Abstract
The objective of this study was to develop an intestinal model of Mycobacterium avium subspecies paratuberculosis (Map) infection in the calf for evaluation of mucosal pathology and local and systemic immunologic responses. Map was inoculated into Peyer’s patches of young calves using a right flank surgical approach in standing calves to exteriorize the ileocecal junction. Inoculum doses ranging from 103 to 109 colony-forming units of strain K10 Map were injected through the serosal surface into Peyer’s patches of the distal ileum near the ileocecal valve. Fecal samples were collected for culture from each calf weekly until termination of the study. Calves were necropsied at 7, 30, 60, and 90 days after infection, when inoculation sites, lymph nodes, spleen, and peripheral blood were collected for evaluation. Ileocecal lymph nodes were consistently colonized by Map in the 105 to 109 groups. The ileocecal valve was also colonized in 107 and 109 groups. This correlated with fecal culture results as infected calves intermittently shed Map in their feces throughout the study. Granulomatous lesions with giant cells and acid-fast bacilli at the ileocecal junction, ileocecal lymph nodes, and lamina propria of high-dose animals (107 and 109) were identified from each time point. Flow cytometry was used to detect antigen-specific production of interferon-γ and interleukin-4 locally (ileocecal lymph node) and systemically (peripheral blood mononuclear cells), which defined distinct immunologic profiles in low-dose and high-dose calves. This study demonstrates intestinal Map infection via Peyer’s patch inoculation, a novel model with many shared features of natural Map infection.
Mycobacterium avium subspecies paratuberculosis (Map) is the causative agent of ruminant paratuberculosis (Johne’s disease), a disease widely distributed in domestic and free-range ruminants throughout the world. The prevalence is unknown in many parts of the world, but in the United States it has been recently estimated to affect as high as 68% of dairy herds. 17 Economic losses result directly from reduced milk yield, premature culling, and reduced slaughter value and indirectly as shedding adults serve as a source of infection for susceptible herd mates and young calves. 12
A considerable amount of research using a variety of experimental model systems has been devoted to this disease in recent years, yet the pathogenesis of intestinal Map infection remains incompletely understood. It is generally accepted that infection occurs primarily during the neonatal period when calves ingest an infectious dose of Map from feces or colostrum. The organism is likely endocytosed by epithelial M cells overlying ileal Peyer’s patches and subsequently phagocytosed by subepithelial macrophages and dendritic cells. 25 A major challenge for studying the disease is the lengthy subclinical infection period of Map (from 2 to 5 years) and the difficulty associated with accurate identification of infected animals during this period. A number of recent studies in cattle have documented infection by tissue culture, fecal culture, and/or polymerase chain reaction (PCR) following oral inoculation 8,9,16,19,20,22 –24,26 or intratonsillar inoculation; 24 however, intestinal histopathologic lesions containing acid-fast bacteria are not a consistent feature of experimental infection.
In a recent review of experimental model systems for studying Map infection, Hines et al 6 articulated pressing knowledge gaps regarding Map infection, which include the following questions. How does early tissue colonization relate to protection or lack of protection? Is there correlation between tissue colonization and fecal shedding? What is the optimal but practical number of necropsy tissues to collect for culture and histopathology?
Given the lengthy time course and cost associated with studying natural disease and in view of recent recommendations for studying Map infection, our objective for the current study was to develop a short-term experimental calf model to adequately address these unanswered questions relevant to the immunopathogenesis of intestinal Map infection. With the rationale that submucosal Peyer’s patches of the distal ileum are the initial site of Map infection in the calf, our hypothesis was that direct delivery of an infectious dose of live Map into this anatomic location is sufficient to induce rapid intestinal infection with pathologic and immunologic similarity to naturally occurring disease. We set out to establish intestinal Map infection by injecting bacteria directly into the proposed site of natural infection in young calves, and we thus designed the experiment to test a range of appropriate inoculation dosages. Our overall study goals were twofold. First, we used bacteriology and histopathology to determine the optimal Map dosage that results in intestinal colonization after direct inoculation. Second, we evaluated key T-helper cytokine expression patterns (interferon-γ [IFNγ] and interleukin-4 [IL-4]) both locally and systemically to characterize the progression of local and systemic immune responses in infected calves.
Methods
Experimental Design and Animals
The experimental design is outlined in Table 1 . Briefly, 40 five-week-old castrated male dairy breed calves (Holstein, Jersey, and Brown Swiss) were acquired for the study from various local private dairy herds. All herds were actively participating in the voluntary Johne’s disease certification program and had not had positive cases for at least 2 years. Animals were housed within the biosafety level 2 large animal housing facilities on the campus of Fort Dodge Animal Health in Fort Dodge, Iowa, for the duration of this study. Calves were housed in groups of 7 to 9 animals based on randomly assigned Map dosage. Control calves were housed separate from infected calves and their feces. Live Map was prepared in colony-forming units (CFU) per dose, including 103 (n = 7), 105 (n = 7), 107 (n = 9), 109 (n = 9), and uninfected negative control (n = 8). Each group was separately housed, but for logistical purposes the 2 low-dose groups (103 and 105 CFU) were infected simultaneously and the 2 high-dose groups (107 and 109 CFU) were infected simultaneously. For each group, individual calves were randomly selected for euthanasia at the following times: postinfection day (PID) 7 (n = 1 or 2), PID 30 (n = 2), PID 60 (n = 2), and PID 90 (n = 2 or 3). A single uninfected control animal was euthanatized at each time point for low-dose and high-dose groups. All live animal protocols were approved prior to the experiment by the internal Institutional Committee on Animal Care and Use at the Fort Dodge Animal Health Company.
Experimental Design
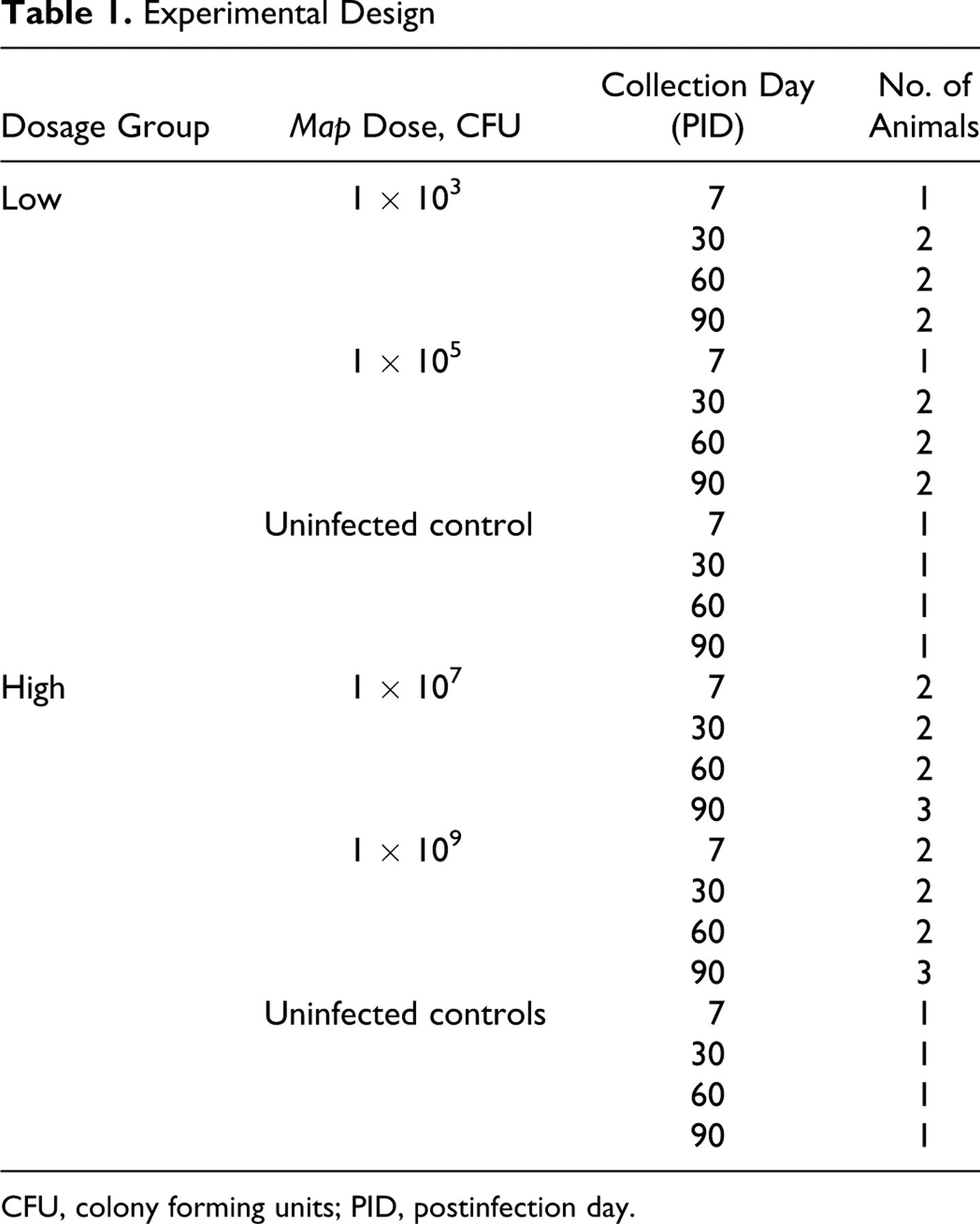
CFU, colony forming units; PID, postinfection day.
Bacterial Inoculum Preparation
The Map strain K10 was obtained from the American Type Culture Collection (Manassas, VA) and maintained in Middlebrook 7H9 broth supplemented with mycobactin J. Logarithmic growth-phase bacteria were washed and suspended in sterile saline. Bacterial concentration was determined by measuring absorbance at 540 nm and comparing the optical density to a standard curve generated in our laboratory. By dilution or concentration, inocula were adjusted to the appropriate final concentration of 103 to 109 CFU/250 μL in 0.9% sterile saline solution. The inoculum used in these studies was shown to have greater than 90% viability using fluorescein diacetate staining with analysis by flow cytometry prior to inoculation. Challenge inocula were confirmed to be acid fast by Ziehl-Nielson (ZN) staining and negative for contaminants by streaking onto sheep blood agar plates 24 hours prior to injection.
Animal Inoculation
Animals were inoculated via direct subserosal injection into the Peyer’s patch regions in the distal ileum adjacent to the ileocecal valve. Briefly, a right-sided approach to the ileocecal valve was used through a 4- to 5-cm vertical incision in the paralumbar fossa using local anesthesia in standing calves. Once access to the peritoneum was achieved, the cecum and distal ileum were isolated and exteriorized. The antimesenteric Peyer’s patches of the distal ileum were visualized and 250 μL containing the appropriate dosage of live Map K10 was injected. The intestine was then replaced into the abdominal cavity and the body wall was sutured using a standard 3-layer closure. Sterile instrumentation and surgical techniques were used, and perioperative systemic antibiotics were not administered.
Bacterial Fecal and Tissue Cultures
Following inoculation, fecal samples were collected per rectum from each calf (PID 1–2) and then weekly for the duration of the study. Fecal samples and tissue samples collected at necropsy were cultured using the BACTEC MGIT 960 system (BD Biosciences, Sparks, MD). All samples positive by the BACTEC culture system were subsequently subjected to PCR for confirmation by identification of the unique Map sequence IS900. Within the text of this manuscript, culture-positive status is only used to indicate fecal or tissue samples that were positive by both the BACTEC culture system and confirmatory PCR testing.
Polymerase Chain Reaction
Tissue and fecal samples identified as positive using BACTEC liquid culture system were verified as Map using real-time PCR to detect the Map-specific sequence IS900. Genomic DNA was extracted from egg yolk media-grown bacterial cultures using a beadbeater (4,500 rpm for 5 minutes), high-speed centrifugation (16,000 × g for 10 minutes), followed by alcohol precipitation of the bacterial nucleic acid. Commercially available oligonucleotide primers were derived from the IS900 DNA insertion sequence unique to Map (forward 5′-CCGCTAAATGAGAGATGGGATTGG-3′ and reverse 3′-ATTCAACTCCAGCAGCGCGGCCTCG-3′, Integrated DNA Technologies, Coralville, IA). A commercially available IS900 fluorescent probe (Integrated DNA Technologies) was used for detection.
Tissue and Sample Collection
At each time point small groups of 5 to 6 calves were humanely euthanatized, and the following tissue samples were collected during a complete necropsy examination: ileocecal valve, ileocecal lymph node (ICLN), mesenteric lymph node, prescapular lymph node, liver, spleen, and ileum. All tissues were aseptically collected and placed either into sterile containers for mycobacterial culture or into 10% neutral buffered formalin for histopathologic evaluation. An additional 4- to 5-g section of ileocecal lymph node, mesenteric lymph node, and prescapular lymph node was placed into sterile phosphate-buffered saline (PBS) with antibiotic and antimycotic agents (penicillin, streptomycin, amphotericin B) and transported to the laboratory for lymph node cell collection, cell culture, and flow cytometric evaluation. Thirty milliliters of peripheral blood from each calf was obtained by jugular venipuncture and aseptically collected into 2× acid citrate dextrose anticoagulant for peripheral blood mononuclear cell (PBMC) culture, antigen stimulation, and flow cytometric analysis.
Histopathology and Acid-Fast Staining
Formalin-fixed, paraffin-embedded tissues were sectioned (4 μm) and stained with hematoxylin and eosin (HE) for histopathologic evaluation and ZN acid fast staining per standard laboratory protocols. All tissue sections were examined thoroughly for evidence of mycobacterial infection by identification of appropriate lesions (granulomatous inflammation) and presence of acid-fast bacteria.
Tissue Processing for Flow Cytometry
For PBMCs, the buffy coat was separated from whole blood, the remaining erythrocytes were lysed, and the PBMCs were washed with PBS as previously described. 14 For lymph node-derived lymphocytes, fresh ICLN sections were minced, filtered through 7-μm mesh filter, and washed twice with PBS. All cells were counted using a Becton-Dickinson Coulter counter (Becton-Dickinson, Franklin Lakes, NJ), and diluted to 5 × 106 cells/ml in culture medium.
Multi parameter Flow Cytometry
PBMCs and lymphocytes from ICLN of each animal were activated as previously described, 13 with minor alterations using Map K-10 whole-cell sonicate (WCS, 20 μg/ml final dilution) as recall antigen. Six fluorophores were used to simultaneously identify T-lymphocyte subsets (CD4, CD8, and γδ), the activation marker CD25 (IL-2α receptor), and the intracellular cytokines IFNγ and IL-4 as previously described. 14
Flow Cytometric Data Collection and Analysis
Flow cytometric data were collected using a FACSCanto flow cytometer (Becton Dickinson Biosciences, San Jose, CA), and FlowJo cell analysis software (Tree Star Inc., San Carlos, CA) was used to analyze data. Expression index values were calculated to reflect the percentage gated positive events and mean fluorescence intensity for each parameter from antigen-stimulated samples as described earlier. 14 Index values for each parameter were then normalized to stimulated cells of the same tissue harvested from noninfected (negative control) calves at the same time point. Using normalized values, Map-specific immune response locally (ICLN) was compared with the systemic response (PBMC) at each time by comparing expression of CD25 and cytokine (IFNγ and IL-4) production by T-cell subsets (CD4, CD8, and γδ).
Statistical Analysis
Statistical analysis was performed using JMP 8.0 (SAS Institute, Cary, NC). Data are presented as the mean values ± standard error of the mean. Student’s t-test and 1-way analysis of variance were used for the statistical analysis. Group mean differences were considered significant if the P value was <.05 and highly significant if the P value was <.01. Because multiparameter flow cytometry data from low- and high-dose groups were collected on different days, it was not statistically valid to compare the 2 groups to each other given significant daily variation inherent in these assays. A single noninfected control animal was analyzed for each Map dosage group each day, so values for infected versus noninfected animals could not be statistically compared. Instead, animals were grouped as low dose (103 and 105 CFU) and high dose (107 and 109 CFU), and the data were normalized by subtracting values for the control animals from the values for the infected animals prior to statistical analysis. We then compared local (ICLN) to systemic (PBMC) cellular immune responses within each T-cell subset and dosage group each day after Map infection.
Results
Postsurgical Health Status
Calves demonstrated minimal postsurgical effects. Incisions healed normally and minimal soft tissue swelling of the incision site occurred in some calves, but these tissue changes resolved during healing. No calf developed clinical signs of disease attributable to intestinal Map infection (diarrhea, weight loss, or inappetence) during the study.
Gross Pathology
Gross lesions uncommonly observed during the necropsy procedures were limited to fibrous adhesions between the omentum and the body wall at the surgical site.
Histopathology
We examined ileocecal valve, ileum, spleen, liver, and lymph nodes from control and infected calves at each time point to identify significant microscopic pathology. Tissue histopathology results are summarized by inoculation dose group (low dose = 103 and 105 CFU and high dose = 107 and 109 CFU) and by day (PID 7–90). Significant histopathologic lesions were not identified in uninfected or low-dose calves at any time point during the study. Mild lymphofollicular hyperplasia of ileal Peyer’s patches and lymph nodes as well as multifocal ileocecal crypt abscesses were present; however, Map was not identified by culture or acid-fast staining in these calves.
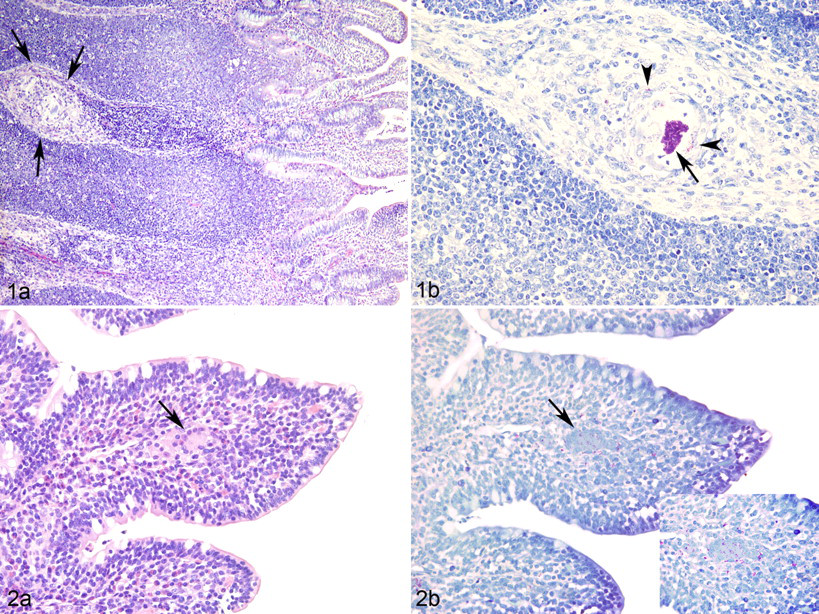
In contrast, the high-dose group calves had significant morphologic changes. High-dose calves had consistent granulomatous lesions in which acid-fast organisms were identified in the intestinal mucosa and draining lymph node. As early as PID 7, intestinal tissues harvested from high-dose calves had mucosal and submucosal macrophage aggregates containing acid-fast bacteria. In calf No. 917, a focal submucosal pyogranuloma with central necrosis and large numbers of intracellular and extracellular acid-fast bacteria occurred at the site of Map injection. At PID 7, multifocal and coalescing inflammatory cellular aggregates composed of macrophages, epithelioid macrophages, and occasional Langhans-type multinucleated giant cells were present within the intestinal submucosa (Fig. 1a ) and ZN staining showed intralesional acid-fast bacteria (Fig. 1b). By PID 60, inflammatory cells also extended into the lamina propria of villous tips of infected animals (Fig. 2a ), and ZN staining revealed myriad intracellular and extracellular acid-fast positive short bacterial rods in these areas (Fig. 2b). Low numbers of intracytoplasmic acid-fast organisms were also identified within macrophages in the draining (ileocecal) lymph node at PID 7, 30, and 90 (not shown).
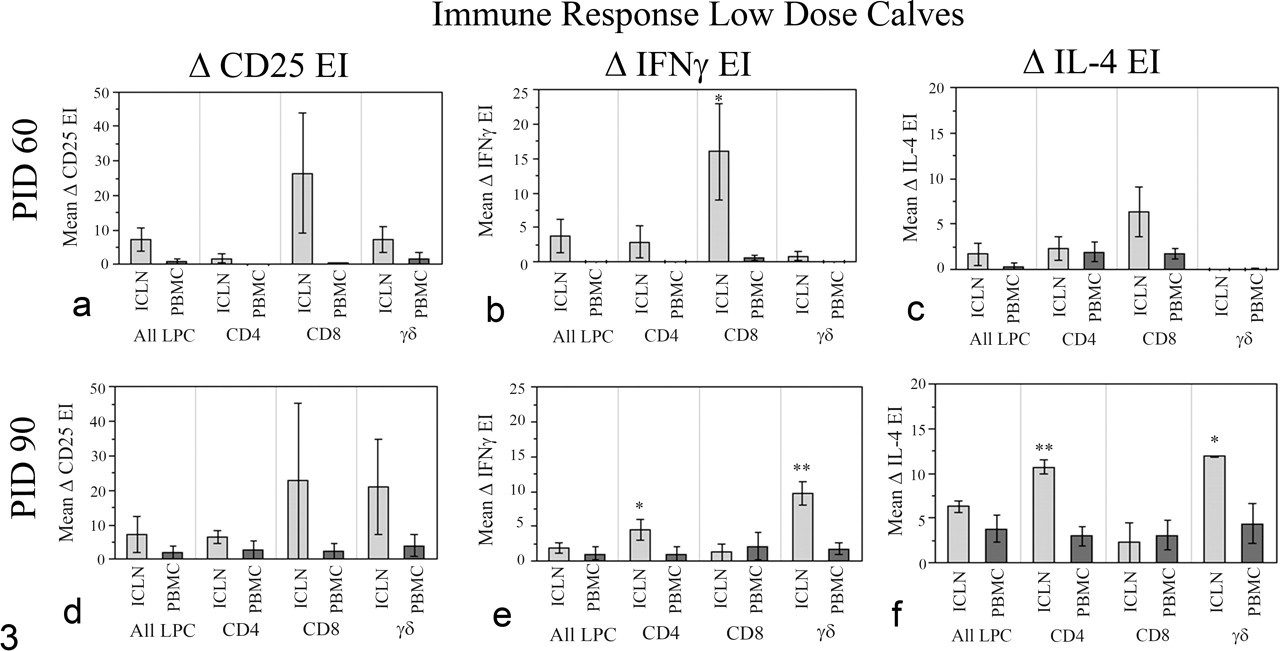
Immune responses, low-dose Map-infected calves. Flow cytometric detection of local (ICLN) versus systemic (PBMC) immune responses in low-dose calves is shown. Net expression indices (mean ± standard error of mean) of CD25, IFNγ, and IL-4 of ICLN and PBMCs from the low-dose Map-infected calves are shown for each T-cell subset. EI, expression index; ICLN, ileocecal lymph node; IFNγ, interferon-γ; IL, interleukin; PBMCs, peripheral blood mononuclear cells; LPCs, lymphocytes. Significantly higher responses within the same T-cell subset are indicated, *P < .05 and **P < .01.
Bacteriology
We collected a complete set of tissues during necropsy and collected feces weekly from each calf for Map culture and PCR to determine tissue colonization and fecal shedding. Map culture was performed using the BACTEC system with IS900 PCR as a confirmatory test. Tissue culture results were grouped by inoculation dose (low dose = 103 and 105 CFU and high dose = 107 and 109 CFU) and by time after inoculation (PID 7–90); the data are summarized in Table 2 . All tissue and fecal samples from noninfected control calves were Map culture negative. Map cultures from low-dose calf tissues were sporadically positive, whereas fecal cultures from this group were consistently negative throughout the study. In contrast to findings in low-dose calves, live Map was detected in 1 or more tissues harvested at necropsy from all high-dose calves (18/18). A majority of calves in the high-dose group had multiple tissues culture positive for Map (13/18). Of high-dose calf tissues, the ileocecal lymph node (16/18) and ileocecal valve (14/18) were most frequently Map culture positive, whereas the prescapular lymph node (4/18) and spleen (4/18) were less frequently Map culture positive. High-dose calves also displayed intermittent Map shedding in fecal samples. We recognize that a small percentage of the inoculum may have been inadvertently deposited into the intestinal lumen or the peritoneal cavity during intestinal Map inoculation. As a result, culture-positive feces acquired from experimentally infected animals during the first 2 days after inoculation (6/32) were interpreted to represent inoculum flow-through rather than active fecal shedding and were thus excluded from the final analysis.
Tissue and Fecal Culture Summary
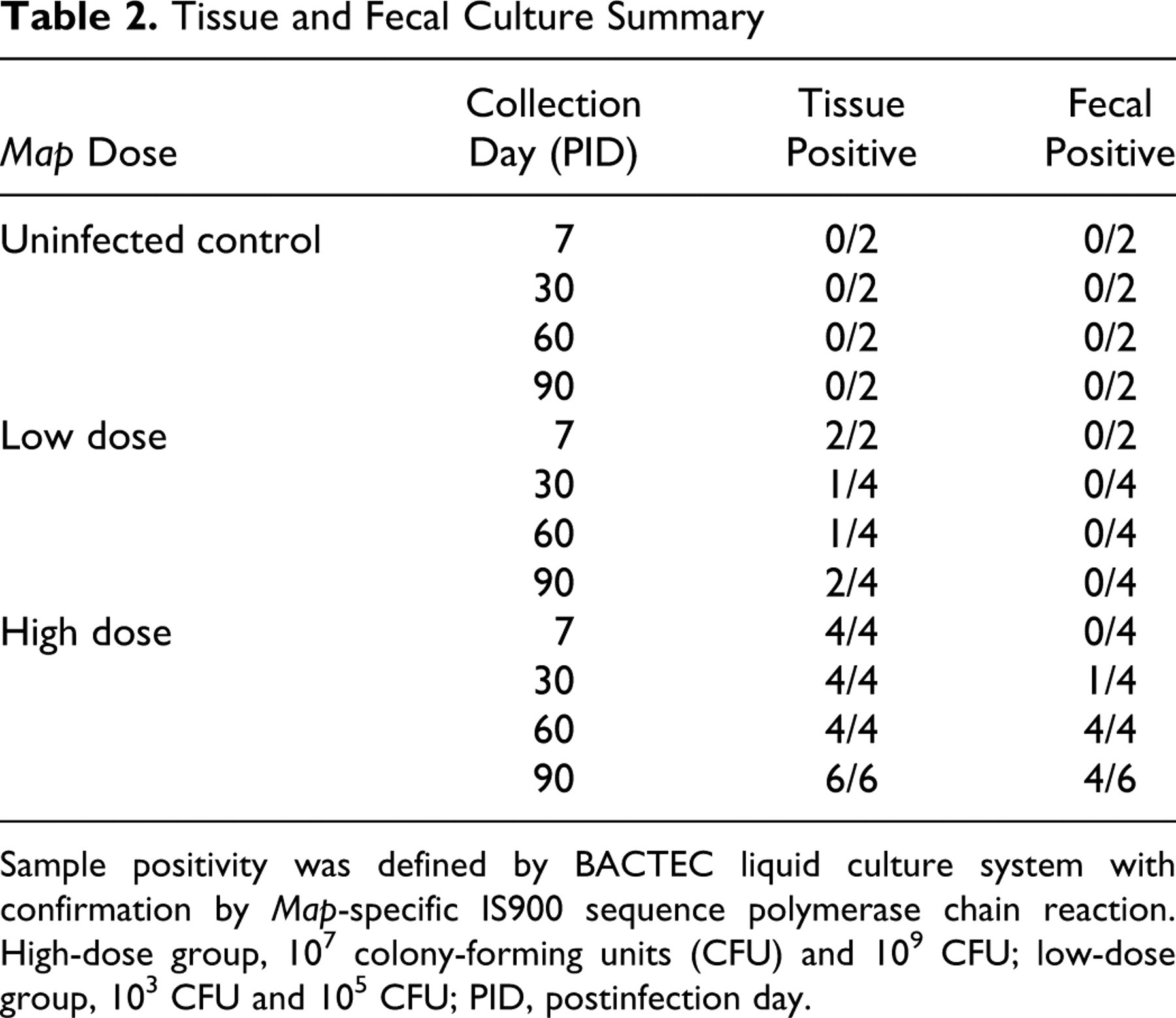
Sample positivity was defined by BACTEC liquid culture system with confirmation by Map-specific IS900 sequence polymerase chain reaction.
High-dose group, 107 colony-forming units (CFU) and 109 CFU; low-dose group, 103 CFU and 105 CFU; PID, postinfection day.
Map-Specific Cell-Mediated Immune Responses (Multiparameter Flow Cytometry)
The mean percentages of lymphocyte subsets (CD4, CD8, γδ, and non–T cells) within unstimulated PBMCs and ICLN cells from uninfected and Map-infected calves are shown in Table 3 . The lymphocyte subset profiles of each tissue at 6 days incubation were similar between infected and noninfected animals. The major difference was that the γδ T-cell population was high in PBMCs but low in ICLN. To evaluate Map-specific immune responses, we used immunofluorescence and flow cytometry to compare lymphocyte activation and cytokine secretion from PBMCs (systemic) and ICLN cells (local) during recall assays. Flow cytometry data were grouped by challenge dose (low, high) and evaluated within each day (PID 30–90). In low-dose calves, significant differences between local and systemic immune responses were limited to PID 60 and PID 90 (Fig. 3 ). Compared with PBMCs, lymphocyte subsets from ICLN generated significantly greater amounts of Map-specific cytokines IFNγ (CD8 T cells at PID 60, Fig 3b; CD4 and γδ T cells at PID 90, Fig. 3e) and IL-4 (CD4 and γδ T cells at PID 90, Fig. 3f). Overall, in low-dose calves, systemic immune responses to intestinal Map infection detected in PBMCs remained significantly lower than local immune responses detected in ICLN throughout the study.
Percentage Cell Types in Lymph Nodes or Peripheral Blood
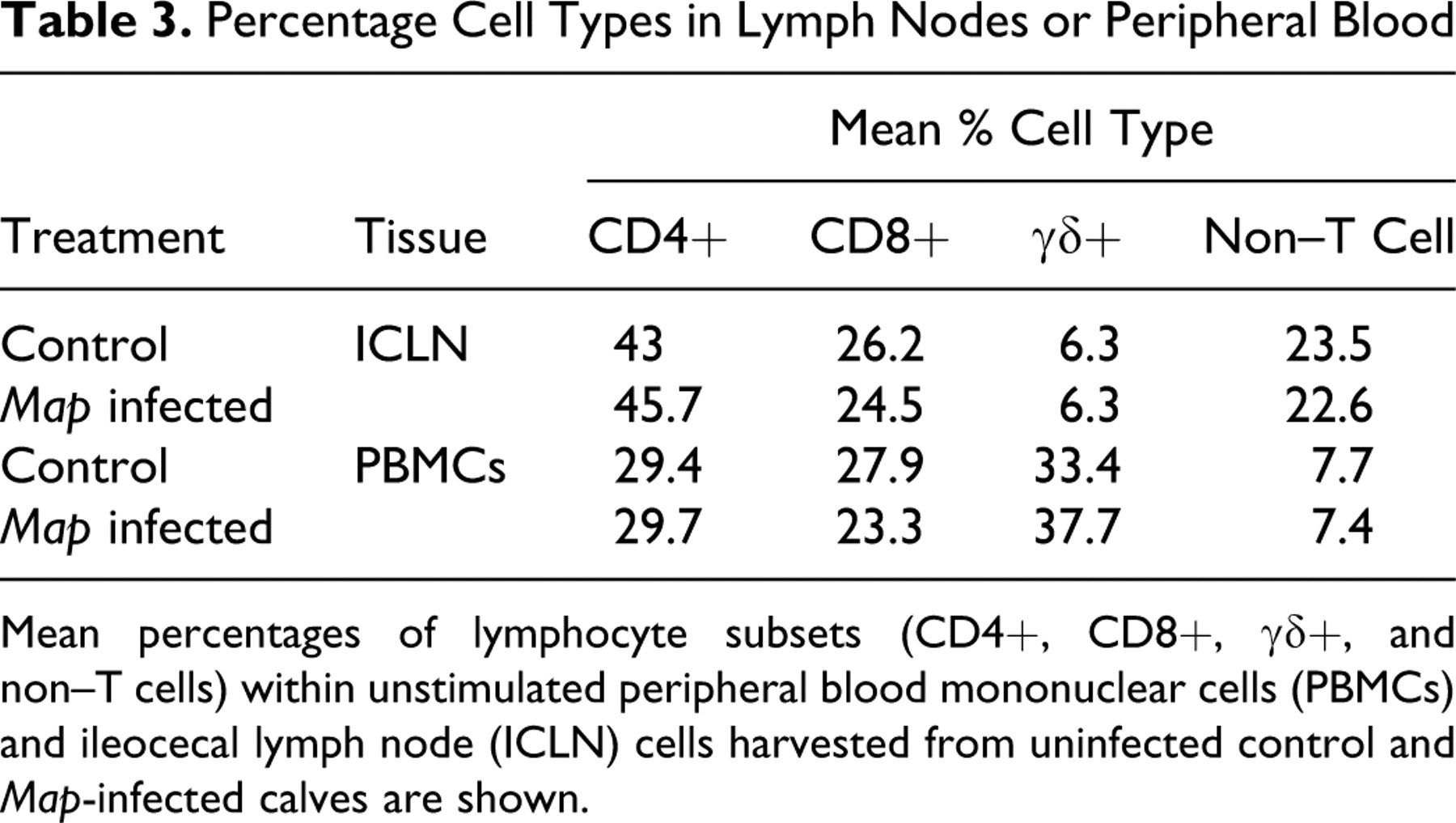
Mean percentages of lymphocyte subsets (CD4+, CD8+, γδ+, and non–T cells) within unstimulated peripheral blood mononuclear cells (PBMCs) and ileocecal lymph node (ICLN) cells harvested from uninfected control and Map-infected calves are shown.
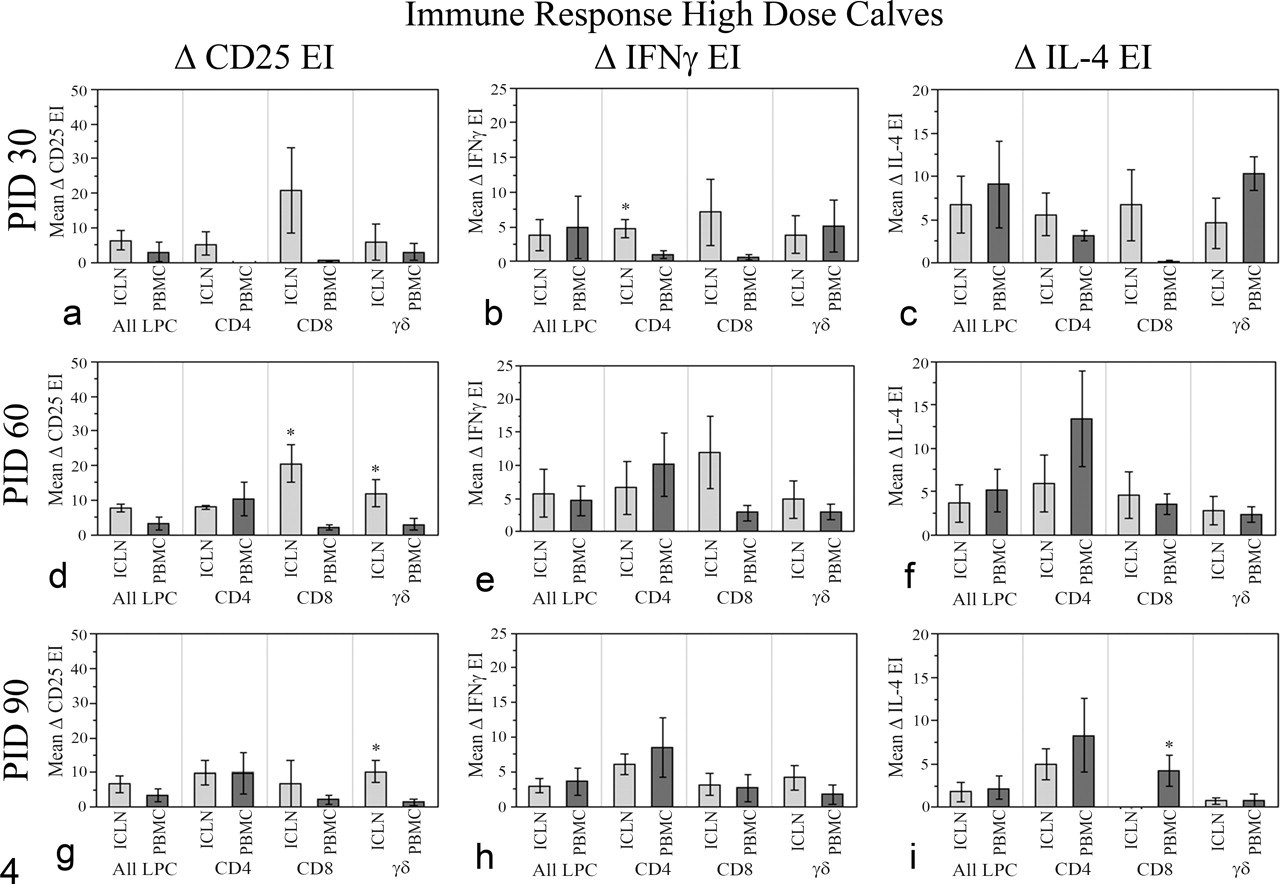
Immune responses, high-dose Map-infected calves. Flow cytometric detection of local (ICLN) versus systemic (PBMCs) immune responses in high-dose calves is shown. Net expression indices (mean ± standard error of mean) of CD25, IFNγ, and IL-4 of ICLN and PBMCs from the high-dose Map-infected calves are shown for each T-cell subset. EI, expression index; ICLN, ileocecal lymph node; IFNγ, interferon-γ; IL, interleukin; PBMCs, peripheral blood mononuclear cells; LPCs, lymphocytes. Significantly higher responses within the same T-cell subset are indicated, *P < .05.
Significant differences between the local and systemic immune responses were detected as early as PID30 in high-dose calves (Fig. 4 ). Increased generation of Map-specific IFNγ was detected in ICLN cells (CD4 T cells, Fig. 4b) at PID 30. Although not statistically significant, ICLN CD8 T cells tended to generate more IFNγ and IL-4 at PID 30 compared with PBMC CD8 T cells (Fig. 4b, c). Peripheral blood-derived CD4 T cells tended to generate elevated IFNγ and IL-4 at PID 60 and PID 90 compared with ICLN cells (Fig. 4e, f, h, i). At PID 90, peripheral blood CD8 T cells generated significantly more IL-4 than ICLN CD8 T cells (Fig. 4i). Overall, in high-dose calves (in contrast to low-dose animals), systemic responses to intestinal Map infection were detected in PBMCs during later time points (PID 60, PID 90) and did not remain significantly lower than local immune responses detected in ICLN.
Discussion and Conclusions
Difficulty in controlling Map-related disease in ruminants throughout the world is related to poor sensitivity and specificity of available diagnostic assays and the lack of completely protective vaccines. 12 Progress in improving diagnostic assays and vaccines is greatly hindered by challenges associated with the study of subclinical disease when infected animals lack clinical signs, typical intestinal pathology, and definitive cellular or humoral evidence of their infection status. 11,12 This gap in understanding the early immune response to Map infection in cattle underscores the need to develop models by which factors that shape the early response to Map and development of Map-associated disease can be clarified.
A wide range of oral dosages (103-2.5 × 1010 live Map organisms) has been used in ruminant model studies with variable success, 2,6 and Hines et al 6 recently suggested that 109 live Map organisms be administered on each of 2 consecutive days for experimental studies in cattle. We have used a novel approach to directly inoculate live Map into Peyer’s patches of the ileocecal valve to successfully induce intestinal Map infection in young calves in a dose-dependent manner. Unlike oral or intratonsillar infection models, 2,6,9 high-dose calves consistently and rapidly developed characteristic intestinal lesions with intralesional acid-fast mycobacteria throughout the mucosa, ileal Peyer’s patches, and draining lymph nodes. In addition, Map was cultured from tissues of high-dose animals, and these calves intermittently shed live Map in their feces. Importantly, the morphologic and bacteriologic features identified in high-dose calves in this study that mimic aspects of natural infection in the bovine have not been collectively demonstrated in an experimental Map intestinal infection model.
Dose-dependent responses (histology and bacteriology) are evident in our study. We were unable to histologically demonstrate intestinal infection in low-dose animals although up to 50% of these calves were tissue culture positive for Map at necropsy. Mild intestinal inflammation and lymphofollicular hyperplasia of Peyer’s patches and draining lymph nodes were observed in low-dose calves but were not significantly different from control calves.
The host immune response to Map infection has been studied locally during the clinical stages of Map infection and systemically using peripheral blood cells, 4,10,25 but few studies have evaluated the local response during early or subclinical infection. 1,7 The classic immunologic paradigm associated with mycobacterial disease is biphasic and characterized by an initial T-helper 1 (Th1) response. Effective Th1 polarization mediated by IFNγ is considered to be integral for controlling mycobacterial infections, 18 and detection of Map-specific IFNγ has been successfully used diagnostically during early stages of mycobacterial disease including Map infection in cattle. 21 Clinical Map infection in cattle is associated with a strong T-helper 2 (Th2) polarized response characterized by dominance of Th2 cytokines. IL-4 and IL-5 play roles in activation and proliferation of B lymphocytes and subsequent immunoglobulin production, which is prominent during clinical disease and serves as the basis for commercially available serum antibody diagnostic assays. The shift from Th1 to Th2 dominance of Map-associated disease has been well documented, yet the classic Th1/Th2 paradigm may not be completely accurate in the bovine, and based on the work of several laboratories, a more complex interplay seems to be a more likely scenario. 4,5
In the current study, distinct immunologic profiles were evident in the low- and high-dose groups. In general, Map-specific immune responses were strongest in the draining lymph nodes for the study duration in low-dose Map-infected calves. In the high-dose Map-infected animals, the local immune response dominated initially but diminished in relation to the systemic response as the study progressed. In both dose groups, we observed mixed Th1-like and Th2-like responses that were characterized by the production of both IFNγ and IL-4 from multiple lymphocyte subsets. This response was identified sooner following inoculation in the high-dose group. Interpretation of the immunologic data is limited because of animal numbers and statistical power in this proof of principal study. However, we are intrigued by the dose-dependent responses observed in the draining lymph node and systemic circulation in our system. Because they coincide temporally with tissue colonization, development of intestinal lesions, and active fecal shedding in high-dose but not low-dose calves, we believe that the data provide preliminary clues regarding the evolution of the immunopathologic response during early intestinal Map infection in calves. Given the complexity of host immune responses to mycobacterial pathogens, 3,5 our model affords a unique opportunity to analyze the interplay of specific cellular subsets and cytokines occurring at the infection site and systemically during the initial stages of intestinal infection.
Other models have been successfully developed to study intestinal Map infection in cattle, and these systems offer invaluable opportunities to study the immunopathogenesis of Map-associated disease. A ligated intestinal loop model with direct luminal Map inoculation has been used to elucidate mechanisms of initial intestinal invasion by Map and local host transcriptional events. 7,26 Use of this model appears limited to the very early stages (hours) of infection, and the procedure requires prolonged surgical anesthesia of experimental animals. Allen et al 1 describe the development of an ileal cannulation model in cattle to study the local and systemic immune response to Map in an oral challenge system. Rare Map-infected macrophages were observed in the intestinal propria within 24 hours after oral infection, and Map DNA was identified using PCR in sequentially biopsied intestinal tissues; however, significant differences in the activation status of intraepithelial lymphocytes of infected versus uninfected animals were not demonstrated in the study. 1 The characteristic Map-induced lesions, acid-fast organisms, and active fecal shedding of Map have not been observed in either the ligated intestinal loop or ileal cannulation models. Regardless, a clear advantage of intestinal loop, ileal cannulation, and oral infection models is the ability to potentially investigate interactions of live Map with intestinal epithelia and/or M cells, which are proposed to play a significant but incompletely explored role in the pathogenesis of natural infection. 15 A unique and significant advantage of the experimental system reported here is that we have described a method to accelerate the pathogenesis of intestinal Map infection via direct delivery of a known infectious dose to the ileocecal valve, the proposed natural infection site in young calves. An objective of the current study was to develop a model resulting in consistent intestinal Map infection, and although this was successfully achieved, we did not observe classic lesions or clinical signs of naturally occurring disease in cattle such as diffuse granulomatous enteritis leading to chronic diarrhea and emaciation. Future work focusing on the disease progression of intestinal Map infection using this model should be performed; however, a reliable model to study lesion development and immune responses during establishment of initial intestinal infection and progression of disease is important. Given the current challenges associated with efficacious vaccination and accurate diagnosis of the subclinically infected animal, use of this model’s unique features including granulomatous intestinal inflammation and intermittent fecal Map shedding is expected to prove useful for evaluation of novel vaccine candidates and/or diagnostic approaches in subclinically infected animals.
In conclusion, we have developed a reproducible model of intestinal Map infection in calves that shares important microbiologic, pathologic, and immunologic features of natural Map infection in cattle, all within a short time period. The model has significant potential for not only expanding our understanding of the pathogenesis of Map-induced disease but also for identifying factors that influence the development of protective immune responses to infection.
Footnotes
Acknowledgements
We thank Dr Shawn Rigby and Christine Deal of the ISU Cell Facility for assistance with flow cytometry data collection. We also thank Dr Norman Wonderlich, the veterinary staff, and the many animal caretakers of the Fort Dodge Animal Health company for expert animal care and assistance during surgical and necropsy procedures.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The study was funded by Fort Dodge Animal Health Company, Fort Dodge, IA. Yu-Wei Chiang and Jean Zylstra were employees of Fort Dodge Animal Health Company, Fort Dodge IA during the study.
