma.
The PCR analyses confirmed the identity of the fungal elements as Aspergillus sp. The primer sets used amplify sequences of several Aspergillus spp, such as A fumigatus, A flavus, A terreus, and A niger. 9,10 The sequence of the PCR product suggested that the species was A flavus; however, because of the limited number of sequences of Aspergillus spp deposited in the GenBank database, it is possible that the PCR amplified DNA of another Aspergillus sp whose sequences are not included in the database.
The route of infection and the cause of colonization or invasion were an important concern in this case. Aspergillus sp is a ubiquitous saprophytic fungus with a worldwide distribution. It is an opportunistic agent and is usually nonpathogenic in immunocompetent mammals. 2,4,6,8,12 In our case, no clinical signs were observed before death, and the lesions were restricted in the thoracic mass, tracheobronchial lymph nodes, and parts of the organs adhered to the mass. Histopathologic examination detected no abnormalities in the spleen or thymus. Thus, it is suggested that the protective reaction of the host against the pathogen was maintained. A partially or mildly immunocompromised status of the monkey might have led to the localized invasion of Aspergillus sp through the airborne route of exposure. Unfortunately, because hematologic and biochemical examinations in this animal were not performed, the accurate evaluation of immunologic function for this animal was not possible. Also, there were no clinical parameters available to explain the death of this animal. The cause of death and its relation to aspergillosis was unclear.
The origin of the mass was unclear; however, given the location and structure of the mass, the lesion may have originated in a lymph node and may so represent a primary lymph node infection. In humans, a case of primary lymph node aspergillosis was reported in which the patient had no predisposing factors for opportunistic diseases. 7 A silent lung infection of Aspergillus sp and a selective macrophage defect against the species were suggested as the cause of the case.
The possibility of concurrent involvement of tuberculosis in aspergillosis should be considered. Tuberculosis is a well-known underlying disease in chronic necrotizing pulmonary aspergillosis and aspergilloma, with the clinical symptoms of aspergillosis being characterized by limited invasiveness that occurs in mildly immunocompromised patients. 2,12 In addition, concurrent tuberculosis has been reported in a case of mycotic lymphadenitis in cattle. 3 In our case, results of 4 tuberculin tests were all negative, as were histopathologic examination for mycobacteria using Ziehl-Neelsen and Gram stains. Thus, it is unlikely that tuberculosis is an underlying disease in our case. However, further analysis, such as PCR testing, is necessary to definitively exclude the possibility of the concurrent occurrence of aspergillosis and tuberculosis.
Although some specific types of aspergillosis and their predilection have been reported in dogs and other mammals, 11 cases of spontaneous invasive aspergillosis in purpose-bred monkeys are not well known. The clinical signs of aspergillosis are nonspecific and vary with the location of the infection site or the degree of the immune suppression of the host. 4 Note that asymptomatic fungal infection can occur spontaneously in purpose-bred nonhuman primates and thus affect the results of preclinical toxicologic studies.
Footnotes
Acknowledgements
We thank Dr Rui Kano, Nihon University School of Veterinary Medicine, and Dr Tokuma Yanai, the United Graduate School of Veterinary Sciences, Gifu University, for their helpful advices on polymerase chain reaction–based identification of Aspergillus spp and the morphologic diagnosis, as well as Emiko Maeda, Yusuke Sudo, and Yoshiyuki Furukawa, Takeda Pharmaceutical Company Limited, for technical assistance in histopathology, technical advice of polymerase chain reaction experiments, and animal management, respectively. We also thank the members of the Kansai Conference of Toxicologic Pathology for critical discussion.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
