Abstract
Bighorn sheep sinus tumors are a recently described disease affecting the paranasal sinuses of Rocky Mountain bighorn sheep (Ovis canadensis canadensis). Several features of this disease suggest an infectious cause, although a specific etiologic agent has not been identified. To test the hypothesis that bighorn sheep sinus tumors are caused by an infectious agent, we inoculated 4 bighorn sheep lambs and 4 domestic sheep lambs intranasally with a cell-free filtrate derived from a naturally occurring bighorn sheep sinus tumor; we held 1 individual of each species as a control. Within 18 months after inoculation, all 4 inoculated domestic sheep (100%) and 1 of the 4 inoculated bighorn sheep (25%) developed tumors within the ethmoid sinuses or nasal conchae, with features similar to naturally occurring bighorn sheep sinus tumors. Neither of the uninoculated sheep developed tumors. Histologically, the experimentally transmitted tumors were composed of stellate to spindle cells embedded within a myxoid matrix, with marked bone production. Tumor cells stained positively with vimentin, S100, alpha smooth muscle actin, and osteocalcin, suggesting origin from a multipotent mesenchymal cell. A periosteal origin for these tumors is suspected. Immunohistochemical staining for the envelope protein of JSRV (with cross-reactivity to ENTV) was equivocal, and PCR assays specific for these agents were negative.
Bighorn sheep sinus tumors have recently been described within the paranasal sinuses of Rocky Mountain bighorn sheep. 2 –4 Lesions range from a thickening of the sinus lining to solid, gelatinous masses filling the sinus cavities. 4 Cystic expansions of the sinus lining are commonly associated with these masses 2,3 although it is unknown if cystic structures and tumor formation are part of the same disease process.
Histologically, bighorn sheep sinus tumors are reported to contain proliferative epithelial and stromal cells, with both populations ranging from hyperplasia to neoplasia. 4 The vast majority of cases are predominated by benign-appearing spindle cells that are diffusely distributed within the submucosa of the sinus lining, and embedded within a myxoid matrix. Lymphoplasmacytic inflammation is often prominent. 3,4 These inflamed fibromyxomatous masses often invade and destroy the underlying bone, 4 and cell cultures of affected tissues demonstrate features of neoplasia including a lack of contact inhibition. 2 Metastasis has not been observed. 4
The original description of bighorn sheep sinus tumors was based on a small sample size of cases (n = 10). 4 Following publication of these 10 index cases, at least 30 additional paranasal sinus tumors have been identified in bighorn sheep and examination of these cases has provided further insight to pathogenesis. For example, additional cases have demonstrated the variable occurrence of bone production, suggesting a possible origin from bone or periosteum (Fox, unpublished data). Additional cases have also demonstrated less robust epithelial proliferation than described for the index cases, suggesting that epithelial proliferation may be less of a defining feature than previously thought and may possibly represent a secondary lesion (Fox, unpublished data). Geographic clustering of cases has been restricted to discrete herds of sheep, supporting the hypothesis for an infectious etiology. 2
Due to similarities between bighorn sheep sinus tumors and oncogenic retroviral diseases of domestic sheep and goats, these tumors were originally hypothesized to be caused by enzootic nasal tumor virus (ENTV), Jaagsiekte sheep retrovirus (JSRV), or a closely related oncogenic retrovirus. However, initial screens of naturally occurring bighorn sheep sinus tumors were negative for these viruses by PCR and IHC 4 and virus particles have not been detected by electron microscopy in nasal secretions or cell cultures of sinus tumor tissues. 2
Although a specific etiology had not been identified for bighorn sheep sinus tumors, we suspected involvement of an infectious agent. We attempted to reproduce the disease through experimental infection, providing an opportunity for further characterization of this disease.
Materials and Methods
Animals and Facilities
A total of 10 animals were used for this study, including 5 Rocky Mountain bighorn sheep and 5 domestic sheep (Ovis aries, 5 male). The bighorn sheep (4 female, 1 male) were born at the Colorado Division of (Parks and) Wildlife, Foothills Wildlife Research Facility (FWRF) in Fort Collins, Colorado, USA and descended from wild stock originating in Colorado and Wyoming, USA. The domestic sheep (5 male) were a mixed breed of Dorset, East Friesian, and Lacaune breeds, and were acquired from a sheep dairy herd in Bushnell, Nebraska, USA. All experimental animals were housed at the FWRF throughout the study.
Lambs were allowed to suckle colostrum from their dams, but were separated from dams within 48 hours of birth. On arrival at the FWRF, domestic sheep were treated with a broad-spectrum antibiotic for respiratory bacterial pathogens (Draxxin, Zoetis, Florham Park, NJ). Both species were bottle raised using a commercial lamb milk replacer (domestic sheep) or evaporated milk (bighorn sheep) until approximately 8 weeks of age, when animals were transitioned to a mixture of grass and/or alfalfa hay and a pelleted diet specific to the dietary needs of each species.
Domestic and bighorn sheep were housed approximately 100 m apart, with separation by 2.5 meter solid wooden fencing. For each species, lambs were separated into groups of treatment (n = 4) and control (n = 1) animals. Treatment animals were housed together, but treatment and control pens were separated by at least 2 meters and wire double fencing. Biosecurity protocols were approved by the Colorado State University Institutional Biosafety Committee at biosafety level 2 (IBC 10-034B) and protocols for animal use were approved by both the Colorado State University and Colorado Division of Parks and Wildlife Animal Care and Use Committees (CSU 10-1783A, CPW 07-2010).
Preparation of Inoculum
Tumor material and associated seromucinous exudates were collected postmortem from an adult female bighorn sheep with a naturally occurring sinus tumor, within 2 hours of death. The ewe was part of a captive herd for which sinus tumors are endemic but respiratory disease is otherwise not observed. This ewe died from causes unrelated to this study. At necropsy, a sinus tumor was identified unilaterally within the frontal sinus, with extension into the cornual sinus causing a deformity of the horn on that side (Supplemental Figure 1). Cystic expansions of the maxillary sinus lining were also noted (Supplemental Figure 2). Histopathology demonstrated typical features of bighorn sheep sinus tumors including proliferation of mature, uniform spindle cells within a myxedematous background in the submucosa (Supplemental Figure 3), and submucosal cystic structures lined by ciliated epithelial cells (Supplemental Figure 4). Tumor tissue and associated exudates were collected using sterile instruments, and homogenized in sterile phosphate-buffered saline using a Dounce homogenizer. The resulting homogenate was clarified by centrifugation at 8000 × g for 30 minutes, and the supernatant was passed through a 0.45 micron filter, creating a cell-free filtrate. The filtrate was divided into 1.75 ml aliquots, and frozen at –80°C until inoculation (within 8 weeks of preparation).
Inoculation of Animals
At approximately 48 hours of age, all treatment lambs were inoculated intranasally (unilaterally) with 1.75 ml of filtrate prepared as described above and thawed on ice. The bighorn lambs were inoculated in the right nasal cavity, and the domestic lambs were inoculated in the left nasal cavity. For each animal, the contralateral side was not inoculated to serve as a level of internal control.
The inoculum was administered using a rigid plastic nasal vaccine applicator, inserted through the nostril to approximately the level of the medial canthus. This level of the nasal cavity corresponds to the junction of the rostral ethmoid turbinates/middle nasal concha with the dorsal and ventral nasal conchae. The inoculum was administered into the nasal cavity of each lamb while the head was positioned parallel to the ground to avoid swallowing of the inoculum. Lambs were calm and breathing normally during inoculation. The inoculum was heard bubbling within the nasal cavity with the animals’ breaths. An identical procedure was used to administer sterile saline intranasally (unilaterally) to each of the negative control lambs.
Monitoring of Animals
All animals were clinically assessed at least every 2 weeks for any signs of respiratory distress, discomfort, or nasal exudate. Every 2 months for the first 8 months, all animals were assessed for sinus tumor formation using standard radiographic films. Due to low resolution of lesions by standard radiology, at 9 months postinoculation (mpi) all animals were assessed by computed tomography (CT, Gemini Time-of-Flight Big Bore PET/16 slice CT, Philips Healthcare, Andover, MA). Studies were acquired in transverse plane with 1 mm and 2 mm slice thickness in bone and standard algorithms. CT scans were repeated at the anticipated termination of the study, 18 mpi. For 3 bighorn sheep, we extended the study past 18 months, and CT was repeated at the termination of the study 22.5 mpi.
Postmortem Examinations and Histopathology
Sheep were euthanized and necropsied immediately following final screening by CT. At necropsy, tissues were collected including samples of the frontal and maxillary sinus linings, ethmoid turbinates, and nasal conchae, including any masses or other abnormal tissues. Tissues were collected in 10% neutral buffered formalin, and also frozen at –80°C for molecular diagnostics. After fixation in 10% neutral buffered formalin, samples containing bone or mineral were decalcified in 10% formic acid for 7–14 days prior to histologic processing. Selected fixed tissues were embedded in paraffin blocks, sectioned at 4 to 6 µm, and stained with hematoxylin and eosin.
Histochemical and Immunohistochemical Stains
For tumors identified in the nasal conchae or paranasal sinuses, additional staining was performed including Alcian blue (pH 2.5) stain, and periodic acid-Schiff (PAS) reaction, as well as the immunohistochemical (IHC) stains vimentin (PA0033, monoclonal, Leica Biosystems, Buffalo Grove, IL), S100 (PA0900, polyclonal Leica Biosystems), alpha smooth muscle actin (SMA, PA0943, monoclonal, Leica Biosystems), and osteocalcin (MA1-20786, monoclonal, 1:100, Thermo Scientific Pierce Antibodies, Rockford, IL). The above IHC staining was performed using a Leica BOND-MAX automated IHC stainer (Leica Biosystems). Samples that were positive for SMA were also stained for p63 (Indiana Animal Disease Diagnostic Laboratory) to investigate possible myoepithelial origin.
For the above immunohistochemical stains, positive and negative tissue controls (from normal bighorn sheep tonsil or normal bighorn nasal concha), as well as internal controls and a negative control with no primary antibody, were used to confirm specificity of staining (Supplemental Methods). Based on similarities of bighorn sheep sinus tumors to oncogenic retroviral diseases of domestic sheep and goats, additional IHC was performed to detect the envelope proteins of these viruses. Staining was performed as previously described 4 using a mouse monoclonal antibody against the surface (SU) domain of JSRV envelope (Env), 9 with demonstrated cross-reaction for ENTV Env. 8 Tumor tissue from a domestic sheep with ENTV was used as a positive control. Isotype controls were performed for each slide to evaluate nonspecific staining.
Polymerase Chain Reaction
Tissues from the upper respiratory sinuses were screened for the presence ENTV-1, ENTV-2, and JSRV, as well as the respiratory pathogens Mycoplasma ovipneumoniae and leukotoxigenic Pasteurellaceae using PCR methods as previously described. 3 –6 DNA extraction was performed using a commercially available extraction kit (QIAmp DNA mini kit, QIAGEN) per manufacturer’s instructions. PCR primers and cycling conditions are given in Supplemental Table 1.
Results
Clinical Assessment
Details of the findings are given in Supplemental Table 2. Mild nasal exudates were occasionally noted in animals throughout the study. The degree and character of exudate did not differ between treatment and control animals, and these findings were attributed to transient rhinitis within both treatment and control groups. There was no evidence of respiratory distress, discomfort, or other significant clinical findings throughout the duration of the study.
Computed Tomography and Gross Necropsy
Bighorn Sheep
For case No. 1, CT findings were normal at 9 mpi. At 18 mpi CT demonstrated a hyperattenuating nodule in the ethmoid turbinates on the right side, near the site of inoculation. The nodule was approximately 0.5 cm × 0.6 cm and filled 1 small-diameter ethmoid turbinate. The lesion had mixed soft tissue attenuation, with a hyperattenuating rim surrounding internal soft tissue/fluid attenuating material. In addition, hyperattenuating stippling was noted within the lesion, consistent with calcification or ossification in these areas (Supplemental Figure 5). Because of the small size of this nodule, case No. 1 was not euthanized at 18 mpi to assess for possible progression. Case No. 1 was reexamined by CT at 22.5 mpi and no progression of the lesion was noted. The animal was euthanized and necropsy findings confirmed observations made by CT. A soft tissue nodule with a gelatinous texture was identified near the site of inoculation (Supplemental Figure 6).
For case No. 2, at 9 mpi an approximately 0.5 cm diameter cystic expansion of the sinus lining was identified by CT in the maxillary sinus on the inoculated side. No significant progression was noted by CT at 18 mpi and case No. 2 was euthanized. Necropsy findings confirmed CT observations. The cystic structure within the maxillary sinus was filled with mucinous to gelatinous material, and was completely contained within the sinus lining. The sinus lining with the cystic structure could be peeled away from the underlying bone, and the exposed bone was remodeled, with apparent bone resorption beneath the cyst.
Case No. 3 showed no lesions by CT at 9 and 18 mpi and was euthanized. No lesions were noted in the upper respiratory tract at necropsy. Case No. 4 showed no lesions by CT at 9 and 18 mpi, but was given an extended incubation period to assess for possible lesion formation. At 22.5 mpi, no lesions were noted and case No. 4 was euthanized.
The uninoculated control bighorn sheep (case No. 5) showed no lesions by CT at 9, 18, and 22.5 mpi. Case no. 5 was euthanized at 22.5 mpi and no lesions were noted at gross necropsy.
Domestic Sheep
For case No. 6, at 9 mpi CT demonstrated a 1.6 cm × 1.9 cm nodule near the site of inoculation in the middle nasal concha on the left side. The nodule contained a rim of hyperattenuating tissue and a core of less attenuating tissue, with multifocal hyperattenuating stippling (Supplemental Figure 7), similar to findings for case No. 1. At 12 mpi, case No. 6 died for reasons unrelated to the study. Postmortem examination demonstrated a soft tissue mass partially filling the middle nasal concha on the left side, with multifocal ossification consistent with the CT interpretation (Supplemental Figure 8).
For case No. 7, at 9 mpi a 1.4 cm × 1.1 cm nodule was detected by CT in the ventral nasal concha, near the site of inoculation at the junction of the dorsal, ventral, and middle conchae. Features were similar to those described above for case Nos. 1 and 6. At 18 mpi, this lesion showed progression, with growth to 4.7 cm × 1.2 cm and increased mineralization/ossification (Figure 1). Gross necropsy confirmed CT findings (Figure 2).
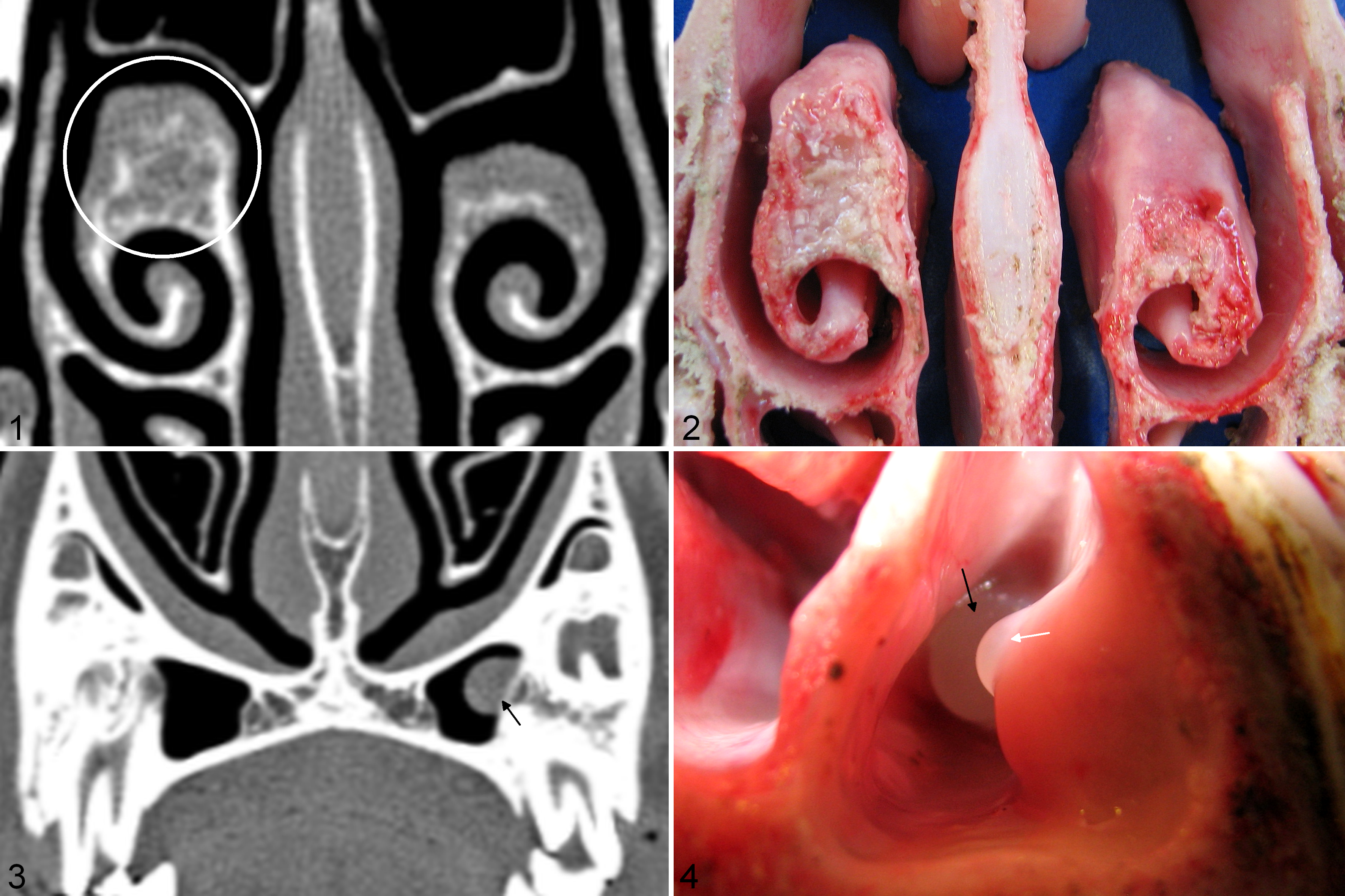
Experimentally induced nasal tumors. Domestic sheep.
For case No. 8, no lesions were seen by CT at 9 mpi. At 18 mpi, a hyperattenuating nodule was identified near the site of inoculation within the middle nasal concha near the junction with the ethmoid turbinates. The nodule measured 3.2 cm × 1.5 cm and features were similar to those described for case Nos. 1, 6, and 7 (Supplemental Figure 9). Gross necropsy confirmed CT findings (Supplemental Figure 10). In addition, a second, smaller (1.9 cm × 1.0 cm) nodule with similar features was detected by CT within the ventral nasal concha, rostral to the site of inoculation. This finding was also confirmed by gross necropsy.
For case No. 9 we did not detect a tumor by CT near the site of inoculation, but at 18 mpi we did detect a 2.0 cm × 0.75 cm nodule contralateral and rostral to the site of inoculation, within the ventral nasal concha (Supplemental Figure 11). Features were similar to those described for case Nos. 1, 6, 7, and 8. CT findings were confirmed by gross necropsy (Supplemental Figure 12). For case No. 9 we also detected 3 cystic expansions of the maxillary sinus lining (0.4 cm, 0.3 cm, and 0.7 cm diameter) on the uninoculated side by CT at 18 mpi (Figure 3), and one 0.8 cm diameter cystic expansion of the maxillary sinus lining on the inoculated side. Findings were confirmed at gross necropsy (Figure 4). Features of the cystic structures were similar to those described for case No. 2 with mucus-filled cysts confined to the sinus lining, and lysis of the subjacent bone. No lesions were identified by CT at 9 mpi or 18 mpi, or at gross necropsy in the uninoculated domestic sheep (case No. 5).
Histopathology
Histologically, the experimentally induced tumors in this study had characteristics similar to those observed in naturally occurring bighorn sheep sinus tumors. In each case, the proliferative cell population was composed of spindle to stellate cells embedded within a myxoid matrix (Figure 5a) that stained light blue with Alcian blue (pH 2.5) (Figure 5b) and lacked magenta staining by PAS reaction, consistent with acid mucopolysaccharides found within other myxomatous tumors. For most cases, tumors were predominated by spindle to stellate cells that surrounded bone spicules. Spindle cells lacked atypia, with uniform shape and size, and small hyperchromatic nuclei that lacked mitotic figures (Supplemental Table 2, Supplemental Figures 13–16).
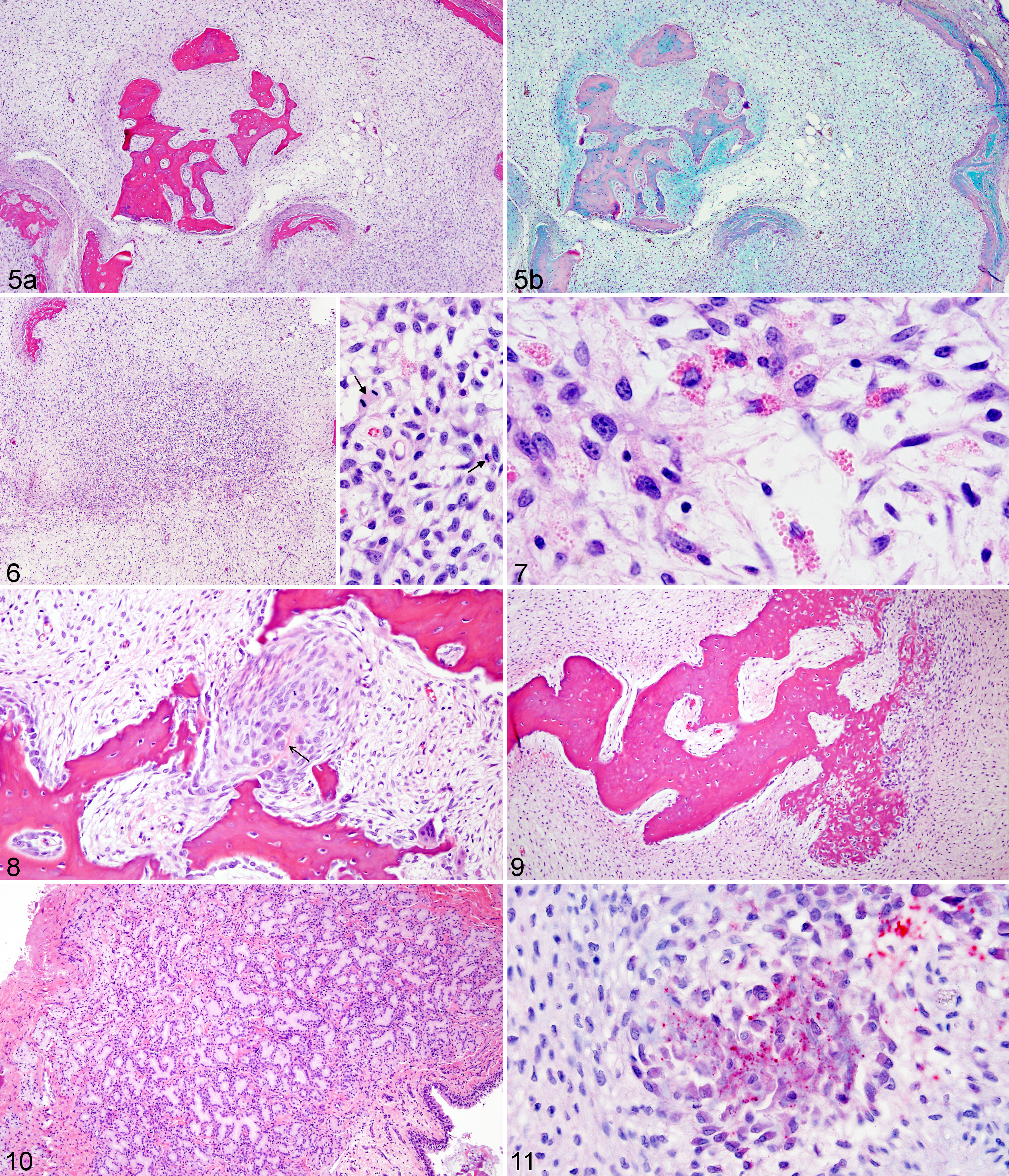
Experimentally transmitted tumors, nasal tissue, domestic sheep.
For case No. 7 similar cytologically bland spindle to stellate cells predominated, but rare foci within the mass demonstrated atypia including increased cellular density. These foci were composed of stellate cells with moderate anisocytosis and anisokaryosis, coarsely clumped chromatin with prominent nucleoli, and frequent mitotic figures (Figures 6, 7). Stellate cells within these atypical foci often contained intracytoplasmic round hyaline eosinophilic globules that were PAS negative (Figure 7).
One prominent finding in the experimentally transmitted tumors was marked production of bone, consistent with the pattern of hyperattenuating stippling seen on CT scans. Multiple progressive stages of bone formation were observed, suggesting orderly maturation. Nests of suspected osteoblastic cells were either clustered randomly within the stellate mesenchyme of the tumor, or clustered at the periosteal surfaces of bone spicules. These cells were oval with eccentric nuclei and amphophilic to basophilic cytoplasm. Occasionally, these clusters of cells contained small central streams of osteoid (Figure 8). Larger islands of bone formation were composed of woven bone with gradual differentiation to mature bone (Figure 9).
Epithelial involvement was limited, with hyperplasia of submucosal glands occurring in 2 cases (case Nos. 6 and 9). Hyperplastic glands were located adjacent to a cystic expansion of the maxillary sinus lining in 1 case (case No. 9) and adjacent to the site of tumor formation in another case (case No. 6). In both cases hyperplastic serous glands formed focal, unencapsulated, nodular expansions within the submucosa, with maintenance of acinar architecture and no evidence of anisocytosis, anisokaryosis or other cellular atypia. For case No. 9, hyperplastic glands were mildly dilated and filled with eosinophilic (proteinaceous) product (Figure 10). Cystic expansions of the maxillary sinus lining (case Nos. 2 and 9) were filled with mucous product and lined by ciliated cuboidal epithelial cells with scattered goblet cells, consistent with attenuated respiratory epithelium. Hyperplasia of surface epithelium was not observed, and inflammation was not a prominent finding. No lymphoplasmacytic inflammation was seen, and only mild neutrophilic inflammation was observed associated with submucosal gland hyperplasia in 1 case (case No. 9).
Immunohistochemistry
Immunohistochemical staining for osteocalcin demonstrated variable positive staining within the cytoplasm of suspected osteoblastic cells and associated extracellular matrix (Figure 11). In 1 case (case No. 8) rare clusters of spindle to stellate cells adjacent to bone spicules stained intensely positive for osteocalcin (Supplemental Figure 17).
The stellate to spindle stromal cells that predominated in the experimentally transmitted tumors were strongly positive for vimentin, SMA, and S100 (Figures 12a–c). Based on IHC positivity for SMA, we also evaluated these cases with IHC staining for p63 to investigate a possible myoepithelial cell origin for the tumors. 1 The proliferative cell populations were uniformly negative for p63. IHC staining for the envelope (Env) protein of JSRV (with cross-reactivity for ENTV) demonstrated punctate, granular staining of the cytoplasm of scattered mucus-producing surface epithelial cells in case Nos. 1, 6, 7, and 9. Although staining occurred only in sinus lining tissues from sheep that developed tumors, positive staining was observed only in surface epithelial cells and not within the proliferative mesenchymal cell populations.
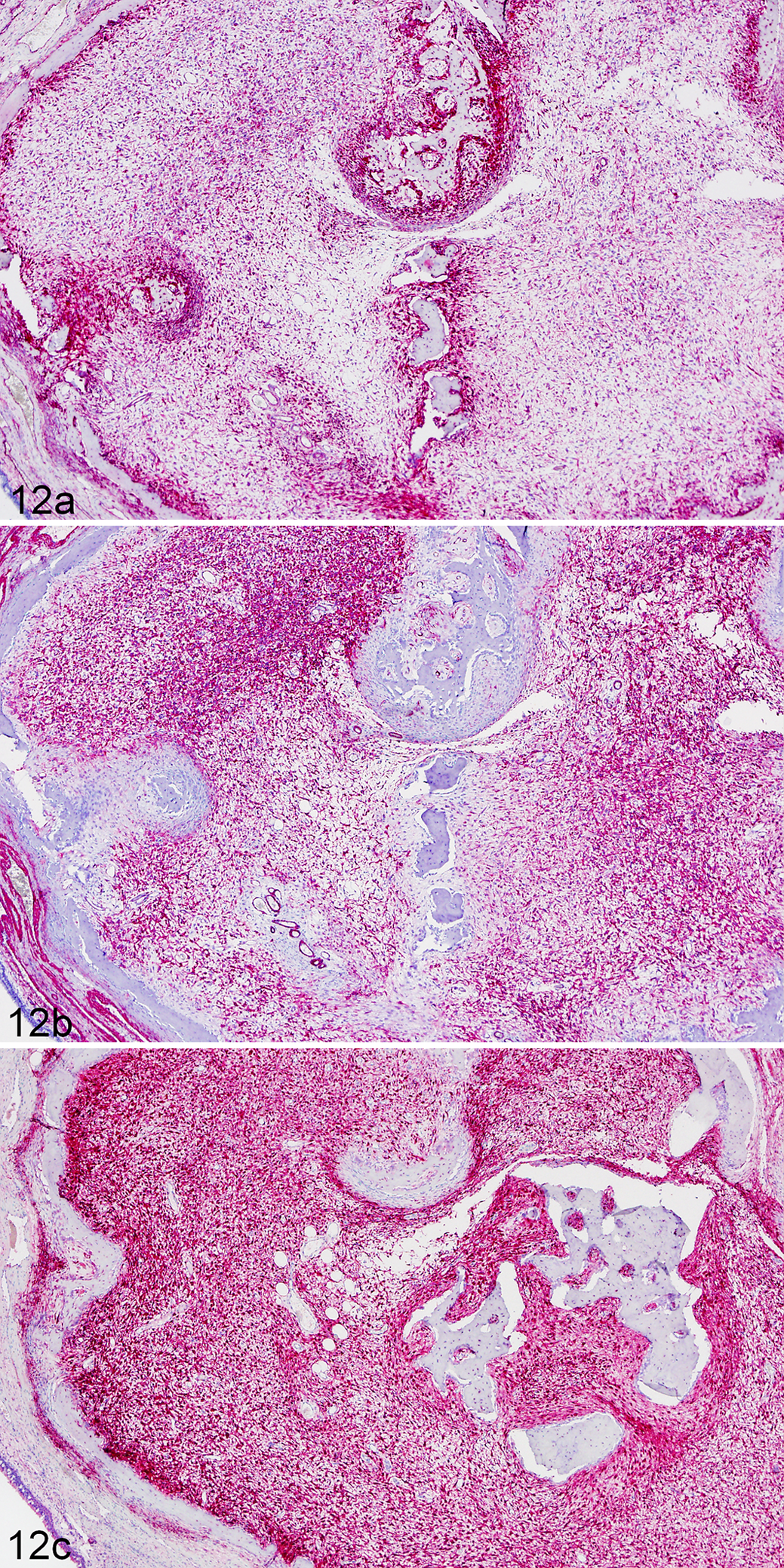
Experimentally induced tumor, nasal tissue, domestic sheep, case No. 7. Immunohistochemical staining demonstrates probable origin from multipotent mesenchyme, with positive staining for vimentin (a), alpha smooth muscle actin (b), and S100 (c).
PCR Results
The tissues used to prepare the inoculum, and all tissues from the affected and unaffected sinus linings of the experimental animals were PCR negative for ENTV-1, ENTV-2, JSRV, and the common ovine respiratory pathogens Mycoplasma ovipneumoniae and leukotoxigenic Pasteurellaceae. Tissues examined included maxillary sinus lining from each animal, as well as tumor material or unaffected ethmoid turbinates from each animal.
Discussion
Our results demonstrate transmission of sinus tumors to bighorn and domestic sheep. Only inoculated animals developed tumors, and in most instances the tumors formed specifically at the site of inoculation. Two animals developed tumors within the nasal conchae unassociated with the site of inoculation. In 1 of these cases (case No. 8), while a tumor did form at the site of inoculation, a second, smaller mass also formed rostral to this site, and may represent local spread of infection. For 1 case (case No. 2), no tumor developed at the site of inoculation, but tumor formation on the contralateral side may represent animal-to-animal transmission between the treatment animals, or spread of infectious challenge material through mechanisms such as aspiration and expectoration at the time of inoculation.
Skull radiography was uniformly insensitive for identifying sinus tumors within the nasal and paranasal sinuses of the sheep. However, CT identified all of the lesions accurately. The CT characteristics of the tumors were consistent with gross and histologic findings, and the CT features described here may be helpful as a reference when making a diagnosis of sinus tumors in living sheep.
Gross, histologic, histochemical, and immunohistochemical findings for the experimentally transmitted tumors in this study are not definitive for the diagnosis of a specific neoplastic or hyperplastic process. The tumors that developed demonstrated several features of myxoma or myxomatous fibroma (as previously reported for naturally occurring cases) including a gelatinous gross appearance, and histologic features of stellate to spindle cells embedded within a myxoid matrix. Positive IHC staining for vimentin, S100, SMA, and osteocalcin suggested a multipotent mesenchymal cell origin, and we suspect that proliferative cells may originate from the primitive mesenchyme of the periosteum.
The production of bone matrix was a prominent finding in the experimentally transmitted cases described here. This finding helped to interpret the occasional observation of prominent bone production in naturally occurring bighorn sheep sinus tumors (Supplemental Figure 18), and to identify bone production as a feature of bighorn sheep sinus tumors in addition to those previously described. Based on the presence of bone production, differential diagnoses should include tumors of bone, such as parosteal osteosarcoma. A diagnosis of mesenchymoma may also be appropriate as more than 1 mesenchymal cell type is present within the masses.
Although several of the experimentally induced masses did demonstrate bone invasion, and 1 of the masses possessed cellular features suggestive of neoplasia (poor cellular differentiation, anisocytosis, anisokaryosis, and increased mitotic figures), the orderly proliferation of cells in most cases may suggest a nonneoplastic process. Several features of the masses including cytologically bland spindle cells, thin woven bone trabeculae or fragments, and presence of myxoid stroma or myxoid change are comparable to proliferative lesions, such as fibrous dysplasia, for which the classification of neoplastic or nonneoplastic is undefined. 7
The mucus-filled, epithelial-lined cystic expansions of the maxillary sinus lining seen in 1 bighorn sheep and 1 domestic sheep in this experiment have been seen in association with naturally occurring bighorn sheep sinus tumors, but it remains uncertain if tumors and cysts are part of the same disease process. In this experiment, the locations of the cysts were not consistent with the locations at which the sheep were inoculated, but cysts developed only in inoculated animals. Discovery of the specific etiology for this disease will help to further define whether these cystic structures are nonspecific findings or are related to tumor development.
Because naturally occurring sinus tumors are often markedly invaded by chronic inflammatory cells, we considered chronic inflammation from bacterial infections (particularly Mycoplasma ovipneumoniae) as a possible factor driving tumor formation. Our successful transmission of tumors using an inoculum that excluded bacteria, the lack of inflammation in the experimentally transmitted tumors, and PCR-negative results for common respiratory pathogens of bighorn sheep suggest that bighorn sheep sinus tumors are not caused by bacterial infections leading to chronic inflammation. Although a bacterial cause for sinus tumors cannot be entirely ruled out, we suspect that bacterial infections seen in naturally occurring cases of bighorn sheep sinus tumors are a secondary process, due to decreased clearance of agents by the altered sinus lining. 3
Marked epithelial hyperplasia and rare epithelial neoplasia previously described for naturally occurring cases of bighorn sheep sinus tumors were not observed in the experimentally transmitted tumors. Epithelial proliferation was limited to mild submucosal gland hyperplasia in 2 cases, and no proliferation of surface epithelial cells was observed. We suspect that proliferation of epithelial cells, particularly surface epithelial cells, in naturally occurring cases may in part be secondary to chronic inflammation.
We continue to pursue retroviruses among other causes for bighorn sheep sinus tumors, although IHC staining for the Env protein of JSRV/ENTV was equivocal in the animals that developed tumors and specific PCR assays designed to detect these agents were negative. If bighorn sheep sinus tumors are caused by a retrovirus, more rigorous sequence analysis will be necessary to detect novel retroviral sequences, and to discriminate between exogenous and endogenous sequences.
The low transmission of sinus tumors to bighorns as compared to domestic sheep was surprising, and may suggest species differences in susceptibility to this disease. Measures to prevent transmission to domestic sheep may be warranted, including exclusion of domestic sheep from the natural range of bighorn sheep.
Footnotes
Acknowledgements
We thank the Colorado Division of (Parks and) Wildlife, the Colorado bighorn sheep auction and raffle fund, Colorado State University College of Veterinary Medicine and Biomedical Sciences College Research Council, and the Boone and Crockett Club for financial contributions. We thank Mindy Meyers, Laura Bernhardt, and Olivia Hutton for assistance with animal husbandry, Lisa Wolfe for assistance with anesthesiology and veterinary care of experimental animals, Nicolle Linnerth-Petrik and Lisa Santry for ENTV/JSRV IHC staining, and Todd Bass for slide preparation and assistance with other IHC stains.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
